Abstract
Post-traumatic stress disorder (PTSD) is associated with increased cardiovascular disease (CVD) morbidity and mortality, yet the biological mechanisms linking psychological trauma to cardiovascular risk remain incompletely understood. Disruptions in one-carbon metabolism, reflected by elevated homocysteine and altered folate and vitamin B12 levels, contribute to vascular inflammation and endothelial dysfunction and may represent a modifiable pathway linking PTSD to CVD. The purpose of this study was to examine relationships among physiological biomarkers (homocysteine, folate, vitamin B12), PTSD diagnosis, PTSD treatment participation, and CVD risk in a veteran population. A quantitative, comparative retrospective chart review was conducted among 279 U.S. veterans with documented homocysteine levels. CVD risk was categorized as high or low-to-moderate based on metabolic and vascular risk factors. Logistic regression, odds ratios, and chi-square analyses were used to examine predictors of CVD risk and elevated homocysteine. The Neuman Systems Model guided variable selection and interpretation. Elevated homocysteine was significantly associated with age, gender, race, systolic blood pressure, folate, vitamin B12, PTSD diagnosis (OR = 4.31, 95% CI [1.36–13.61]), and CVD risk (OR = 3.50, 95% CI [1.01–12.05]). Participation in PTSD treatment was significantly associated with homocysteine levels (OR = 6.43, 95% CI [2.02–20.45]). Findings support homocysteine as a clinically relevant biomarker linking PTSD and cardiovascular risk. The association between PTSD treatment and homocysteine suggests psychological interventions may influence biological pathways relevant to cardiovascular health, underscoring the value of biomarker-informed nursing assessment in trauma-exposed populations.
Introduction
Cardiovascular disease (CVD) remains the leading cause of death globally and in the United States (World Health Organization, 2025). Each year, it claims over 17.9 million lives worldwide and more than 900,000 lives in the U.S., accounting for roughly one in three deaths (Centers for Disease Control and Prevention, 2024). Beyond mortality, the burden of CVD includes chronic illness, disability, and significant healthcare costs. More than 85.6 million Americans are currently living with some form of CVD, and projections suggest that by 2050, over 60% of the U.S. population will be affected (American Heart Association, 2024).
While these statistics are alarming, they are even more concerning when viewed through the lens of veteran health. Veterans face a unique convergence of risk factors including combat exposure, chronic stress, traumatic experiences, and psychological comorbidities, which increase their susceptibility to both acute and chronic cardiovascular conditions. Within the Veterans Affairs (VA) healthcare system, CVD is not only the most common cause of hospitalization, but also a leading contributor to disability and diminished quality of life (U.S. Department of Veterans Affairs, Office of Research & Development, 2025). The societal and economic implications are substantial. CVD-related conditions account for nearly 38% of U.S. healthcare expenditures, with costs projected to increase exponentially over the coming decades, from $393 billion to $1.49 trillion by 2060 (Kazi, 2024).
Although CVD has been extensively studied in the general population, the mechanisms that contribute to its disproportionate burden among veterans remain insufficiently understood. One area of growing interest is the intersection between post-traumatic stress disorder (PTSD) and cardiovascular health. PTSD is highly prevalent among veterans, and a growing body of evidence links the disorder to adverse cardiovascular outcomes (Padhi et al., 2024). A dose–response relationship has been observed between PTSD symptom severity and cardiovascular risk, suggesting that biological mechanisms, in addition to behavioral factors, may underlie this association (Kubzansky et al., 2009; Merkas et al., 2023). Emerging genetic and neurobiological research provides further insight into these mechanisms. Individuals with PTSD demonstrate heightened adrenergic activity both at baseline and in response to stress, resulting in sustained elevations in heart rate and blood pressure that impose chronic hemodynamic burden on the cardiovascular system (Shen et al., 2025). Shared genetic architecture between PTSD and cardiovascular traits supports dysregulation of stress-response pathways as a contributor to long-term vascular vulnerability. PTSD has also been associated with increased prevalence of dyslipidemia and impaired glucose regulation, suggesting trauma-related neuroendocrine alterations may accelerate metabolic risk. However, the precise physiological pathways linking PTSD to CVD remain incompletely characterized, and it is unclear whether treatment of PTSD can meaningfully alter these biological processes.
Chronic activation of stress-response systems in PTSD (including dysregulation of the hypothalamic–pituitary–adrenal (HPA) axis and sustained sympathetic nervous system activity) promotes oxidative stress, endothelial dysfunction, and vascular inflammation, which are central mechanisms in CVD development. These processes may influence one-carbon metabolism, a pathway critical to homocysteine regulation. Elevated homocysteine has been independently associated with endothelial injury and atherosclerosis, and increased levels have been observed in individuals exposed to chronic psychological stress (Yuan et al., 2023). Because homocysteine metabolism depends on adequate folate and vitamin B12 availability, deficiencies in these cofactors may contribute to elevated homocysteine and increased cardiovascular risk. In addition to their vascular effects, folate and vitamin B12 are involved in methylation processes affecting neurotransmitter synthesis, suggesting a potential biological intersection between PTSD-related psychological symptoms and cardiovascular vulnerability. Together, these associations support examination of homocysteine, folate, and vitamin B12 as physiological variables that may link PTSD diagnosis and treatment participation to CVD risk in veterans.
