Abstract
Purpose:
To report outcomes of a sutureless dehydrated amniotic membrane for persistent epithelial defects (PED).
Methods:
This retrospective study included consecutive patients with a PED (⩾14 days) treated with a sutureless dehydrated amniotic membrane and bandage contact lens (BCL). Included were patients with an epithelial defect that did not respond to treatment with a BCL. Excluded were patients with a follow-up time of less than 3 months.
Results:
Nine eyes of eight patients with a mean age of 54.6 ± 10.9 years (range 38–73 years) were included in this study. The main etiology of the PED was limbal stem cell deficiency (n = 5/9) due to Stevens-Johnson Syndrome (n = 2/5), glaucoma procedures (n = 1/5), graft-versus-host disease (n = 1/5) and severe allergic reaction (n = 1/5). Additional etiologies included neurotrophic cornea (n = 2/9), post keratoplasty and severe dry eye disease (n = 2/9). Time from PED presentation to amnion treatment was 65.9 ± 60.6 days (range 15–189 days) with the area of the PED being 11.0 ± 12.2 mm2 (range 1.0–36.0 mm2). The amnion was absorbed within 2 weeks in 100% of the cases. Following insertion of the amnion, resolution of the PED was achieved in 8/9 eyes (89%) without the need for additional interventions within 17.8 ± 9.6 days (range 7–35 days). LogMAR BCVA improved from 0.94 ± 0.88 to 0.37 ± 0.25 (p = 0.036) with no complications or recurrences recorded.
Conclusions:
Sutureless dehydrated amniotic membrane achieved resolution of PEDs secondary to various etiologies in 89% of eyes with a significant improvement in vision demonstrated. Further studies are needed to assess long term safety and effectiveness.
Introduction
The corneal epithelium acts as a protective barrier to infectious agents and maintains a smooth optical surface. 1 An insult to the epithelium leading to a defect (corneal erosion) predisposes the cornea to infection and reduces quality of vision. In normal conditions after an insult to the epithelium leads to a defect (corneal erosion), the epithelial layer undergoes an active repair process involving regulated growth factors, cellular signaling, proliferation, migration, and extracellular matrix remodeling. 2
Persistent corneal epithelial defects (PEDs) result from the failure of re-epithelialization and closure within 10–14 days after a corneal injury despite standard treatment. 3 Potential etiologies include defective epithelial adhesion, limbal stem cell deficiency (LSCD), inflammation, neurotrophic components, and idiopathic/hereditary disorders. 1 Management involves a stepwise approach depending on etiology. Non-surgical modalities include optimization of the ocular surface, bandage contact lenses (BCLs), blood derived products (e.g. autologous serum and platelet-rich plasma), punctal plugs, scleral contact lenses, and cenegermin. 2 Subsequent surgical treatment options include epithelial debridement, amniotic membrane transplant (AMT) and corneal stem cell transplant. 4
Amniotic membranes contain a plethora of growth factors that facilitate wound healing, provide a scaffold for re-epithelialization, and have anti-inflammatory properties 5 and have proven to be effective for the treatment of PEDs.6,7 Commercially available amniotic membrane implantation can be performed without the need for sutures. Most reports concerning commercial amniotic membranes have reported on the cryopreserved ProKera (Bio-Tissue, Miami, FL) with encouraging results for the treatment of bacterial keratitis, 8 alkali burns, 9 partial LSCD, 10 acute toxic epidermal necrolysis, 11 as well as PEDs. 12
More recently, commercially available freeze and vacuum dehydrated amnion tissue were introduced, and these allow for ease of storage and transportation at room temperature. 13 To the best of our knowledge, this is the first study to assess outcomes of an in-office sutureless dehydrated amniotic membrane for PEDs.
Methods
This study was conducted in compliance with the tenets of the Declaration of Helsinki and received Research Ethics Board approval from University Health Network (Toronto Western Hospital, Toronto, Canada).
Study participants
This is a retrospective study conducted by means of a chart review of consecutive patients who were treated in office with a sutureless dehydrated amniotic membrane for persistent epithelial defects from July 2019 to December 2019 at one of two tertiary cornea clinics, Toronto, Canada. Included were patients that did not demonstrate sufficient resolution of the ED following optimization of the ocular surface and treatment with a BCL. Optimization of the ocular surface in our clinic follows the guidelines of the Global Consensus of the Management of Limbal Stem Cell Deficiency. 14 It includes addressing any underlying eyelid and conjunctival comorbidities, reduction of epithelial toxicity from topical medications, reduction of inflammation (low dose cyclosporine, tacrolimus, lifitegrast, or low-potency corticosteroids), optimization of tear aqueous function (preservative-free artificial tears and/or autologous serum) and treating underlying meibomian gland dysfunction (lid hygiene, topical macrolides, and omega-3 fatty acid supplements). Excluded were patients with a follow-up time of less than 3 months.
