Abstract
Purpose:
Patients who have suffered an ocular trauma may present with varying degrees of injury to the anterior segment. In this retrospective interventional case series, we report the outcome of seven patients who underwent complete anterior segment reconstruction in a single surgery.
Methods:
All patients with posttraumatic corneal decompensation or scar, aphakia, and iris defect underwent human donor corneal graft transplantation and implantation of an intraocular lens combined with a flexible silicone iris prosthesis. Postoperative examinations included assessment of best corrected distance visual acuity, objective refraction, and intraocular pressure. Sensitivity to glare and subjective discontent with the eye’s appearance was rated on a scale from 1 to 10, with 1 standing for low and 10 for high severity.
Results:
Mean best corrected distance visual acuity (BCDVA) was 1.51 ± 0.26 logMAR preoperatively and 1.29 ± 0.36 logMAR postoperatively. Mean IOP was 15.71 ± 8.94 mmHg pre-surgery and 13.57 ± 6.52 mmHg post-surgery. The mean sensitivity to glare was reduced from 7.17 ± 2.91 to 3.80 ± 3.43 and subjective cosmetic disfigurement was reduced from 5.33 ± 3.35 to 1.80 ± 1.60.
Conclusions:
A single surgery technique for entire anterior segment reconstruction in trauma patients can effectively reduce glare and patient discontent with the eye’s appearance.
Keywords
Introduction
Traumatized eyes often show multiple structural damages. In many cases, almost the complete anterior segment of the eye is affected, including cornea, iris, and lens. Most of the injuries result from foreign material that hits the eye from anterior, with only the eyelid protecting the globe. Due to the direction of the impact, anatomical structures are involved consecutively in the following order: first the cornea, secondly the iris, lastly the lens. In even more severe cases, also the posterior segment of the eye is affected. 1 It is sometimes challenging to reconstruct these structures in one single surgery.
However, nowadays, there are implants which allow surgeons to reconstruct anterior ocular structures: human donor corneal grafts or corneal prostheses 2 can substitute the cornea, the human lens can be replaced with an intraocular lens, and the human iris can be substituted with an iris prosthesis. 3 We present our results in trauma cases in which there is a complete reconstruction of the anterior segment – with corneal transplantation, implantation of an iris prosthesis, and implantation of an IOL – all performed in one single surgery.
Material and methods
Inclusion criteria were (1) the need for a corneal transplant, IOL, and iris reconstruction, (2) visual acuity of at least light perception, and (3) stable posterior segment with an attached retina. Eight patients were included in this retrospective, interventional case series. From eight cases, outcomes can be reported for seven patients. One patient was excluded from the investigation after failure to attend eye-examinations. Patient consent to publish this case series including images was obtained.
Written informed consent was obtained, and the patient was especially informed about the long surgery time and the risk for intra- and postoperative complications (e.g. retinal detachment, IOP rise, and corneal graft failure). The study was approved by the local Ethics Committee and made under the tenets of the Declaration of Helsinki. The corneal donations were obtained from the responsible eye bank, no tissues were procured from vulnerable populations, for example prisoners.
Material
The corneal grafts were standard not HLA-matched human grafts. Therefore, we depended on the availability of human donor donations. Single continuous sutures, double-running cross-stich sutures (according to Hoffmann) or single interrupted sutures were used for fixation of the graft.
For treatment of aphakia or traumatic cataract, we used the standard IOL used at our center for routine patients with aphakia. In patients who were already pseudophakic, the IOL was explanted and replaced. We chose a flexible acrylic IOL, the Aspira MC6125AS-Y (HumanOptics, Erlangen, Germany), which because of the soft material is amenable for suturing and its haptics can be readily severed.
We used the flexible, ArtificialIris (AI) implant (HumanOptics, Erlangen, Germany, Figure 1(d)), which features an overall diameter of 12.8 mm and a fixed pupil aperture of 3.35 mm. The thickness decreases from the pupillary margin (0.4 mm) to the peripheral edge (0.25 mm). The surface of the prosthesis is individually handcrafted with colored silicone, based on a photographic documentation of the patient’s iris in the healthy fellow eye. The posterior consists of a smooth, opaque, and black silicone layer. This device received European Conformity (CE) marking for use within the European Economic Area in 2011, and Food and Drug Administration (FDA) approval in the United States in 2018. One advantage of this silicone-iris device is that most commercially available IOLs can be sutured to its posterior side (Figure 1(f)—(h)).
