Abstract
Purpose
Keratoconus (KCN), classically defined as a noninflammatory corneal ectasia, was recently associated with chronic inflammation. This study aimed to further evaluate the association between inflammation and KCN severity by assessing patient tear films.
Methods
Retrospective chart review of consecutive patients (July 2016–February 2020) referred to a tertiary KCN centre. Using tomography Kmax values, the worst-eyes of patients with a diagnosis of KCN and tear film test results were included. Eyes were stratified as matrix metalloproteinase-9 (MMP9) positive or negative and analyzed using independent t and Pearson chi-squared tests. A p-value ≤ 0.05 was considered significant.
Results
551 (51.40%) unilateral eyes were included. Mean age was 30.1 years and 70.1% were male. 55.5% of the eyes were MMP9 positive. Mean corrected distance visual acuity was 20/30<συπ>−3</sup> Snellen and Kmax was 60.1 Diopters. MMP9 positive eyes had a higher Kmax (p = 0.048), and were more likely from patients who were male (p < 0.001), had a paediatric history of asthma (p = 0.042), and used glasses (p = 0.041). MMP9 negative eyes more likely corresponded to soft contact lens users (p = 0.012). No other significant differences were found in risk factors, topography, tomography, and tear film osmolarity.
Conclusion
MMP9 positive keratoconic eyes had significantly higher Kmax readings which may correlate with increased disease severity, supporting an association between keratoconus and inflammation. Further research is warranted to evaluate the role of targeted therapy and contact lens use on MMP9 levels in keratoconic eyes and whether disease progression is affected.
Introduction
Keratoconus is classically defined as an irreversible, noninflammatory, progressive corneal ectasia.1,2 It commonly starts in the second decade of life with an estimated prevalence of 1.4–54.5 per 1000 population, greater in those of South Asian and Arab ethnicity.3,4 Keratoconus leaves patients with bilateral irregular myopic astigmatism, which can be high and lead to visual impairment even with the use of glasses. In some areas of the world, early detection and corneal cross-linking (CXL) have been paramount in preventing further progression and decreasing the frequency of keratoconic eyes needing keratoplasty. 5 To devise further targeted treatment, the etiology of keratoconus must be better understood.
Keratoconus’ pathogenesis is multifactorial: a result of interaction between environmental, behavioural, and genetic factors. Risk factors include atopy, contact lens use, collagen vascular disorders, ocular inflammation and trauma (e.g. via eye rubbing and nocturnal eye compression), floppy eyelid syndrome, obstructive sleep apnea, and genetic alterations.1,2,4–6 Bawazeer et al. (2000) concluded that chronic eye rubbing was the most significant risk factor of keratoconus, likely associated with atopy. 1 More recent studies have implicated low-level, chronic inflammatory components detected in tear films including cytokines, growth factors, and enzymes in keratoconus.7–9 Fodor et al. (2020) followed 42 patients over one year and found that tear film levels of interleukin-13 (IL-13) and nerve growth factor (NGF) can predict keratoconus progression with 100% specificity and 80% sensitivity. 10 Matrix metalloproteinases (MMP) are associated with cellular apoptosis and extracellular matrix degradation; several studies have demonstrated significantly enhanced upregulation of MMPs, specifically subtypes 1, 2, 3, 7, 9, and 13, in keratoconus tear films.11,12 Further to this, Shetty et al. (2015) identified cyclosporine A (CyA), an immunosuppressive steroid-sparing agent, to inhibit MMP9 (gelatinase-B) which the authors also found to be elevated in tear films of keratoconus patients. 13
Paucity of consensus regarding the role of inflammation in keratoconus warrants further investigation, especially since the progressive and irreversible disease has an insidious onset and is often asymmetric, affecting one eye before the other. 14 Tear film analysis is a noninvasive biomarker that can be used for diagnostic testing and staging purposes. A better understanding of inflammatory markers implicated in keratoconus may allow for the earlier diagnosis, prompt treatment, and development of targeted therapies. To our knowledge, this is the largest study of its kind to assess MMP9 in tear films of keratoconic eyes and to compare patient ocular features.