Data collection
Preoperative demographics that were recorded included gender, age, and laterality. Additional baseline data included best-corrected visual acuity (BCVA), etiology of PED, systemic comorbidities, PED size (area in mm2 measured with fluorescein staining and cobalt flue filter), PED time to amnion (time from PED presentation to amnion treatment), time to PED resolution following amnion treatment, follow-up time, and BCVA following resolution. Recurrences of an ED, complications, or adverse events occurring during the follow-up time were recorded.
Amniotic membrane
The sutureless dehydrated amniotic membrane BioDOPTIX (Labtician Ophthalmics, Oakville, Ontario, Canada) was utilized and stored at room temperature prior to usage. Patients received a drop of proparacaine hydrochloride 0.5% (Alcaine, Alcon, Canada). After 5 min, an eyelid speculum was placed and cellulose sponges were used to dry the cornea. In our experience, the corneal surface must be dry in order for the amniotic membrane to properly adhere to its surface. The 9.0 mm circular amniotic membrane disc was removed from the sterile packaging with curved non-toothed forceps and placed over the center of the cornea covering the entire epithelial defect. At this stage, care must be taken as the amnion tends to fold on itself once placed on the cornea. To prevent the membrane from folding on itself a second non-toothed curved forceps may be used to smooth the amnion onto the corneal surface immediately after placement. After 2 to 5 min, a sterile BCL was dried with cellulose eye sponges and placed over the amnion. Adequate positioning of the amnion and BCL was confirmed at the slit lamp 5 min later (Figure 1).
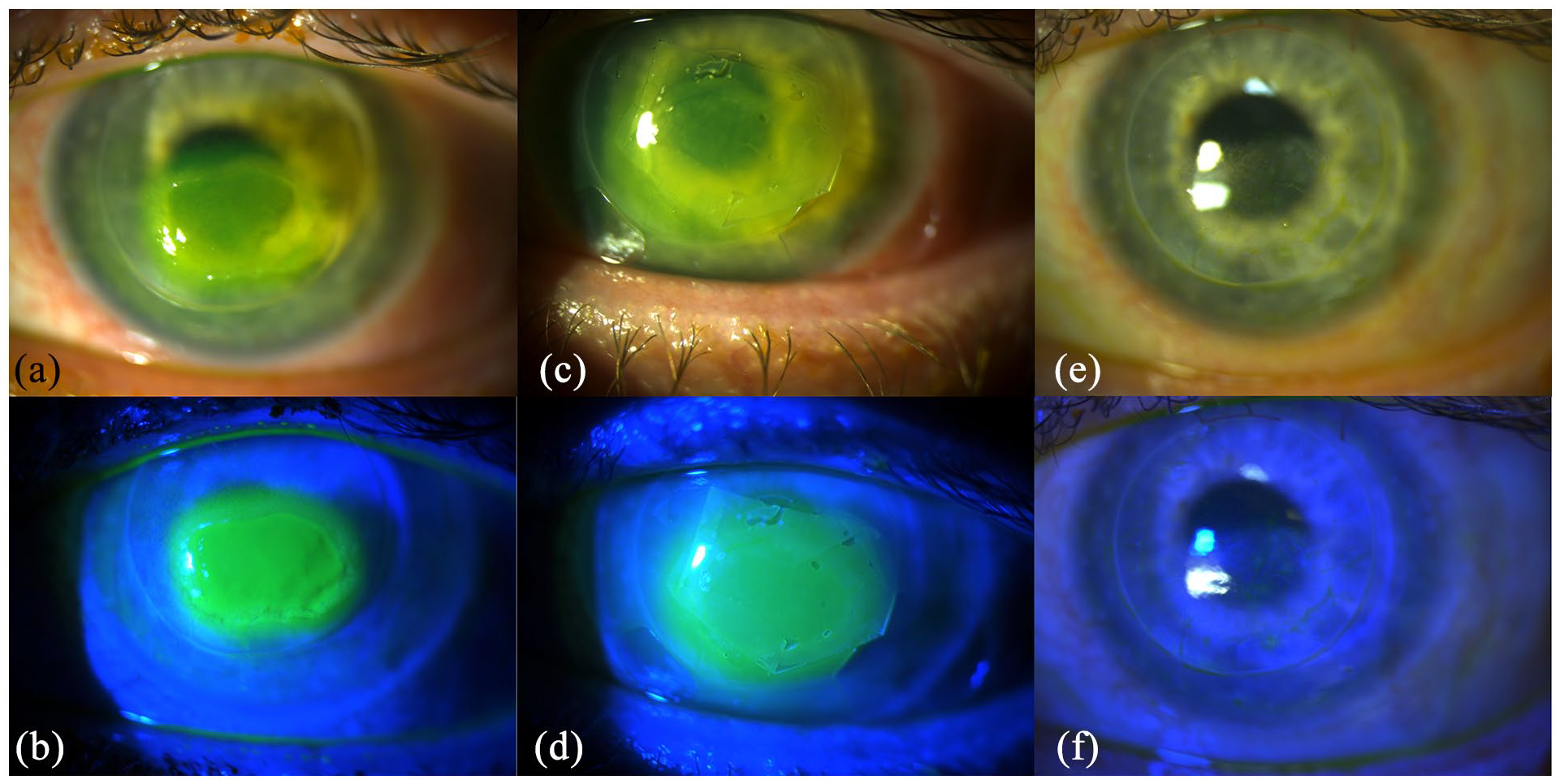
Slit lamp images of a patient before (a, b), with (c, d) and following absorption of (e, f) the amniotic membrane. It is worth noting the scrolled-up edges (c, d) of the amniotic membrane disc may be avoided if a meticulously dry ocular surface is maintained prior to placement of the amniotic membrane. Drying the bandage contact lens with a cellulose spear before application is also critical to avoid slippage and folding of the amniotic membrane. Nonetheless, this patient’s persistent epithelial defect healed completely following the absorption of the membrane.
Patient follow-up
Following insertion of the amniotic membrane, patients were followed every 1–3 weeks until resolution of the PED was achieved. At every visit BCVA and area of PED was recorded. If following absorption of the amnion, adequate but not complete resolution of the PED was achieved, then the BCL treatment was maintained.
