Abstract
Purpose
Bietti crystalline dystrophy (BCD) is a rare autosomal recessive retinal dystrophy caused by pathogenic variants of CYP4V2 gene and characterized by shiny yellow deposits in the retina and progressive atrophy of the retinal pigment epithelium (RPE) and choriocapillaris. The main aim of the present study is to describe the optical coherence tomography angiography (OCTA) characteristics of a patient affected by BCD.
Methods
A 59-years-old female with genetically confirmed BCD underwent an ophthalmological examination complete of OCTA performed in the atrophic retina, the junctional zone and the apparently normal retina. The area of choriocapillaris (CC) atrophy was compared to the area of RPE atrophy on fundus autofluorescence (FAF) imaging.
Results
A severe vessel density (VD) deficit at the level of superficial and deep capillary plexa as well as CC was registered in atrophic areas, which resulted deeper with respect to the junctional area, whereas the apparently preserved retina revealed VD values similar to that of control eyes. The area of RPE atrophy on FAF was larger (55.90 mm2 in right eye and 48.76 mm2 in left eye) than the area of CC atrophy on OCTA imaging (51.86 mm2 and 42.44 mm2 respectively in right and left eye).
Conclusions
Our findings suggest that VD impairment of retinal plexa and CC follows the degeneration of RPE as demonstrated by the greater size of the area of RPE atrophy compared to CC atrophy. Further investigations based on OCTA imaging are necessary to enhance our knowledge of this rare disease.
Introduction
Bietti crystalline dystrophy (BCD) is a rare autosomal recessive inherited retinal dystrophy (IRD), caused by pathogenic variants in CYP4V2 gene, 1 playing a role in fatty acid and steroid metabolism, 2 BCD is typically characterized by shiny yellow deposits in the retina and sometimes in the