Abstract
Purpose
To assess the prevalence of Cystoid macular edema (CME) in children with early onset retinal dystrophies (EORD) and to evaluate if there are associated factors and/or response to early treatment.
Methods
Consecutive, retrospective case series. Medical records of patients, 18 years or younger, diagnosed with EORD were included in the study. Optic coherence tomography (OCT) scans, clinical and genetic characteristics as well as other associated factors were analyzed. Main outcome was the presence of CME on OCT scans
Results
One hundred and two children with EORD (aged 1–18 years, mean 9.7 ± 4.2) were recruited. OCT was performed in 60/102 and among them, 19/60 had CME (31.7%). The disease-causing gene was identified in 13 children with CME; autosomal-recessive inheritance was found in 88.3% of those with an identified genotype. Children with Usher syndrome had CME in 44.4% of the cases. Early treatment of CME resulted in variable response.
Conclusions
Our results show that 31.7% of children with EORD who underwent OCT have macular edema. CME prevalence was found to be relatively higher in children with Usher syndrome. Autosomal recessive was the most prevalent inheritance identified in the EORD group as well as in the CME group. Additional prospective research is needed to assess the efficacy of early CME treatment in pediatric EORD patients.
At a glance
Cystoid macular edema (CME) is considered a common feature in patients with retinal dystrophies. Our results show that as many as one-third of children early onset retinal dystrophies (EORD) such as RP and Leber Congenital Amaurosis (LCA), who underwent OCT, may manifest retinal cystic changes.
Introduction
Hereditary retinal diseases are known as the leading cause of blindness in the working age population (age 16–64 years) in England and Wales, of which retinitis pigmentosa (RP) is the most common disorder. 1 In the pediatric population, inherited eye diseases (mainly inherited retinal dystrophies) are also being described during the last decade as the most common causes for childhood blindness in developed countries.2,3
Cystoid macular edema (CME) is considered a common feature in patients with retinal dystrophies. Usually, central vision may be preserved until late stages in the disease. However, the development of CME may exacerbate central vision loss, affecting its earlier course. 4 The prevalence of CME among retinal dystrophies patients varies throughout different studies from 8% to 58%, probably due to different factors including variable definitions of CME based on Optic coherence tomography (OCT), exclusion criteria or type of OCT machine used (time domain vs. spectral domain). 5 The greater the width of the macular area affected by CME, the worse the vision will be. 6
CME in the pediatric population with EORD such as RP and LCA has been insufficiently addressed in the literature. The aim of our study was to assess the prevalence of CME (detected by OCT) in children with EORD and to evaluate associated factors as well as response to early treatment.
Methods
This is an observational, retrospective, cross-sectional study describing the prevalence of CME among children with early onset inherited retinal dystrophies with a nested retrospective cohort analysis of the response to treatment in those patients with CME. Data were collected from medical records of children up to age 18 years old who were examined at the department of Ophthalmology, Hadassah Medical Center, Jerusalem, Israel, between January 2013 and January 2021 with a diagnosis of EORD. The clinical diagnosis of EORD was based on different clinical characteristics of retinal dystrophy: history of poor visual acuity in early childhood, nystagmus, nyctalopia, high refractive errors, and/or atrophic retinal findings on clinical examination. Confirmation of diagnosis was performed by full field electroretinogram (ffERG) in all cases, and genetic testing in some of them. Methods used for genetic testing included whole exome sequencing (WES), panel for inherited retinal diseases and Sanger sequencing of already known mutations. Only pathogenic and likely pathogenic variants were considered as a positive genetic result. Variants of unknown significance (VUS) were not recorded as causing variants.
CME diagnosis was based on spectral domain (SD)-OCT scans and was defined as more than one hyporeflective cystic macular space seen in horizontal cross-sections of the macular area as determined by retina specialist. SD-OCT was performed with Heidelberg Spectralis, Heidelberg, Germany and analyzed with Heidelberg workstation to evaluate cross-sectional images. The location of the foveola in the OCT scan was centered by the examiner.
Due to the retrospective nature of the research, age at examination was determined to be the age at the first exam or OCT scan. Additional clinical data were obtained from medical records including best-corrected visual acuity (BCVA) that was assessed for distance using a computerized monitor (linear Lea symbols for cooperative young children and Snellen optotypes for older children), inheritance pattern and genotype when available, and treatment for CME. The study was approved by the Institutional Review Board at Hadassah-Hebrew University Medical Center.
