Abstract
Aims:
To present an authoritative, universal, easy-to-use morphologic classification of diabetic maculopathy based on spectral domain optical coherence tomography.
Methods:
The first draft of the project was developed based on previously published classifications and a literature search regarding the spectral domain optical coherence tomography quantitative and qualitative features of diabetic maculopathy. This draft was sent to an international panel of retina experts for a first revision. The panel met at the European School for Advanced Studies in Ophthalmology headquarters in Lugano, Switzerland, and elaborated the final document.
Results:
Seven tomographic qualitative and quantitative features are taken into account and scored according to a grading protocol termed TCED-HFV, which includes foveal thickness (T), corresponding to either central subfoveal thickness or macular volume, intraretinal cysts (C), the ellipsoid zone (EZ) and/or external limiting membrane (ELM) status (E), presence of disorganization of the inner retinal layers (D), number of hyperreflective foci (H), subfoveal fluid (F), and vitreoretinal relationship (V). Four different stages of the disease, that is, early diabetic maculopathy, advanced diabetic maculopathy, severe diabetic maculopathy, and atrophic maculopathy, are based on the first four variables, namely the T, C, E, and D. The different stages reflect progressive severity of the disease.
Conclusion:
A novel grading system of diabetic maculopathy is hereby proposed. The classification is aimed at providing a simple, direct, objective tool to classify diabetic maculopathy (irrespective to the treatment status) even for non-retinal experts and can be used for therapeutic and prognostic purposes, as well as for correct evaluation and reproducibility of clinical investigations.
Keywords
Introduction
Diabetes, currently affects 415 million people worldwide, and the number is projected to increase to 750 million by 2030. 1 The prevalence of diabetic retinopathy and diabetic macular edema (DME) is expected to increase as well. 2 DME represents the main cause of visual impairment in diabetic patients, and approximately 50% of patients with DME lose two or more lines of visual acuity within 2 years if left untreated. 3
At present, fluorescein angiography (FA) and spectral domain optical coherence tomography (SD-OCT) represent the techniques of choice for assessment and follow-up of diabetic maculopathy (DM). 4 In particular, SD-OCT offers both quantitative and qualitative information in a non-invasive and repeatable way,5,6 and provides the central subfoveal thickness (CST), which has been widely used both in randomized clinical trials (RCTs) and in clinical practice. 7 The state of the ellipsoid zone (EZ) and the external limiting membrane (ELM), the presence of hyperreflective intraretinal foci (HF), 8 the features of the vitreoretinal interface, 9 the presence of subfoveal fluid (SF), 10 the size of intraretinal cysts, 10 and the occurrence of disorganization of the inner retinal layers (DRIL), alone or in different combinations have been used to categorize DME. 11
Despite attempts to classify DME according to its location (center- vs non-center-involving), 12 extent (focal vs diffuse), 13 or nature (vasogenic vs non-vasogenic),14,15 a morphologic categorization of the disease, including all its relevant features visible on SD-OCT is missing.
An international panel of experts elaborated and here proposes a SD-OCT-based classification, centered on standard figures, which considers specific morphologic features and quantitative indices of the entire spectrum of macular involvement in diabetic retinopathy, globally referred as DM.
Methods
A Pubmed-based research was carried out by the first author (G.P.) using the keywords “diabetic macular edema,” “diabetic maculopathy,” “classification,” and “optical coherence tomography.” All reports published in English up to January 2019, including those available online prior to publication, were reviewed. The project was based on previously conducted studies and did not contain any direct involvement of human or animal participants; therefore, ethical approval was not necessary.
The first draft of this DM classification was elaborated and sent for review to an international panel of experts with recognized and long-standing experience in medical retina and SD-OCT technology. The document was modified according to the experts’ comments and the panel was invited to participate to a consensus meeting, held at the European School of Advanced Studies in Ophthalmology (ESASO) headquarters in Lugano (Switzerland) on 30 March 2019, to discuss and finalize the definitive classification.
Method of acquisition
The classification was based on structural SD-OCT figures, meant to be used as references for the grading system.
All the SD-OCT were acquired using a high-resolution scan (length of 9 mm or more) with the modalities “raster scan” and/or “radial scan” centered to the foveola. The grade was judged from the scan crossing the foveola with the worse morphology or, in case of media opacities, with the best visibility of the retinal structures. In case of retinal signal “shadowing,” due to vitreous hemorrhages, hard exudates, or microaneurysms, the grade was judged from the scan crossing the foveola with the least shadowing.
Quantitative measurement
Normative retinal Early Treatment Diabetic Retinopathy Study (ETDRS) maps available in every SD-OCT machine were used as reference and provided quantitative measurement of the CST and the macular volume (MV) in the 500-μm-radius ETDRS ring centered to the foveola.
The images shown at the consensus conference and reported in this article were obtained through the Spectralis® SD-OCT (Heidelberg Engineering, Vista, CA, USA), using the six-radial scans, centered at the fovea at equally spaced angular orientations. The in-built Spectralis® software automatically provided the CST and MV for each of the nine subfields of the inner, intermediate, and outer rings (with a diameter of 1, 2.22, and 3.45 mm, respectively); normal range for the CST in the 1-mm-diameter ring was considered 225–315 µm. 16 Conversely, the MV normal range in the 1-mm-diameter ring was considered 0.17–0.26 mm3. 17
Qualitative parameters
Morphologic parameters on the SD-OCT scans were considered an integral, essential part of each stage; they included: (1) the size of the intraretinal cysts; (2) the visibility of ELM at the fovea; (3) the visibility of EZ at the fovea; (4) the presence of DRIL; (5) the presence of SF; (6) the presence and the number of HF; and (7) the vitreoretinal relationship.
Intraretinal cysts were defined as round, minimally reflective spaces within the neurosensory retina, located in the outer nuclear layer, inner nuclear layer, or ganglion cell layer. Cysts’ size was graded using the figures as references, considering the dimension of the largest intraretinal cyst identifiable on the scan.
