Abstract
Ocular toxoplasmosis, a disease of the eye caused by the protozoan parasite Toxoplasma gondii, represents a common cause of posterior uveitis. The Authors review the current Literature regarding the uncommon presentation of ocular toxoplasmosis as macular serous retinal detachment (SRD). It is imperative to keep in mind that inflammatory SRD is a possible presentation of toxoplasmic retinochoroiditis. Underestimation of this clinical scenario and treatment with steroids alone without appropriate antiparasitic drugs, could lead to devastating consequences.
Introduction
An otherwise healthy 20 year-old-male was referred by his ophthalmologist with a diagnosis of “atypical” central serous retinopathy in the right eye (OD). Best corrected visual acuity (BCVA) was 20/400 OD and 20/20 in the left eye (OS). Anterior segment examination was unremarkable in each eye while fundus examination disclosed mild vitritis, two superior extramacular scars (dotted arrows) and a new whitish choroidal lesion (arrow) in the papillomacular bundle OD (Figure. 1). A wide serous macular detachment was shown on Optical Coherence Tomography (OCT) OD (Figure. 2), while fundus examination and OCT were unremarkable OS. Signs of arteritis were seen on fluorescein angiography and indocyanine green angiography (Figure. 3). He was treated with pirimetamine, sulphadiazine and folic acid with gradual resolution of symptoms and signs. At three months follow up BCVA improved to 20/20 and OCT showed a normal foveal thickness with a residual chorioretinal scar (Figure. 4).
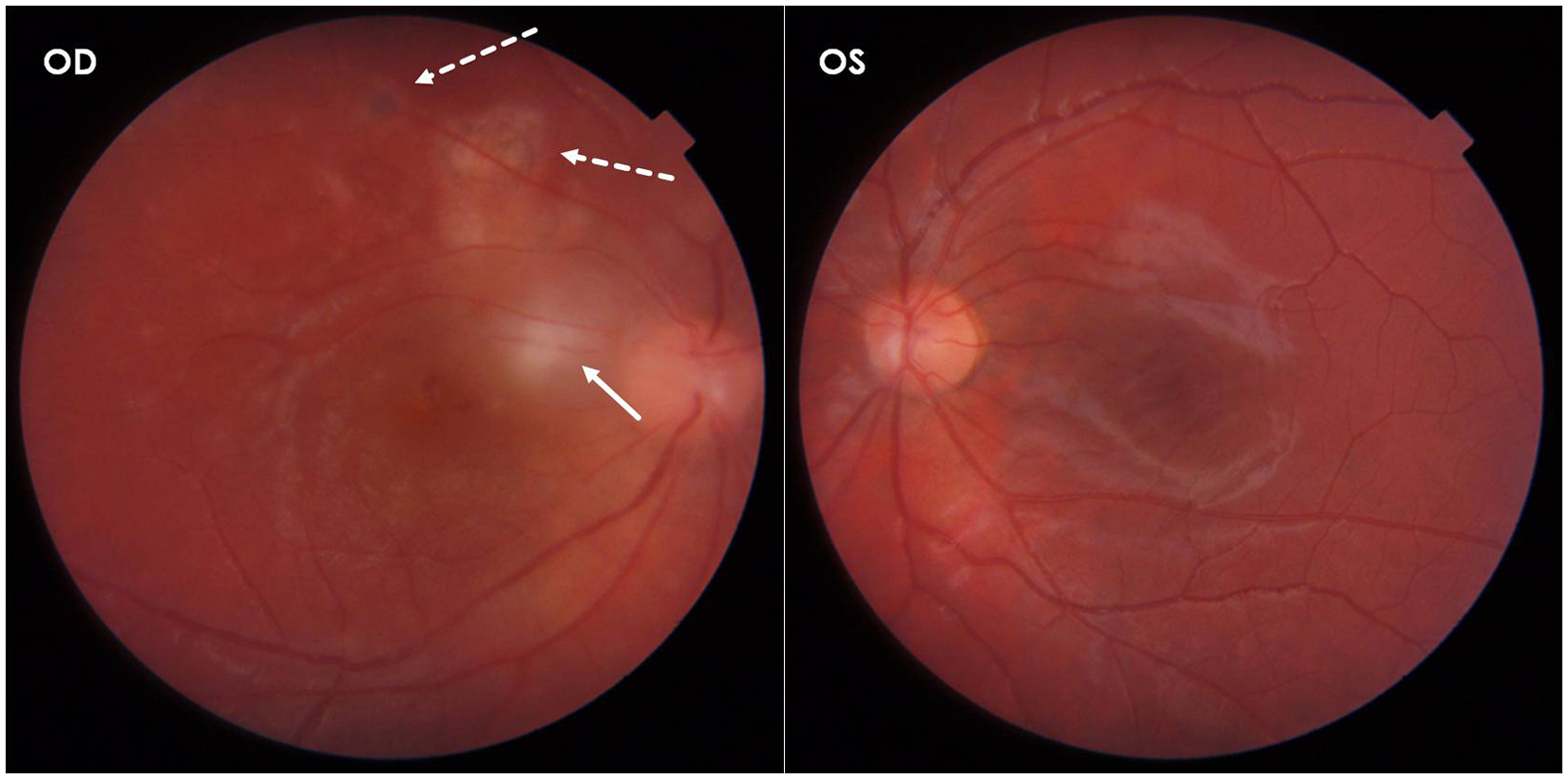
Fundus examination disclosed mild vitritis, two superior extramacular scars (dotted arrows) and a new whitish choroidal lesion (arrow) in the papillomacular bundle in the right eye.