Although biological mechanisms are central to understanding PTSD-related cardiovascular risk, PTSD is a multidimensional condition encompassing psychological, behavioral, and physiological processes. Symptoms such as sleep disturbance, hyperarousal, and chronic stress exposure may contribute to cardiovascular vulnerability through behavioral and autonomic pathway (Straus et al., 2022). The present study focuses specifically on inflammatory and metabolic biomarkers as one component of this broader biopsychosocial framework.
Statement of the Problem
Veterans face a disproportionate burden of CVD, with PTSD emerging as a significant yet incompletely understood contributor. While prior research confirms an association between PTSD and increased cardiovascular risk, the specific biological pathways that drive this relationship, particularly those involving inflammatory and metabolic biomarkers such as homocysteine, folate and B12 remain insufficiently characterized in veteran populations. In addition, it’s unclear whether participation in evidence based PTSD treatment such as cognitive processing therapy is associated with measurable changes in cardiovascular risk markers. This gap in knowledge limits the development of biomarker informed, evidence-based strategies for early detection, risk stratification and interventions within the veteran population.
Purpose of the Study
The purpose of this study was to evaluate associations among homocysteine, folate, vitamin B12, PTSD diagnosis, Cognitive Processing Therapy (CPT) participation, and cardiovascular disease (CVD) risk in a veteran population. By investigating how these variables interact in the veteran population, this study seeks to clarify potential psychobiological pathways linking PTSD and CVD risk to explore whether treatment participation is associated with differences in CVD risk markers. These findings may inform nursing assessment, enhance CVD risk stratification, and guide future research and interventions tailored to veterans. In doing so, it contributes to the broader field of veterans’ studies by addressing a pressing issue at the intersection of trauma, biology, and long-term health outcomes.
Theoretical Framework: Neuman Systems Model
The Neuman Systems Model (NSM) served as the theoretical framework for this study by providing a structured approach to examining relationships among psychological stressors, physiological responses, and health outcomes. The NSM conceptualizes the individual as an open system that interacts dynamically with internal and external stressors. When stressors penetrate the flexible and normal lines of defense, physiological responses are activated, potentially influencing system stability and long-term health (Fawcett & DeSanto-Madeya, 2013).
Within this framework, PTSD diagnosis was conceptualized as a primary psychological stressor. Cognitive Processing Therapy (CPT) participation was conceptualized as a protective intervention corresponding to the flexible line of defense, representing a factor that may strengthen system stability. Physiological biomarkers (homocysteine, folate, and vitamin B12) were operationalized as measurable biological response indicators reflecting inflammatory and endothelial processes. CVD risk, defined using metabolic and vascular criteria, represented system stability and served as the primary dependent variable in the main analytic model.
Consistent with this conceptualization, two analytic models were specified. In the first model, CVD risk (high vs. low–moderate) was treated as the dependent variable, with PTSD diagnosis, CPT participation, homocysteine, folate, and vitamin B12 entered as independent variables. Age, gender, and race were included as covariates to account for internal and external system factors. In the second model, elevated homocysteine served as the dependent variable to examine its association with psychological factors, demographic characteristics, and cardiovascular risk. This approach reflects the NSM’s emphasis on dynamic relationships among stressors, physiological responses, and system stability without assuming direct causal pathways. This structured operationalization ensured that abstract NSM constructs were explicitly translated into measurable psychological, biological, and clinical variables consistent with the study aims and analytic strategy.
