Abstract
Introduction
The most common site of obstruction in the lacrimal duct is the junction between the lacrimal sac and the nasolacrimal duct (NLD). 1 Primary Acquired Nasolacrimal Duct Obstruction (PANDO) is the most common cause of obstruction of NLD in adults and occurs more frequently in women between the fifth and sixth decades of life. Epiphora and dacryocystitis occur in 1% of adults and in 3–5% of children with obstruction of the NLD.2,3
Dacryocystorhinostomy (DCR) is the procedure of choice for the treatment of NLD obstruction (NLDO) and consists in the creation of a bypass between the lacrimal sac and the ipsilateral nasal cavity. There are three different approaches to this surgery: the ab externo, the intranasal and transcanalicular diode laser-assisted.4,5
Anatomic variants and intranasal pathologies (deviation or adhesion of the nasal septum, nasal polyps, rhinosinusitis) are the most common cause of nasal narrowing, resulting in failure of the DCR. Other causes are the formation of granulation tissue and scar tissue, inappropriate localization of the ostium, inadequate size of the fistula, lacrimal sump syndrome, previous maxillofacial trauma. 6 For this reason, the pre-operative ENT assessment is of crucial importance in identifying possible causes of failure of the DCR. Several common causes of DCR failure are reported: cicatricial closure of the ostium, inadequately sized osteotomy, inadequate lacrimal sac marsupialization, common canalicular obstruction, intervening ethmoids, inappropriately placed osteotomy with respect to the lacrimal sac leading to sump syndrome, turbinoseptal synechiae in and around the ostium, inappropriate granulation tissue, and internal ostium stenosis. Not rarely, multiple causes for failure may be noted.7–9
Epiphora could also be present in case of anatomical patency of the lacrimal drainage apparatus. These cases are referred to as ‘‘functional block’’ and the diagnosis is one of exclusion. 10
Several studies have investigated the proinflammatory action and fibrotic changes produced by glaucoma eye drops on the conjunctival surface.11,12 These changes can be caused directly by the active ingredients or by the preservatives present in commercial preparations.13–15
Hereby we report our clinical experience and physiopathological hypothesis of patients on prostaglandin analogues presenting with persistent epiphora after EN-DCR.
Methods
This study was conducted in compliance with the Declaration of Helsinki and Health Insurance Portability and Accountability Act. Disclosure of identifiable health information was given with written patient consent.
In this prospective observational cohort study, we included all consecutive patients: (1) with a dacryo-CT documented diagnosis of PANDO, (2) who underwent for the first time EN-DCR, with intubation of lacrimal pathway and without mitomycin-C, at Humanitas Research Hospital, Rozzano, Milan, Italy, from June 2018 to June 2020, (3) who were affected by glaucoma and treated with either topical monotherapy PGs (PG group) or other antiglaucoma molecules (namely, beta blockers, adrenergic agonists, cholinergic agents, and carbonic anhydrase inhibitors) in monotherapy or in combination therapy (non-PG group), (4) who attended 3-month, 6-month and 12-month postoperative follow-up, and (5) who were older than 65 years. It should be noted that only patients who did not have associated osteomeatal complex alterations at the dacryo-CT were taken into consideration to standardize the surgical procedure.
Patients with any of the following conditions were excluded: (1) history of associated tear flow disturbances like punctal or canalicular obstruction, (2) lid malposition.
Two groups were created based on the regimen of eye drops containing PGs or not to investigate possible differences in post-surgical epiphora incidence depending on antiglaucoma medication type. Prostaglandin eyedrop therapy was commenced 2 years and 4 months previously on average. Nine patients were on Bimatoprost, four on Latanoprost and two on Tafluprost administered as per technical specification. Thirteen patients belonging to the control group were on Timolol, of which three associated with Brimonidine, and two were on Carteolol.