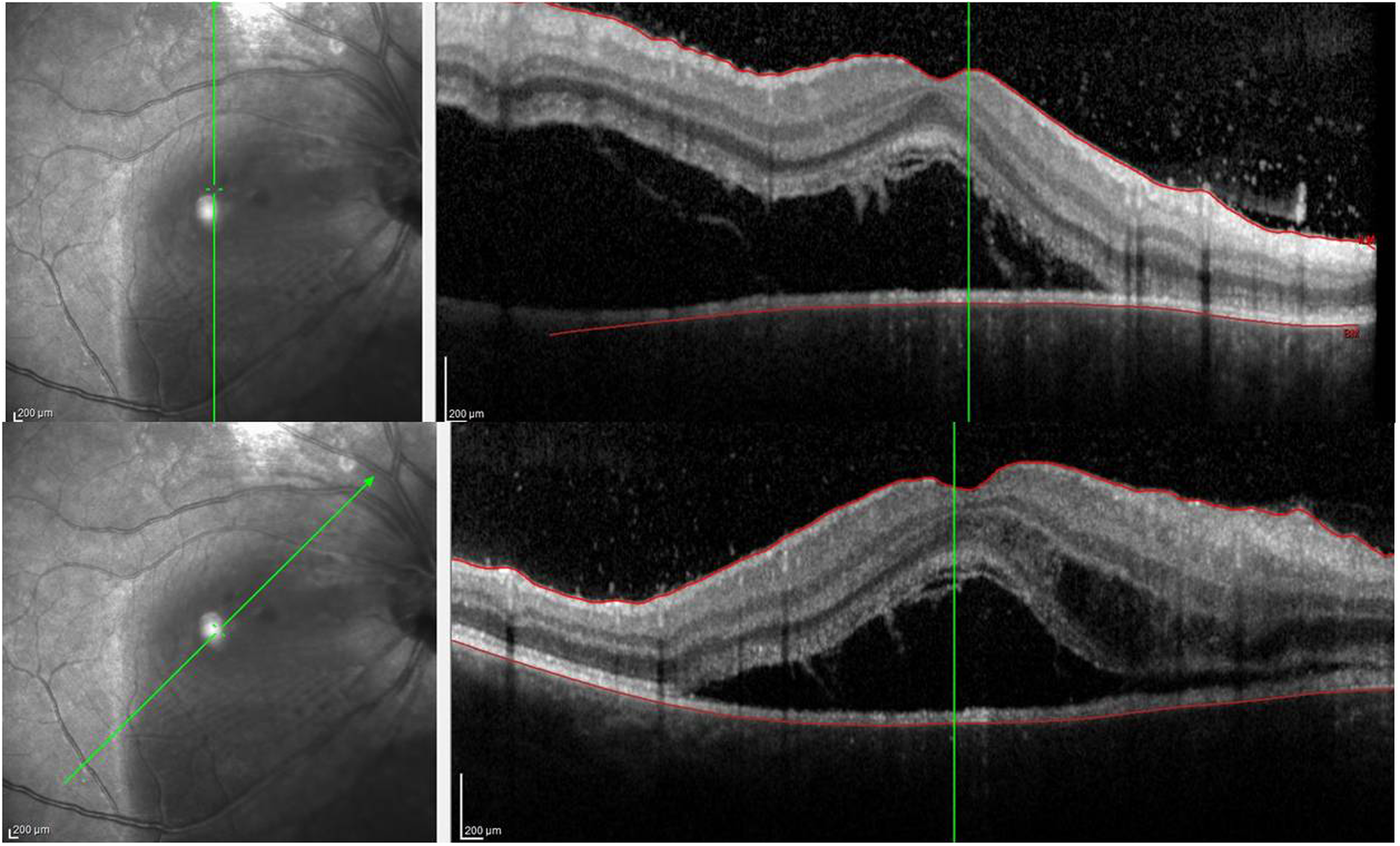
A wide serous macular detachment was shown on Optical Coherence Tomography.