Figure 1 illustrates the structured mapping of NSM constructs to study variables and their roles in the analytic models. The NSM was selected because it integrates psychological stress exposure, biological response, and protective interventions within a unified explanatory framework, supporting examination of biomarker-informed cardiovascular risk assessment in trauma-exposed veteran populations. Conceptual and Analytic Model Derived from the Neuman Systems Model. This figure illustrates the structured mapping of Neuman Systems Model constructs to study variables. PTSD diagnosis is modeled as the primary psychological stressor and CPT participation as a protective intervention. Homocysteine, folate, and vitamin B12 represent physiological response variables. CVD risk is the primary dependent variable, with age, gender, and race included as covariates. Homocysteine is examined as both a dependent and independent variable across models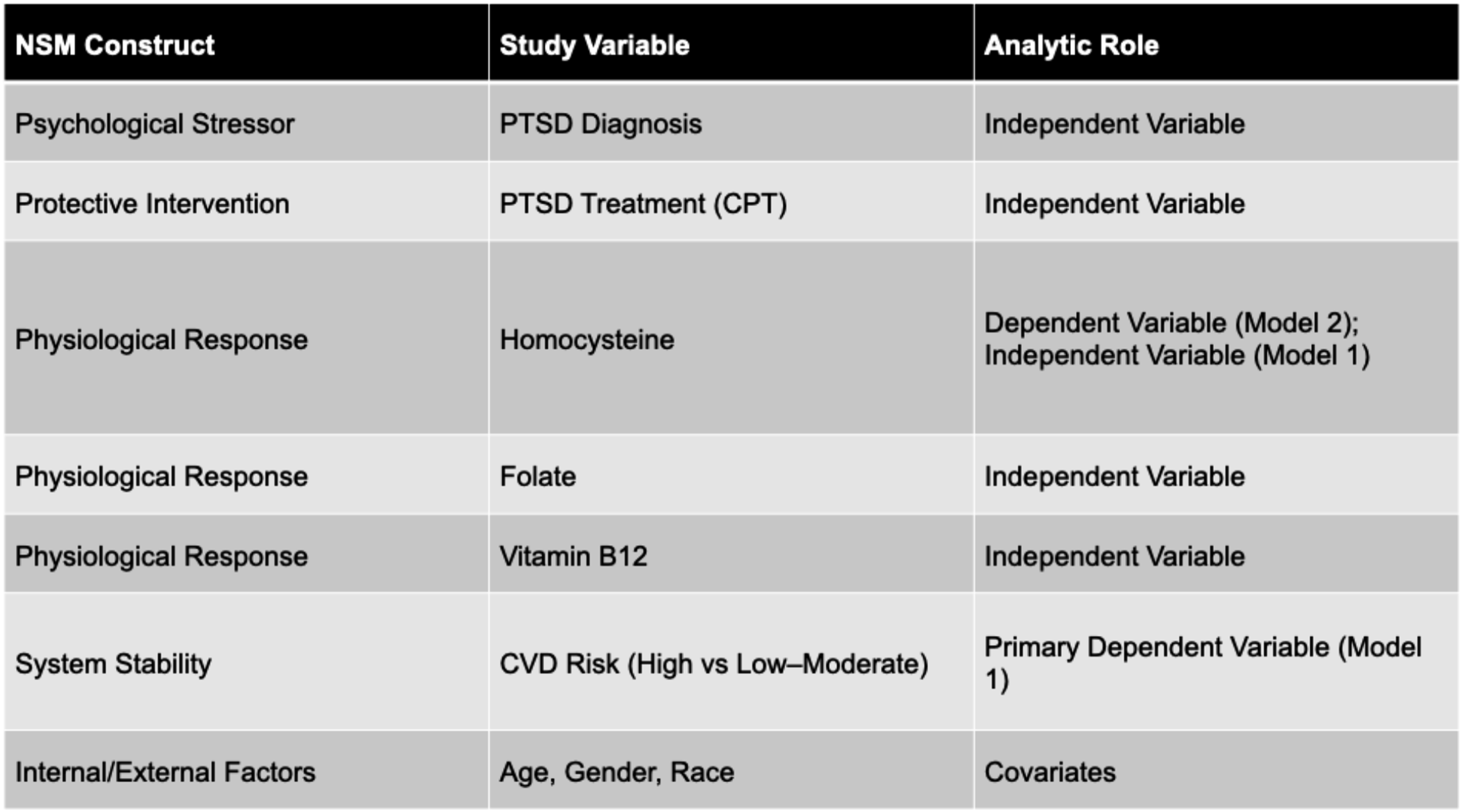
Literature Review
Search Strategy and Aims
A review of peer-reviewed literature published between 1997 and 2025 was conducted using CINAHL, OVID, MEDLINE, and PubMed. Search terms included post-traumatic stress disorder, cardiovascular disease, veterans, homocysteine, metabolic syndrome, folate, and vitamin B12. Clinical guidelines from the American Heart Association, American Diabetes Association, and National Institutes of Health were reviewed to define cardiovascular risk and biomarker thresholds. Landmark studies older than five years were included when relevant. The review aimed to summarize evidence defining CVD risk, identify relevant biological pathways, and highlight gaps regarding biomarkers and treatment effects.
Defining Cardiovascular Disease Risk
CVD risk reflects the cumulative burden of metabolic and vascular abnormalities, including hypertension, dyslipidemia, impaired glucose regulation, and obesity. These factors frequently cluster, producing synergistic effects that substantially increase cardiovascular risk (D’Agostino et al., 2008). Veterans may experience accelerated cardiovascular aging due to the combined effects of chronic stress, trauma exposure, and comorbid conditions.
Metabolic syndrome provides a clinically practical framework for categorizing cardiovascular risk by identifying clusters of these factors (Dhondge et al., 2024), therefore CVD risk was operationalized using metabolic syndrome criteria consistent with National Heart et al., 2024 guidelines. Veterans were classified as high CVD risk if they met at least three of the following criteria. • BMI ≥30 kg/m2 (obesity) • Systolic BP ≥130 mmHg or diastolic BP ≥85 mmHg • Fasting glucose ≥100 mg/dL or HbA1c ≥ 5.7% • Triglycerides ≥150 mg/dL • HDL <40 mg/dL (men) or <50 mg/dL (women)
Veterans meeting two or less criteria were classified as low–moderate risk.
Physiological Pathways Linking PTSD and CVD
Building upon evidence linking PTSD to dysregulated stress physiology, increasing attention has focused on downstream inflammatory and metabolic pathways that may contribute to cardiovascular vulnerability (Dell’Oste et al., 2023; Smith & Vale, 2023; Sumner et al., 2023; Tian et al., 2023). Among these, disruption of one-carbon metabolism represents a biologically plausible mechanism relevant to vascular risk. This pathway regulates homocysteine metabolism and depends on adequate folate and vitamin B12 availability.
Hyperhomocysteinemia has been independently associated with endothelial dysfunction, oxidative stress, and atherogenesis and is recognized as a risk factor for myocardial infarction and stroke (Clarke et al., 2019; Graham et al., 1997). Homocysteine may also interact with traditional cardiovascular risk factors, including blood pressure and lipid metabolism, potentially amplifying vascular injury. Because folate and vitamin B12 are essential cofactors in homocysteine regulation, deficiencies in these vitamins may contribute to both elevated homocysteine levels and increased inflammatory burden (Etcheverry & Gonzalez, 2024). These interrelated biological processes support examination of homocysteine, folate, and vitamin B12 as measurable indicators of cardiovascular risk in trauma-exposed populations.