Results
A total of 102 children (57 males and 45 females) with diagnoses of EORD (RP or LCA) were reviewed. Age ranged from 1–18 years, mean 9.7 ± 4.2. Only in one of the 102 children studied, there was a recorded “clinical suspicion of CME” during the different follow up visits in their medical records. All children aged 3 years and above were referred for OCT imaging, but the scans were not performed in all of them for different reasons: lack of child cooperation or high-speed nystagmus precluding quality scans.
Sixty patients (27 females and 33 males) had at least one OCT scan. Their age ranged from 3 to 18 years old (mean 9.47 ± 4.3). Nineteen of these 60 children were identified to have CME (31.7%) as seen by SD-OCT; 17/19 (89.4%) had bilateral CME and 2 patients had unilateral CME (patients number 11 and 16). Age range was similar in the CME group (range 3–18; mean 9.8 (±4.3), and in the group without CME (mean age 9.5 (±4.2)). Regarding gender, CME was present in 40.6% of males and only 24% of females (Fisher test: 0.260; not statistically significant) (Figure 1,Table 1).
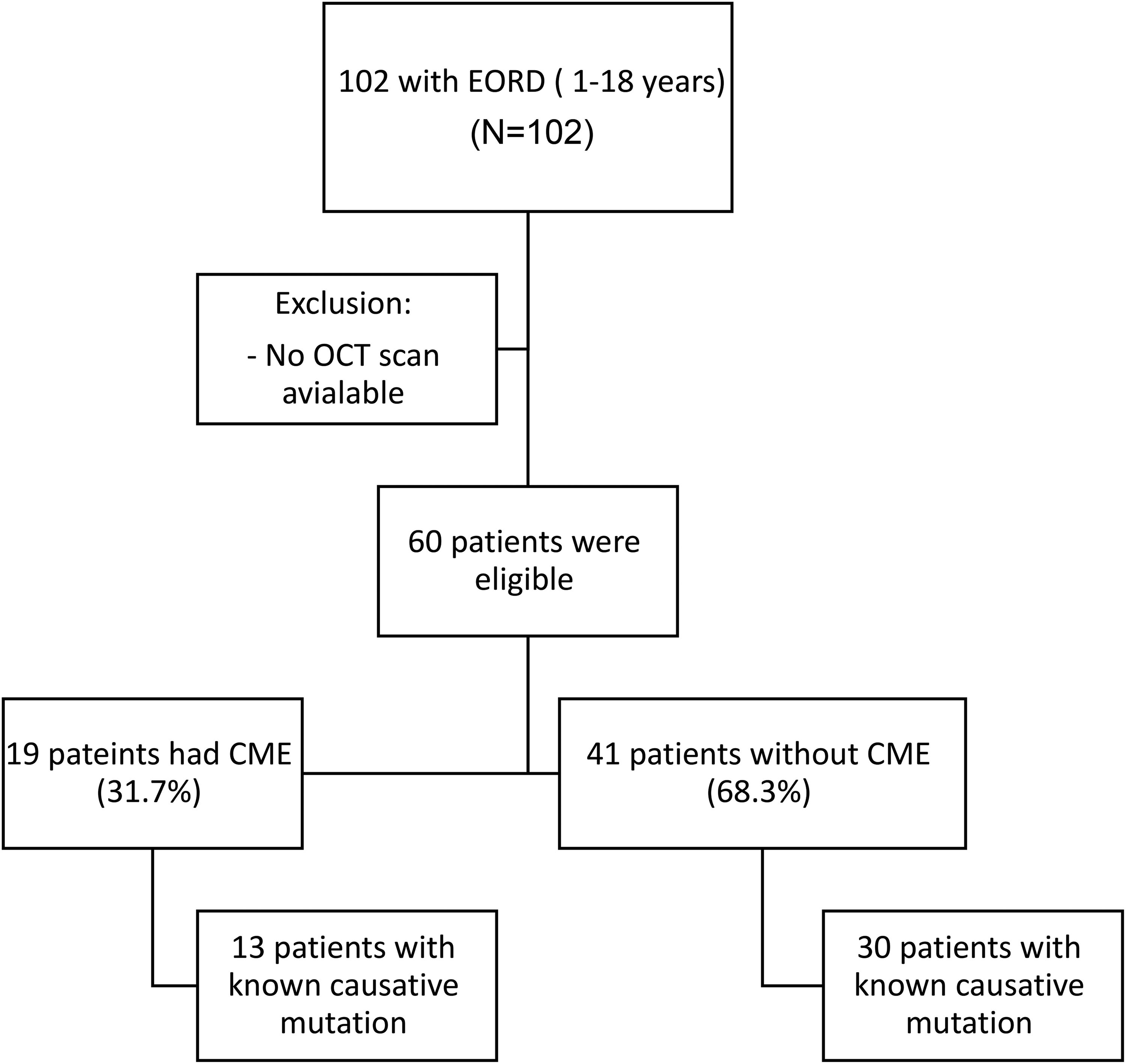
Flowchart with study design details.