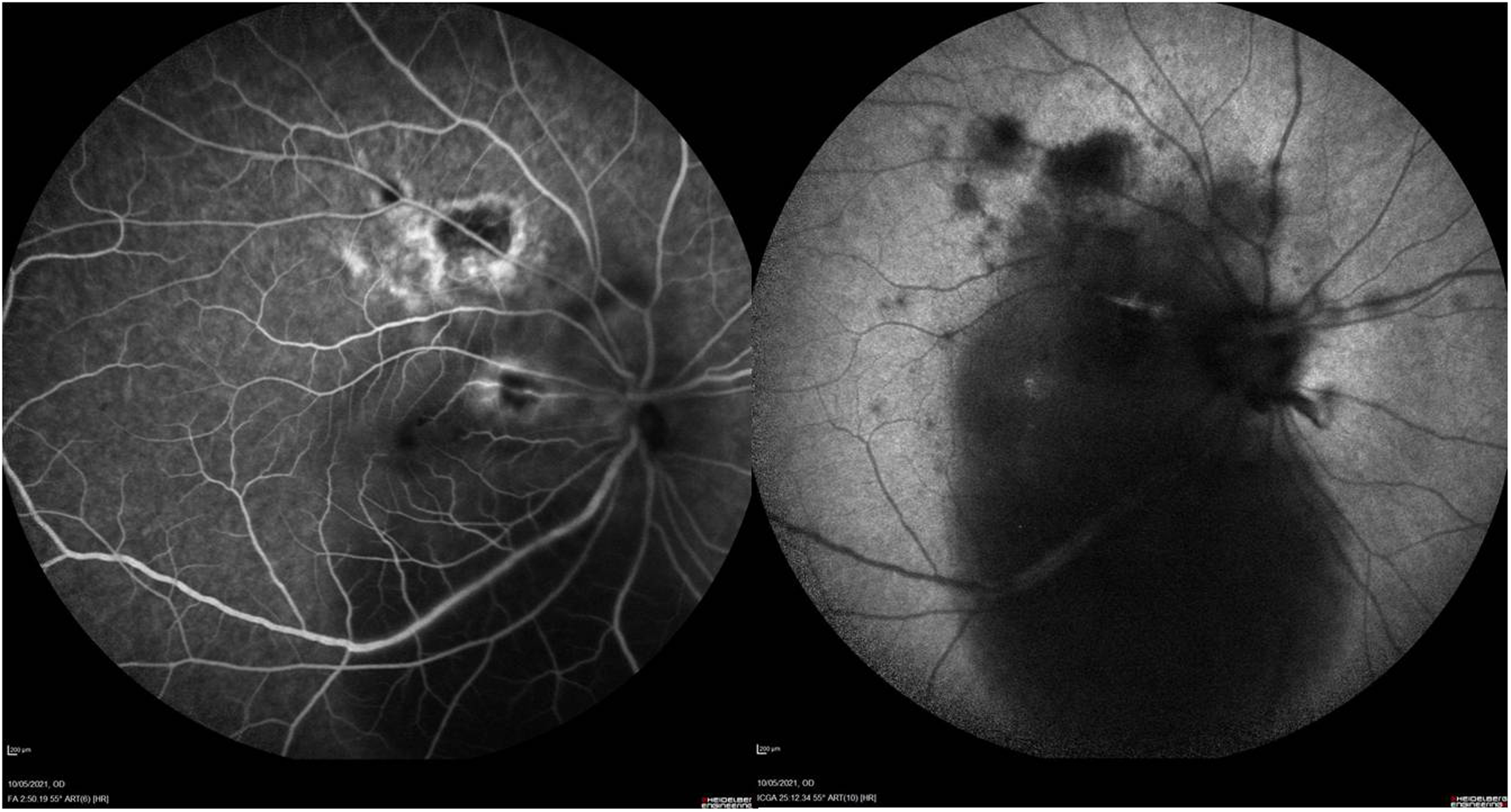
Arteritis were seen on fluorescein angiography and indocyanine green angiography.