Biomarkers of Inflammation
Homocysteine is a well-established biomarker of inflammation and endothelial dysfunction and improves cardiovascular risk prediction when incorporated into traditional risk models (Zhou et al., 2025). While traditional clinical definitions classify hyperhomocysteinemia at levels ≥15 µmol/L, evidence suggests that vascular risk may increase at lower concentrations within the upper-normal range (Etcheverry & Gonzalez, 2024; Liu et al., 2024). Because the present study focuses on cardiovascular risk rather than metabolic disorder diagnosis, homocysteine was categorized using a risk-sensitive threshold (≥ 8.5 µmol/L) to capture subclinical elevations potentially relevant to endothelial injury.
Folate and vitamin B12 are essential cofactors in one-carbon metabolism and play a central role in homocysteine regulation; deficiencies in either nutrient can contribute to elevated homocysteine levels and may independently influence vascular and neuropsychiatric processes relevant to cardiovascular risk (Maynard et al., 2023). Established laboratory reference ranges were used for folate (2–20 ng/mL) and vitamin B12 (200–900 pg/mL).
PTSD Treatment and Biological Outcomes
Despite strong evidence supporting the effectiveness of trauma-focused psychotherapies for reducing PTSD symptoms, limited research has examined whether psychological treatment influences biological markers associated with cardiovascular risk. Cognitive Processing Therapy (CPT) is a widely validated, standardized intervention with broad implementation in veteran healthcare settings, making it particularly suitable for examining potential psychobiological effects. Emerging work suggests that improvements in PTSD symptoms may be associated with reductions in incident CVD; however, specific effects on inflammatory and metabolic biomarkers remain largely unexplored (Scherrer et al., 2020). Evaluating whether CPT participation is associated with differences in homocysteine and related biomarkers may provide insight into potential psychobiological pathways linking treatment and cardiovascular risk.
Gaps in the Literature
Although PTSD has been linked to CVD, few studies have examined how cardiovascular risk stratification varies by PTSD status or how biomarkers associated with inflammation and endothelial dysfunction contribute to this relationship in veterans. Importantly, no studies have evaluated whether evidence-based PTSD treatments such as CPT are associated with changes in biological markers relevant to cardiovascular risk. Addressing these gaps may inform biomarker-informed nursing assessment and integrated strategies for CVD prevention in trauma-exposed populations.
Research Questions
This study addressed the following questions: (1) What demographic, psychological, and physiological factors are associated with high versus low–moderate cardiovascular disease (CVD) risk among veterans? (2) Are PTSD diagnosis and Cognitive Processing Therapy (CPT) participation independently associated with CVD risk after adjusting for homocysteine, folate, vitamin B12, age, gender, and race? (3) What demographic, psychological, and physiological factors are associated with elevated homocysteine levels among veterans? and (4) Are PTSD diagnosis and CPT participation independently associated with elevated homocysteine levels after adjusting for folate, vitamin B12, CVD risk, and demographic covariates?
Methodology
Study Design
This study employed a quantitative, comparative retrospective chart review to examine psychological (PTSD diagnosis), physiological (serum homocysteine, vitamin B12, folate), demographic (age, gender, race), and interventional (CPT) factors associated with CVD risk among veterans within a large southern California healthcare system. Institutional Review Board (IRB) approval was obtained, and inter-rater agreement was implemented to ensure data reliability.
Sample Selection and Size
A random purposive sampling strategy was used to promote diversity and generalizability to the veteran population. Participants were categorized as high risk (≥ 3 abnormalities in BMI, blood sugar, lipids, or blood pressure) or low–moderate risk (≤ 2 abnormalities). Using a significance level of p < .05, 80% power, and effect size estimates based on Cohen’s d, the required sample size was calculated as 245 veterans.
Exclusion Criteria
Exclusions included hospitalization for myocardial infarction or stroke at the time of homocysteine draw, homocysteine levels >100 µmol/L (to rule out rare genetic mutations), and diagnosis of type 1 diabetes due to differing inflammatory processes.
Operational Definitions
The Neuman Systems Model (NSM) guided conceptual framing, and operational definitions ensured consistent measurement of study variables within the retrospective dataset. CVD risk was operationalized using metabolic syndrome criteria consistent with National Heart, Lung, and Blood Institute guidelines. Veterans meeting three or more abnormalities (BMI ≥3 0 kg/m2, systolic BP ≥130 mmHg or diastolic BP ≥85 mmHg, fasting glucose ≥100 mg/dL or HbA1c ≥ 5.7%, triglycerides ≥150 mg/dL, HDL <40 mg/dL for men or <50 mg/dL for women) were classified as high risk. Veterans meeting two or fewer criteria were classified as low–moderate risk. In the primary analytic model, CVD risk (high vs. low–moderate) served as the dependent variable. Psychological status was operationalized as documented PTSD diagnosis (yes vs. no) in the electronic medical record. Although PTSD is a multidimensional condition encompassing symptom severity and behavioral manifestations, diagnostic status was used as the measurable psychological variable available within the retrospective dataset. Treatment participation was operationalized as documented participation in Cognitive Processing Therapy (CPT) versus no documented CPT participation within the medical record. Homocysteine, folate, and vitamin B12 were extracted from laboratory records. Homocysteine was categorized as normal (< 8.5 µmol/L) or elevated (≥ 8.5 µmol/L) using a risk-sensitive threshold selected to capture subclinical elevations relevant to cardiovascular risk stratification. Folate (2–20 ng/mL) and vitamin B12 (200–900 pg/mL) were evaluated using established laboratory reference ranges.