Materials and methods
A retrospective, noncomparative chart review was conducted on consecutive patients referred to a keratoconus clinic at Kensington Vision and Research Centre from July 2016 to February 2020. This study received approval from the University of Toronto Research Ethics Board and was carried out in compliance with the tenets of the Declaration of Helsinki.
Data abstraction from patient charts treated by cornea specialists (D.R., N.S., C.C.C.) was performed by two independent authors (P.A.H.N., B.S.), with any unresolved conflicts addressed in collaboration with another two independent arbitrators (T.T., M.M.). Data included demographics, past medical history, and risk factors collected via a standardized intake form (atopy, symptoms, ocular history, systemic comorbidities, medication use, eye-rubbing habits, Symptom Assessment Questionnaire iN Dry Eye (SANDE) responses), and corrected distance visual acuity (CDVA). Corneal tomography measurements were obtained using the Pentacam® (Oculus Inc., Wetzlar, Germany) and topography simulated keratometry (SimK) measurements were obtained using the Nidek OPD3 corneal analyzer (Nidek Technologies, Gamagori, Japan). Automated flat keratometry (K1), steep keratometry (K2), and astigmatism axis (K2 axis) readings were obtained using the Nidek ARK-1S autorefractor/keratometer (Nidek Technologies, Gamagori, Japan). Central corneal thickness (CCT) was measured using a handheld pachymeter (Pachpen, Accutome, Malvern, Pennsylvania, USA). Tear film osmolarity (I-pen Osmolarity System, I-Med Pharma, QC, Canada) was abstracted as a continuous variable. Matrix metalloproteinase-9 (MMP9; InflammaDry®, Quidel Corporation, CA, United States) level was collected as positive (>40ng/mL) or negative.15–21 Patient records were excluded in a stepwise fashion: (1) duplicates; (2) unattended rescheduled appointments with absent data; (3) missing MMP9 test results; and (4) diagnosis of a clinically significant ocular pathology apart from keratoconus in the study eye. All patients with a confirmed diagnosis of keratoconus and available MMP9 results were included. Keratoconus was diagnosed as irregular astigmatism with corneal thinning at the steepest point along with a subjective and/or objective decrease in vision. Disease progression was defined as an increase of >1D of manifest refraction astigmatism, an increase of >1D in steepest keratometry reading, or documented historical progression (change in vision or prescription eyewear). Given keratoconus is often asymmetric with the potential for one eye to have a diagnosis while the other does not, a worst-eye analysis was employed throughout to avoid potential confounding with the inclusion of eyes with unilateral and bilateral disease. The worst eye was selected based on a priority algorithm with preference given to the higher tomography Kmax value, then SimK steep if Pentacam keratometry values were unavailable, followed by any clinical notes indicating keratoconus dominance in one eye over the other by the diagnosing ophthalmologist, and, finally, by using the spreadsheet function = CHOOSE(RANDBETWEEN(1,2),“Right”, “Left”) if the clinical note indicated that both eyes of one patient had keratoconus that was clinically symmetric and if no tomography or topography data was available.
Microsoft® Excel version 16.44 (Microsoft Corporation, WA, United States) was used to manage the collection process and Statistical Package for the Social Sciences (SPSS) Statistics Version 24.0 (IBM Corporation, NY, United States) was used for all data analyses. In the reporting of baseline demographics and study endpoints, continuous data was summarized into means ± standard deviations (SDs), while categorical data was summarized into percentages of the total sample population. Independent samples T tests were used for the comparison of continuous endpoints and Levene's test for equality of variances was used to determine the assumption of equal variances. Pearson chi-squared tests were used for categorical variables. In the primary analysis, eyes were stratified by MMP results (positive or negative). In the case that a significant difference was found in this primary analysis, subgroup analyses were performed for that implicated variable. For CDVA analysis, Snellen values were converted to LogMAR using
Results
1295 appointments were scheduled from July 2016 to February 2020. After the removal of duplicate patient records (n = 223), unattended and/or rescheduled appointments (n = 228), no available MMP9 test results (n = 211), and eyes without keratoconus (n = 81), 551 (42.55%) eyes were included in this analysis. In a worst-eye analysis of the 551 patients, 386 (70.05%) were from male patients, mean age was 30.14 ± 9.76 years, 271 (49.18%) were right eyes, mean CDVA was 0.24 ± 0.32 LogMAR (20/30<συπ>−3</sup> Snellen), mean CCT was 486.76 ± 50.37 μm, mean Pentacam thinnest local was 454.89 ± 59.54 μm, mean Pentacam maximum keratometry was 60.12 ± 10.22 Diopters, mean SANDE score was 3.60 ± 2.71, mean tear film osmolarity was 311.21 ± 20.03 Osm, 354 eyes (64.25%) wore glasses, 115 eyes (20.87%) wore contact lenses, and 306 eyes (55.5%) were MMP9 positive (Tables 1 and 2).
