Abstract
Purpose
To describe a new surgical technique designed to reduce the risk of tube exposure following glaucoma drainage device (GDD) surgery.
Methods
A retrospective, single-center study included all patients who underwent GDD surgery by a single surgeon between January 2018 and December 2019, using a double scleral tunnel technique to secure the tube to the sclera. A 23-gauge needle was used to create a 3 mm partial thickness scleral tunnel near to the GDD plate, with a second partial thickness scleral tunnel created 2.5 mm from the surgical limbus into the anterior chamber (AC). The silicone tube was passed through both scleral tunnels and into the AC.
Results
46 GDD surgeries were performed during the time frame. The mean age of patients was 65 ± 13 years. Mean follow up duration was 27.7 ± 8.15 months (range 11 to 44). Significant reductions in intraocular pressure and number of glaucoma medications were achieved. Zero exposures were recorded occurring during follow-up.
Conclusion
This double scleral tunnel technique to secure the GDD silicone tubing firmly to the sclera may reduce micro-movements which have been purported to be associated with tissue erosion post GDD surgery. The addition of this simple technique to existing GDD surgery may reduce the risk of post-operative tube exposure.
Introduction
Glaucoma drainage devices (GDDs) are increasingly being used in the surgical management of patients with previous failed glaucoma filtering surgery or as a primary surgical intervention in refractory glaucoma.1–3 In standard GDD surgery, the length of the tube between the GDD plate and the limbus is around 8–9 mm, typically with the last 1–2 mm lying within a short scleral tunnel before entering the anterior chamber (AC), leaving 6–8 mm of exposed tube in the subconjunctival space. Tube erosion tends to occur in the exposed section, and given that erosion presents a serious infection risk, 4 various techniques are currently used to cover this exposed section, including; suturing the tube to the sclera with non-absorbable suture, using patch graft (such as human donor sclera or cornea, Tutoplast® Pericardium or fascia lata), and creation of partial thickness scleral flap to cover the tube. Despite this, tube erosion in the Tube Versus Trabeculectomy (TVT) Study showed a cumulative rate of 4.7% at 5 years. 5 The cause of tube erosion is likely to be multifactorial but it has been suggested that micro-movements of the tube secondary to ocular pulsation and blinking may be an important contributing factor. 6 These micro-movements may contribute to erosion of the overlying patch graft and conjunctiva subsequently, due to friction between the device and the overlying tissue.
With the aim of reducing micro-movements of the tube, we propose a simple additional step of creating a partial thickness double tunnel using a blue 23-gauge needle to firmly secure the tube portion of the GDD to the sclera once the plate has already been secured. The external diameter of the 23-gauge needle is similar to the silicone tubing of the Baerveldt/Ahmed GDDs. The silicone tubing can thus be passed through the scleral tunnels into the AC and secured to the sclera with resultant minimal horizontal micro-movements, conferring a theoretical advantage over existing techniques. We describe our preferred surgical technique for implanting GDDs and our preliminary results.
Methods
This is a retrospective, single-center study involving a consecutive series of patients who underwent GDD surgery using a double scleral tunnel technique to secure the tube to the sclera. Surgeries were performed between January 2018 and December 2019 by a single surgeon (KSL). The study was performed in accordance with the Declaration of Helsinki. All patients underwent a standard ophthalmic examination preoperatively including best-corrected visual acuity, slit lamp examination, Goldmann applanation tonometry, anterior segment and dilated fundus examination. Demographics, diagnoses, operative details, number of glaucoma medications were also recorded.
Statistical analysis was performed with STATA SE version 17.0 software (StataCorp LLC). Snellen VA was converted to logMAR units. Continuous variables were expressed as mean (SD) and were compared with two-sided t-test. A p value of < 0.05 was considered significant. Categorical variables were expressed as numbers (percentages).
Surgical technique
The following describes the technique for superior-temporal Baerveldt tube placement in the AC (see video, Supplemental Digital Content 1, for demonstration of double scleral tunnel technique).
A 7-0 silk traction suture is passed through the superior cornea followed by a broad conjunctival peritomy. Subtenons dissection down to bare sclera is performed with blunt dissection between the superior and lateral rectus muscles. The extraocular muscles are isolated with muscle hooks and the tube plate inserted under the muscles. The plate is positioned approximately 9–10 mm posterior to the limbus and 7-0 Prolene is used to secure the GDD plate to sclera. A 3-0 polyamide (Supramid) intraluminal stent is passed approximately 3 mm into the tube with a 7-0 vicryl ligature to constrict the tube to completely occlude flow in the early postoperative period. Fenestrations are created on the tube proximal to the points of occlusion using the head of the 7-0 vicryl needle passed into the side of the tube one to three times.