Results
Statistical Analysis
Descriptive statistics were used to summarize demographic, psychological, and physiological characteristics of the sample. Continuous variables were summarized using means and standard deviations, and categorical variables were summarized using frequencies and percentages.
Two logistic regression models were specified to examine associations among psychological, physiological, and demographic variables. In Model 1, CVD risk (high vs. low–moderate) served as the dependent variable. Independent variables included PTSD diagnosis, CPT participation, homocysteine, folate, and vitamin B12, with age, gender, and race entered as covariates. Bivariate associations between CVD risk and categorical variables were initially examined using odds ratios with 95% confidence intervals and chi-square analyses as appropriate. Variables demonstrating statistical relevance were subsequently entered into multivariable logistic regression models to identify independent predictors of CVD risk.
In Model 2, elevated homocysteine (≥ 8.5 µmol/L) served as the dependent variable. Independent variables included PTSD diagnosis, CPT participation, folate, vitamin B12, and CVD risk, with age, gender, and race included as covariates. Bivariate analyses were conducted to examine associations between homocysteine status and demographic, psychological, physiological, and cardiovascular variables. Variables meeting criteria for inclusion were entered into multivariable logistic regression analyses to determine independent predictors of elevated homocysteine levels. Statistical significance for all analyses was set at p < .05. This analytic structure reflects the Neuman Systems Model by examining associations among psychological stressors, physiological response indicators, protective interventions, and system stability without assuming direct causal pathways.
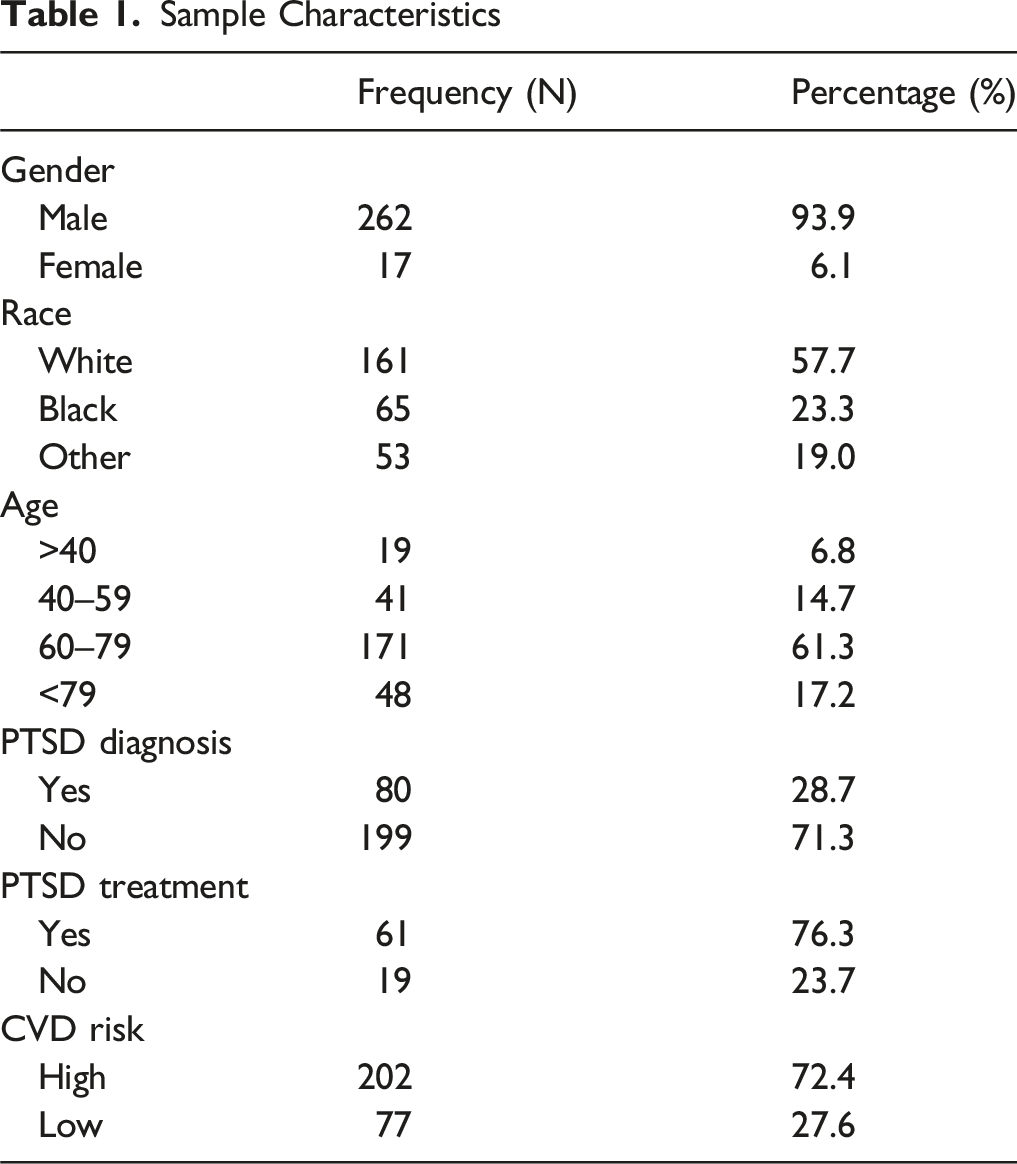
Sample Characteristics
Sample Characteristics
Cardiovascular Disease Risk Associations
Odds Ratio of Characteristics Among Veterans by CVD Risk
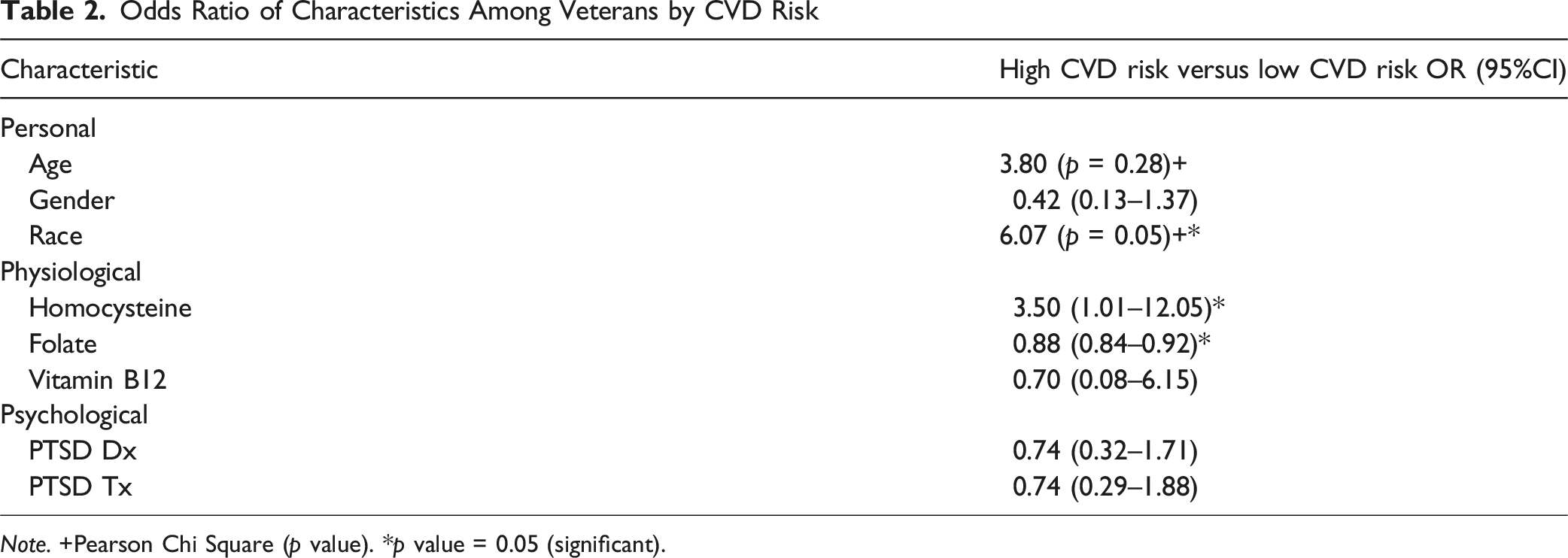
Note. +Pearson Chi Square (p value). *p value = 0.05 (significant).
Binary Logistical Regression to Predict Cardiovascular Disease Risk
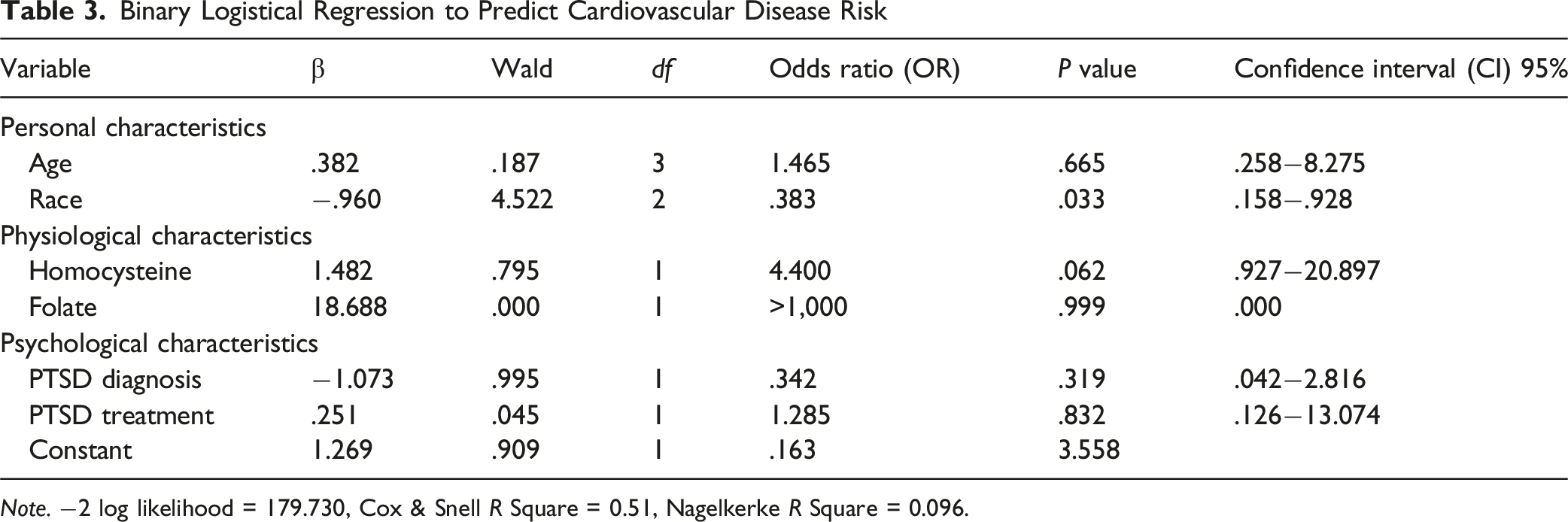
Note. −2 log likelihood = 179.730, Cox & Snell R Square = 0.51, Nagelkerke R Square = 0.096.
