Abstract
Purpose
to report an uncommon presentation of Encorafenib-Binimetinib retinal side effects
Case report
A 56-year-old Caucasian woman, naïve to previous chemotherapies, was started on Encorafenib/Binimetinib for metastatic melanoma. After seven hours from the first 45 mg Binimetinib dose, the patient developed blurry vision with coloured halos. The symptoms were transient and the following day a complete ophthalmological examination revealed the presence of subretinal fluid (SRF) at Optical coherence tomography (OCT). After one week, the patient remained asymptomatic, with no signs of SRF at the follow up reevaluation. However, six weeks later, the symptoms originally experienced with the first drug intake appeared again. This time fundus examination revealed an irregular macular region. At infrared OCT an almond shaped hyporeflective lesion, surrounded by hyperreflectivity, was demonstrated without signs of SRF. Encorafenib/Binimetinib was continued at the same dose and strict monitoring was scheduled, according to the European Medicine's Agency indication to withhold the drug only in presence of symptomatic retinal pigment epithelial detachment.
Conclusion
Visual symptoms associated with SRF induced by Binimetinib have been described in the literature. In our case, visual symptoms were experienced by the patient at different times, both with and without evidence of SRF. This finding seems to suggest that while Binimetinib-induced SRF is an asymptomatic finding in most cases, with excellent outcome and rapid resolution, visual symptoms could be initially triggered by detectable SRF, yet persist without any further evidence of abnormal fluid accumulation and manifest intermittently.
Introduction
In the last few years, the combination of BRAF (v-Raf murine sarcoma viral oncogene homolog B) and MEK (mitogen-activated protein kinase) inhibitors has been widely used in patients with advanced melanomas, showing remarkable results in both adjuvant and metastatic settings.1,2 This combined targeted therapy acts by inhibiting the proliferative pathway induced by an altered protein encoded by the BRAF gene, found in approximately 50 − 60% of melanoma patients, which provides the tumour with an externally independent proliferative stimulus. 3 Overall, these drugs are well tolerated, with mild to moderate toxicities which can be generally handled rapidly. 3 As for MEK inhibitors, such as Trametinib, Cobimetinib and Binimetinib, the association with subretinal fluid (SRF) accumulation has been documented and its relatively benign nature has been described in the literature.4,5 Herein, we describe the uncommon development of intermittent visual symptoms, with and without concomitant funduscopic and OCT findings, in a patient treated with MEK inhibitors.
Case description
A 56-year-old Caucasian woman diagnosed with metastatic cutaneous melanoma (pT4b, N0, pM1a) was started on the standard dose of Encorafenib (450 mg q.d.) and Binimetinib (45 mg b.i.d.). The patient had no past ophthalmologic history and was naïve to both chemotherapy and corticosteroids. Family history was positive for glaucoma. After seven hours from the first Binimetinib dose (i.e., three tablets of 15 mg each), the patient suddenly developed blurry vision with coloured halos. These symptoms were reported to be bilateral and last for three hours, with complete resolution afterwards. The following day, she presented to our ophthalmology clinic and underwent complete ophthalmological evaluation. At the time of the visit, she did not complain of any visual symptoms. Natural visual acuity was 20/20, Amsler grid negative, Intraocular pressure (IOP) 19 mmHg (Goldmann applanation tonometry). The anterior segment was normal. Dilated fundoscopy revealed an augmented cupping of the optic disc and a bilateral macular lesion similar to bull's eye macula. Optical coherence tomography (OCT) was performed (Heidelberg Spectralis), revealing foveal and parafoveal subretinal fluid in both eyes (Figure 1). No other pockets of fluid were found along the arcades. Both visual field 30-2 (Zeiss, Humphrey Field Analizer 3) and Retinal Nerve Fiber Layer (RNFL) analysis at OCT were normal and glaucoma was ruled out. At the 1-week reevaluation, the patient was asymptomatic, fundus examination was normal and there were no signs of SRF at OCT. However, seven weeks later, due to the recurrency of intermittent visual symptoms, the patient sought again medical attention. The patient complained of waxing and waning bilateral visual symptoms, experienced at the time of the evaluation as well, lasting for four days. While visual acuity and anterior segment examination were normal, fundoscopy showed a bilateral mild almond-shaped retinal depigmented area surrounding the macula, detected at Infrared-OCT (Figure 2). Autofluorescence was normal. Interestingly, after 3 more weeks, the patient still complained of occasional bilateral visual symptoms (this time not present at the follow-up visit), and both fundoscopy and infrared-OCT revealed the same lesion of the previous time. After one more month, symptoms subsided permanently, while the patient was still on medical treatment at the same dose regimen.