To perform the double tunnel scleral entry technique, a 23-gauge needle is bent with bevel facing up. The course of the initial 3 mm tunnel is marked on the sclera using callipers, starting ∼1.5 mm from the GDD plate and directed towards the cornea. The 23-gauge needle is used to create an initial partial thickness scleral tunnel (Figure 1(a)). A feather blade is used to mark an entry point for the second tunnel, 2.5 mm posterior to the surgical limbus ‐ using the 2.5 mm needle bevel as a guide (Figure 1(c)). A small exposed tube section will be present between the end of the first and the second tunnel. The 23-gauge needle is used to create a 2.5 mm scleral tunnel directed towards the limbus (Figure 1(d)). Upon reaching the surgical limbus, the needle is directed downwards to enter the AC. The direction of the stay suture can be rotated toward the operator prior to AC entry to provide counter traction (Figure 1(e)). AC entry of the silicone tube should be at the level of the trabecular meshwork, aiming towards the iris. This assists with tube positioning away from corneal endothelium. The needle is removed and AC inflated with balanced salt solution before inserting the tube. Two forceps are used to insert the tube into the AC. The position of the tube is checked - aiming to be as close as possible to the iris plane and directed away from the corneal endothelium. For AC tube positioning, ∼3mm of silicone tube should be visible in the AC with the distal end of the tube trimmed bevel facing upwards to prevent iris plugging. The tube is sutured in place using 10-0 nylon sutures over the area of the silicone tube between the two scleral tunnels (Figure 1(f)). A patch graft of Tutoplast® Pericardium (Innovative Ophthalmic Products, Inc., Costa Mesa, CA, USA) is cut in a rectangular shape to cover the length of the tube between the limbal entry and GDD plate in a double layer, ensuring it is streamlined at the limbus to avoid dellen formation. 