Study outcomes
Primary outcomes included PED resolution rate, time to PED resolution, change in BCVA and serious adverse events.
Statistical analysis
Data were analyzed with the Minitab Software, version 17 (Minitab Inc, State College, PA). For the comparison of continuous data, before, and after amnion membrane, the Wilcoxon Signed Rank test was used. In all analyses, a two-sided p value <0.05 was considered statistically significant. All presented means are accompanied by their respective standard deviations.
Results
Nine eyes of eight patients with a mean age of 54.6 ± 10.9 years (range 38–73 years) of which 50% (n = 4) were of female gender were included in this study. There was a mean follow-up time of 164.9 ± 64.1 days (range 90.0–265 days). Table 1 depicts the demographic and clinical information of the patients included in this study.
List of patients that received sutureless dehydrated amniotic membrane for persistent epithelial defects (PEDs).

PED time: time from epithelial defect to amnion treatment, PED Heal Time: time it took for ED to heal after placing amnion, LSCD: limbal stem cell deficiency, DED: dry eye disease, SJS: Stevens-Johnson Syndrome, BCVA: best-corrected visual acuity, HM: hand motion.
PED etiologies
The main etiology of the PED was limbal stem cell deficiency (n = 5/9) due to Stevens-Johnson Syndrome (n = 2/5), glaucoma procedures (n = 1/5), graft-versus-host disease (n = 1/5) and severe allergic reaction (n = 1/5). Additional etiologies included neurotrophic cornea post keratoplasty (n = 2/9) and severe dry eye disease (n = 2/9).
PED duration
Time from PED presentation to amnion treatment (PED Duration) was 65.9 ± 60.6 days (range 15–189 days) with the area of the PED being 11.0 ± 12.2 mm2 (range 1.0–36.0 mm2). The amnion was absorbed within 2 weeks in 100% of the cases. Following insertion of the amnion, resolution of the PED was achieved in 8/9 eyes (89%) without the need for additional interventions within 17.8 ± 9.6 days (range 7–35 days). For the 8/9 eyes where complete resolution was achieved, the PED resolution time was significantly lower than the PED duration prior to amniotic membrane application (17.8 ± 9.6 vs 70.4 ± 63.2, p = 0.04), suggesting the positive impact the amniotic membrane had on resolution of the PED. The one eye of one patient that did not heal is currently waiting to undergo limbal stem cell transplant surgery.
Visual acuity
Compared to baseline, following resolution of the PED, the BCVA significantly improved from 0.94 ± 0.88 logMAR (20/174 Snellen) to 0.37 ± 0.25 logMAR (20/47 Snellen) (p = 0.036).