NLD patency was assessed in all patients through endonasal probing and irrigation of lacrimal pathway preoperatively, early postoperatively at month 3, and late postoperatively at months 6 and 12. At the same time, both objective and subjective measures of epiphora were evaluated respectively through Fluorescein Dye Disappearance Test (FDDT) and Munk scale (
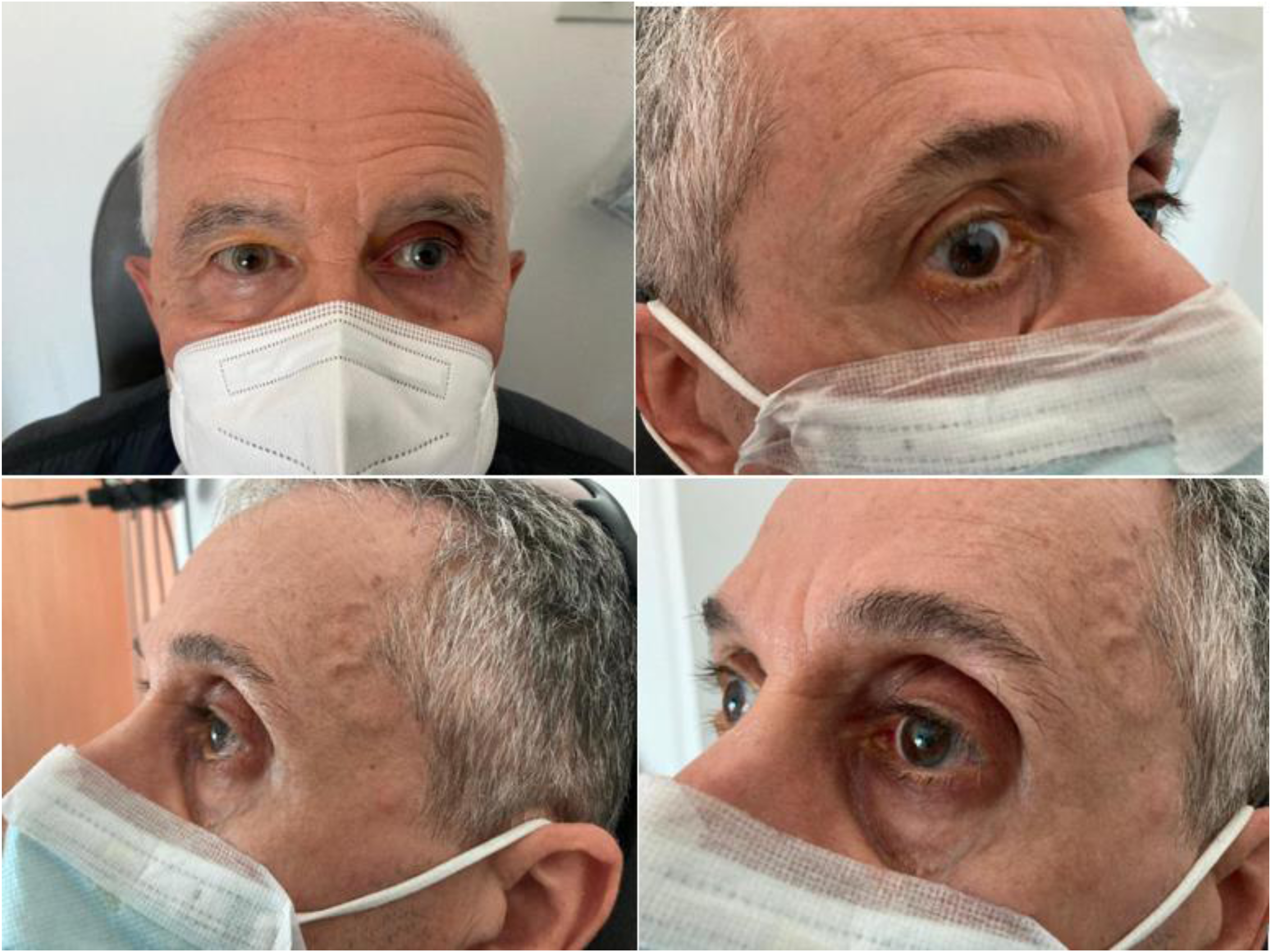
shows an example of a patient treated with right DCR for epiphora and examined with fluorescein dye disappearance test signs of long-term treatment with prostaglandin-based eyedrop, orbital fat atrophy and droopy eyelid are evident. FDDT shows impaired dye drainage despite drainage surgery.
Definition of fluorescein Dye disappearance and munk scale
Epiphora was assessed objectively with Fluorescein Dye Disappearance test (FDDT). Most specifically, after a drop of 2% fluorescein was placed in the fornix, the fluorescein remaining in the conjunctival cul-de-sac was examined with the cobalt blue light after five minutes. The amount of remaining fluorescein was graded using a scale from 0 to 4, where 0 indicates no remaining dye, and 4 indicates all the dye remains. This was also compared to the opposite eye. Retention of the fluorescein indicated a delay in tear flow. Grades 0 and 1 were considered as negative FDDT, and grades from 2 to 4 as positive FDDT.
Epiphora was also assessed subjectively with Munk scale. All patients were asked to rate subjectively their epiphora severity based on the 0–4 scale developed by Munk et al. 16 ranging from 0 (no watering) to 4 (constant watering). Grades 0 and 1 were considered as negative Munk, and grades from 2 to 4 as positive Munk.
Statistical analysis
We used STATA software (Stata Corp., College Station, TX, USA) for the statistical analysis. All tests were two-sided. P-values <0.05 were considered statistically significant.
The primary outcome measure was the presence of post-surgical epiphora in the two groups, as objectively (FDDT) and subjectively (Munk scale) detected in early to late postoperative follow-up.