Characteristics of Children with EORD and CME.
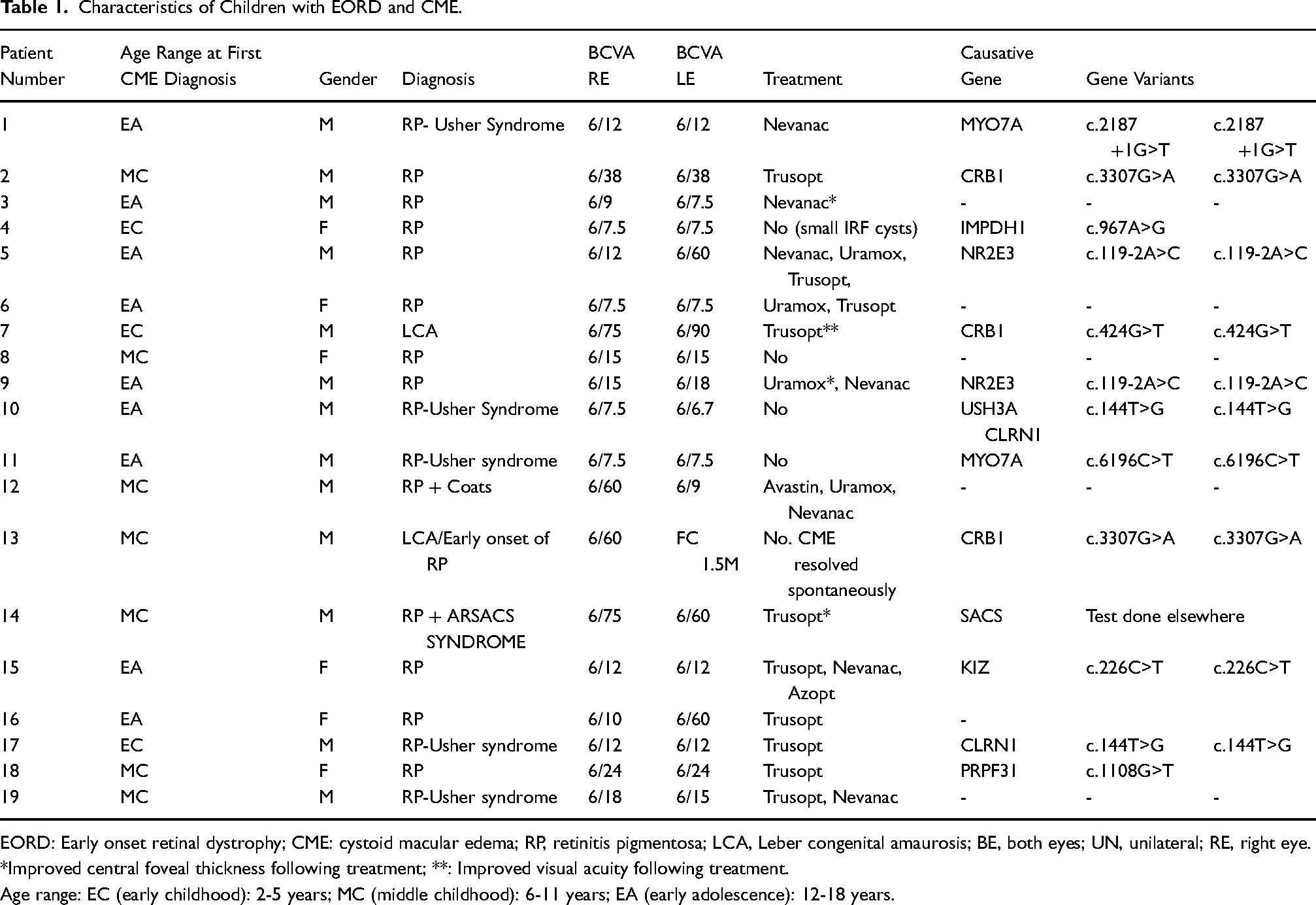
EORD: Early onset retinal dystrophy; CME: cystoid macular edema; RP, retinitis pigmentosa; LCA, Leber congenital amaurosis; BE, both eyes; UN, unilateral; RE, right eye.