10-0 nylon is used to suture the patch graft in position with one mattress suture at the limbus and one compression suture over the patch graft. Prior to conjunctival closure, the free end of the supramid suture is trimmed and tucked under the subtenons/subconjunctival space inferotemporally. The conjunctiva is then closed with two purse-string sutures using 10-0 nylon and additional sutures to close the radial conjunctival incisions.
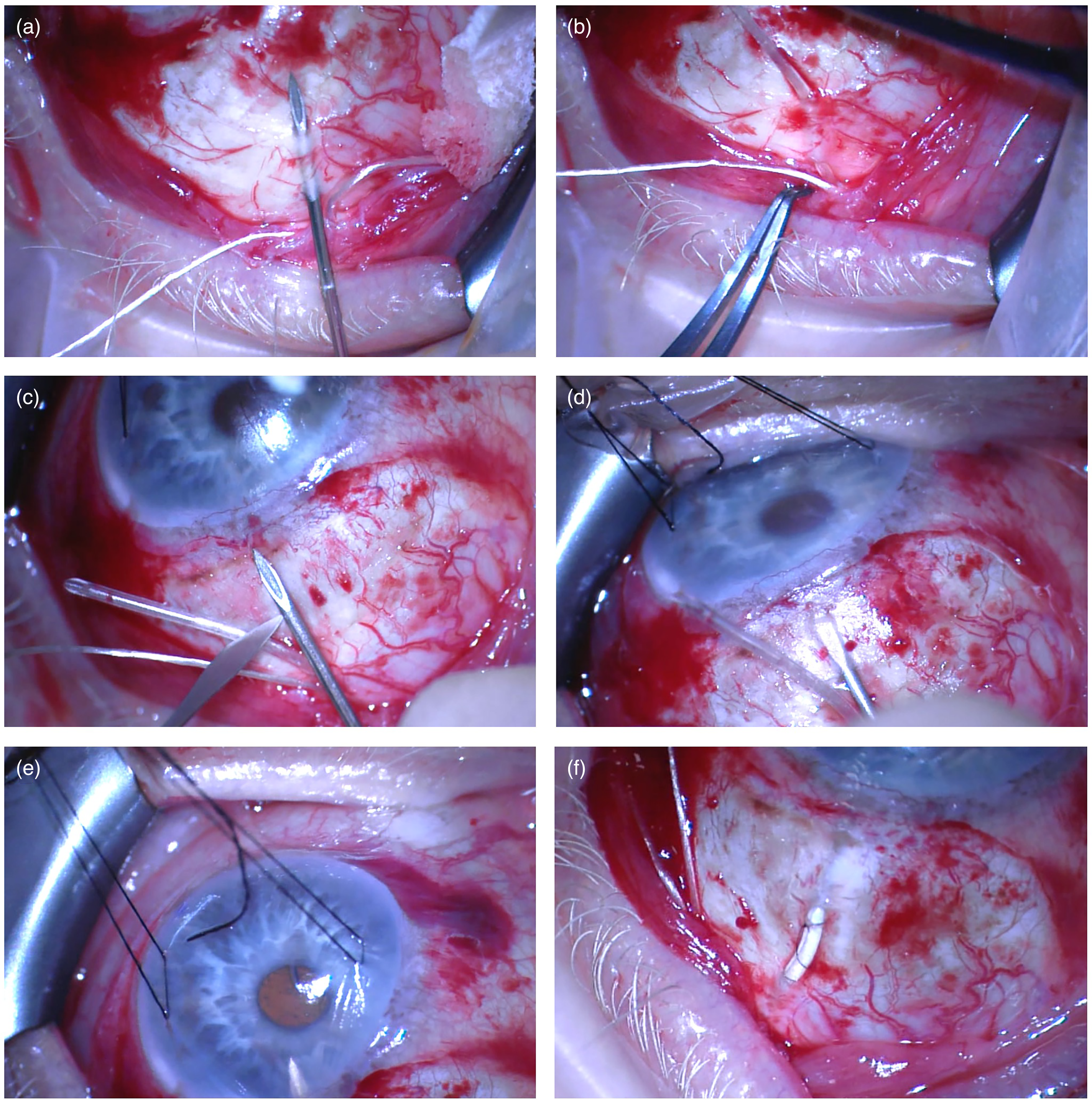
Double tunnel technique. (a). Sclera is marked and 23G needle used to create initial 3mm tunnel close to tube plate. (b). Tube is passed through initial tunnel. (c). Feather blade used to mark entry point for second tunnel, 2.5 mm posterior to limbus, using the bevel of 23G needle as a guide. (d). 2.5 mm tunnel is created with 23G needle. (e). Direction of stay suture is rotated to provide counter traction and direction of needle angled towards towards AC to enter AC. (f). Tube in double tunnel with 10-0 nylon mattress suture to secure exposed area of tube to sclera and prior to covering with Tutoplast/donor sclera.
Results
Since January 2018, we have performed 46 GDD surgeries with the double scleral tunnel technique described above. There were no cases of conjunctival erosion during the follow up period. The demographics, clinical characteristics and postoperative results are summarised in Table 1. The mean age was 65 in a predominantly male cohort (67% male). The majority of patients were of Afro Caribbean ethnicity (56%), followed by Caucasian (30%). POAG was the most common diagnosis (56%) as well as secondary open angle glaucoma (24%) and neovascular glaucoma (10.9%). Of the 11 patients with secondary open angle glaucoma, 1 had uveitic glaucoma, 5 developed glaucoma post-vitrectomy, 2 post-silicone oil, 1 post-complex phacoemulsification, and 1 patient had Reiger's anomaly. A further 2 patients had primary angle closure glaucoma and 2 patients had secondary angle closure glaucoma.
Demographic and clinical characteristics of patients.