Absence of the EZ and/or the ELM was defined as a complete loss of foveal reflectivity at this level, identified as the first and the second hyperreflective bands of the four outermost layers on OCT, respectively. These layers were categorized as disrupted if they were not perfectly discernible, but still partially visible in the fovea; if EZ was non-gradable (N/G) due to the presence of SF, ELM only was considered.
DRIL was defined as the loss of clear demarcation between the ganglion cell–inner plexiform layer complex, the inner nuclear layer, and the outer plexiform layer in the central fovea. SF was identified by subfoveal neurosensory hyporeflective detachment, due to fluid accumulation between the retina and the retinal pigment epithelium line; SF could be isolated or associated with any stage of DM. Patients were classified into two dichotomic groups according to the presence of DRIL and SF (yes/no). HFs were manually counted in each scan. Only HFs with the following morphologic characteristics were evaluated, in order to exclude hard exudates and microaneurysms from the analysis: (1) reflectivity similar to that of nerve fiber layer; (2) absence of back-shadowing; and (3) <30 μm diameter. All scans were divided into two groups (high HF/low HF) based on the average number of HF, using the arbitrary number of 30 as a cut-off value.
The vitreomacular relationship was classified according to a simplified version of the International Vitreomacular Traction Study Group classification. 18 Complete posterior vitreous detachment (PVD) was defined as no residual vitreoretinal adhesion, demonstrated by a scan including the optic disk; if the optic disk was not visualized, the scan was labeled as absence of any visible adhesion or traction between vitreous cortex and retina, as complete PVD was not evident. In case of single or multiple points of residual vitreoretinal adhesion, without influence on retinal profile (no traction), the scan was labeled as incomplete posterior vitreous detachment (IVD). Vitreomacular traction (VMT) was identified as residual macular vitreous attachment exerting antero-posterior traction, while epiretinal membrane (ERM) was defined as evidence of epiretinal tissue adhering to the macular surface, irrespective of the effect on the underlying retina.
Results
Preliminary definitions
DM included all the phenotypes of macular involvement in diabetic retinopathy irrespective to the presence of macular thickening. DME was identified by accumulation of intra and/or subretinal fluid in the macular area associated with retinal thickening on OCT.
The classification was designed for center-involving edema, that is, retinal thickening and/or intraretinal cysts and/or neurosensory retinal detachment involving the first ring of the ETDRS macular map. Furthermore, it was valid for both naïve and treated eyes, irrespective of the nature of the treatment. Prior focal or grid macular laser was also acceptable, as the status of the EZ/ELM was assessed only subfoveally (and laser spots were presumed not to be localized under the fovea).
SD-OCT grading system of diabetic maculopathy
Seven parameters were considered: foveal thickness (T), corresponding to either CST or MV, the size of intraretinal cysts (C), the EZ and/or ELM status (E), DRIL (D), hyperreflective foci (H), subfoveal fluid (F), and vitreoretinal relationship (V) (Table 1). The acronym can be easily memorized as “To Classify Edema Discerning Hidden Functional Variables.”
Spectral domain optical coherence tomography grading system of diabetic maculopathy.

EZ: ellipsoid zone; ELM: external limiting membrane; DRIL: disorganization of the inner retinal layers; IVD: incomplete posterior vitreous detachment; PVD: posterior vitreous detachment; VMT: vitreomacular traction; ERM: epiretinal membrane.
SD-OCT Staging of diabetic maculopathy
Four different stages of disease, that is, early DM (Figure 1), advanced DM (Figure 2), severe DM (Figure 3), and atrophic maculopathy (Figure 4), were discerned based on the first four variables (Table 2).
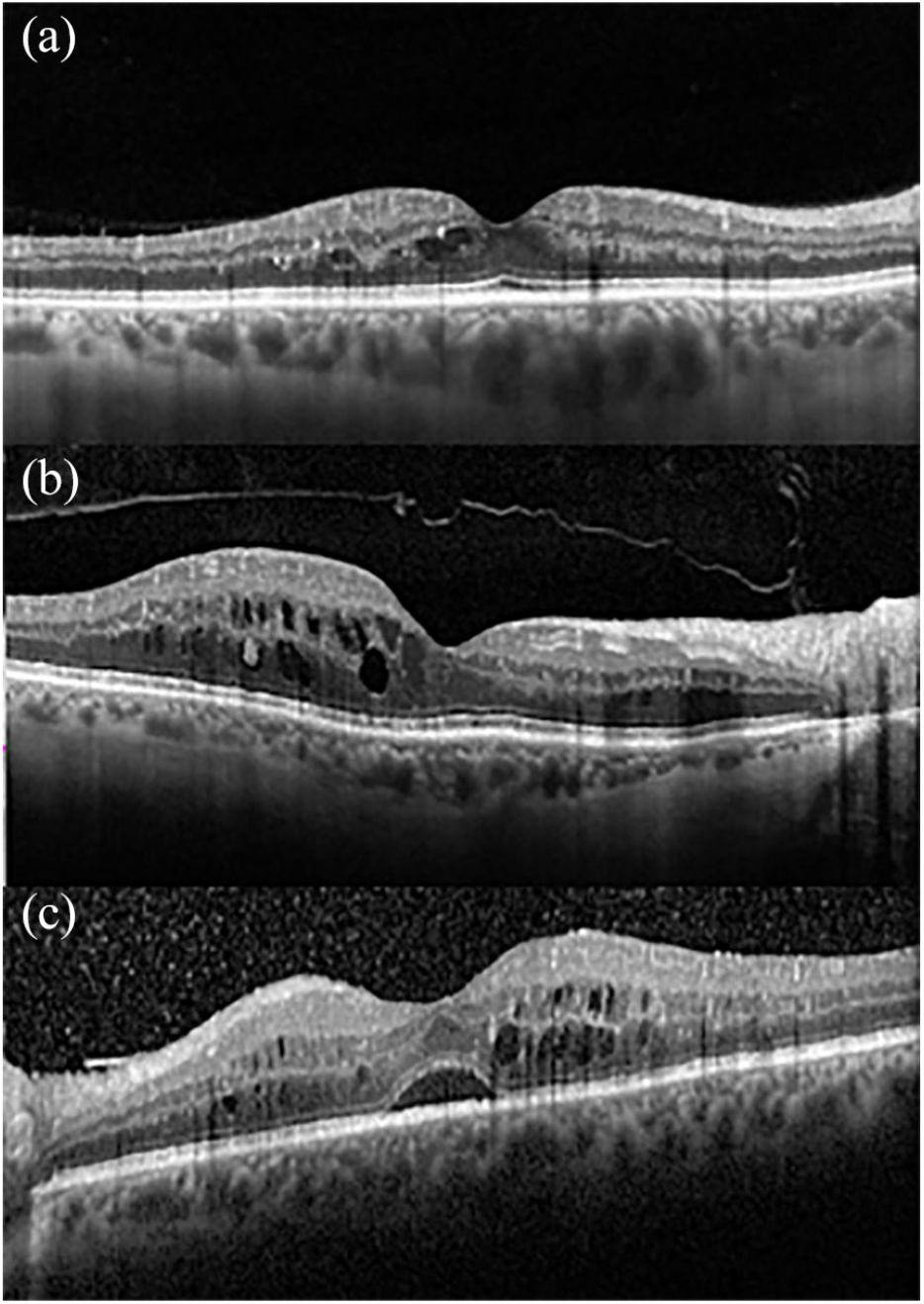
Early diabetic maculopathy.