*Improved central foveal thickness following treatment; **: Improved visual acuity following treatment.
Age range: EC (early childhood): 2-5 years; MC (middle childhood): 6-11 years; EA (early adolescence): 12-18 years.
Of note, patient number 17 (a five-year-old child) referred for ophthalmological examination due to suspected poor visual performance according to parents with a BCVA of 6/12 Snellen in each eye. Eye exam was within normal limits; hence the child was referred for a SD-OCT test to ascertain normal retinal structure. CME was then seen raising suspicion of an inherited retinal dystrophy that was later confirmed by electrophysiology of the retina. Later on, Usher syndrome was diagnosed by genetic testing. Hearing was found to be normal.
OCT
Mean central foveal thickness (CFT) was 268.52 μm (± 149.58) in the no-CME group and 409.73 μm (± 160.53) in the CME group.
Genotype and inheritance
The causative mutation was identified in 42 out of 60 patients with an available SD-OCT scan. Based on genetic testing, 38/43 harbored autosomal recessive (AR) mutations (88%), two patients had X-linked mutations (4.7%) and another three patients had autosomal dominant (AD) mutations (7.1%). Of note, a similar pattern of inheritance was found within the CME group. Among 19 patients, the causing mutations were identified in 13 patients while 11 out of 13 patients (84.6%) harbored AR mutations, two patients had AD mutations. Of note, three patients harbored bi-allelic CRB1 mutations.