Proportions were compared by means of the Fisher exact test We also computed the relative risk (risk ratio) and the respective 95% confidence interval (95% CI) for each comparison. When zero events occurred in one group of patients, we used a continuity correction that was inversely proportional to the relative size of the opposite group.
Approval of the present study was issued by IRCCS Humanitas Research Hospital, via Manzoni 56, Rozzano, Milan, Italy.
Results
Fifteen right eyes from 15 PG-group patients and 15 right eyes from 15 non-PG-group patients were considered in this study.
No statistically significant differences between the 2 groups were detected in terms of age, gender, treatment duration and IOP at the enrollment. Median age was 69.9 + /- 4.3 SD, 17 were female and 13 male patients. Prostaglandin eyedrop therapy was commenced 2 years and 4 months previously on average. Mean IOP was 19.6 + /- 2.3 mmHg.
There was always concordance between FDDT and Munk grading for epiphora evaluation at different follow-up visits. Epiphora occurred in 1/15 (7%) of PG-group patients at 3 months, in 8/15 (53%) at 6 months, and in 11/15 (73%) at one year. In comparison, only 1/15 of non-PG-group patients developed epiphora at 6 months.
The relative risk of epiphora in PG-group patients versus non-PG-group patients was 3.00 (95% CI, 0.13‒68.1; p = 0.99) at 3 months, 8.00 (95% CI, 1.14‒56.3; p = 0.014) at 6 months, and 11.0 (95% CI, 1.62‒74.9; p < 0.001) at 12 months. The histograms display epiphora frequency distribution in the two groups at different follow-up visits (
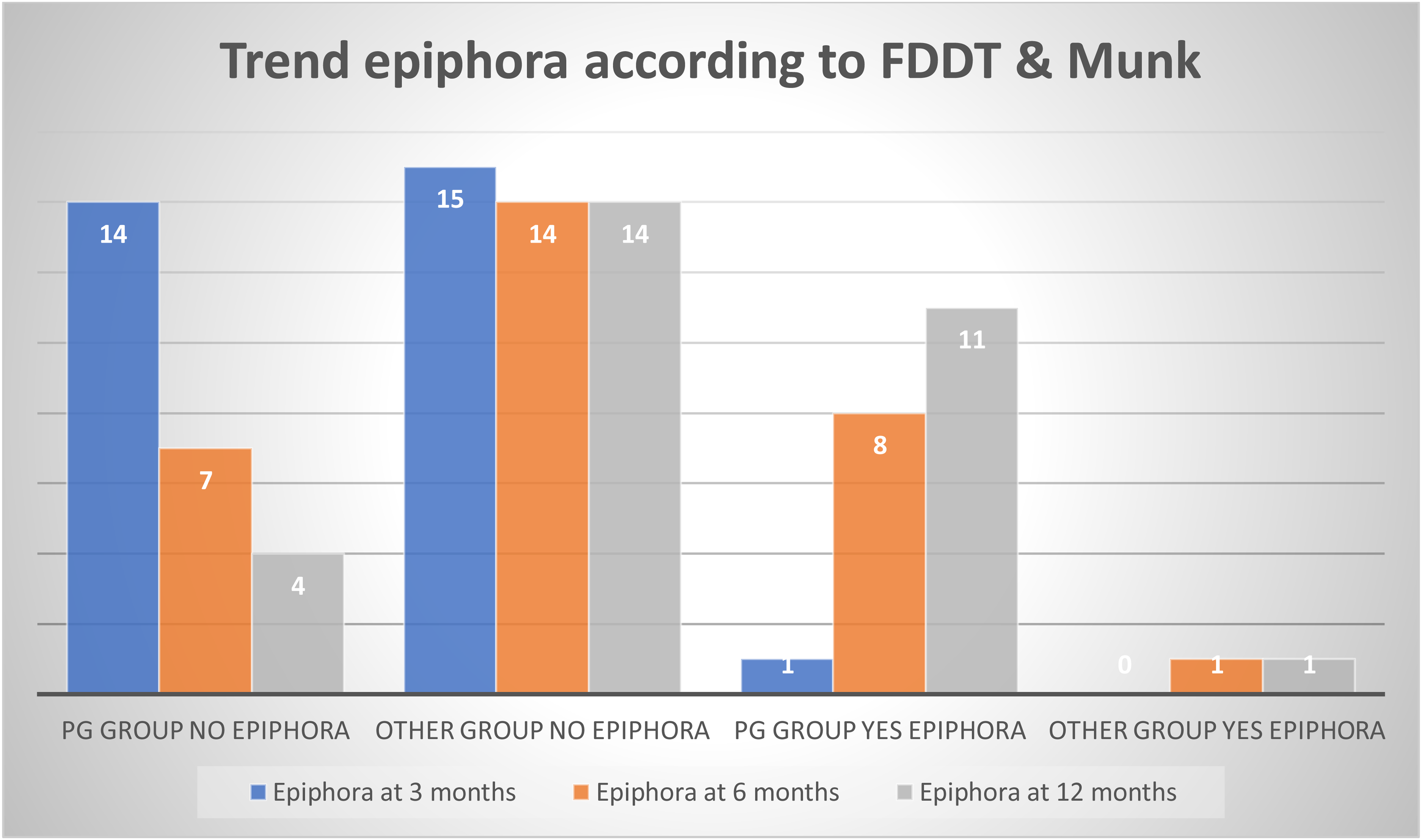
shows epiphora frequency distribution in the two groups at different follow-up visits. PG = Prostaglandin-based eyedrops.