Demographic characteristics of patients with keratoconus at first assessment, stratified by MMP9 positivity.

CL, contact lens; FHx, Family History; MMP, Matrix Metalloprotinease; N = Population Size; n = Sample Size; OD, Oculus Dexter/Right Eye; SD, Standard Deviation; μ, Mean; x̄, Median.
Independent sample T-test analysis, equal variances assumed as per Levene's test (significance > 0.05).
Pearson's Chi-Square Test analysis.
Rating scale and test results of patients with keratoconus at first assessment, stratified by MMP9 positivity.
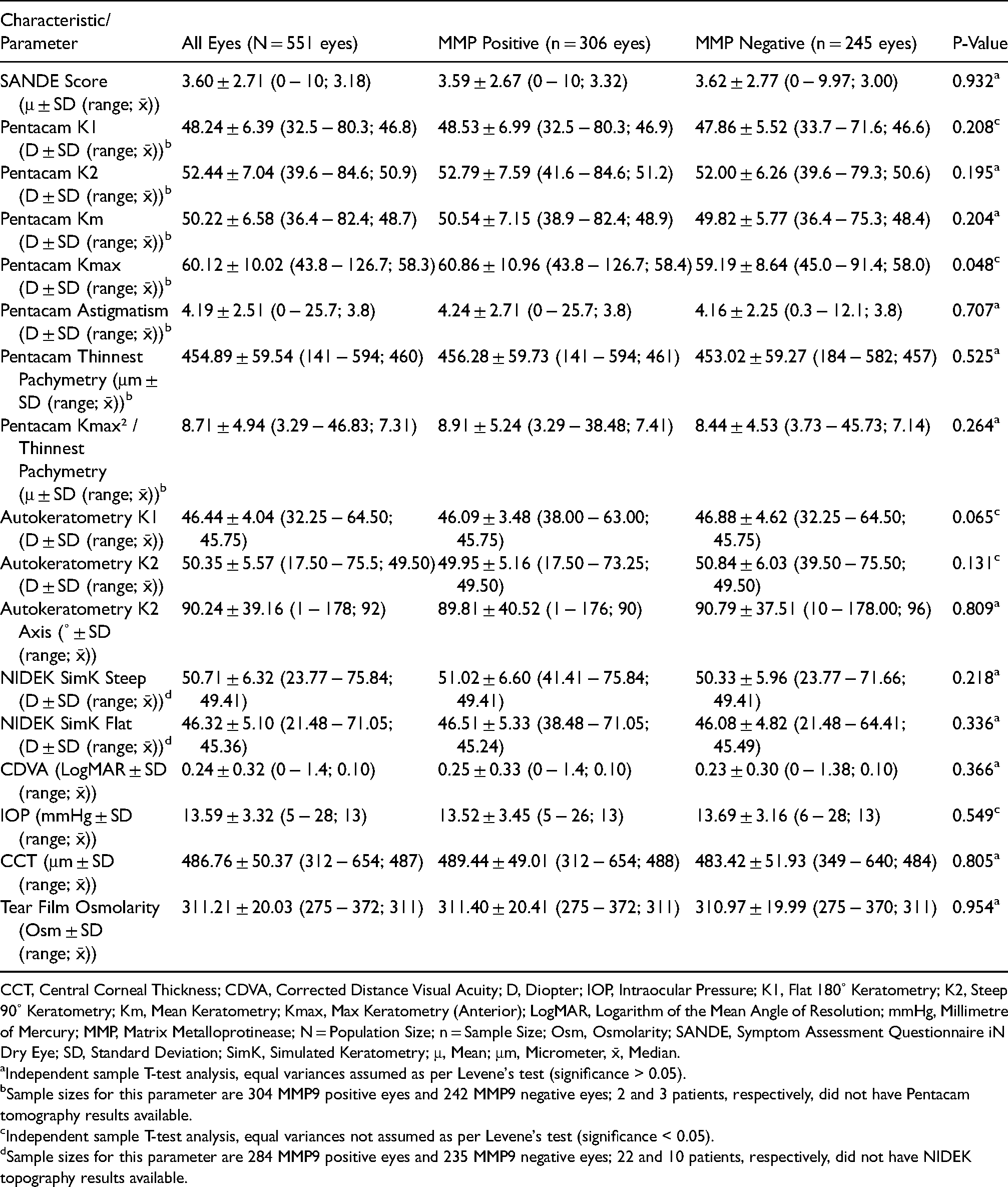
CCT, Central Corneal Thickness; CDVA, Corrected Distance Visual Acuity; D, Diopter; IOP, Intraocular Pressure; K1, Flat 180˚ Keratometry; K2, Steep 90˚ Keratometry; Km, Mean Keratometry; Kmax, Max Keratometry (Anterior); LogMAR, Logarithm of the Mean Angle of Resolution; mmHg, Millimetre of Mercury; MMP, Matrix Metalloprotinease; N = Population Size; n = Sample Size; Osm, Osmolarity; SANDE, Symptom Assessment Questionnaire iN Dry Eye; SD, Standard Deviation; SimK, Simulated Keratometry; μ, Mean; μm, Micrometer, x̄, Median.
Independent sample T-test analysis, equal variances assumed as per Levene's test (significance > 0.05).
Sample sizes for this parameter are 304 MMP9 positive eyes and 242 MMP9 negative eyes; 2 and 3 patients, respectively, did not have Pentacam tomography results available.
Independent sample T-test analysis, equal variances not assumed as per Levene's test (significance < 0.05).
Sample sizes for this parameter are 284 MMP9 positive eyes and 235 MMP9 negative eyes; 22 and 10 patients, respectively, did not have NIDEK topography results available.