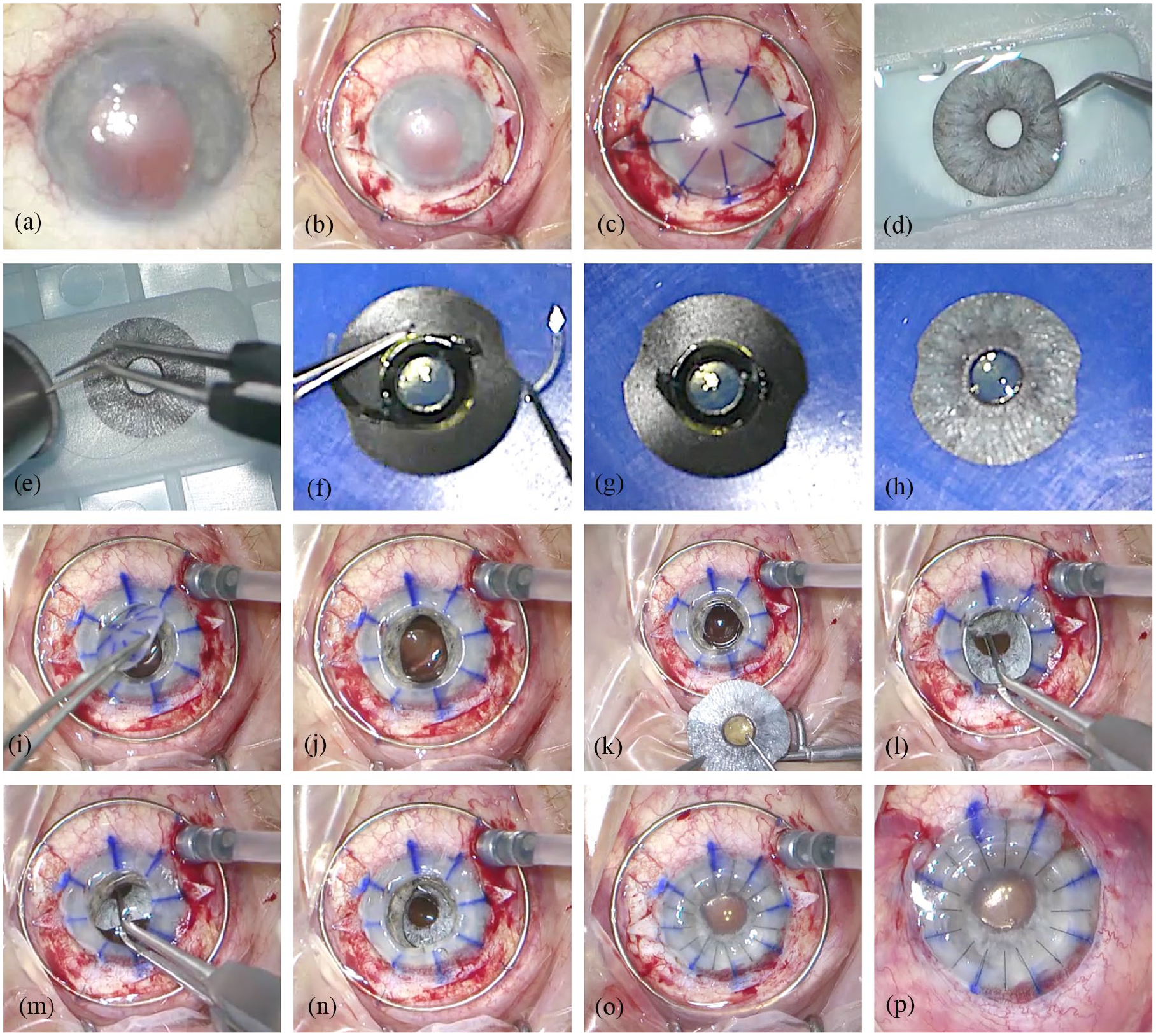
Surgical steps: (a) anterior segment at the beginning of surgery shows corneal scarring, traumatic iris defect mydriasis, and aphakia, (b) suturing of the Flieringa ring onto the sclera and preparation of the scleral flaps at the 3 and 9 o’clock position, (c) corneal marking, (d) ArtificialIris with individually colored anterior surface, (e) trephination of the iris prosthesis, (f) suturing of the IOL on the backside of the iris prosthesis, (g) cutting of the IOL haptics, (h) photo of the combined ArtificialIris-IOL-“sandwich,” (i) trephination and removal of the altered cornea, (j) open sky, (k) placement of the sutures for scleral fixation through the central opening beneath the scleral flaps, (l) and (m) implantation of the ArtificialIris-IOL-“sandwich” through the large central opening, (n) placement and fixation of the combined implant in the ciliary sulcus by tightening of the sutures, (o) insertion and suturing of the corneal graft, and (p) anterior segment at the end of surgery.