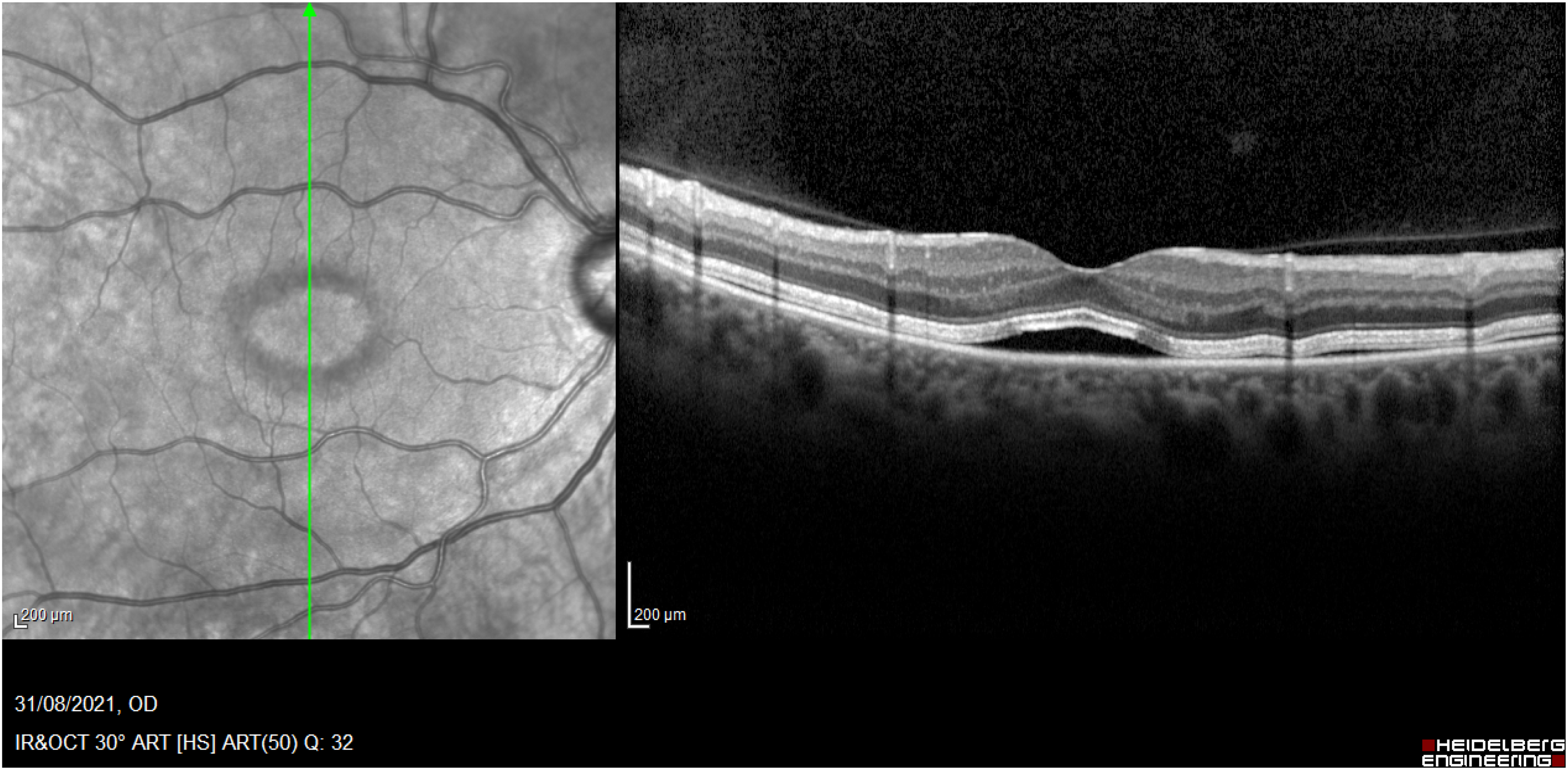
IR-OCT scan performed at symptoms onset. A well-demarcated round-shaped hyporeflective area can be seen at the infrared. SRF is present in the foveal and parafoveal region.