After stratification of the eyes by MMP9 test results, eyes that tested positive were more likely to be of male patients (p < 0.001) and from patients who had a paediatric history of asthma (p = 0.042) and wore glasses (p = 0.041). Further, MMP9 positive eyes were significantly more likely to have a higher Pentacam maximum keratometry reading (p = 0.048) than MMP9 negative eyes. Eyes that were MMP9 negative were significantly more likely to be of patients who used contact lenses (CL; p = 0.012). No other significant differences in atopic history, family history, SANDE scores, topography, tomography, or tear film osmolarity were observed between MMP9 positive versus negative eyes. To note, in the MMP9 positive group, corneal tomography and topography results were unavailable for 2 and 22 patients, respectively; in the MMP9 negative group, corresponding values were 3 and 10 patients.
Current CL use was reported in 115 eyes and specific data on the type of CL used was available for 106 eyes (Table 1). Patients with MMP9 negative eyes were more likely to report use of soft CLs (63.79% vs. 35.42%; p = 0.004); no significant differences were found in the use of piggyback CLs, rigid gas permeable CLs, and scleral CLs. Compared to non-CL eyes, eyes which used soft CLs had a significantly lower Kmax (54.85 ± 5.82 D vs. 65.29 ± 9.21 D; p < 0.001) and SimK Steep (47.84 ± 2.95 D vs. 56.25 ± 7.18 D; p < 0.001). Eyes which used rigid gas permeable CLs and scleral CLs had a significantly higher Kmax and SimK Steep (rigid gas permeable: Kmax 64.78 ± 9.52 D vs. 58.06 ± 8.50 D; p = 0.001, and SimK Steep 56.77 ± 8.21 D vs. 49.83 ± 5.02 D; p < 0.001 | scleral: Kmax 67.25 ± 8.78 D vs. 58.30 ± 8.58 D; p < 0.001, and SimK Steep 56.27 ± 5.46 D vs. 50.90 ± 6.70 D; p = 0.003). No Kmax differences were found between non-CL eyes and eyes which used piggyback CLs (p = 0.577). No differences in a history of atopy, eye rubbing habits, and floppy eyelid syndrome were found between patients who were CL and non-CL users (p = 0.803, 0.548, and 0.240).