Description of surgical procedure (Figure 1, Supplemental Video):
All patients were aphakic, pseudophakic or had traumatic cataract as well as a traumatic iris defect and corneal decompensation or corneal scar after ocular trauma.
All seven reconstructive procedures were performed with general anesthesia and by the same surgeon (CM). All procedures occurred a sufficiently long time after the initial trauma in an interval in which the eye was free of inflammation and in a stable condition. The time between the trauma and the reconstructive surgery varied between 9 months and 37 years. None of the surgeries were performed as primary repair of the trauma.
After opening of the conjunctiva, a Flieringa ring was sutured onto the sclera for intra-operative stabilization of the globe. Scleral flaps were prepared in the 3 and 9 o’clock position. After trephination of the cornea with handheld trephines, the tissue bridges were dissected with Troutman corneal scissors. In pseudophakic patients, the IOL was removed through the opening of the trephined cornea. In patients with traumatic cataract, we proceeded with capsulorhexis and phacoemulsification. The AI was trephined to the required diameter and two prophylactic iridectomies were made on the prosthesis. We used 9.0 or 10.0 polypropylene sutures to attach the AI to the IOL. These sutures penetrated the IOL haptic at a point close to the optic-haptic junction from the posterior direction; then the IOL and AI together were turned around on the front side to go back through the iris. A knot was placed on the posterior side. To reduce the size of the AI and IOL combination and to keep the incision size as small as possible, the distal part of the IOL haptics were severed with surgical scissors. The sutures fixing the combined implants to the sclera were then attached at the 3 and 9 o’clock positions, making sure to set the attachment points in opposing directions and thus achieve a well centered pupil. The combined AI + IOL was then inserted as a folded “sandwich” into the eye through the large opening in the trephined cornea (open-sky-procedure). The combined implants were positioned in the ciliary sulcus and attached with 9.0 or 10.0 polypropylene sutures onto the sclera. In the final step, the corneal graft was firmly sutured with 10.0 nylon sutures.
Follow-up and ocular examination
Main outcome measures were subjective impairment from glare, subjective cosmetic disfigurement, patient satisfaction, and intraocular pressure (IOP). Best-corrected distance visual acuity (BCDVA) was only a secondary outcome measurement because of the recognition that long-term rehabilitation is required.
We measured BCDVA at every examination visit with a Snellen-chart at 6 m distance. Postoperative refraction was determined using objective refractometry.
For the subjective evaluation of the patients’ complaints and the evaluation of the postoperative result, we used a questionnaire in German language, that we designed specifically for use in patients who underwent AI implantation.
The first part of the questionnaire consists of two numerical rating scales from 1 to 10, with 1 standing for low and 10 for high severity, that allows the patient to rate their sensitivity to glare and their discontent with their eye’s appearance.
The second part of the questionnaire is composed of a numerical rating scale from 1 to 10, where 1 represents minimum satisfaction and 10 is maximum satisfaction, so that the patients can indicate their overall satisfaction.
We performed the first part of the assessment preoperatively and postoperatively and the second part of the questionnaire was used at the postoperative follow-up visits.
IOP was measured at every examination using Goldmann applanation tonometry.
Results
Table 1 summarizes the patient characteristics and outcomes of the seven patients included in this study.
Patient characteristics and outcomes.