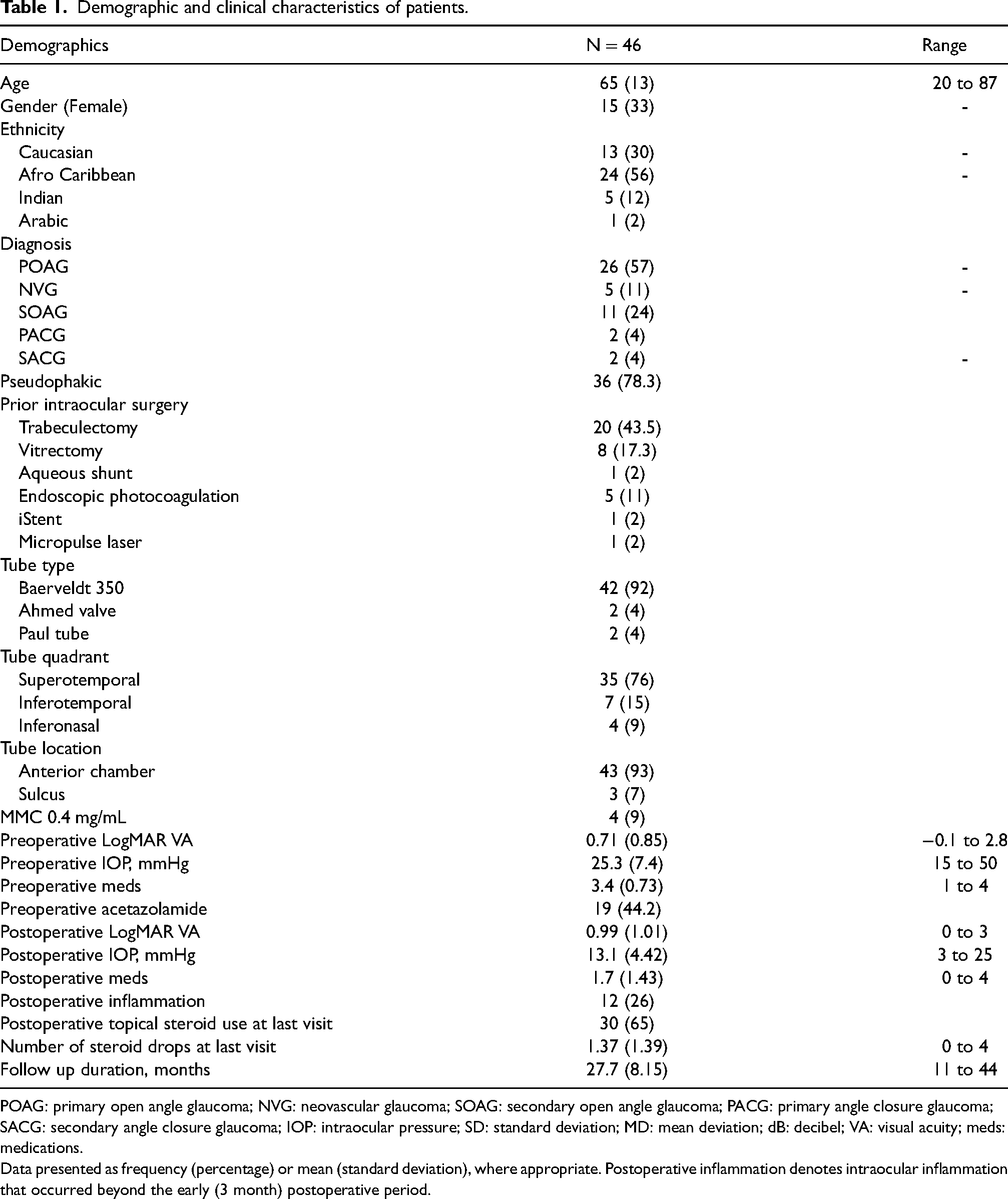
POAG: primary open angle glaucoma; NVG: neovascular glaucoma; SOAG: secondary open angle glaucoma; PACG: primary angle closure glaucoma; SACG: secondary angle closure glaucoma; IOP: intraocular pressure; SD: standard deviation; MD: mean deviation; dB: decibel; VA: visual acuity; meds: medications.
Data presented as frequency (percentage) or mean (standard deviation), where appropriate. Postoperative inflammation denotes intraocular inflammation that occurred beyond the early (3 month) postoperative period.
78.3% of patients were pseudophakic at the time of GDD surgery. Regarding other previous intraocular surgeries, 20 patients (43%) had undergone trabeculectomy, 8 patients (17.3%) had vitrectomy, 1 patient had a previous aqueous shunt, 5 patients had endoscopic photocoagulation, 1 patient had iStent implantation and 1 patient had Micropulse laser. 7 patients (15%) had undergone more than one surgery intraocular surgery prior to GDD insertion (excluding phacoemulsification).
The majority of GDDs were Baerveldt 350 implants (92%), with the plate positioned in the superotemporal quadrant (76%) and the tube located in the AC (93%). MMC 0.4 mg/mL was used intraoperatively for 4 patients (9%).
12 patients (26%) experienced significant postoperative inflammation, beyond the early (3 month) postoperative period. 30 patients (65%) remained on a low frequency of topical dexamethasone at the last postoperative visit recorded, with patients on a mean of 1.37 drops per day of dexamethasone at this visit.
Mean preoperative VA was 0.71 (LogMAR), which was significantly different from mean postoperative VA of 0.99 (p = 0.0012, paired t-test). Mean preoperative IOP was 25.3 and was significantly lower postoperatively at 13.1 (p < 0.001, paired t-test). The mean number of topical glaucoma agents used preoperatively was 3.4 and was significantly lower postoperatively at 1.7 (p < 0.001, paired t-test). The mean follow up duration was 27.7 months, with a range of 11 to 44 months.