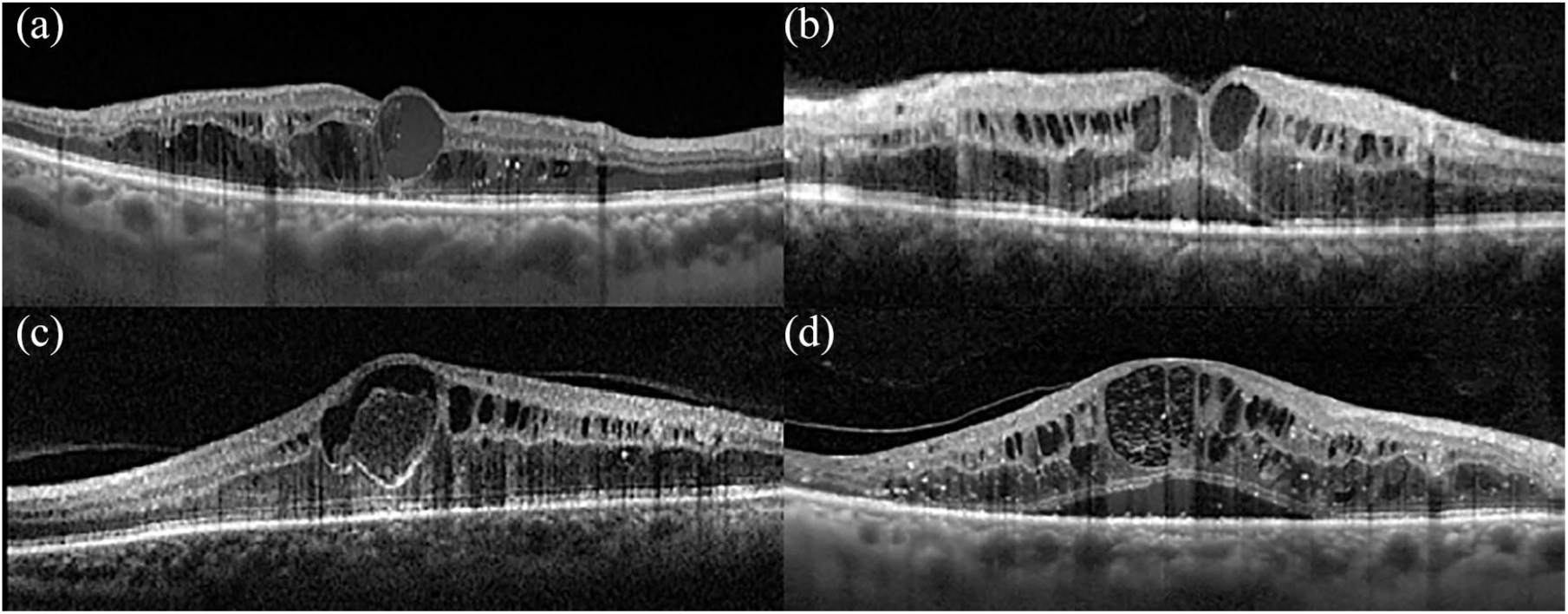
Advanced diabetic maculopathy.