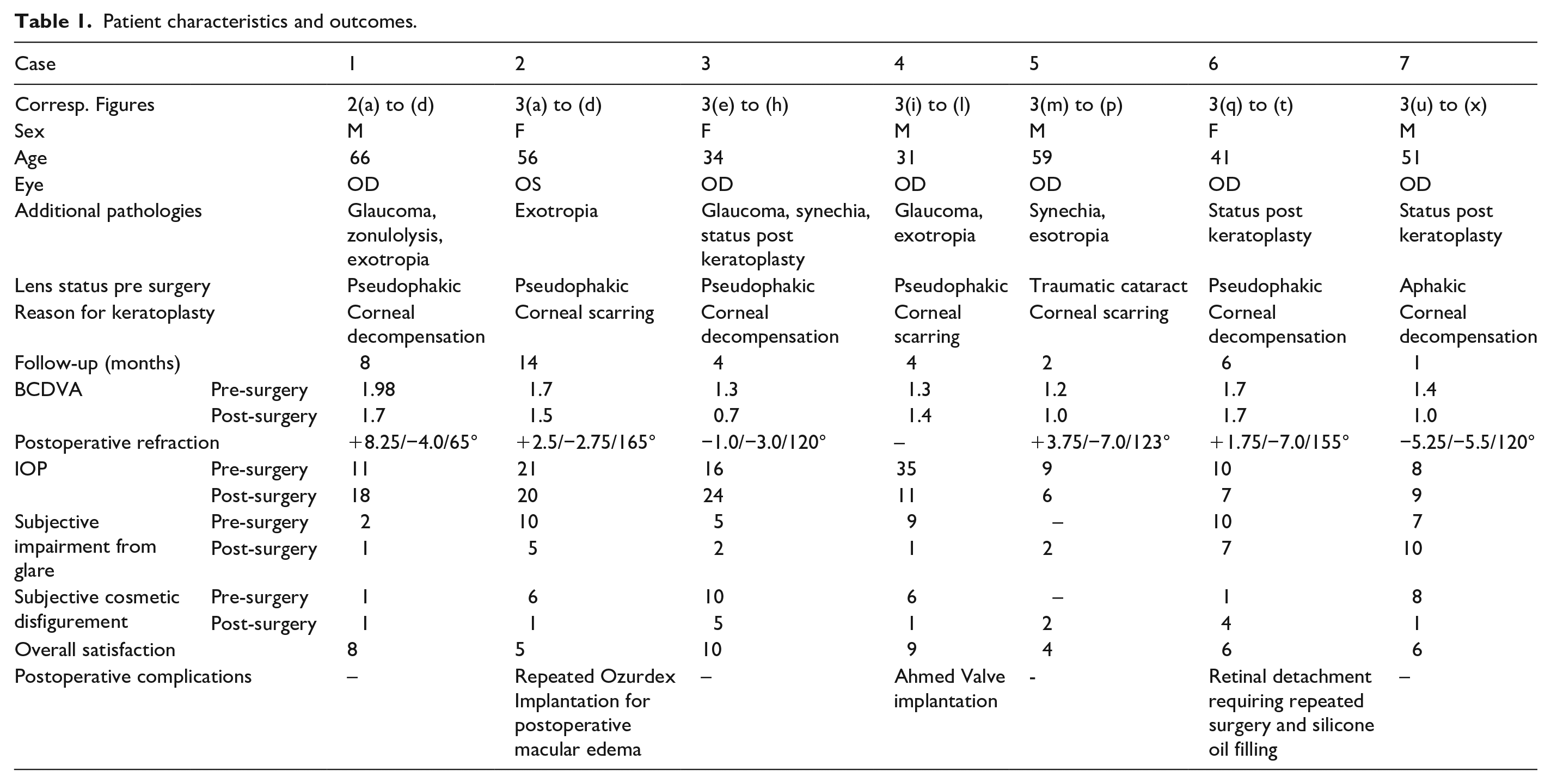
Figure 2(a) and (c) and Figure 3 (left column) show the preoperative findings of all patients.