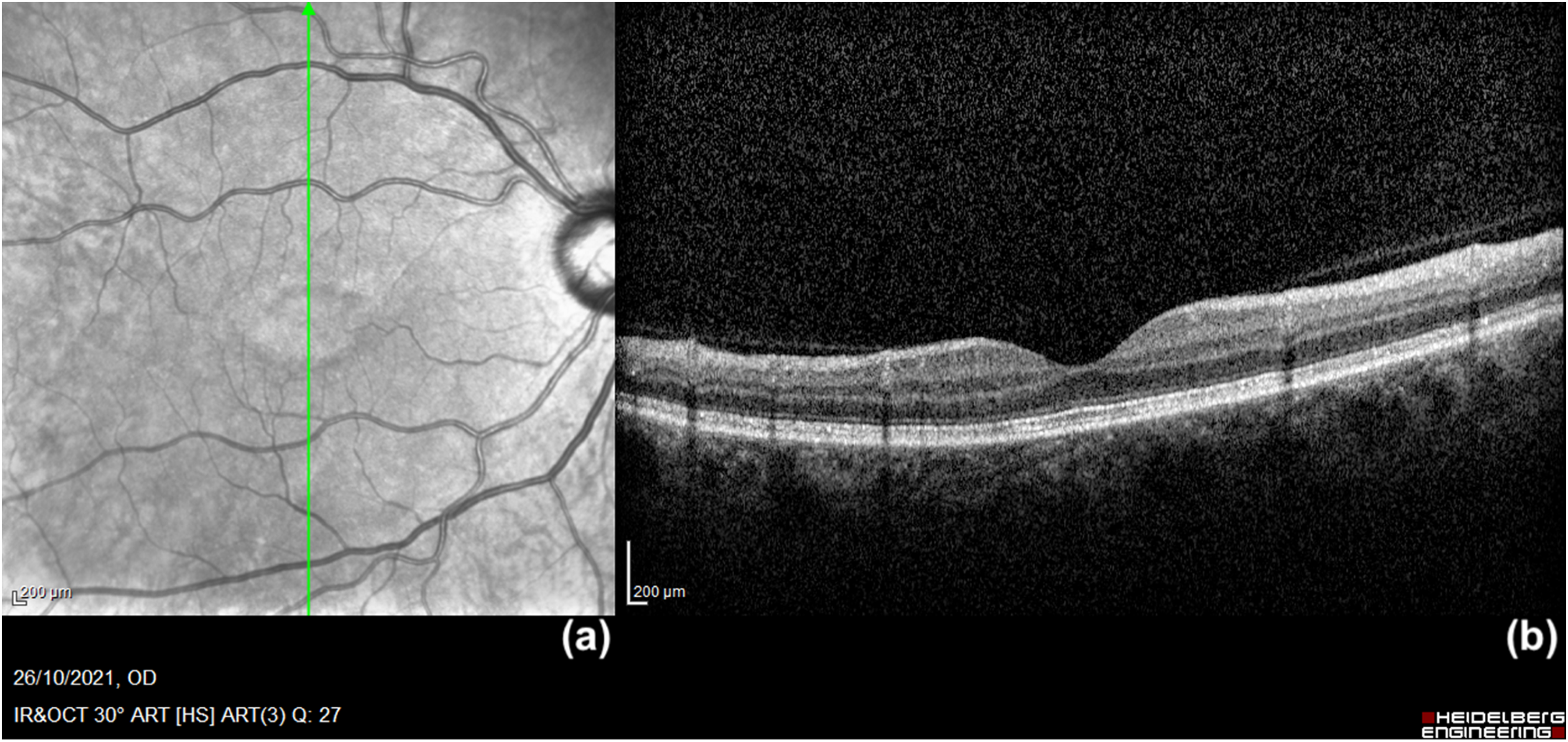
IR-OCT scan performed seven weeks after fluid reabsorption, when symptoms were reported to be experienced again. (a) IR shows an almond-shaped hyporeflective lesion in the macular region surrounded by hyperreflectivity. (b) The OCT does not reveal any alteration of the retinal layers nor fluid pockets.
Discussion
In the largest trial carried out so far, Weber et al. reported a 90% prevalence of SRF in MEK-inhibitor-treated patients, with no permanent ocular sequelae nor need for drug discontinuation. 6 Remarkably, all 9 patients (20%) with visual symptoms had a confirmed diagnosis of subfoveal SRF.
Moreover, Chancellor et al reported a case report of a patient with persistent SRF even after drug discontinuation. 7 Though ocular disturbances secondary to BRAF inhibitors have been also reported, their occurrence is uncommon when Encorafenib is administered as single agent. Moreover, according to the European Medicines Agency, this side effect usually consists in uveitis, 8 while SRF accumulation has been strictly related to concomitant MEK-inhibitor administration. 9 In our case, visual symptoms were experienced by the patient at different times, both with and without evidence of SRF (i.e., at day 2 and at week 8). This finding seems to suggest that while Binimetinib-induced SRF is an asymptomatic finding in most cases, with excellent outcome and rapid resolution, visual symptoms could be initially triggered by detectable SRF, yet persist without any further evidence of abnormal fluid accumulation and manifest intermittently. According to the reported data in the literature, in which the transitoriness of visual symptoms and SRF has been well described, therapy was continued at the same dose and strict monitoring scheduled.4–6 In our patient, symptoms’ onset was early, but transient. The resolution of SRF was rapid, yet after 7 weeks symptoms reappeared. Considering that visual acuity was unchanged and that only mild signs of retinal involvement were present, Encorafenib-Binimetinib was continued, in accordance with the European Medicines Agency's recommendations to withhold the drug only in case of symptomatic retinal pigment epithelial detachment. 9 The patient is currently asymptomatic. The novelty of this report lies in the finding that MEK-inhibitor related visual symptoms, after being originally triggered by SRF detectable through examination, can later persist without any additional evidence of abnormal fluid accumulation, and occur intermittently. Although this discovery did not change the clinical management of the patient, its peculiarity should not be overlooked either and clinicians should be aware of this possible presentation. Our case consolidates the relative safety of the combined targeted therapy and suggests the importance of strict follow-up, that could avoid unnecessary therapy discontinuation, in those patients that happen to experience intermittent visual symptoms.
Patient consent
The patient in this manuscript has given written informed consent to publication of her case details
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