Complications and adverse events
There were no complications during the placement of the amniotic membrane or afterwards during follow-up. None of the patients reported discomfort with the amnion and BCL placed on the ocular surface. There were no recurrences of PEDs during the follow-up time.
Discussion
This study assessed the outcomes of an in-office sutureless dehydrated amniotic membrane for PEDs. Overall, resolution of PEDs secondary to various etiologies was achieved in 89% of eyes with a significant improvement in vision. To the best of our knowledge, this is the first study to report outcomes of an in-office sutureless dehydrated amniotic membrane for PEDs.
Amniotic membrane transplant has been documented as an effective and safe treatment for PEDs.5,15,16 The aforementioned studies have employed amnion grafts that require a more invasive and time-consuming procedure involving either sutures or fibrin glue. As such, in-office, self-retained sutureless amnion grafts are of interest as they are less invasive and less time consuming. However, a paucity of data exists regarding the efficacy of commercial amnion grafts for the treatment of PEDs. In 2009, Pachigolla et al. 12 reported on the outcome of 20 eyes treated with the ProKera of which only two were for PEDs. They reported that after an average duration of 25 days, 25% of the eyes had residual epithelial defects. In addition, they reported that 30% of patients reported eye pain or headache. In their ProKera study, one patient requested to have the device removed due to intolerable pain. Pachigolla et al. hypothesized that the conformer ring surrounding the membrane was the cause of discomfort. This hypothesis was confirmed in a subsequent study. 17 Suri et al. 17 reported on the short-term results of ProKera for non-healing corneal ulcers (n = 9) and neurotrophic cornea (n = 11) and found that 44% and 64%, respectively, had complete or partial success. They also reported that 17.1% of patients reported discomfort form the device. 17 The current study assessed the BioDOPTIX amnion graft in patients with PEDs and after an average duration of 17.8 days, 89% of the eyes achieved resolution of PEDs with none of the patients experiencing discomfort from the graft. We speculate that the use of a simple and readily available BCL to retain the BioDOPTIX amnion graft on the ocular surface and the lack of a need for a conformer ring led to better patient comfort.
In the current study, there was an improvement in logMAR BCVA from 0.94 ± 0.88 to 0.37 ± 0.25 (p = 0.036). This is likely due to the fact that in many of the cases the PED involved the visual axis and therefore subsequent resolution of the PED led to improvement in vision. This is supported by the findings of Pachigolla et al. 12 that reported that with the ProKera there was an improvement in logMAR VA from 1.47 to 1.05 (p = 0.007) when treating various ocular surface disorders. They did not provide a sub-analysis of results for patients with PEDs (n = 2). In the current study, by 2 weeks all patients demonstrated complete absorption of the BioDOPTIX. This is considerably less time for absorption then that reported for the ProKera which has been reported to take up to 125 days to absorb. 12 This may be due to the difference in thickness between the two amnion grafts, the ProKera being 100–200 μm and the BioDOPTIX 40 μm thick. Therefore, a BioDOPTIX may be more appropriate for a monocular patient whereas the ProKera may be more appropriate for indications where a thicker amnion is required (e.g. highly inflammatory conditions).
There is a question of cost regarding commercial amniotic membranes. Prices range from 300 CAD to 900 CAD per implant depending on the manufacturer, distributor, and geographic location. Furthermore, these devices may not be covered by a patient’s health plan. These price ranges are not very far off from those of 3–6 months supply of autologous serum and several scales more affordable than the recently reported potent cenegermin. In any event all of these different treatment options should be weighed against the economic capability of a patient.
This study has several limitations, first of which is its small sample size and retrospective nature. However, the preliminary results of this pilot study are encouraging. An additional limitation is the lack of assessment of long-term safety; however, the overall safety of amniotic membranes has been well documented and we do not anticipate long term adverse events with this modality. Last, this was a single arm study with no control group, yet all of these eyes did not achieve resolution following standard treatment with a BCL only. Given the aforementioned, further, larger, prospective studies evaluating long term outcomes with a control group are warranted.
Nevertheless, to the best of our knowledge this is the first study to report outcomes of an in-office sutureless dehydrated amniotic membrane for PEDs. The BioDOPTIX achieves resolution of PEDs secondary to various etiologies in an overwhelming majority of eyes with a significant improvement in vision. No patients reported discomfort with the graft as it is held in place with a readily available BCL. The graft is absorbed within 2 weeks with no complications recorded in this short-term study.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Michael Mimouni is a cornea fellow funded by the Marta and Owen Boris Endowed Chair in Cornea and Stem Cell Research as well as the Gerald Schwartz and Heather Reisman Foundation.