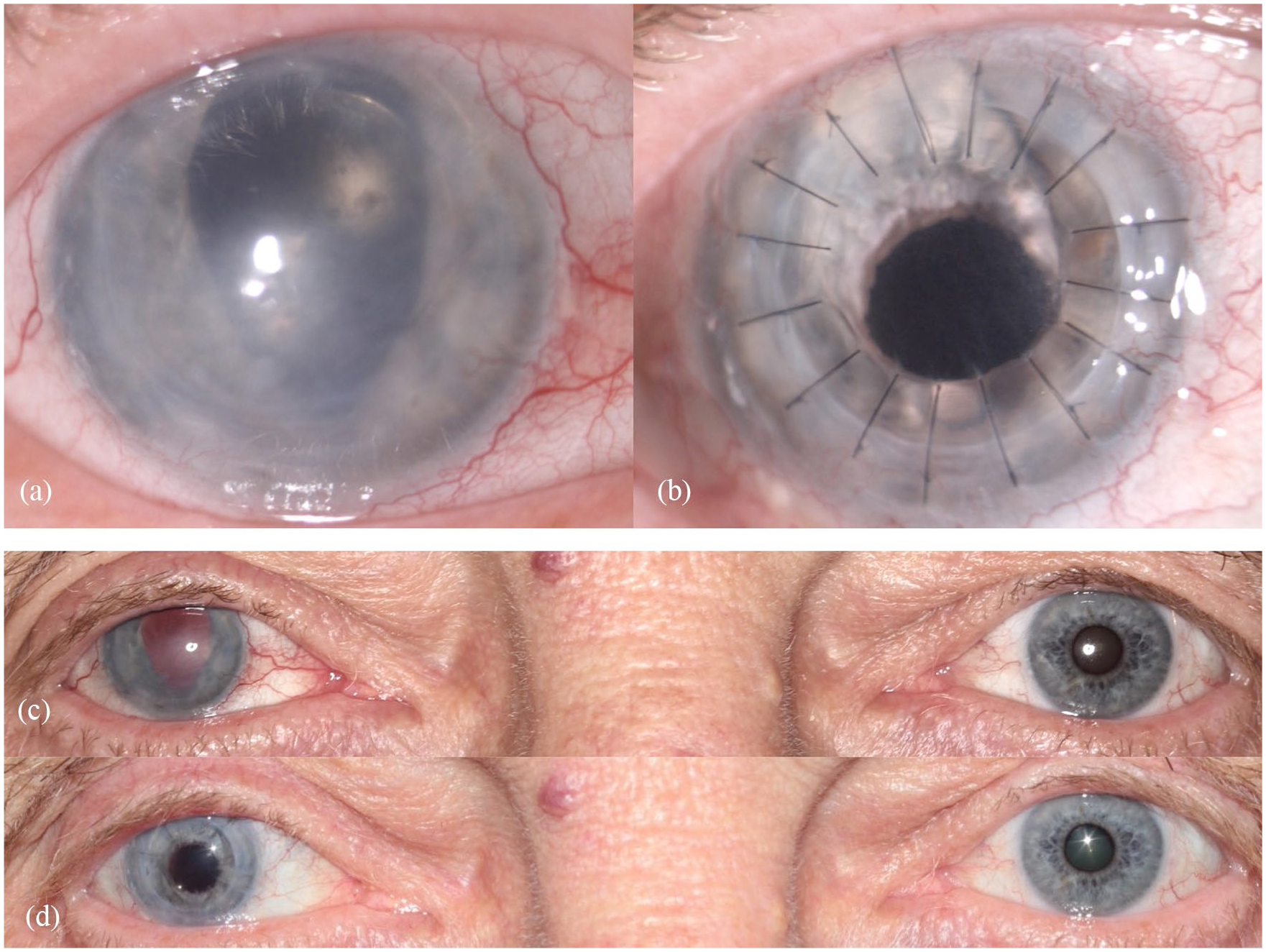
Findings in patient 1: (a) preoperative close-up photograph of the traumatized right eye, (b) postoperative close-up photo after combined surgery (perforating keratoplasty and implantation of an artificial iris and IOL), (c) preoperative binocular photograph, and (d) postoperative binocular photograph.