Discussion
Tube erosion through the conjunctiva is an important long-term complication which may be mitigated by modifying GDD insertion technique. The traditional technique is to employ a short entry (1–2 mm) scleral tunnel before entering the AC and to suture a patch graft in place over the tube to prevent erosion. Use of a longer scleral tunnel has been described by several groups and has been shown to have a very low rate of long-term erosion. 7 We describe the use of a double scleral tunnel to secure the tube against sclera. The potential advantage offered by using scleral tunnels in GDD surgery may be related to improved stability of the tube and reduction of micro-movements, which has been posited as a mechanism for tube erosion and exposure.6–8
Albis-Donado et al. reported on low rates of exposure using a needle-generated single scleral tunnel with the Ahmed Glaucoma Valve, and suggested their observed lack of extrusions may have been related to reduced tube micro-movements with their technique. 7 They cited a further possible benefit of this technique in producing a flatter transition between conjunctiva and cornea, which may also assist in reducing the risk of exposure long term, by more evenly distributing pressure over the scleral surface as the upper lid moves during blinking.
The length of a long scleral tunnel varies between studies, from 3–4 mm7,9 to 5–6 mm, with some groups describing the use of longer tunnels with and without the use of overlying patch grafts.9,10 Our experience is that it is difficult to create a 5–6 mm tunnel safely and so we have modified our technique to use a double tunnel technique. We believe this technique strikes a balance between intraoperative safety through ease of surgery and long-term safety of the tube through improved stability. Various materials can be used as a patch graft over the tube, and this is a crucial step to further prevent erosion of the tube through conjunctiva. Our preference is for Tutoplast® Pericardium, which is a commercially available dehydrated human tissue graft. In units where donor tissue is readily available, donor sclera can be used as well as clear cornea.
An alternative approach to creating double scleral tunnels was described by Brouzas, et al., 11 with Ahmed valve implantation, in which a crescent blade was used to create both scleral tunnels, without the use of an overlying patch graft. In their study, they reported two cases of tube exposure, which further highlights the ongoing need for an overlying patch graft, even in the context of scleral tunnels.
We also considered other risk factors for tube erosion in the postoperative period, based on studies which have investigated these. Younger age was identified by Chaku et al. 12 as a risk factor in a case-control study. Mean age in their tube exposure group was 48.2 years compared to 67.3 years in the control group. The mean age of our cohort was 65 years, which would suggest a lower risk of exposure. Female gender has also been associated with an increased risk of tube exposure13,14 – in our cohort 33% were female. White ethnicity was reported by Muir et al. 14 as a risk factor for erosion, and our study included 70% non-white patients. Al-Beishri et al. found prior surgery to be a risk factor for erosion. 13 30 patients (65%) in our cohort had previous intraocular surgery (excluding phacoemulsification), with 7 patients having more than one prior surgery (15%).
Postoperative inflammation has also been highlighted as a risk factor for exposure. Chaku et al., found a significantly higher rate of inflammation at equivalent postoperative time points in patients who had tube exposure (65.6%) compared to a control group (9.4%). 12 While a significant proportion of our patients also had inflammation beyond the early (3 month) postoperative period (26%), we did not observe any cases of exposure across our cohort. Topical steroid use beyond the early postoperative period has also been associated with a significantly increased rate of exposure. 12 65% of our cohort remained on topical steroids at their most recent postoperative visit, albeit at a low mean number of drops per day (1.37).
Limitations of this study include a small sample size of 46 patients, as well as a relatively short follow up time at an average of 28 months. The literature suggests average time to tube exposure varies from 7 months 15 to 5.3 years, 16 with various time points in between.12,17–20 Further study is needed to evaluate longer-term outcomes and erosions may be captured in our cohort at later time points. Follow up intervals were based on clinician discretion, and varied depending on clinical need. It is possible that conjunctival erosions may have been missed in between follow up visits.
Notwithstanding the preliminary nature of these results, this technique offers a theoretical advantage in improving stability of the tube and minimising micro-movements in the postoperative period. Our view is that this improved stability may contribute to lower rates of conjunctival erosion and tube exposure in the long-term.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
EF is supported by the Hector Maclean Scholarship from the Royal Victorian Eye and Ear Hospital, Melbourne and the Centre for Eye Research Australia.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