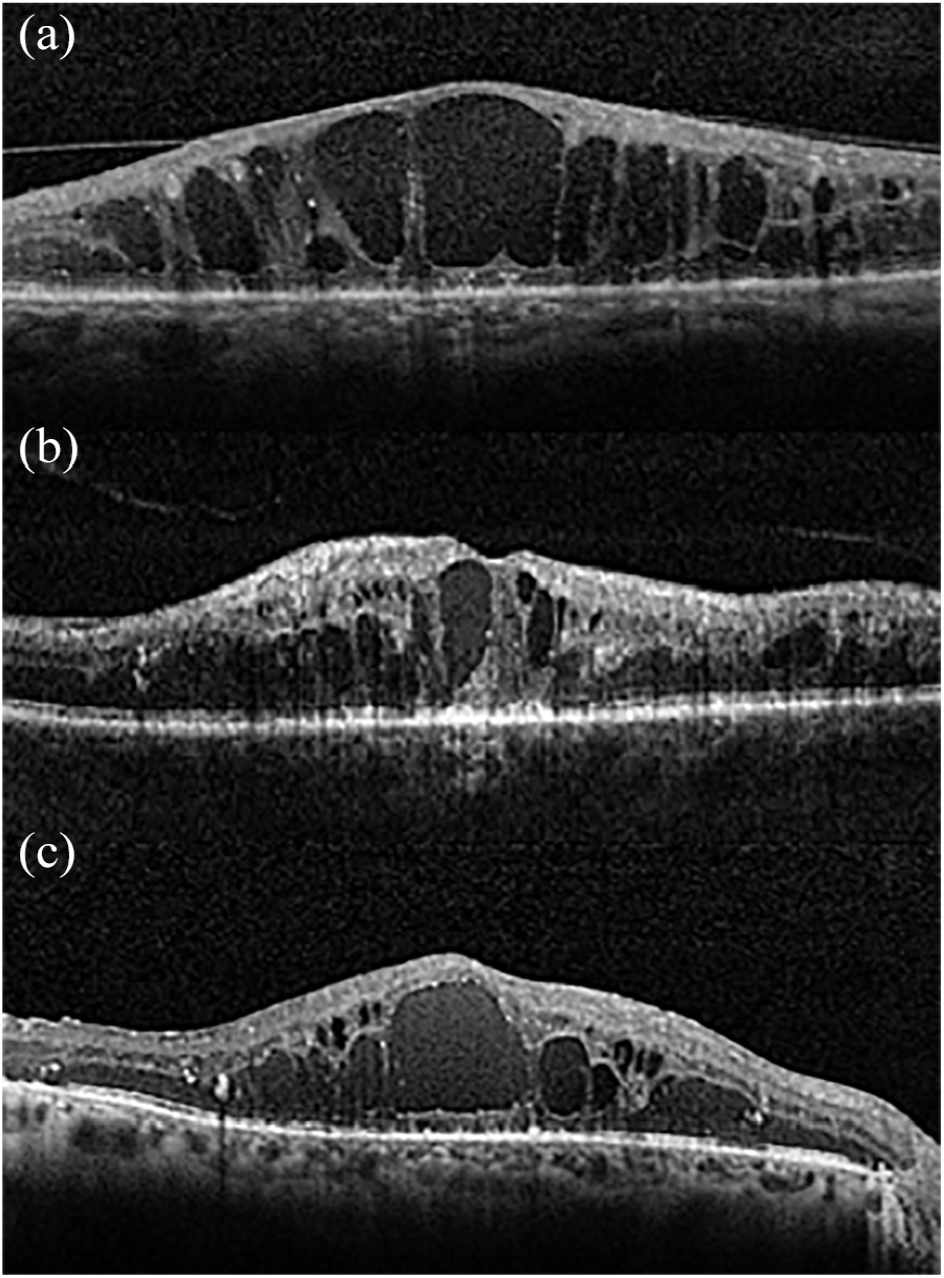
Severe diabetic maculopathy.
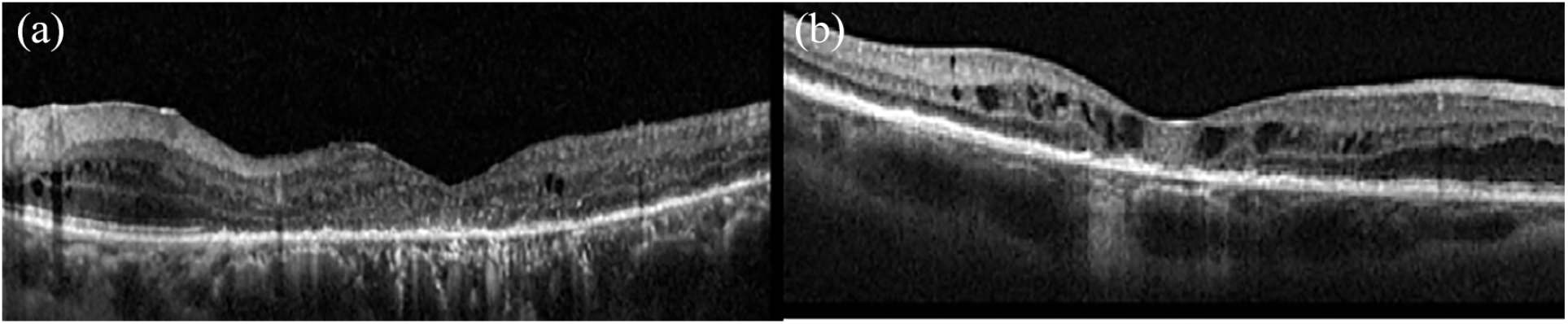
Atrophic diabetic maculopathy.
Staging of diabetic maculopathy (DM) according to the different combinations of the first four variables: thickening (T), cysts (C), EZ and ELM status (E), and DRIL (D).