Notably, a subgroup of 11 children with Usher syndrome demonstrated CME in 5 patients, while 2/5 had Usher type 1 and 2/5 had Usher type 3
Causing gene and inheritance among studied cohort
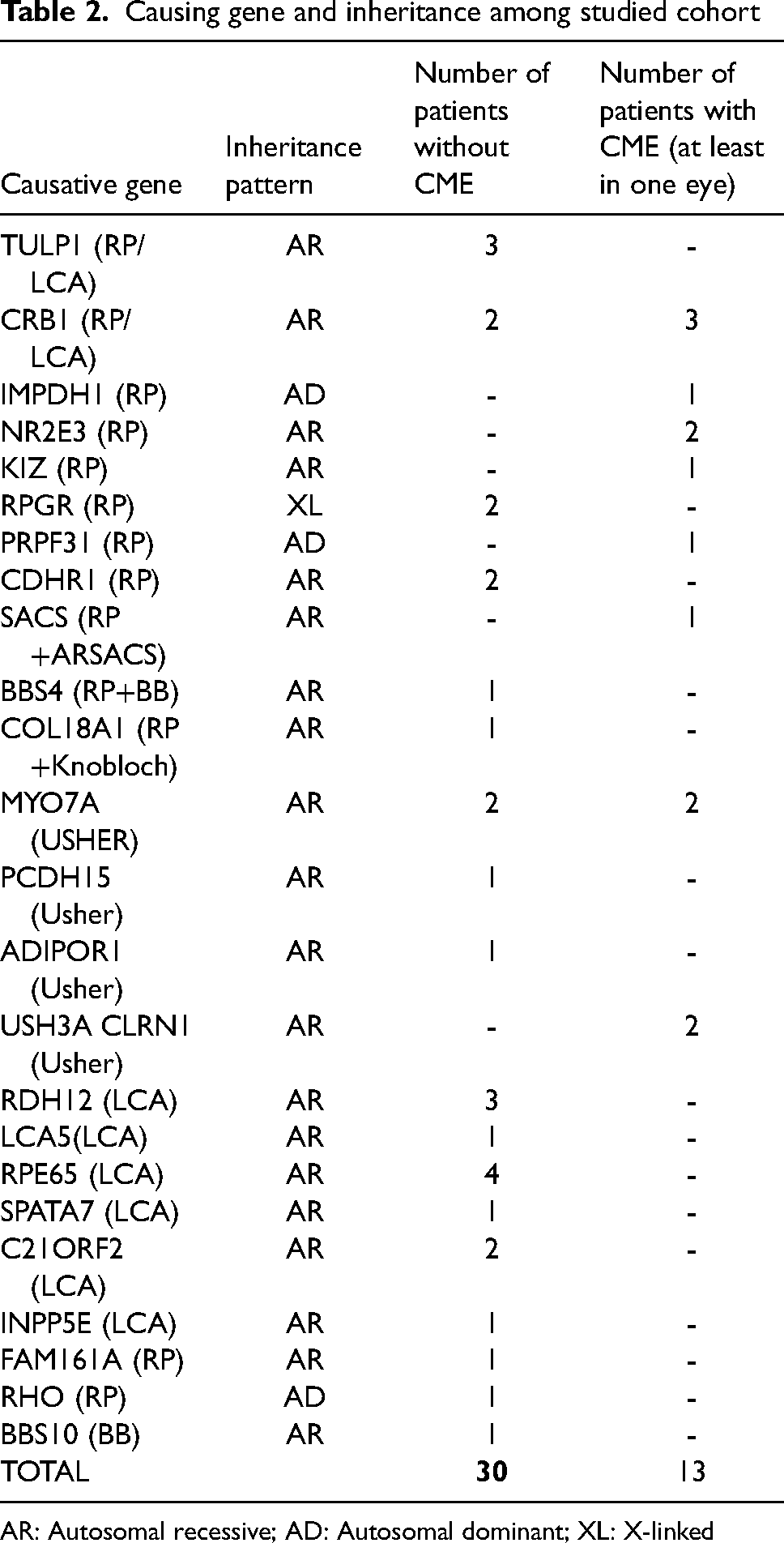
AR: Autosomal recessive; AD: Autosomal dominant; XL: X-linked
Treatment
Most children received topical treatment: nepafenac (Nevanac) in 7 children, dorzolamide (Trusopt) in 10 children and brinzolamide (Azopt) drops in 1 child; one child (#12) received intravitreal Avastin injections as an off-label supplementary therapy for macular edema secondary to concurrent Coats disease previously treated with ablation therapy. Four patients underwent a trial of oral acetazolamide treatment (125 mg three times a day) which was halted due to systemic side effects. Mean CFT in the CME group before treatments was 409.73 μm (± 160.53) and after treatment was 360.27 μm (± 174.78). Only 4 out of 13 treated patients showed some sort of improvement: patient number 7 showed visual acuity improvement 3 months following topical dorzolamide as demonstrated by one line gain for distance (from 6/75 to 6/60 in the right eye and 6/90 to 6/60 in the left eye) and near visual acuity: C3 at 10 cm to C2 at 15 cm on the right eye and unchanged (C3) on the left eye. Three children experienced relatively reduced CFT with stable vision treated by variable agents (dorzolamide in one and nepafenac in 2 subjects) and treatment periods (Figure 2).