Discussion
In this prospective observational cohort study, we explored the relationship between topical antiglaucoma therapy and persistence of epiphora in glaucoma patients who underwent EN-DCR surgery for PANDO. We chose EN-DCR instead of external DCR because we believe that the first allows to correct any pathologies of the osteomeatal complex that might cause the stenosis or would condition a restenosis.
Further, we investigated the difference in postsurgical epiphora incidence between PG-group patients and non-PG-group. Glaucoma patients exposed to PG topical eyedrops showed a higher risk of developing post-surgical epiphora compared to patients exposed to other antiglaucoma eyedrops, with a greater difference at month 12.
Prostaglandin analogs are one of the most efficient topical treatments for glaucoma with a good safety profile. Benzalkonium chloride (BAC) is usually used as a preservative because it has an antimicrobial function and it increases the prostaglandin biodisponibility, but it could also induce proapoptotic and necrotic effects involving oxidative stress and reactive oxygen species (ROS).
Guenon et al. 17 showed that the cytotoxicity is proportional to the BAC concentration in eyedrops: bimatoprost contains the lowest BAC concentration, so it could be less toxic than latanoprost and travoprost 18
Some authors 19 demonstrated that β blockers can induce a higher conjunctival inflammation than prostaglandin analogues, as evaluated by an increase in inflammatory marker expression and a decrease in mucin production. Other studies 20 conducted in patients receiving monotherapy with latanoprost or β blockers, showed that an early inflammatory condition (3–4 months) after prostaglandin treatment could develop but its intensity would not increase over time and a long therapy with β blockers would finally reach higher inflammatory levels.
Kashkouli et al. 21 studied the effect of antiglaucoma eyedrops on the lacrimal system: two drugs combination (timolol + dorzolamide and timolol + dorzolamide + pilocarpine) had a strong association with obstruction of the lacrimal drainage system, even if their patients on timolol alone did not show a significant epiphora.
We wish to emphasize that patients on PGs showed a significantly lower success rate than previously reported for EN-DCR while the other treatments group revealed high success rates as described for this technique. This might in fact be explained by the proinflammatory effect of PGs which might be more intense than that of β blockers in the long term.
It is curious that there has always been an agreement between the objective and subjective parameters and the authors should discuss this fact in Discussion Section. In general, the studies that approached the several techniques of DCR revealed important differences between the objective and subjective measures of success. 22
Recently, C. Pinto et al. 23 hypothesized that the use of topical prostaglandin analogues to lower intraocular pressure could influence the success rates promoting occlusion of the neo-ostium obtained after TCL-DCR. However, they showed no significantly different success rates in patients under these drugs after a 36-month follow-up.
We know that PG analogues induce a pro-inflammatory status and that there is evidence of its impact on lacrimal pathology. However, the possible effect of BAC was not ruled out and we cannot exclude a significant impact of these preservatives in the results obtained. It is then crucial to highlight that the next step should be to study their possible impact comparing two groups, one with preservative-free PG formulations and another with BAC or other preservative containing PG formulations.
Another of the major limitations of this study is the small number of patients we managed to enroll. Further studies are necessary to underpin our finding, possibly with more numerous cohorts.
Conclusion
Based on our results, we speculate that prostaglandin eye drops may negatively affect the outcome of EN-DCR in terms of increasing epiphora recurrence from early to late postoperative follow up. The toxicity of antiglaucoma eye drops might damage spiral fibers of the mucous membrane of the lacrimal system inducing fibrosis by a proinflammatory mechanism with subsequent loss of propulsive ability.
Accordingly, despite a post DCR patent lacrimal drainage apparatus, confirmed by irrigation of lacrimal duct, patients keep showing epiphora. The knowledge of this possible cause of DCR failure allows to inform patients correctly before surgery, modulating their expectations.
Future studies are warranted to further define the role of topical antiglaucoma treatments as predictors of epiphora persistence in patients with PANDO undergoing DCR surgery.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Disclosure statement
The authors report no conflicts of interest The authors alone are responsible for the content and writing of the article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