cornea, with progressive atrophy of the retinal pigment epithelium (RPE) and sclerosis of the choroidal vessels.3,4 Clinically, the affected patients experience decreased vision, night blindness, and constriction of the visual field, usually occurring around the third and fourth decade of life.
Optical coherence tomography angiography (OCTA) can provide useful insights regarding the retinal and choroidal vascular network. 5 A reduced choriocapillaris (CC) blood flow has been reported in patients affected by BCD. 6
The aim of the present case report is to quantitatively describe the OCTA findings obtained from a BCD patient, analysing both affected and not affected retina (Figure 1).
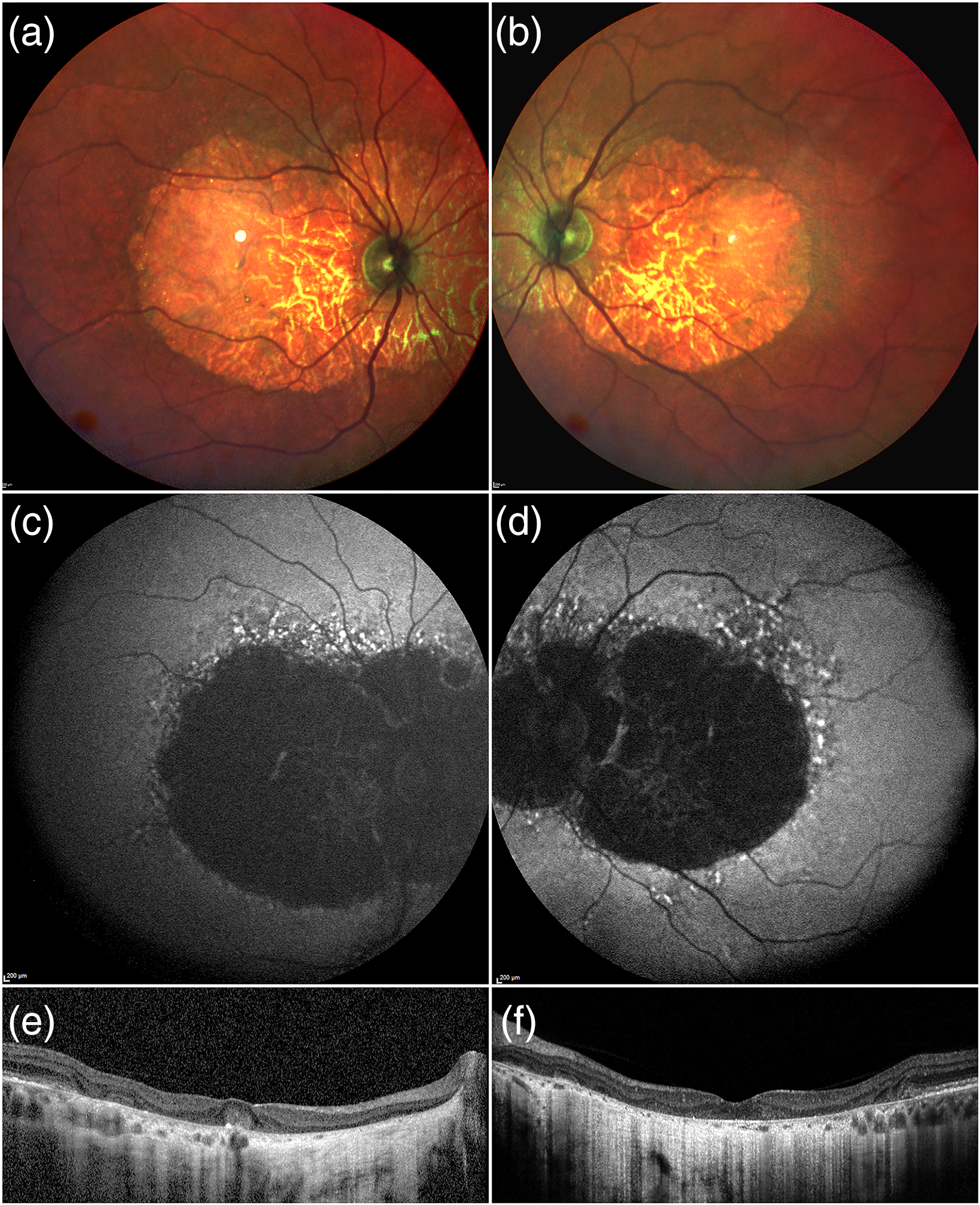
Multimodal imaging of BCD. Multicolor images show the presence of retinal atrophy involving almost the entire posterior pole (a,b). This is confirmed by the hypoautofluorescence signal detected by FAF imaging (c,d). Structural OCT shows extensive outer retinal atrophy with choroidal hypertransmission and other alterations including hyperreflective foci and outer retinal tubulation (e,f).
Case report
A 59-years-old Caucasian woman was referred to the IRD Units at the Department of Ophthalmology of Scientific Institute San Raffaele in Milan for a progressive visual impairment in both eyes starting from the third decade of life. She had a negative family history of ocular disorders. Best corrected visual acuity on ETDRS charts was 0.6 LogMAR and 0.4 LogMAR in right and in the left eye, respectively. The anterior segment was normal in both eyes, with no crystal deposits at the corneal limbus. Intraocular pressure was 16 mmHg in both eyes. Fundus biomicroscopy disclosed a bilateral extensive chorioretinal atrophy associated with multiple crystalline deposits, manly located at the margins of the area of chorioretinal atrophy (Figure 2).