Homocysteine Associations
Odds Ratio of Characteristics Among Veterans by Homocysteine Level
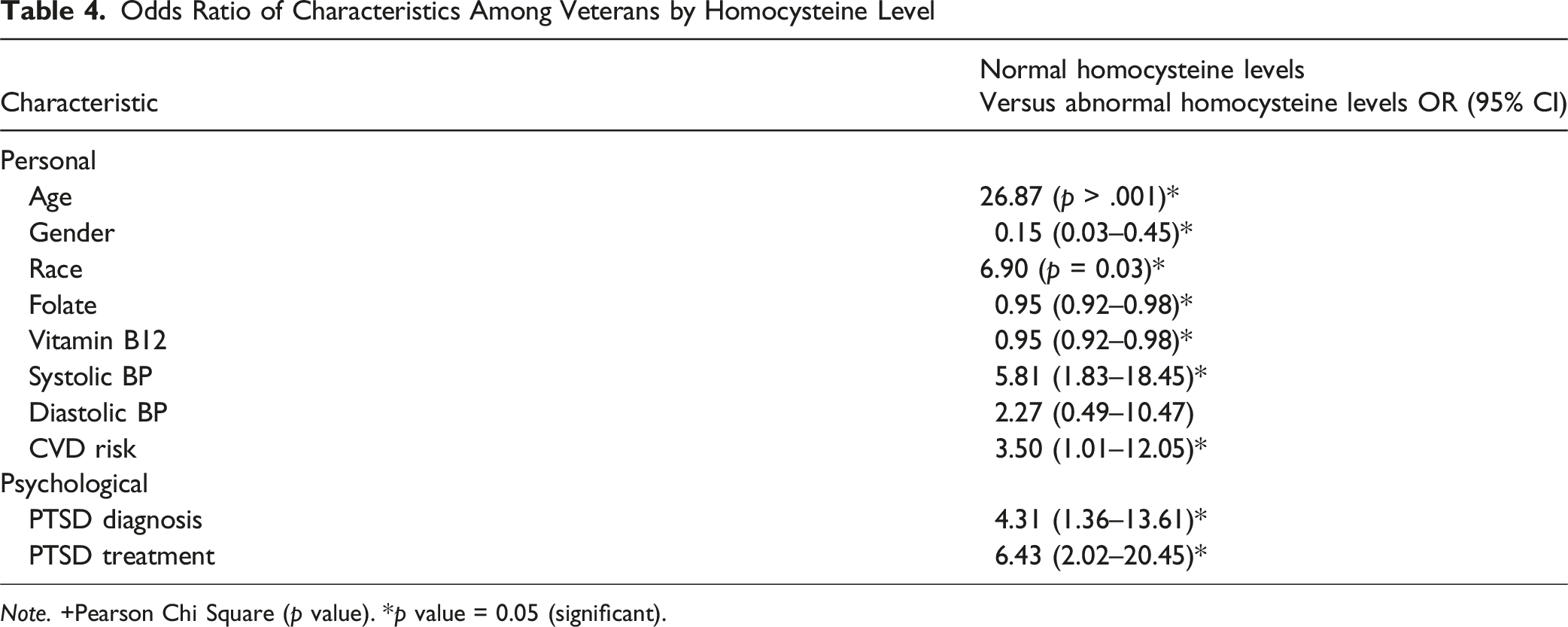
Note. +Pearson Chi Square (p value). *p value = 0.05 (significant).
Predictors of Elevated Homocysteine Levels
Binary Logistical Regression to Predict Homocysteine Levels
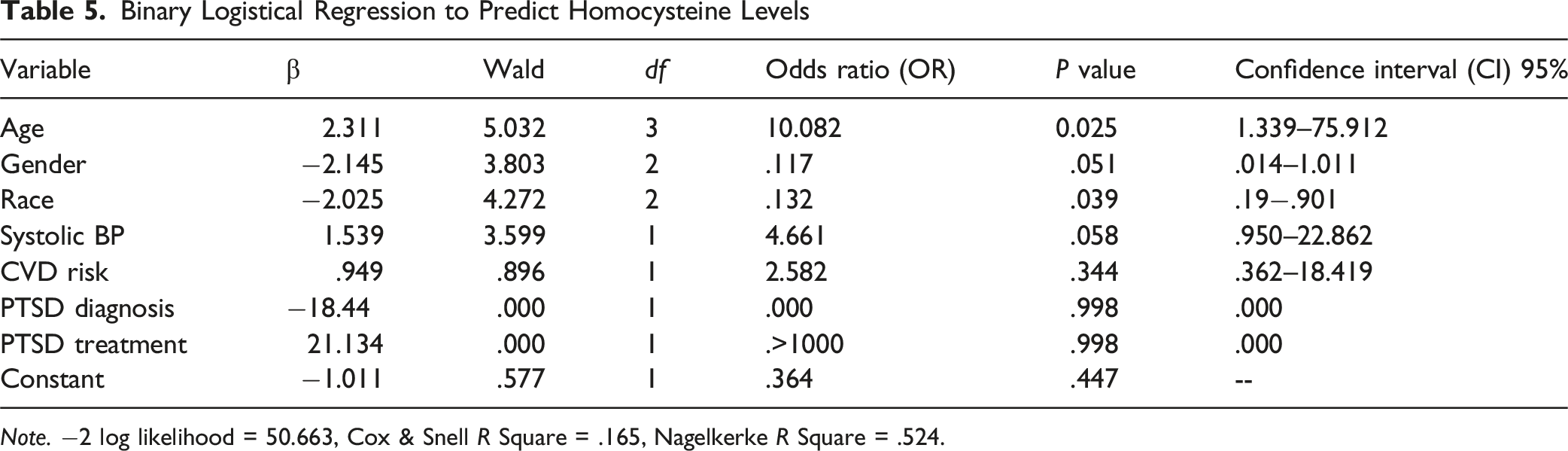
Note. −2 log likelihood = 50.663, Cox & Snell R Square = .165, Nagelkerke R Square = .524.