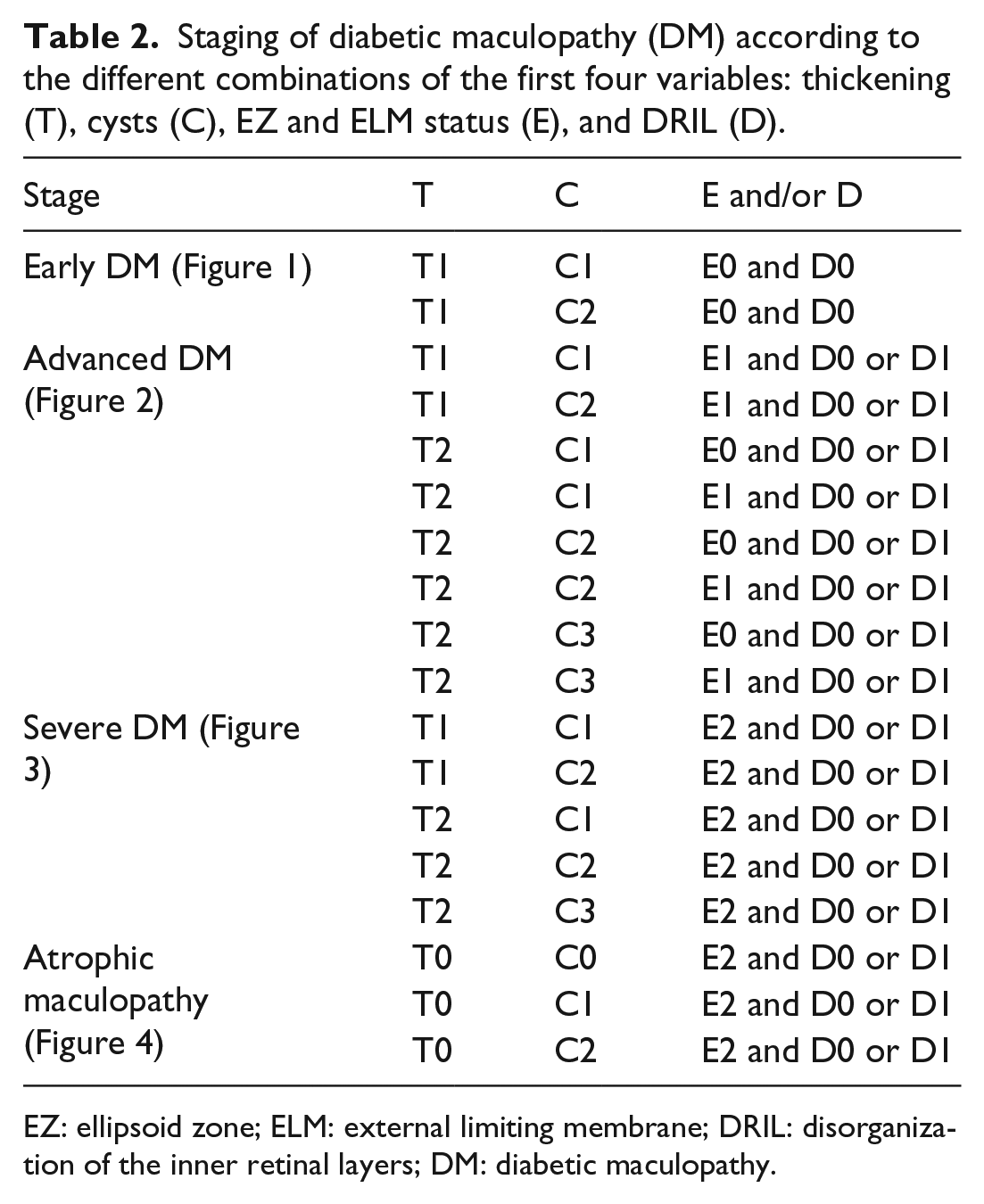
EZ: ellipsoid zone; ELM: external limiting membrane; DRIL: disorganization of the inner retinal layers; DM: diabetic maculopathy.
Each stage was additionally described according to the presence of the variables described in Table 3 (Figure 5).
Adjunctive features of each stage of diabetic maculopathy.
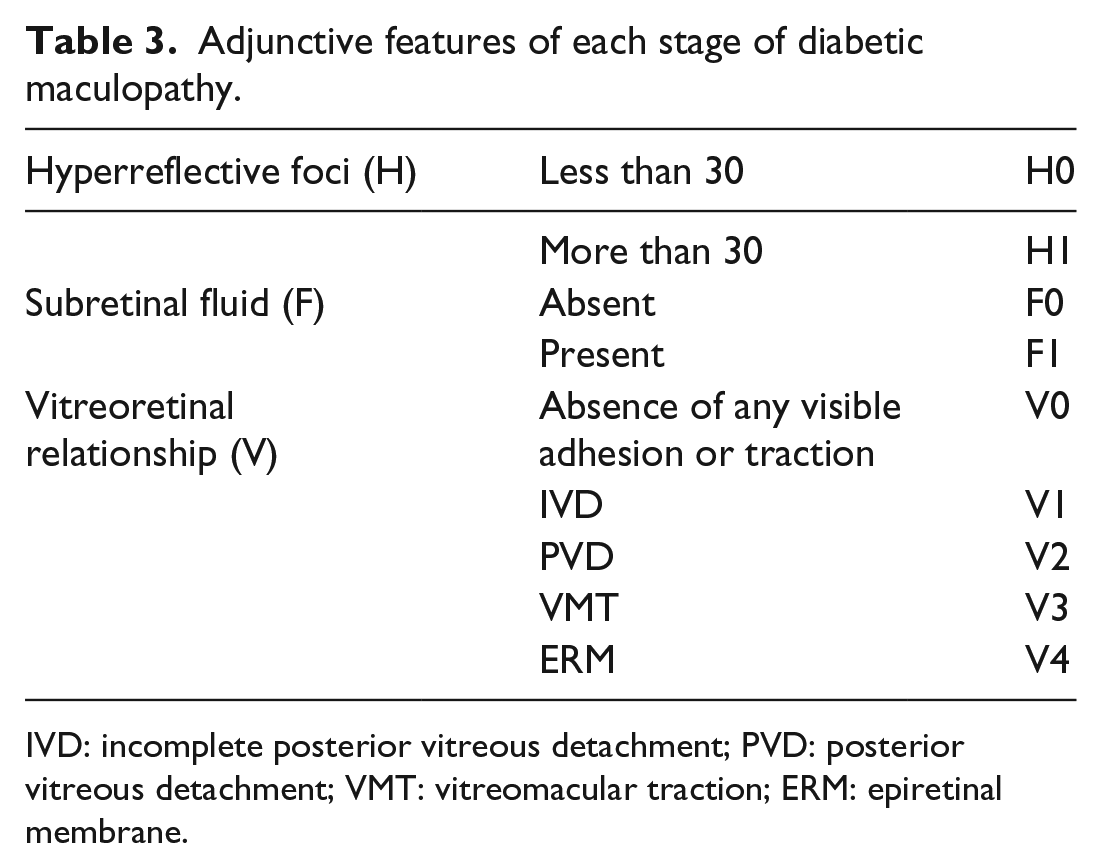
IVD: incomplete posterior vitreous detachment; PVD: posterior vitreous detachment; VMT: vitreomacular traction; ERM: epiretinal membrane.