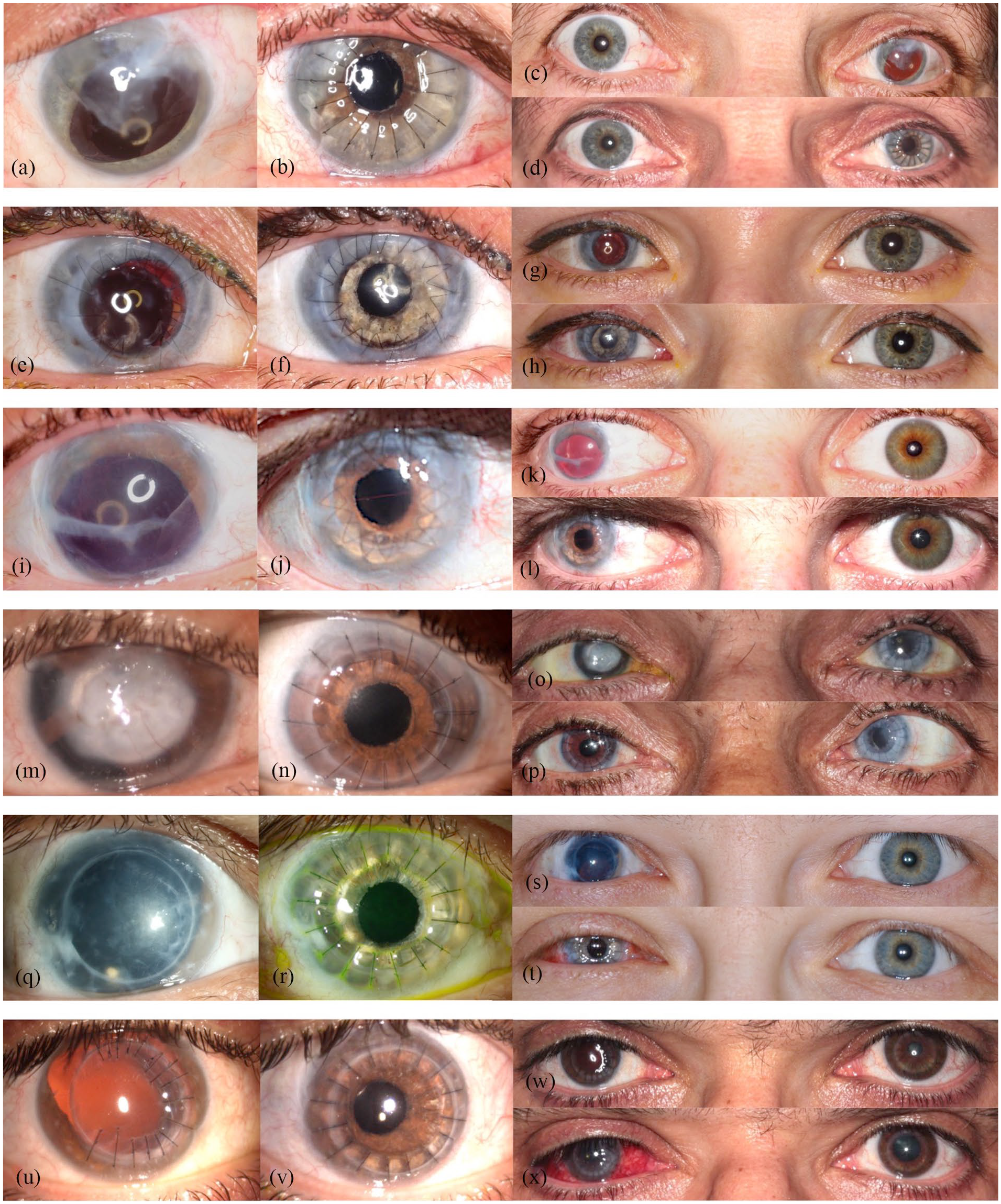
Preoperative close-up photographs of the traumatized eyes (left column) and respective postoperative close-up photographs of the same eyes (middle column) in the remaining six patients. Preoperative binocular photographs (upper sections of the right column) and postoperative binocular photographs (lower sections of the right column) ((a)–(x)).
The mean follow-up time was 5.43 ± 4.24 months, ranging from 1 to 14 months. Mean BCDVA was 1.51 ± 0.26 logMAR preoperatively and 1.29 ± 0.36 logMAR postoperatively. BCDVA did not decrease in any patient. All patients had a considerable postoperative astigmatism. The median value for diopter cylinder was −4.75 D with a range from −2.75 to −7.0 D. Mean IOP was 15.71 ± 8.94 mmHg pre-surgery and 13.57 ± 6.52 mmHg post-surgery. Patients rated subjective visual impairment from glare 7.17 ± 2.91 on average preoperatively and 4.00 ± 3.21 on average postoperatively. Mean subjective cosmetic disfigurement was 5.33 ± 3.35 pre-surgery and 2.14 ± 1.55 post-surgery. The cosmetic result for each patient is shown in Figure 2(b) and (d) and Figure 3. Patients were very satisfied with the cosmetic result of surgery (Figure 2(d)). Mean patient satisfaction on the numerical scale was 6.86 ± 2.03.