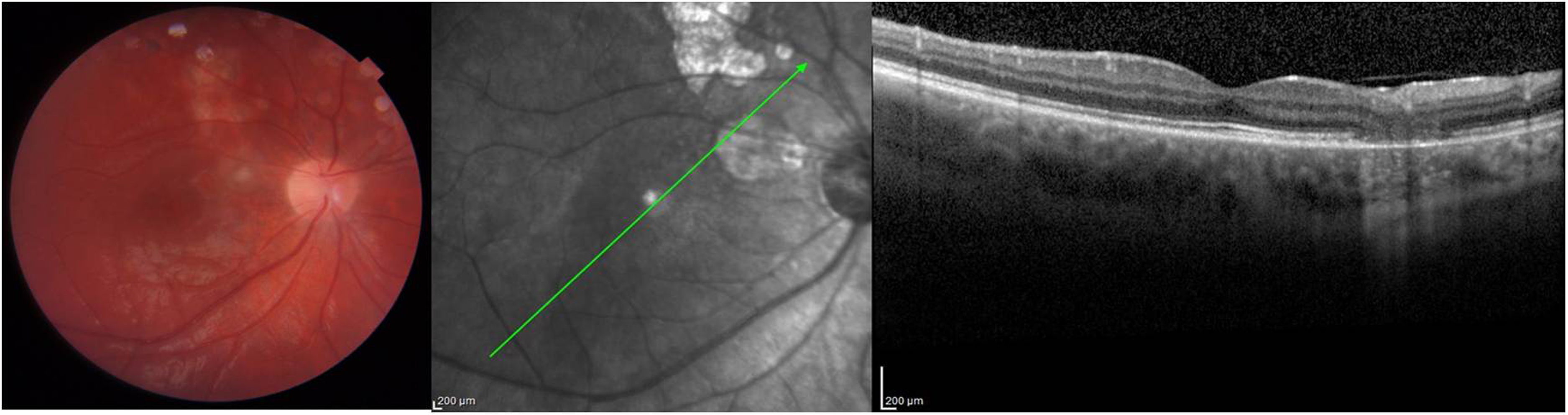
At three months follow up OCT showed a normal foveal thickness with a residual chorioretinal scar.
Toxoplasmosis represents the most common infectious cause of posterior uveitis worldwide. 1 It results from the obligate intracellular protozoan parasite Toxoplasma gondii (T. gondii) that can affect all warm-blooded vertebrates, including humans. 2
The infectious stages of T. gondii include sporozoites (contained within oocysts), tachyzoites and bradyzoites. Oocysts originate in feline intestines and sexual reproduction is required. Sporulated oocysts contain two sporocysts, and each sporocyst contains four sporozoites enclosed in a cell wall. After being spread out with defecation, these forms become infectious after 1–5 days by sporulation. Tachyzoites, which represent the fastest reproducing form, can invade almost any type of host cell, and replicate until the cell is filled with parasites. This leads to cellular lysis and systemic disseminations of tachyzoites, followed by re-entry into a new host cell; multifocal tissue necrosis may occur as a result of this cycle. In most cases, the immune system of the affected individual limits the infection and T. gondii enter the dormant form of bradyzoites, characteristic for chronic infection. Bradyzoites are isolated in tissue cysts; every cyst contains hundreds of bradyzoites, and its size may reach up to 60 microns. Usually, the cysts do not cause host reaction and remain inactive in the host. 3
The humans act as intermediate hosts and the transmission may occur by various routes, including ingestion of infected raw or undercooked meat; water, soil or vegetables contaminated with oocysts; drinking infected unpasteurized milk; blood transfusion or organ transplantation; and intrauterine or transplacental transmission. 4
The typical ocular presentation of toxoplasmosis is characterized by necrotizing focal retinochoroiditis adjacent to a pigmented chorioretinal scar and vitreous inflammation. In addition, atypical manifestations of ocular toxoplasmosis have been reported; these include aggressive (large, multifocal and/or bilateral) retinochoroiditis, neuroretinitis, punctate outer retinal toxoplasmosis (PORT), retinal vascular disease, pigmentary retinopathy, optic neuropathies, rhegmatogenous or tractional retinal detachment, serous retinal detachment (SRD), and scleritis. 1
This review aims to describe clinical features, diagnosis, and treatment of ocular toxoplasmosis (OT), focusing on SRD as a rare but possible manifestation of the disease.
Methods
The literature review for this article was based on a search in PubMed/Medline and Google Scholar database to select reviews, original articles and case report published in English language from January 1974 to July 2022 about OT. The following keywords were used: “Toxoplasma gondii”, “Ocular toxoplasmosis”, “Atypical presentations”, and “Serous retinal detachment”. Additionally, we used the images of one of our patients with SRD and vitreitis as presenting symptoms of OT.