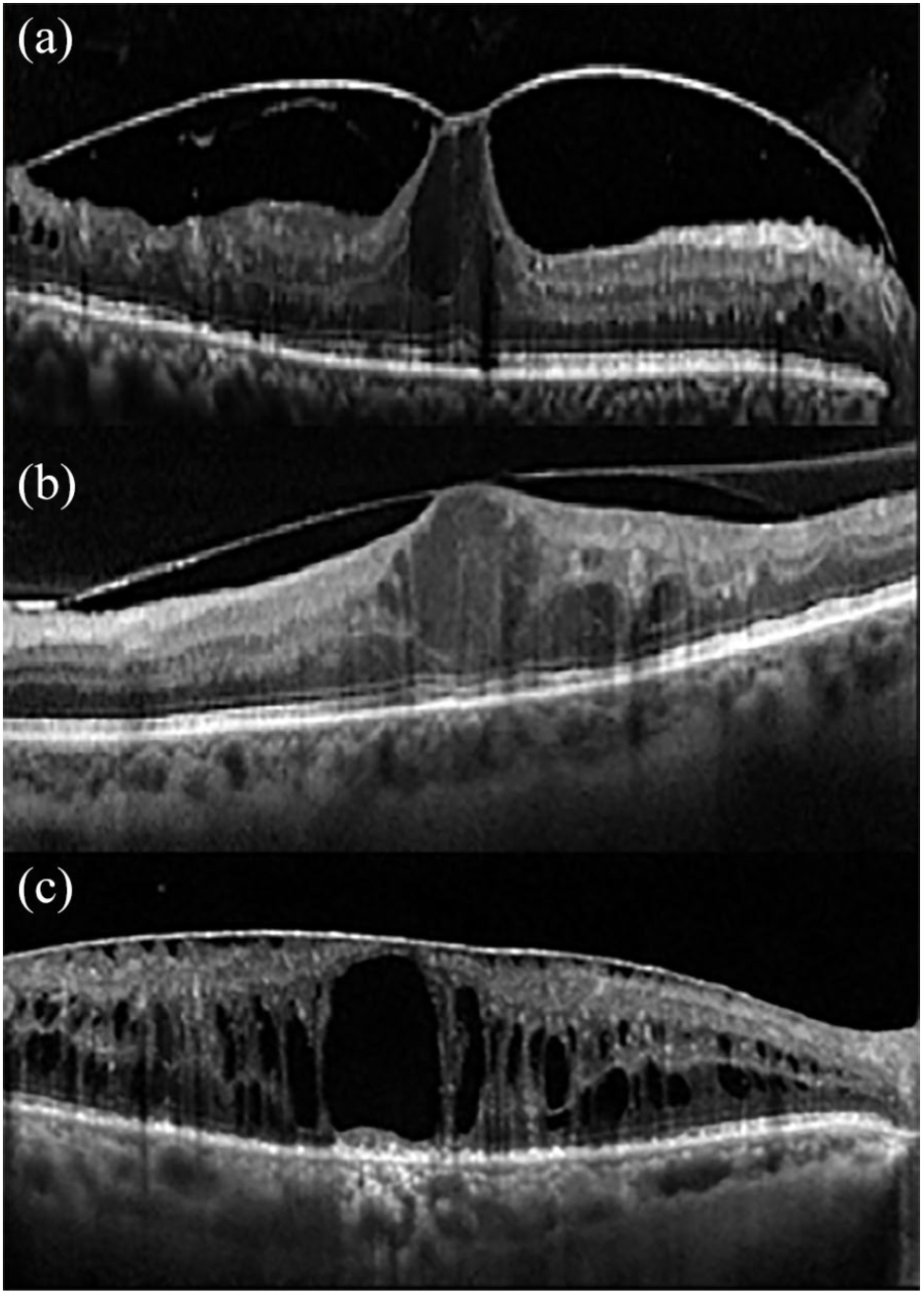
Vitreoretinal relationships associated with the spectrum of diabetic maculopathy.
Discussion
The recent innovations in OCT technology, with its high-resolution of retinal images, have contributed to advancements in our understanding of the pathophysiology of DM. As a result, there is a need to further classify the disease based upon not only the location of the retinal thickening, as first proposed by the ETDRS, 19 but also features associated with differing severities, response to treatment, and visual prognosis.
Several groups have proposed DM classifications, 20 often combining quantitative measurements and qualitative microstructural alterations of the outer retina, the inner retina, and the vitreoretinal interface (Table 4).
Morphological grading classification of diabetic macular edema based on optical coherence tomography (OCT).