In a male versus female analysis, males were more likely to report snoring (55.7% vs. 38.2%; p < 0.001) and have floppy eyelid syndrome (16.1% vs. 8.5%; p = 0.018). Females were more likely to report using glasses (70.9% vs. 61.4%; p = 0.033) and CL use (26.1% vs. 18.7%; p = 0.05). Male eyes tested positive for MMP9 significantly more than female eyes (62.2% vs. 40.0%; p < 0.001) and had significantly lower autokeratometry flat K readings (46.14 ± 4.07 vs. 47.04 ± 3.92; p = 0.039). Male patients had significantly lower SANDE questionnaire scores (3.30 ± 2.53 vs. 4.31 ± 2.98; p = 0.001). While male eyes had a higher mean Kmax than female eyes, the difference was nonsignificant (60.28 ± 10.36 vs. 59.74 ± 9.20; p = 0.567).
Discussion
Given recent findings that keratoconus may be associated with an inflammatory component, we aimed to assess the efficacy of MMP9 positivity as a predictor of keratoconus severity.23–25 To our knowledge, this is the largest study to date that evaluates inflammation in a cohort of keratoconus patients using MMP9 and tear film osmolarity.
MMP9, a 92 kDa gelatinase, is one of the primary proinflammatory cytokine modulators synthesized by the corneal epithelium and responsible for matrix degradation. 26 Upregulation of MMP9 is triggered by an insult to the corneal epithelium; rather than consistently promoting epithelial regeneration and stability, MMP9 has been found to inhibit re-epithelialization, exacerbate extracellular matrix degradation, and precipitate sterile corneal ulceration.27,28 Increased expression of MMP9 has been implicated in the initiation and progression of ocular surface disease, causing progressive inflammation. 15
In keratitis sicca and related syndromes, use of an MMP9 immunoassay has been reported as an efficacious tool to prognosticate patients and monitor their response to treatment. 24 Given that patients with keratoconus report comparable corneal dysesthesias (burning, pruritus, foreign body sensation) and epiphora, MMP9 may similarly be useful in the diagnosis and management of keratoconus. 29
Several studies have reported the degree of corneal inflammation to be demonstrated by the presence of MMP9 in the tear film and further detected increased levels of MMP9 and MMP-1 in keratoconic eyes up to 10-fold compared to normal controls which correlated with disease severity.13,16,30,31 This aligns with enzymatic analyses revealing increased collagenolysis and gelatinolysis with associated upregulation of MMPs, therefore causing proteolytic imbalances due to collagen degradation in keratoconus.12,30,32 However, other studies are discordant with these findings where the level of MMP9 in tear films did not differ amongst healthy controls, subclinical, and diagnosed keratoconus.12,16,33 Considering this lack of consensus and that the presence of inflammation can be assessed by markers such as MMP9 in the tear film, our study aimed to assess for correlation between MMP9 and keratoconus disease severity.
After stratification of the eyes of patients diagnosed with keratoconus into MMP9 positive versus negative groups, our results revealed that MMP9 positive eyes were significantly more likely to exhibit worse disease based on the maximum anterior sagittal curvature (maximum keratometry; Kmax) obtained via Pentacam tomography (p = 0.048). Kmax is often used in the clinic to identify keratoconus progression, especially after CXL which aims to prevent disease progression.34,35 As such, the correlation between MMP9 and Kmax may prove useful as an additional metric for earlier identification of keratoconus which is at higher risk of progressing to advanced disease. 36 On the other hand, Kmax in isolation has also been described as a substandard measure to diagnose keratoconus, assess progression, and determine CXL efficacy as it fails to address the posterior cornea or the extent of ectasia. 37 Toprak et al. (2015) described the ratio of Kmax 2 to thinnest pachymetry as a more robust method of gauging progression with a sensitivity greater than 99% and specificity greater than 94%. 38 When analyzing our data using this method, no significant difference was noted between the MMP9 groups (p = 0.264).