There were no significant intraoperative complications. The postoperative complications observed are also listed in Table 1. One patient (Case 2) developed postoperative macular edema and was treated with four consecutive Ozurdex implantations. Another patient (Case 4) with pre-existing glaucoma needed glaucoma surgery for IOP control and was treated with an Ahmed valve implantation. In Case 6, a retinal detachment occurred 2 months postoperatively. The patient was treated with vitrectomy and C2F6 endotamponade but had a recurrent retinal detachment which was treated with silicone oil endotamponade. We observed that the new, iris-lens-diaphragm effectively prevented the migration of silicone oil into the anterior chamber. Dislocation of lens and/or artificial iris was not observed.
Discussion
We report on the outcomes of seven cases of anterior segment reconstruction following traumatic injury. So far, only a few cases of combined keratoplasty, IOL and AI implantation have been published. Forlini et al. 4 reported four cases of surgical correction of posttraumatic aphakia and aniridia out of which only two patients underwent keratoplasty in the same surgery session using a technique similar to the one described in this study. The authors used the same ArtificialIris that we used but different models of IOL, and they found a good visual rehabilitation as well as a satisfying cosmetic result. Yoeruek et al. 5 treated five patients with combined AI + IOL implantation and keratoplasty. They observed an increase in BCDVA, reduction of glare sensitivity and a stable position of the implants in all cases. The postoperative complications included graft failure and IOP elevation in patients with pre-existing glaucoma.
Another approach to treat patients with corneal decompensation, aphakia, and iris defects would be the implantation of a standard iris reconstruction lens in combination with a corneal graft transplantation. An advantage of this approach would be the fact that the iris prosthesis does not have to be sutured to the IOL before the implantation and there is no need to adapt it in size. This method provides good functional results.6–8 However, these lenses are only available in black, for example the Morcher iris reconstruction lens (Morcher GmbH, Stuttgart, Germany), or in a limited number of colors, as for the Ophtec iris reconstruction lens (Ophtec B.V., Groningen, The Netherlands). Therefore, the result cannot be as esthetically pleasing as with the AI that we used, which is individually hand painted using a photograph to match the remaining iris tissue and/or the iris of the fellow eye. Furthermore, the Morcher aniridia implants are made of rigid poly ethyl methacrylate (PEMA) which would not be an ideal material for our technique. However, suturing of a PEMA IOL onto the sclera can also be done.
Penetrating keratoplasty in combination with cataract removal and IOL implantation is known as “triple procedure.” A problem in triple procedures is the refractive outcome. This is mostly due to unpredictable postoperative keratometry.9,10 It is postulated that a delayed sequential approach may be beneficial in terms of achieving the desired refractive outcome. However, contrary to what was expected, the refractive outcome after delayed sequential surgeries is not superior to the outcome after triple procedure and the triple procedure offers a faster visual rehabilitation.11,12 Therefore, the patient can benefit from a single procedure, because this minimizes the cumulative risk of several surgeries and the additional trauma to the corneal transplant.
The technique we used entails several challenges: Depending on the preexisting conditions, the occurrence of certain postoperative complications is more likely in patients with traumatized eyes. Traumas affecting the retina can lead to an increased permeability of the blood-retinal barrier, leading to a higher risk of macular edema.13,14 The risk of postoperative IOP elevation following other surgical procedures such as cataract surgery or vitrectomy is higher in glaucoma patients.15–17 The same applies to anterior segment reconstruction surgery. Furthermore, the varying degree of traumatized tissue makes it necessary to adapt the surgical procedure individually.
Another difficulty in combined AI and IOL implantation and keratoplasty, is the need to minimize the interaction of the AI + IOL combination with the donor graft. The combination needs to be attached in a stable position to prevent contact between it and the corneal graft, to avoid endothelial cell loss.
We used 9.0 or 10.0 polypropylene sutures to attach the AI to the IOL and to attach the implants onto the sclera. Gore-tex sutures proved to be a safe option for scleral fixation of IOLs 18 and can be used alternatively for both purposes.