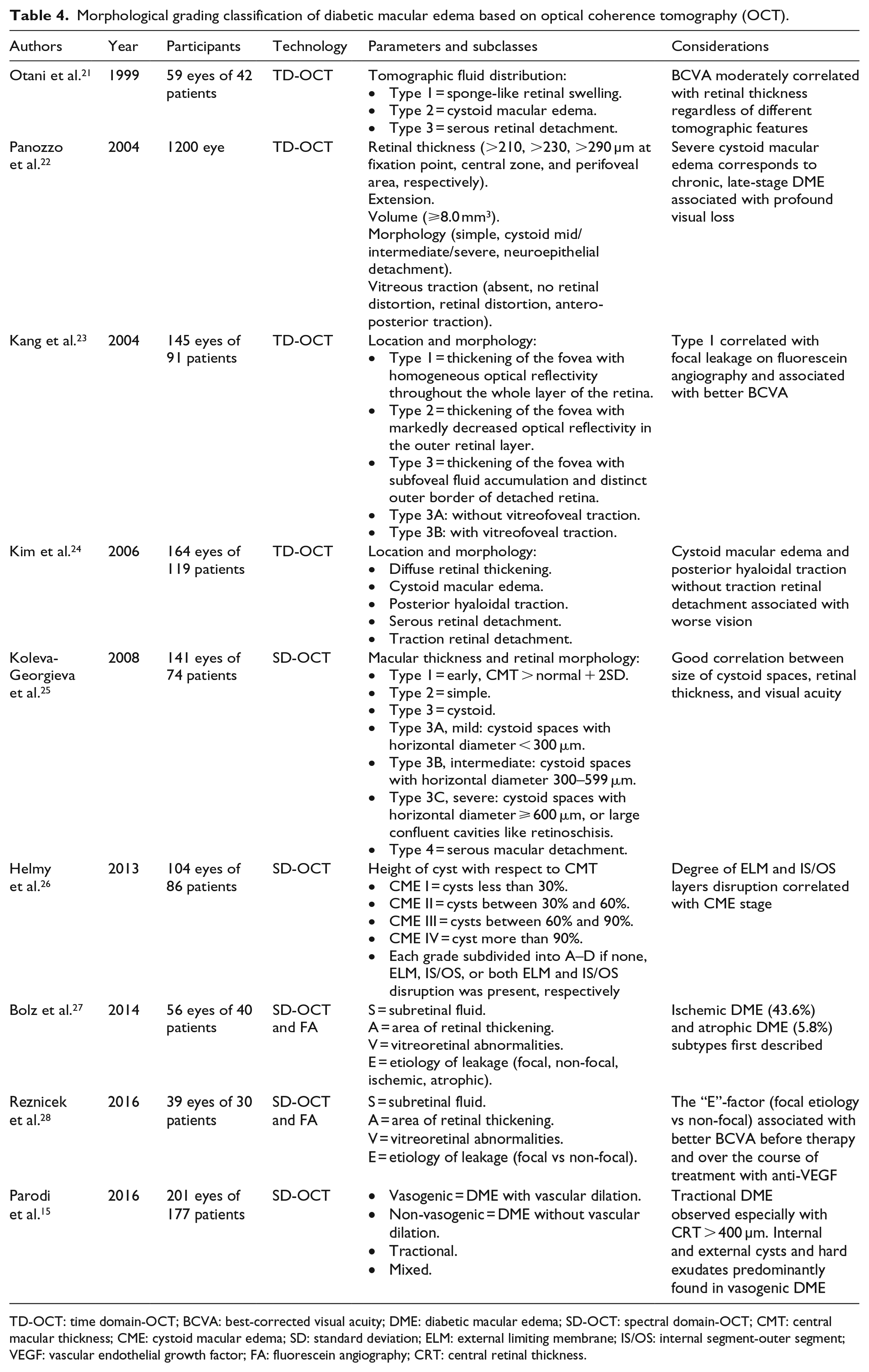
TD-OCT: time domain-OCT; BCVA: best-corrected visual acuity; DME: diabetic macular edema; SD-OCT: spectral domain-OCT; CMT: central macular thickness; CME: cystoid macular edema; SD: standard deviation; ELM: external limiting membrane; IS/OS: internal segment-outer segment; VEGF: vascular endothelial growth factor; FA: fluorescein angiography; CRT: central retinal thickness.
Quantitation of the CST, the central foveal thickness (CFT), the central retinal thickness (CRT) and the central macular thickness (CMT), have been used to evaluate disease activity, progression, and treatment response in RCTs as well as real-life. 29 However, evidence has shown only a weak correlation between these values and both baseline and final outcomes, especially at long-term follow-up. 30 A sub-analysis of the RIDE and RISE trials including patients with non-significant CFT modifications after anti-vascular endothelial growth factor (VEGF) therapy showed visual acuity gains and DR improvement similar to those who had immediate retinal thinning. 31 Bressler and associates demonstrated that the proportion of patients with chronic persistent macular edema gaining at least 10 letters from baseline to 24 weeks was similar to patients with a dry macula, irrespective of the therapeutic agent administered. 32 Patients with visual acuity of 20/40 or better who were enrolled in the bevacizumab arm of the Protocol T experienced the same amount of functional gain as the subjects treated with the other two anti-VEGF drugs, even though the retina remained thicker. 33
Additional information is available from the presence and the pattern of other qualitative features; in this study, the size of intraretinal cysts, the presence of DRIL, the number of HF, the occurrence of SF, and vitreoretinal alterations have been evaluated. The location and the size of intraretinal cysts have been correlated not only with the visual acuity at baseline, 34 but also with the anatomic response to treatment and improvement in macular function over the course of intravitreal anti-VEGF therapy. 35 There have been limited attempts to classify DME on the basis of the characteristics of intraretinal cysts. Koleva-Georgieva and Sivkova subdivided DME into mild, moderate, and severe according to the size of cystoid spaces, 25 while Panozzo et al. have first hypothesized that severe DME might correspond to chronic, late-stage exudative disease, and must be differentiated from advanced typical petaloid edema. 22 Helmy and Atta Allah then confirmed that the size and the appearance of the cysts might have different origins. 26 Large coalescent macrocysts in severe, long-standing disease may characterize retinal cystoid degeneration (RCD), an end-stage phenotype described in chronic diseases including central serous chorioretinopathy, 36 age-related macular degeneration, 37 and idiopathic macular telangiectasia, 38 and has been histopathologically correlated with Müller cell dysfunction or necrosis. 39 It has been showed intraretinal cysts located in the inner nuclear layer (INL) to be more responsive to either anti-VEGF or corticosteroids than fluid accumulation in the outer nuclear layer (ONL). 40 Nevertheless, we intentionally did not introduce a further categorization based on the intraretinal location of the cysts; our morphologic classification was aimed to be simple and directly accessible even for the non-retinal expert. We deemed that more detailed sub-grouping of retinal morphology might add complexity to the overall categorizing structure.