Results
Epidemiology and pathophysiology
T. gondii is a ubiquitous microorganism present all over the world and approximately 25–30% of the human population is infected. 5 However, seroprevalence varies widely, ranging from 10 to 80%, probably because of the prevalence of T. gondii cysts and oocysts in the environment . Ocular toxoplasmosis is likely underdiagnosed in many countries with endemic T. gondii infection. The possibility of acquiring toxoplasmosis depends on geographical factors, presence of animal reservoirs and release of oocysts, meat consumption, and other personal habits. Furthermore, virulence varies by strain and susceptibility based on an individual's genetic traits, patient's age and immune status, and parasite-related factors.6,7 Seroprevalence varies widely from 20% to 60–80%; the rates of infection are higher in tropical environments due to humidity and warmer temperature, which favor the maturation of oocysts in the soil.8,9 Imposed stricter hygiene standards and reduced consumption of raw or undercooked meat have significantly reduced the risk of infection in many countries, especially in Europe. 10
In pregnancy, the initial infection may cross the placenta and reach the fetus. Congenital toxoplasmosis can lead to severe complications, including fetal death, congenital malformations or involve retina. Most affected adults have no symptoms, or present fever and lymphadenopathy but any organ can be affected, especially the eye. About 5000 people develop OT in the USA annually. 11 Although OT has been considered a recurrence of congenital infection so far, recent evidence suggests that many cases of ocular involvement are postnatal acquired infections. 12
Clinical features
Retinochoroiditis
The most common site of ocular infection is the retina, but almost always the underlying choroid is secondly involved producing “retinochoroiditis”. Acute OT typically manifests as a yellow-whitish, fluffy, necrotizing lesion adjacent to a previous pigmented scar. Often the lesion is obscured by vitreous inflammation, more prominent over the lesion. Vitritis may be severe causing epiretinal membrane formation and therefore vitreoretinal traction near the lesion. Vitritis can be even absent when the inflammation does not reach the inner limiting membrane. Vasculitis and retinal hemorrhages can be detected near or even distant from the lesion. Periphlebitis is the more common pattern observed; a segmental retinal arteritis, known as ‘Kyrieleis plaques’, can been present. Kyrieleis plaques are most often seen in eyes with active toxoplasma retinochoroiditis but have also been reported in other infections.13–15 They present as nodular or segmental yellow-whitish plaques that do not extend outside the artery. Pichi et al. demonstrated that Kyrieleis plaques are characterized by an inflammatory involvement within the vessel wall, probably the endothelium, and proposed to shift the nomenclature from ‘periarteritis’ to ‘endothelitis’. 16 Anterior segment inflammation can be either granulomatous or non-granulomatous and varies from a quiet anterior chamber to a severe anterior uveitis. Prolonged and intense inflammation in the anterior chamber can lead to posterior synechiae formation.
In immunocompetent patients, the acute lesion spontaneously heals after few weeks with gradual regression from the periphery to the center, leaving scar tissue; the final retinochoroid scar progressively pigments starting from the edges.
Aggressive retinochoroiditis
In some cases, active lesion may be large, multiple and/or bilateral. Severe disease may occur most frequently in individuals with compromised immune system, such as those treated with corticosteroids, cytotoxic medicines and antibodies against tumor necrosis factor alpha; patients with malignancies; and HIV-patients.1,4,17
Neuroretinitis
Neuroretinitis is typically characterized by sudden and profound visual loss, and optic disc inflammation associated to a partial or complete ‘star-shaped’ exudation around the macula, which usually appears up to two weeks after the onset of the disease. Neuroretinitis is most common associated with cat scratch disease (Bartonellosis), but may be seen in other infectious conditions, including syphilis, Lyme disease, Rocky Mountain Spotted Fever, toxoplasmosis, toxocariasis, histoplasmosis, leptospirosis and many others. 18
Punctate outer retinal toxoplasmosis (PORT)
Punctate outer retinal toxoplasmosis (PORT) is a more limited form of ocular toxoplasmosis, located in the deeper layers of the retina and retinal pigment epithelium (RPE). PORT is characterized by multifocal, small, gray-white lesions; because of lesions are limited to the outer retinal and RPE, vitreous inflammation may be minimal or absent. Optic neuropathy may occur secondarily, causing significant visual loss. 19 It has been reported that PORT occurs more frequently in persons in their first twenty years of life; bilateral involvement is observed in one third of cases while others may present typical OT in one eye and PORT in the fellow eye. 20 With an appropriate antiparasitic therapy, prognosis of PORT is usually good, but a granular retinochoroid may develop.