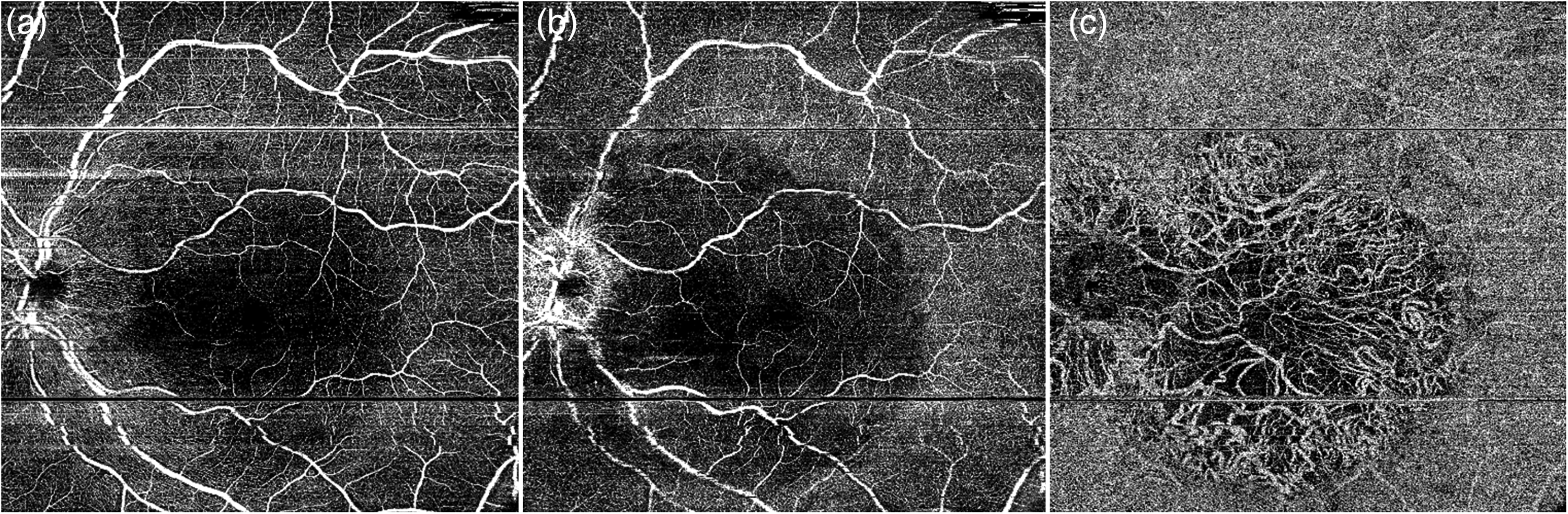
OCTA analysis of BCD. SCP (a) is apparently normal, whereas DCP (b) is characterized by markedly reduced signal at the posterior pole. CC results completely absent in the atrophic retinal area (c) with exposure of choroidal vessels.
Scotopic and photopic full filed electroretinograms resulted significantly reduced, whereas electrooculogram was within normal range. Next Generation Sequencing, performed considering a IRD panel of 44 genes, including TruSight One Enrichment Oligos and Rapid Capture kit-Illumina (NextSeq Illumina platform), with Sanger sequencing confirmation of pathogenic variants (sensitivity >97%; specificity >99%), unveiled two pathogenic variants in CYP4V2 (c.424_427 del (p.(Lys142Glyfs*8)) and c.173C>A (p.(Pro58Gln)), never described in the literature.
Multimodal imaging included fundus photographs (TRC-50DX, Topcon Corporation; Tokyo, Japan), blue-light fundus autofluorescence (FAF), spectral-domain optical coherence tomography (SD-OCT Spectralis HRA + OCT, Heidelberg Engineering; Heidelberg, Germany), and OCTA examination (DRI-Triton OCT, Topcon Corporation, Japan).
FAF precisely outlined the extension of the RPE atrophy and showed as a well-marked region of hypoautofluorescence. SD-OCT revealed the impairment of the outer retinal bands, consisting in reflectivity attenuations,disruption or evident loss of the ellipsoid zone, together with outer retinal tubulations, retinal hyperreflective dots, bright plaque on the top of RPE-Bruch's membrane, and choroidal hyperreflective spots. Central macular thickness (CMT) was 166µm in the right eye and 163µm in the left eye, whereas subfoveal choroidal thickness was 111µm in the right eye and 68µm in the left eye.
On OCTA, vessel density (VD) of superficial capillary plexus (SCP), deep capillary plexus (DCP) and CC were quantified at the level of the apparently preserved retina, the atrophic areas and the junctional zone by means of a previously validated method.7–10 The images were loaded in the Fiji software package and binarized using a “mean” threshold.11,12 VD was calculated as the ratio between the white and black pixel in the areas selected where calculated using in-house scripts. Foveal avascular zone was manually segmented and excluded from VD calculation. The separated calculation performed on preserved retina, junctional zone and atrophic retina was based on the segmentation performed by an expert operation of the three regions on the FAF images. Then, the segmented regions of interest were placed in the OCTA images, using big retinal vessels as reference for the registration of the images.