Since the introduction of SD-OCT, it is possible to assess the integrity of the outer retinal layers, visualized as the four outermost hyperreflective bands on structural B-scan, as well of the inner retinal layers. The extent of damage of the EZ, corresponding to the photoreceptor ellipsoid, and the ELM layer, corresponding to the adherens junctions between the Müller cells and photoreceptor pedicles, has been correlated with the functional outcome in previous studies. 41 Maheshwary et al. had previously demonstrated a strong relationship between visual acuity and foveal photoreceptor layer damage (intended as the percent disruption of the EZ line on SD-OCT) in eyes with DM, regardless the nature of previous treatments. 42 Abnormalities in the EZ OCT reflectivity have been found in patients with non-proliferative DR on both transversal and en face slab reconstructions, suggesting this might be a hallmark of neuronal damage secondary to hyperglycemia.43,44 Interestingly, EZ layer interruption seems to correlate to early microvascular impairment at the level of both the retina and the choroid and choriocapillaris.44,45
As the ELM status is also related to visual acuity, we considered the ELM status only in the presence of SF or ungradable EZ. 46 Similarly, the disorganization of the inner retinal structure on OCT, known as DRIL, with anatomical disruption of the visual pathway, has been correlated with macular ischemia and a worse visual outcome.47,48
The presence of HF, also called hyperreflective spots or dots, is presumed to be a biomarker of local inflammation in different retinal diseases, including exudative age-related macular degeneration, angioid streaks, and vitreoretinal pathology. We considered only HF with <30 μm diameter, reflectivity similar to nerve fiber layer, and absence of back-shadowing; these characteristics should be more specific for clusters of activated intraretinal microglia rather than other hyperreflective focal intraretinal lesions, such as hard exudates or microaneurysms. 49 The clinical and therapeutic importance of HF is still controversial. Zur et al. 45 and Chatziralli et al., 51 independently, found that the presence of HF was inversely correlated with the final visual acuity in eyes treated with intravitreal steroids.45,46 Conversely, Hwang et al. 52 reported that a larger number of HF were associated with a better outcome in eyes treated with dexamethasone. Further studies, assessing the accuracy and the repeatability of this classification in a therapeutic setting would clarify this point.
Accumulation of SF is thought to be either a sign of disruption of the external retinal-blood barrier, secondary to damage in the tight junctions of the retinal pigment epithelium (RPE) or insufficient fluid removal by an impaired RPE pump. 53 The presence of SF has been associated with reduced retinal sensitivity. 54 Its presence is predictive of better treatment response but poorer final vision after anti-VEGF injections. 10
We propose a grading of DM in 4 different disease stages, which reflects progressive severity of the disease. Early DM was defined by the presence of small intraretinal cysts associated with well-recognizable and detectable inner retinal layers, EZ, and ELM, and increase in CST and/or MV less than 30% of maximum normal values. This stage of the disease often corresponds to good visual acuity and a shorter duration of hyperglycemic state. However, it might be a problem in patients undergoing cataract surgery. The effect of surgery on pre-existing DM is not clear. Several studies suggest that cataract surgery is a risk factor for occurrence or worsening of macular edema.55,56 However, real-life studies have reported milder consequences of uneventful procedure on the macular status for eyes featuring early-stage disease. 57 A prospective Spanish study on 132 eyes revealed similar rate of occurrence of macular edema comparing eyes undergoing phacoemulsification and fellow control eyes. Similarly, a multicenter national DR database from UK estimated a relatively low rate of developing treatment-requiring DM after cataract surgery in patients with mild disease. 58
A few reports on the effects of intravitreal steroids or anti-VEGF in preventing post-surgical visual loss have been published.59,60 The better categorization of this initial stage of DM might improve the similarity of the patients included in these trials. Early DM may also represent the eyes studied in the recent Protocol V of DRCR Network. 61
We propose a distinction between advanced and severe DM, both characterized by macrocysts and/or multiple intraretinal cystoid spaces and a CST above 30% of maximum normal values but differing in the EZ/ELM state. While in the former, the EZ/ELM may be compromised but still visible and inner retinal layers segmentation is generally preserved. In the latter subtype, the inner retinal segmentation and/or EZ/ELM are mostly undetectable. The two categories may differ considerably in treatment response and visual outcome and should be distinguished.
Finally, the ESASO classification includes macular atrophy as a stage of DM. Macular atrophy is characterized by complete disruption of the outermost retinal layers (EZ/ELM) and DRIL; macular thickness might be below, within, or slightly above the normal values, if small degenerative cysts persist in the inner retinal layers. Macular atrophy is generally secondary to long-standing macular edema. Only a few authors have evaluated atrophic DM. Bolz et al. 57 introduced atrophic edema in the SAVE protocol, as the result of retinal cystoid degeneration due to degeneration of Müller cells and/or disruption of the photoreceptor integrity in the central subfield. However, the authors included only eyes with macular thickening on OCT, excluding those without macular edema. Moreover, the authors divided “ischemic DME” and “atrophic DME” in two separate categories; as disruption in the horizontal layers’ integrity (namely, DRIL) has been interpreted as an indirect sign of retinal ischemia and loss of normal vasculature in resolved macular edema, we found it more appropriate to merge the two groups. 62
We present the classification based solely on selected morphologic parameters. We recognize that other structural features, as choroidal thickness (CT), or clinical factors, as baseline visual acuity, age at baseline, local and systemic co-morbidities, features on fundus photography, may influence the different aspects of DM. 63 For instance, a recent report has analyzed the differential CT in different stages of DR, finding a progressive thinning towards more advanced stages of disease. 64 In addition, integration of new-generation OCT, the swept-source devices capable of imaging the retina and the choroid, and other diagnostic techniques gathering information on the perfusion status of the periphery and the macula, namely widefield FA and, more recently, OCT angiography, will provide a more precise definition of the grade of ischemia in the different stages of DM (not included in our classification at the present moment).
In conclusion, we propose a novel grading system of DM, classifying the phenotypes of macular involvement based on definitions, grading, and standard OCT figures, including also patients without retinal thickening. In this grading protocol, whose acronym TCED-HFV can be easily memorized as “To Classify Edema Discerning Hidden Functional Variables,” each of the features evaluated can be combined with the others, achieving four clinical stages. The strength of the present classification is its simplicity; it is intended to be kept as a reference for immediate DM grading.
The value of the present classification and its predictive role in treatment planning and functional prognosis must be established with multicentric larger trials.
Footnotes
Authors’ contributions
All the authors contributed to the conception or design of the work, the acquisition, analysis and interpretation of data, drafting the work, revising it critically for important intellectual content and gave final approval of the version to be published.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