Other atypical manifestations
Retinal vasculitis is common in patients with OT and, although venous involvement is the most common pattern, arteritis and Kyrieleis plaques may also develop. In rare cases, vasculitis may be occlusive resulting in retinal venous thrombosis or retinal infarction. Retinal and subretinal neovascularization may rarely occur; frosted branch angiitis and Roth spots have been also reported as rare forms of retinal vascular disease in OT. 1
Unilateral pigmentary retinopathy mimicking retinitis pigmentosa has been reported in few patients with bilateral recurrent ocular toxoplasmosis with both typical retinochoroidal lesions and positive serology for T. gondii. 21
While optic disc swelling and neuroretinitis are common findings of OTs, other optic neuropathies, such as nerve head mass, anterior optic neuropathy, and optic neuritis have been reported. 1
Scleritis is a rare manifestation of toxoplasmosis, which may be more common in HIV-patients and other immunocompromised individuals. 22 Although scleral inflammation generally results as a contiguous spread from a retinochoroidal focus to the overlying sclera, it is even possible to develop scleritis at a distant location.
Retinal detachment is another complication of ocular toxoplasmosis. Most of the retinal detachment is rhegmatogenous or tractional in nature, but SRD may also occur in T. gondii ocular infection.
Serous retinal detachment (SRD)
Although there are only few reports in literature describing SRD in patients with OT, SRD may be an underdiagnosed feature of ocular toxoplasmosis (as the presented case). In some cases, the exudation was severe enough to suggest the diagnosis of Coat's disease but in other patients the exudative reaction was limited to the macula.23–25 When the macula is involved, a substantial deterioration of visual acuity is observed. The role of choroid in the process of fluid accumulation under the macula cannot be overemphasized. An atypical presentation of OT based on exudative retinal detachment associated with choroidal ischemia without vitritis has been recently reported. 26 The authors concluded that “the exudative retinal detachment was likely due a temporary dysfunction in retinal pigment epithelial. Inflammatory processes or choroidal ischemia may contribute to RPE decompensation causing accumulation of subretinal fluid”. Choroidal ischemia can be detected with fluorescein and indocyanine green angiography, which image areas of choroidal hypoperfusion. Possibly, choroidal ischemia may be the underlying causative phenomenon in cases with poor visual acuity and chorioretinal involvement distant from the macula. 26
Khairallah et al. found SRD in 14 of 60 eyes examined with acute OT (23.3%). 27 SRD was visible at funduscopic examination in 6 cases but was detectable only by optical coherence tomography (OCT) in the remaining 8 cases. Choroidal ischemia was a rare manifestation of T. gondii ocular infection. Previously, he had reported SRD in 37.5% of a small cohort of patients with active toxoplasmic retinochoroiditis and signs suggestive for acute choroidal ischemia. 28 In a recent observational case study, SRD was found in approximatively two thirds of patients with toxoplasmic retinochoroiditis in different stages of activity on OCT. 29
SRD is known to be a common feature of posterior uveitis. Recently, SRD was found in 78 of 2761 (2.8%) uveitis patients; while Vogt-Koyanagi-Harada disease by far is the most common cause of clinically detectable uveitic SRD, the second identified etiology is toxoplasmic retinochoroiditis (10.3%). 30
Spectral domain OCT (SD-OCT) allows to obtain a better view of intraretinal microstructure and subretinal space with higher axial resolution and faster acquisition speed. SD-OCT is superior to time domain OCT (TD-OCT) in revealing and delineating retinal abnormalities and features of retinochoroiditis in patients with OT. SD-OCT is a useful tool to detect subtle macular changes related with toxoplasmic retinochoroiditis, such as epiretinal membrane, intraretinal cysts, vitreous retina interface anomalies, and even small SRD not visible either on fundus examination or on TD-OCT. 31 In fact, when SD-OCT is performed, SRD is found as a common feature in patients with OT.32,33 Particularly, Ouyang et al. noted that SRD was present in 45.5% (or 15/33) in eyes with typical active OT and 51.3% (or 20/39) in eyes with all active toxoplasmic retinochoroiditis, using an extramacular OCT scanning protocol. 32 Most cases of SRD was associated with retinal necrosis and reacted well to conventional therapy regardless total fluid volume. Nevertheless, when accompanied with central macular edema or choroidal neovascularization (CNV), SRD remained refractory or responded less favorably to conventional or combined intravitreal treatment, even when the amount of fluid was small.