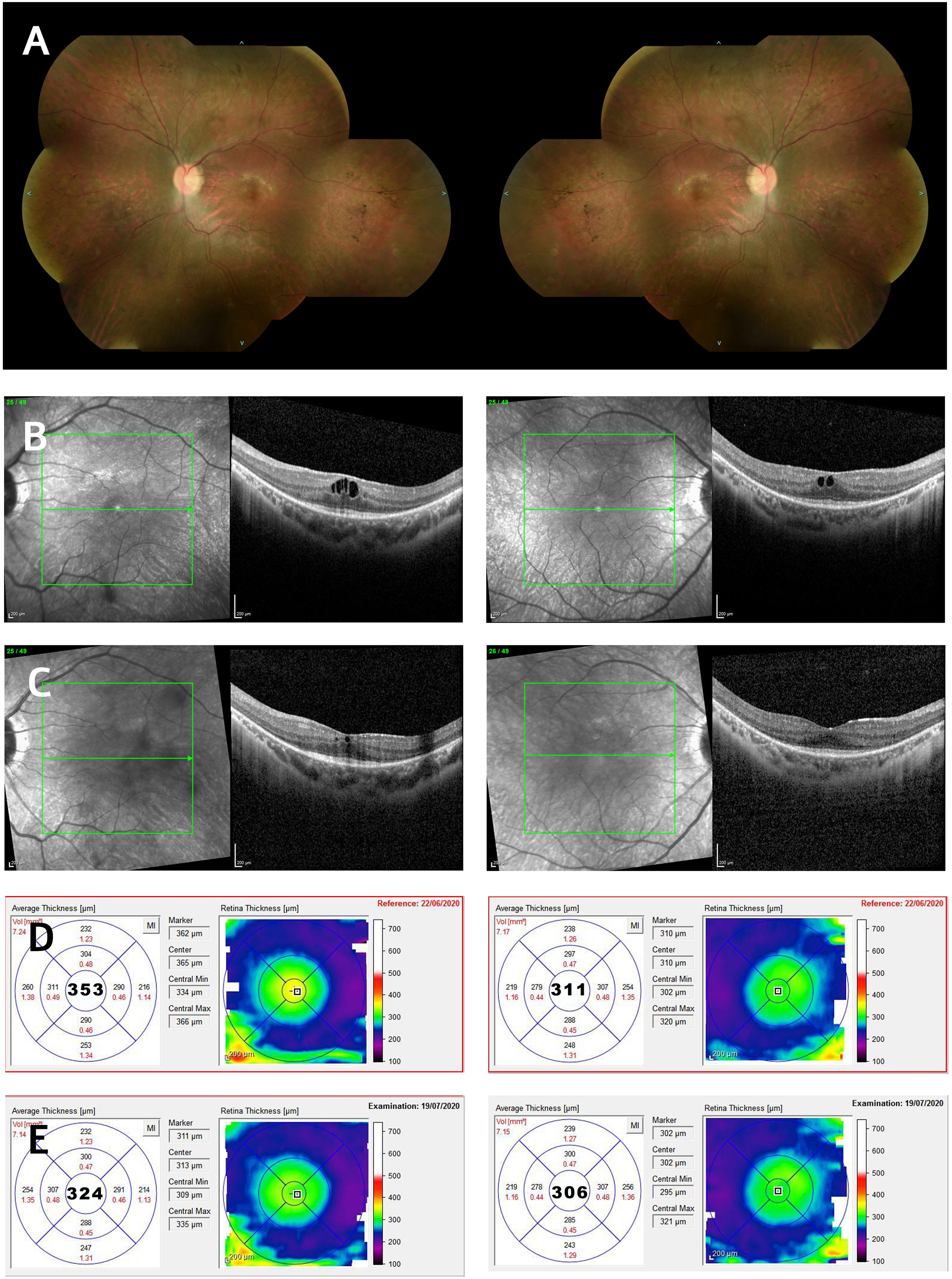
Patient 3 retinal imaging. (A) Color fundus picture of both eyes. (B) Spectral-domain optical coherence tomography showing CME before treatment. (C) OCT demonstrating reduced CME following one month of topical treatment. Average foveal thickness pre- (D) and post-treatment (E).
Discussion
The burden of visual impairment from inherited retinal dystrophies seems to be increasing both in young adults and children and may be partly due to the lack of effective treatments. CME is a relatively common associated feature in these conditions and vision outcome may be potentially improved when these complications are identified and treated early, especially in young ages when children are still in the process of visual development. 7 We hereby report a hospital-based series of 102 children with EORD in whom, only one case of CME could be identified clinically, but by performing routine OCT scans, 31.7% of 60 children showed CME.
Pediatric retina examinations usually reveal limited information due to reduced cooperation, as well as the fact that these children with EORD usually develop nystagmus that precludes the examiner from obtaining fine retina details.