Given a mean age of 30.14 ± 9.76 for the patients included in this analysis, our results confirm past findings that keratoconus primarily affects those between the second to fourth decade of life. 3 To note, this finding may reflect referral bias where our center is less likely to assess paediatric patients.39,40 Results from an analysis of differences by sex in this study generally conformed with the literature: keratoconus was found to be more prevalent in men. 4 Per data published by Fink et al. (2005), who found that females may be more symptomatic from keratoconus and visit their eye care practitioner as a result, the prevalence observed in males may still be an underestimate. 41 Our results reveal a similar pattern where males reported decreased symptom sensitivity, specifically in frequency and severity, as compared to females in their responses to the SANDE questionnaire. 42 On the contrary, while MMP9 positivity was correlated with a higher Kmax value and male eyes, these associations were independent of each other. In other words, males were more likely to test MMP9 positive; albeit, a higher grade of keratoconus along with an increased prevalence of refractive errors in male eyes was not directly observed in this analysis.
Compared to patients with MMP9 positive eyes, those with MMP9 negative eyes were found to have an 8.7% (p = 0.012) higher likelihood of using CLs in general and were 28.4% (p = 0.004) more likely to use soft CLs. Soft CLs may reduce epithelial trauma and deter a patient from eye-rubbing. 43 On the other hand, data identifing CLs as pro-inflammatory devices with measured increases in cytokines such as IL-17A and IL-6 illustrate that CLs may precipitate the progression of keratoconus.43–48 Established downsides of CLs include tear stagnation, potential hypoxic effects, and scarring responses which can be more pronounced with improper CL fitting, potentially causing molding and advancement of ectasias.43,49–51 In this analysis, the use of soft CLs was associated with patients whose eyes tested MMP9 negative. A significantly lower Kmax in the eyes of soft CL users could be related to the fact that less advanced keratoconus patients can still tolerate soft CL wear and achieve adequate vision improvement. Eye rubbing is a strong risk factor for keratoconus and can induce elevated inflammatory markers.6,52–54 Such habits may be reduced in CL users which in turn can prevent progressively increasing Kmax in these keratoconic eyes.53,54 However, our study did not find any significant differences in eye rubbing habits between eyes of patients who were CL and non-CL users. Eyes with more advanced keratoconus, i.e., higher Kmax values, may require rigid gas permeable CLs, scleral CLs, piggyback CLs, or be CL intolerant given potentially increased surface inflammation given a significantly higher MMP9 positive rate in this group. To note, keratoconus was generally advanced amongst included eyes (Kmax 60.12 ± 10.12 D; range: 43.8 − 126.7; median 58.3) for which contact lenses are a more common next step in treatment rather than glasses. Despite this, 64.1% of patients reported wearing glasses versus only 20.9% using contact lenses. We postulate this to result from a patient population who may not readily afford hard contact lenses, topography-guided photorefractive keratectomy, or private CXL therapy. Other causes may include glasses use concurrent with rapid and severe progression of keratoconus and specialists recommending soft CLs which are easier to fit or CXL therapy prior to fitting patients for a trial of hard CLs. Further research is required to better elucidate the relationship between CL wear, types of CLs, ocular surface inflammation, and keratoconus severity.