Additionally, Ouyang and co-authors had first reported an SD-OCT finding of SRD in OT, described as a huge outer retinal cystoid space containing disorganized hyperreflective material and bordered exteriorly by a hyperreflective line continuous both in location and thickness with the ellipsoid. 34 They theorized that this outer retinal cystoid space would represent fluid lying within the photoreceptors. Lujan presented a similar case of OT with a large subfoveal collection of subretinal fluid and weakly reflective material at the base on SD-OCT. 35 Because of the lack of anatomical potential space within the photoreceptor layer, he suggested that this hyperreflective line could represent subretinal fibrin under the SRD. Finally, Mehta et al. described a patient with preexisting pachychoroid disease and recurrent toxoplasmosis chorioretinitis presenting with a photoreceptor splitting detachment. 36 They hypothesized that this SD-OCT finding may precisely represent a split of the photoreceptor layer at the level of the myoid and suggested the term ‘bacillary layer detachment’.
Diagnosis
The diagnosis of OT is generally evident based on typical presentation. Serologic tests including anti-T. gondii titers of IgM and IgG may support the diagnosis. Serum IgM and IgG antibodies develop within one or two weeks after infection. IgM levels progressively rise within one or two weeks after infection and become undetectable after 6–9 months. 37 In clinical practice, the presence of T. gondii-specific IgG in the serum indicates a past contact between the host's immune system and the parasite, but neither elevated levels of IgM nor low levels of IgG should be considered as evidence of a recent infection. The determination of the avidity of IgG by the titration method in patients with detectable IgM antibodies defines most accurately the stage of infection by T. gondii. 38 In immunocompromised individuals, including HIV patients and transplant recipients, serum antibodies response may not be observed, and high IgG levels defines the risk for reactivation of the disease. 8
In some cases, the analysis of intraocular samples is needed to confirm the diagnosis; the Goldmann-Witmer coefficient (GWC), which compares the specific antibodies levels in serum and in ocular fluids, and polymerase chain reaction (PCR) techniques, detecting T. gondii DNA in aqueous humor or vitreous, are useful tools for the diagnosis of OT. Typically, GCW is more sensitive in immunocompetent patients because the immunological response is usually unpredictable in immunocompromised persons. The diagnostic value of PCR in the diagnosis of OT is controversial, due to its low sensitivity despite high specificity. However, PCR may represent an appropriate tool to corroborate diagnosis of OT, especially in immunocompromised patients with seronegativity to T. gondii.
Multimodal imaging can be useful in demonstrating typical and atypical findings of OT. On fundus autofluorescence (FAF), the active lesion shows hyper- and/or isoautofluorescence. As the healing process starts, a hypoautofluorescent rim appears and extends centripetally until the lesion becomes completely dark; SRD is visible as a mild grey area on FAF.
On fluorescein angiography (FA), the typical lesion appears as a masking hypofluorescent area in early phase, followed by progressive leakage from the edges to the center. Vasculitis is seen as hyperfluorescence of the vessel walls, which increases in the late phases of angiography. Kyrieleis plaques are hypofluorescent in the early phase and become hyperfluorescent in the intermediate and late phases with no leakage. On FA, SRD is observed as a dark area with late dye staining at the borders. Pigmented scars have a persistent mask defect, often bordered by hyperfluorescence. An increasing hyperfluorescence of the optic disc is seen when papillitis is present. FA is also useful to detect possible complications of OT, such as retinal vessel occlusion, retinochoroidal vascular anastomosis, and inflammatory CNV. Ultra-widefield angiography (UWFA) is a novel technique able to detect subtle peripheral changes and total extent of the inflammation, and to monitor modifications over time. Wherever available, UWFA could become the imaging modality of choice in toxoplasmic retinochoroiditis and other posterior uveitis.
Indocyanine green angiography (ICGA) typically shows a hypocyanescent area usually larger than the overlying retinal lesion, corresponding to the underlying choroidal thickening; adjacent or even distant dark spots may be also observed probably due to a more diffuse choroidal inflammation. ICGA is the most accurate imaging method to highlight Kyrieleis plaques which appear markedly hypercyanescent, suggesting the endothelial involvement 16 ; SRD is shown as a grey area on ICGA.