Discussion
This study examined psychobiological predictors of CVD risk in a veteran population, with a particular focus on homocysteine as a biomarker linking PTSD and cardiovascular vulnerability. Findings demonstrate that elevated homocysteine was significantly associated with PTSD diagnosis, CVD risk, systolic blood pressure, and nutritional cofactors involved in one-carbon metabolism, supporting its role as a biological intermediary between psychological stress and vascular pathology.
Homocysteine has been implicated in endothelial dysfunction, oxidative stress, and inflammatory processes that contribute to atherosclerosis and thrombosis. The observed associations between homocysteine and both PTSD and CVD risk suggest that chronic psychological stress may exert downstream cardiovascular effects through disruption of one-carbon metabolism. These findings align with emerging psychobiological models proposing that trauma-related neuroendocrine and inflammatory dysregulation accelerates CVD processes.
Clinical Implications and Significance to Nursing
Findings from this study identify clinically relevant predictors of CVD risk in veterans and underscore the value of integrating biological markers into nursing assessment and preventive care. Elevated homocysteine was associated with PTSD, cardiovascular risk, and multiple physiological factors, supporting its potential utility as a biomarker for identifying trauma-exposed individuals at increased CVD risk. Incorporating homocysteine assessment into routine laboratory panels for veterans with PTSD may (1) enhance early detection of cardiovascular vulnerability and inform targeted risk-reduction strategies and (2) be utilized to assess efficacy in PTSD treatments such as CPT.
A novel finding of this study was the significant association between PTSD treatment participation and homocysteine levels. While psychological interventions are typically evaluated based on symptom improvement, these results suggest that PTSD treatment may also influence biological pathways relevant to cardiovascular health. From a nursing and clinical perspective, physiological biomarkers such as homocysteine may complement symptom-based assessments by providing an objective indicator of treatment impact and long-term health risk. Together, these findings support biomarker-informed, interdisciplinary approaches to cardiovascular risk assessment and management in trauma-exposed populations.
Guided by the Neuman Systems Model, these findings underscore the interaction between psychological stressors, biological responses, and protective interventions in determining cardiovascular risk. Homocysteine may serve as a clinically actionable marker for identifying veterans at elevated cardiovascular risk and for informing targeted preventive strategies.
Strengths
This study utilized a clinically relevant veteran population drawn from a large VA healthcare system, reflecting real-world clinical settings. The integration of psychological, physiological, and demographic variables within a theoretically grounded framework allowed for comprehensive examination of psychobiological associations. The final sample size exceeded the calculated requirement, supporting adequate statistical power for multivariable analyses.
Limitations
CVD risk was operationalized using metabolic risk criteria rather than a comprehensive clinical risk calculator, which may not fully capture cardiovascular risk associated with clinical diagnoses or behavioral factors such as tobacco use. Homocysteine was categorized using a lower threshold (≥ 8.5 µmol/L) than conventional clinical definitions of hyperhomocysteinemia (≥ 15 µmol/L); although selected to capture subclinical cardiovascular risk, this approach may limit comparability with studies using traditional cutoffs. The wide age distribution may have influenced homocysteine levels due to age-related physiological increases, potentially affecting observed associations despite statistical adjustment. The predominance of male participants (93.9%) limits gender-specific inference and generalizability to women veterans, and potential collinearity between PTSD diagnosis and CPT participation may have influenced regression estimates.
Future Research
Future studies should employ prospective designs and comprehensive cardiovascular risk models, including tobacco use, to further clarify psychobiological pathways linking PTSD and CVD. Randomized trials are also needed to evaluate whether PTSD treatment and homocysteine-lowering interventions influence long-term cardiovascular outcomes, particularly in female veteran populations.
Conclusion
This study identified homocysteine as a clinically relevant biomarker associated with PTSD, CVD risk, and related physiological factors in veterans. Findings suggest that biomarker-informed nursing assessments may enhance early identification of cardiovascular risk and provide insight into the biological effects of PTSD treatment, supporting integrated approaches to cardiovascular prevention in trauma-exposed populations.
Footnotes
Acknowledgments
The author would like to acknowledge the Greater Los Angeles Veterans Healthcare Administration and faculty at Azusa Pacific University. No funding was received for this work.
Ethical Considerations
This study was approved by the Azusa Pacific University Institutional Review Board, approval number #Exempt 91-16 and Greater Los Angeles Veterans Healthcare Administration Institutional Review Board #2016-0260550.
Consent to Participate
Waived by Azusa Pacific University Institutional Review Board and Greater Los Angeles Veterans Healthcare Administration Institutional Review Board.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are owned by the Greater Los Angeles Veterans Healthcare Administration and are not publicly available due to privacy and institutional restrictions. Access to these data may be possible upon reasonable request and with appropriate permissions from the Veterans Administration.