This study has some limitations. A chart review may be constrained by missing data and no blinding to the study purpose; other limitations such as potential data conflicts were mitigated by two independent data collectors and two additional data arbitrators. Kmax is one widely accepted method to determine keratoconus severity; nonetheless, it may not be as efficacious in the case of decentered cones, posterior corneal changes, or acentric/non-apical corneal thinning. 34 The InflammaDry protocol provided by the manufacturer does not require a specific tear film volume so MMP9 levels may be undetected and yield false negatives. 55 Also, InflammaDry does not quantify MMP9 tear film levels; rather, samples can only be interpreted as positive or negative negating exploration of detailed correlation between MMP9 levels and disease severity. The single measurement approach of this study could not adequately address test-retest reliability. Further, keratoconus patients may have comorbid dry eye disease and variable tear volumes; patients in this study did not undergo Schirmer's testing to evaluate for aqueous tear deficiency, and there was no reliable method to assess tear film volume consistently. Nevertheless, no significant difference in the SANDE and osmolarity scores between MMP9 positive and negative eyes was observed; therefore, the confounding role dry eye disease may have on keratoconus should not exert a significant effect in this analysis. Information on whether patients rubbed their eyes immediately before MMP9 testing and general patterns, frequency, and duration of eye rubbing habits were not available. Additionally, eye rubbing is a well-known risk factor for keratoconus; therefore, a lack of acknowledgement of eye rubbing by patients may not reflect their true habits when awake or asleep resulting in underestimation of the true prevalence of eye rubbing in this study. Given its retrospective nature, only keratoconic eyes were included and findings could not be compared to healthy controls or nonkeratoconic eyes with a history of atopy; despite an established MMP9 threshold of ≥40ng/mL in the literature, it may be beneficial for future studies to plan for the recruitment of normal controls. Tomography was unavailable for 0.91% of eyes, potentially due to out-of-pocket costs for this non-insured test; topography, a financially covered test for patients, was unobtainable for 5.81% of eyes given advanced disease. There was no analyzable data regarding the financial status of and social determinants of health barriers faced by study patients but having this information may potentially identify avoidable inequities in keratoconus treatment. Finally, keratoconus progression may be influenced by sleeping on one side; this information along with BMI were not collected in our study. 6
In conclusion, our study is the largest to date to evaluate MMP9 in a cohort of keratoconus patients. We found that MMP9 positive keratoconic eyes had significantly higher Kmax readings which may correlate with increased disease severity, further supporting keratoconus to be associated with an inflammatory component. Eyes of patients who used soft contact lenses were less likely to be MMP9 positive and more likely to have lower Kmax values. This is consistent with milder keratoconus where adequate vision improvement was achieved without hard lenses. Further research is warranted to evaluate the role of targeted therapy and contact lens use on a wider range of inflammatory markers in keratoconic eyes and their possible effects on disease progression.
Meeting presentation(s)
Nichani P, Solomon B, Trinh T, Mimouni M, Rootman D, Singal N* and Chan CC*. (Jun 2022). Investigating the role of inflammation in keratoconus: A retrospective analysis of 551 eyes. Canadian Ophthalmological Society Annual Meeting & Exhibition (COS), Virtual Meeting (Oral).
Nichani P, Solomon B, Trinh T, Mimouni M, Rootman D, Singal N* and Chan CC*. (Nov 2021). Investigation of inflammatory markers in patients with keratoconus: A retrospective analysis of 551 eyes. American Academy of Ophthalmology, New Orleans, LA, United States (Poster).
Nichani P, Solomon B, Trinh T, Mimouni M, Rootman D, Singal N* and Chan CC*. (Apr 2021). Investigation of inflammatory markers in patients with keratoconus: A retrospective analysis of 551 eyes. UofT Department of Ophthalmology & Vision Sciences 63rd Research Day, Virtual Meeting (Oral).
Footnotes
Online supplements
None.
Acknowledgements
None.
Declaration of conflicting interests
PN: None. BS: None. TT: None. MM: None. DR: Alcon (H), Johnson & Johnson (H). NS: None. CCC: Alcon (H, R), Allergan (H, R), Bausch & Lomb (H, R), Johnson & Johnson (H, R), Labtician Thea (H, R), Santen (H, R), Shire (H, R), Tearlab (H, R), Zeiss (H). Legend: C – consultant/consulting fees; E – equity owner; H – honoraria; R – research grant/financial support.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author note
Michael Mimouni, Department of Ophthalmology, Rambam Health Care Campus, Haifa, Israel.