As discussed above, SD-OCT is a worthwhile tool to detect subtle macular changes, assess the real extent of the disease in the posterior segment, and follow up the healing process in toxoplasmic retinochoroiditis. OCT scan encompassing a typical active lesion generally shows hyperreflective retinal thickening with disruption of the retinal layers and shadowing on the underlying structures. 39 Posterior hyaloid thickening and/or detachment and vitreitis over the lesion can also displayed and an underlying choroidal thickness as well. Over time, a progressive resolution of the retinal and choroidal thickening is seen; a well-demarcated focal area of retinal thinning, hyperreflectivity and disruption of retinal pigment epithelium (RPE) and outer retinal layers are left in the site of the scar. SD-OCT is also able to show Kyrieleis plaques as hyperreflectivity of the entire wall of the involved segment of vessels with a normal reflective lumen. 16 SD-OCT may easily detect SRD, even if small and located in extramacular sites, and show microstructural changes seen in some cases, such as ‘bacillary layer detachment’ (see above). The characteristic modification of RPE-choriocapillaris and deeper retinal layers in PORT as well as complications of toxoplasmic retinochoroiditis, including cystoid macular edema, epiretinal membrane, vitreoretinal traction, macular hole, and CNV may also be displayed on SD-OCT. Different techniques in SD-OCT imaging, such as enhanced-depth imaging OCT (EDI-OCT) and swept source-OCT (SS-OCT) are developed in the last years. On EDI-OCT, some degrees of diffuse choroidal thickening even distant from the primary retinochoroidal inflammatory focus may be visible, consistent with more diffuse choroidal inflammation often seen on ICGA. In the context of acute toxoplasmic retinochoroiditis, SS-OCT has the advantage of visualizing choroidal, retinal and vitreous changes in a single scan, if compared with SD-OCT which requires different imaging algorithms. 40
OCT angiography (OCTA) is a novel technology which allows noninvasive analysis of choroidal and retinal microvasculature. At the site of the typical acute toxoplasmic retinochoroidal lesion, OCTA usually depicts attenuation of the retinal microvasculature accompanied by underlying focal hypoperfusion of the choriocapillaris corresponding to the area of reactive choroidal inflammatory thickening. 41 During resolution, the inflammatory focus leading to obliteration progressively diminishes, eventually leaving various degrees of focal drop out of the choriocapillaris. OCTA is also able to display vascular occlusion and CNV as complications of OT.
Treatment and prognosis
Treatment of OT is still controversial; typically, in immunocompetent individuals the disease has a benign course, and it is expected to resolve within one or two months. So, considering the potential toxicity of the antiparasitic drugs, the treatment can be adjusted for each patient individually, leading some clinicians to not treat small and peripheral lesions. Treatment of OT generally includes combination of two antimicrobial drugs, most often inhibitors of dihydrofolate reductase (i.e., pyrimethamine and trimethoprim) and dihydropteroate synthetase (sulfonamides) which block folic acid synthesis, plus corticosteroids. 42 The ‘classic therapy’ consists of pyrimethamine, sulfadiazine and systemic corticosteroids maintained for 4–6 weeks. A good alternative in terms of high tolerability, wide availability and low cost is the combination trimethoprim-sulfamethoxazole, also known as ‘co-trimoxazole’; it is also prescribed for secondary prevention to a subset of patients, such as immunocompetent persons with multiple recurrences, especially those at risk of visual loss or irreversibly blind in the fellow eye, and immunocompromised individuals as well. Intravitreal injection of clindamycin with steroids represents an acceptable alternative treatment for OT. Two other antiparasitic drugs, atovaquone and azithromycin, have been used to treat OT; while there are only few data on the clinical utility of atovaquone, azithromycin was found to have promising results in selected cases, mostly in combination with pyrimethamine. 42 Given the high risk regarding the adverse effects of the antiparasitic drugs, spiramycin and sulfadiazine seem to be the safest drugs in pregnancy. Corticosteroid treatment, both topically and systemically, is commonly used in healthy patients and relatively less in immunocompromised persons; it is generally started a few days after initiation of antimicrobial therapy and ceased with gradual tapering before the antimicrobial treatment is stopped. A recent paper reported that two thirds of International Ocular Toxoplasmosis Study Group members prefer the combination trimethoprim-sulfamethoxazole as first line treatment regimen for OT, whilst ‘classic therapy’ is favored by just over the 10% of uveitis specialists. 43 Almost all members associate systemic corticosteroids and about one half of them employ steroid eye drops.
The prognosis of OT is quite variable; a best-corrected visual acuity of less than 20/200 has been associated with atypical OT, lesions larger than one disc-diameter, macular lesions, and any complication. 44
Conclusions
Serous retinal detachment may be caused by a number of retinal diseases of inflammatory, vascular or infectious origin. Ocular toxoplasmosis, the most frequent infectious etiology of posterior uveitis, typically presents with retinochoroiditis, but may also be a cause of serous retinal detachment. Underestimation of this uncommon presentation and treatment with steroid alone without appropriate antiparasitic drugs, could lead to devastating consequences.
Footnotes
Author contributions
All authors contributed to the study conception and design: D.C. and F.P.: writing; E.C and F.L.: case presentation and collection of images; A.C. and L.C.: review of the manuscript. All authors read and approved the final manuscript.
Competing interests
The authors have no relevant financial or no financial interests to disclose.
Consent to publish
Written informed consent was obtained by the patient for publication of the images as they appear in the text
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