A number of factors can potentially hamper the achievement of the desired target refraction. Firstly, the effective lens position is difficult to predict. Also, biometry and thus IOL power calculation can be difficult in patients with corneal opacity. Optical biometry is superior to applanation ultrasound measurement, 19 but it requires clear media and the patient being able to fixate: requirements which are often not met in patients with corneal decompensation. Then IOL power calculation has to be based on ultrasound measurement or, in severe cases, even by relying on the biometry of the fellow eye. After corneal transplantation, patients often have a considerable astigmatism. 20 These factors contribute to a possible deviation from the target refraction. If there is a high astigmatism or total refractive error postoperatively, a contact lens can be fitted. 21
AI implantation has proven to be an effective therapeutic option in patients with high glare sensitivity due to traumatic or atraumatic iris defects. An AI implantation significantly reduces the pupillary aperture and can therefore reduce the sensitivity to glare and improve contrast sensitivity. 22 It also provides excellent aesthetic results. 23 Therefore, the patient satisfaction with the functional and cosmetic result is very high,22,24 although in some cases a preoperative strabismus persisted postoperatively. While it is not a primary goal of AI implantation, BCDVA is reported to increase after the implantation.3,25–28 This is most likely due to simultaneous cataract surgery or secondary IOL implantation in patients with aphakia 29 as well as due to the reduction of glare.
A number of reports of complications following AI implantation including decreased BCDVA, IOP elevation, AI dislocation or decentration, corneal decompensation, retinal detachment, and macular edema have been published.25,26,30 The postoperative complications that we observed in our patients included IOP elevation, macular edema, and retinal detachment. We saw a considerable reduction in visual impairment from glare and in subjective cosmetic disfigurement. BCDVA was stable or increased in all patients. We recorded a very high patient satisfaction with the procedure.
When there is residual natural iris tissue, hemorrhage of the remnant iris or darkening of the natural iris tissue can be observed.25,26 Another complication related to remaining natural iris tissue is the residual iris retraction syndrome (RITS), where in some patients with incomplete aniridia, the original pupillary aperture gradually enlarges over time after the AI implantation. This phenomenon is associated with complications like closed angle glaucoma and chronic inflammation. The cause of RITS is unknown. 31
Similar considerations have to be taken into account for our procedure, penetrating keratoplasty combined with implantation of an AI/IOL complex. As previously mentioned, it is known from triple procedure that the refractive outcome is not strongly influenced by choice of a combined surgery versus sequential approach. The advantage of performing all in one single session is to minimize trauma to the corneal transplant. But another advantage is that the AI in combination with the IOL can be smoothly implanted through the large aperture in the cornea (open sky surgery). Thus a sclero-corneal tunnel incision is avoided and the patient need not return for a second surgery session.
One limitation of this study is the range in follow-up time. For one patient results can only be reported 1 month postoperatively. Longer Follow-up is required to evaluate whether the nature of post-operative complications will change over time.
Another limitation is the lack of a standardized questionnaire to evaluate the subjective impairment from glare and subjective cosmetic disfigurement. We used a questionnaire developed specifically for patients who underwent AI implantation in German language to offer our patients a questionnaire that is easy to understand and tailored to the key issues according to our clinical experience.
Our procedure allows achievement of a functional and aesthetically appealing good result for the patient. The use of an artificial iris, specifically designed for the patient, together with soft IOLs allow an individualized type of anterior segment reconstruction. We therefore recommend this single surgery technique as most feasible in the reconstruction of traumatized eyes with corneal scaring, large iris defects, and aphakia.
Footnotes
Acknowledgements
We would like to thank Mr. Donald J Munro for review of this report.
Author contributions
C.M. contributed to study conception and design, analysis and interpretation of data, drafting the manuscript, and critical revision. I.B. contributed to analysis and interpretation of data and drafting of the manuscript. J.S. contributed to analysis and interpretation of data and critical revision. R.K. contributed to study conception and design, analysis and interpretation of data, drafting of the manuscript and critical revision. All authors contributed to the final manuscript preparation and have approved the same.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: C.M. received travel grants and lecture fees from HumanOptics. I.D.B., J.S., and R.K. declare that they have no competing interests.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Department of Ophthalmology of the University of Heidelberg was supported by the Klaus Tschira Foundation.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