The prevalence of CME in children with retinal dystrophies has not been sufficiently studied in the literature. Liew et al., showed that CME was associated with younger age (OR 0.81, 95% CI 0.67 to 0.98). 8 The mean age of patients in this study was 47.1 years (±18.4); no data were included specifically about children under the age of 18. By contrast, Hajali and colleagues included 10 patients younger than 18 years old, reporting a total age range from 8 to 71, and showed that the presence of CME did not predominate within any age-group category. 9 Kim et al., described early-onset CME in a family with RP due to the p.P347L mutation in the rhodopsin gene (RHO), reporting 4 teen-aged patients out of 5 family members with bilateral CME, with the youngest being only 11 years of age. They reported that this child was the youngest RP patient with CME discussed in the literature by then. 10
We believe that obtaining good quality OCTs in young children is challenging due to the high incidence of nystagmus and poor cooperation, and probably explains the lack of data in the literature regarding the prevalence of CME in children diagnosed with EORD. Our study includes patients presenting with CME as early as 3 and 5 years old. To the best of our knowledge, this is the first report in the literature analyzing the prevalence of CME in a pediatric cohort with EORD as well as the first report of CME in a patient as young as 3 years old
Testa et al., have assessed macular abnormalities in patients with Usher syndrome (type 1 and 2) and showed that CME was significantly associated with young age (patients with CME: 30.1 ± 11.1 years; patients without CME: 36.9 ± 14.9 years; β = − 0.038; p = 0.016). 11 However, no further data about pediatric population was mentioned. 11 The prevalence of CME in Usher type 1 and 2 patients ranged between 15.7% and 25%.11,12 Subira et al, recently published a pediatric case series of Usher syndrome type 1 due to a MYO7A mutation, in which 25% of the studied children had CME. 13 In our study, five children out of eleven with a diagnosis of Usher syndrome had CME, leading to a relatively higher prevalence of CME as compared to other children without Usher in our study. The main types of Usher linked to CME in our study were type 1 and 3. Interestingly, Usher type 3, usually an adult-onset disorder, has not been previously reported in the literature in relation with a young child with CME. One possible explanation for the higher incidence of CME in children with Usher syndrome in our study might be the inclusion of early diagnosed children with Usher type 3 in our case series.
Regarding the inheritance pattern, previous studies showed a strong association between AD-inheritance RP and CME.3,8,12–14 Liew et al., made the assumption that relatively healthy retinal tissue is required to cause CME, which would explain why younger patients with milder forms of RP (such as AD disease) are more likely to have CME than more severe forms (X-linked RP). 8 In the current study, most of the patients included in our cohort harbored AR inheritance pattern (88%). Similarly, in the CME group 10 out of 12 patients presented with AR disease. The high prevalence of AR cases in our cohort can be explained based on the high incidence of AR inheritance among patients with inherited retinal dystrophies in our population. 15 and also due to the late disease onset in patients with an AD inheritance, mostly reaching a diagnosis beyond childhood. 16
Of note, 3 out of 12 children had biallelic genetic mutation in the CRB1 gene,2/3 with the c.3307G>A variant and one child with the c.424G>T variant. Different mutations in this gene have been reported in patients with a variety of phenotypes of retinal dystrophies, including LCA and RP. 17 A recently published analysis of 74 patients from 63 families, assessed characteristic features associated with biallelic variants in CRB1 18 with 17.9% presenting CME. Another published case-report describes a patient with CRB1 mutations with CME associated with the c.4014T>A variant. 17 Further evaluation is required to assess if CRB1 gene mutation predominates in a larger CME pediatric cohort group.
Regarding management of CME in children, of the 19 children who received treatment for CME, 4 showed some relative improvement. The retrospective nature of the study, the small number of patients, and the diversity of treatments used, limits the ability to accurately evaluate the response to treatment or the potential additional "amblyogenic effect" of CME at young ages. The evidence currently available suggests that topical carbonic anhydrase inhibitors (CAIs) may be used as a first-line approach and oral CAIs may be a second-line agent, but with the risk of more side effects. 7
Limitations of our study include its retrospective nature, the small sample size, and the fact that prevalence of CME was calculated in the sub-group who performed OCT scans limiting the ability to ascertain the exact prevalence of CME in our cohort. Of note, all patients aged three years old and older were systematically referred for OCT, but many of them were not able to complete the scans for variable reasons leading to bias due to the possible underrepresented number of CME cases.
In conclusion, our results show that as many as one-third of children with EORD may manifest cystic changes on OCT, comparable to the reported prevalence of CME in adults. In our cohort, the incidence of CME seemed to be higher in children with Usher syndrome. Since clinical assessment of retinal cystic changes is challenging in children, in general, and in patients with low vision and nystagmus in particular, screening of these children using OCT is recommended to exclude the presence of CME. Early treatment of CME may potentially lead to better visual outcome as well as additional amblyopia prevention. Prospective studies with bigger samples are needed to assess recommended treatment and its efficacy on CME in children with EORD.
Footnotes
Abbreviations and acronyms
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article