In detail, no reduction in VD values were detected in the apparently preserved retina with respect to eyes of a control group of 10 patients of the same age (all women; mean age 58 ± 3 years). On the other hand, a slight reduction in VD was registered in the junctional zone, whereas a severe VD alteration was found in the atrophic areas (VD of SCP, DCP and CC were respectively 0.11, 0.12 and 0.34 in the right eye and 0.09, 0.115, 0.35 in left eye) (Table 1).
Vessel density analysis in a patient with BCD and healthy controls.

Mean values are reported. BCD: Bietti crystalline dystrophy, CC: choriocapillaris, DCP: deep capillary plexus SCP: superficial capillary plexus.
The patient was further quantitatively analysed by measuring the area of CC atrophy and comparing it to the area of RPE atrophy on FAF imaging. Measurements of RPE atrophy on FAF were performed using the built-in measurement tool in the Spectralis HRA software, whereas CC atrophy on OCTA imaging was measured using ImageJ software. RPE atrophy on FAF was 55.90 mm2 and 48.76 mm2 respectively in right and left eye, whereas the size of CC atrophy was 51.86 mm2 in right eye and 42.44 mm2 in left eye.
Discussion
Variable degrees of severity of RPE and CC degeneration can occur in BCD,3 and it still is uncertain whether the RPE degeneration precedes the CC impairment. Expression of vascular endothelial growth factor in the RPE is essential for the CC trophism development.13,14 Previous studies strongly suggested a correlation among CYP4V2 pathogenic variant, fatty acid metabolic disorder, and RPE degeneration in patients with BCD. Zhang and colleagues reported that the poly-unsaturated fatty acid overload could provoke mitochondrial damage and RPE degeneration in BCD patient. 15 RPE and CC share a mutualistic relationship, so that if one of the components is compromised, both become dysfunctional and degenerate.
Few studies investigated the OCTA findings in patients with BCD.6,16–18 Miyata et al. identified a CC flow deficit in 92% of eyes using OCTA, demonstrating that the presence of residual subfoveal CC was significantly associated with visual function. 6 Demir and colleagues confirmed the flow deficit in CC, highlighting also a significant deficit in VD of DCP and SCP in atrophic retina in BCD. 19
We performed the OCTA analyses based on high-quality OCTA imaging aiming at the characterization of vascular status in different areas of retina. Our results indicated the presence of a marked VD reduction in SCP, DCP and CC of the atrophic areas, which was deeper with respect to the junctional zone, whereas the apparently preserved retina on color photography, autofluorescence and OCT scans had VD values similar to that of control eyes.
The area of RPE atrophy was larger with respect to that of CC damage, with a mean difference of 5 mm2 between RPE and CC degeneration. This evidence suggests that RPE atrophy occurs first, then followed by CC atrophy, as previously described in ABCA4-related conditions. 20
The study suffers from several limitations mainly related with the “case report design” of the investigation. Hence, no statistical analysis was possible. Furthermore, although the accurateness of multimodal imaging acquisitions, our findings are potentially prone to artefacts. In addition, ours was just a cross-sectional investigation of the alterations characterizing BCD. For all these reasons, further prospective studies including more BCD patients are warranted to draw definite conclusions about this rare condition.
In conclusion, our findings suggest that VD impairment of retinal plexuses and CC follows the primary degeneration of RPE. Further findings are necessary to better understand the steps of the degenerative process and define whether VD on OCTA can be considered a biomarker of the stage and severity of BCD.
Footnotes
Authorship
All authors attest that they meet the current ICMJE criteria for Authorship.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article. Francesco Bandello consultant for: Alcon (Fort Worth, Texas, USA), Alimera Sciences (Alpharetta, Georgia, USA), Allergan Inc (Irvine, California, USA), Farmila-Thea (Clermont-Ferrand, France), Bayer Shering-Pharma (Berlin, Germany), Bausch And Lomb (Rochester, New York, USA), Genentech (San Francisco, California, USA), Hoffmann-La-Roche (Basel, Switzerland), NovagaliPharma (Évry, France), Novartis (Basel, Switzerland), Sanofi-Aventis (Paris, France), Thrombogenics (Heverlee, Belgium), Zeiss (Dublin, USA). All other authors have no disclosures to declare.
Patient consent
Consent to publish this brief report has been obtained from the patients in writing.
