Abstract
Objective
This study aimed to investigate the effects of aerobic interval training and resistance training on anti-inflammatory adipokines, high sensitivity C-reactive protein, and clinical outcomes in sedentary men with metabolic syndrome.
Methods
A total of 33 sedentary men with metabolic syndrome (age: 46.2 ± 4.6 years; body mass index: 35.4 ± 1.9 kg.m2) were randomly assigned to one of 3 groups: aerobic interval training (n = 12), resistance training (n = 10), or control (n = 11). Participants in the exercise groups completed a 12-week training program, 3 sessions per week, while those in the control group maintained their sedentary lifestyle. The levels of high sensitivity C-reactive protein (hs-CRP), omentin-1, adiponectin, lipid profiles, blood pressure, glucose metabolism, body composition, and peak oxygen uptake (VO2peak) were measured at baseline and after the intervention.
Results
Both aerobic interval training and resistance training significantly improved the levels of omentin-1 and adiponectin, as well as reduced inflammation, as indicated by a decrease in hs-CRP levels. Exercise training also led to significant improvements in lipid profiles, blood pressure, glucose metabolism, and body composition. Specifically, the aerobic interval training group had significantly greater increases in high-density lipoprotein cholesterol and VO2peak, as well as greater reductions in low-density lipoprotein cholesterol, triglycerides, and total cholesterol compared to the resistance training group.
Conclusion
Exercise training, particularly aerobic interval training and resistance training, can be an effective non-pharmacological intervention for managing inflammation and improving cardiovascular health in metabolic syndrome patients.
Introduction
Metabolic syndrome is a group of metabolic risks that can contribute to the development of conditions such as diabetes, coronary heart disease, non-alcoholic fatty liver disease, and a number of tumors (Han et al., 2021), and it has been demonstrated that it is more prevalent in men than in women (Grundy, 2008). This cluster of risks includes insulin resistance, visceral adiposity, high blood pressure, and dyslipidemia (Lemos et al., 2023; Mohammad Rahimi et al., 2020). Recent statistics reveal that in 2019, approximately 460 million individuals, accounting to 6.25% of the world’s population, experienced type 2 diabetes (Virani et al., 2021). Additionally, cardiovascular disease (CVD), which is primarily caused by excessive body weight and lack of physical activity, was responsible for approximately 18.6 million deaths worldwide, making it the leading cause of mortality (Virani et al., 2021).
Studies have demonstrated that elevated levels of C-reactive protein (CRP), which is a measurable biomarker of inflammation, are correlated with all parameters of metabolic syndrome. CRP has been found to be a robust and independent predictor of incident diabetes (Pradhan et al., 2001), as well as incident CVD (Ridker et al., 2002). Moreover, the serum level of CRP is associated with other components of metabolic syndrome, such as fasting insulin, microalbuminuria, and impaired fibrinolysis. These components are not commonly assessed in routine clinical practice (Fröhlich et al., 2000).
On the other hand, research has shown that a low level of adiponectin, a peptide produced by adipose tissue (Banu & Elango, 2019), is associated with the prevalence of metabolic syndrome (Banu & Elango, 2019; Mohammad Rahimi et al., 2020), insulin resistance (Adiyaman et al., 2020), coronary heart disease (Lindberg et al., 2017), and diseases related to inflammation (Mohammad Rahimi et al., 2022). Elevated levels of adiponectin promote insulin sensitivity in skeletal muscles by binding to adiponectin receptor type 1 and 2, which triggers the activation of several signaling pathways (Roy & Palaniyandi, 2021). Adiponectin also has anti-inflammatory effects by decreasing the production of proinflammatory adipokines in different cell types (Ouchi & Walsh, 2007).
Omentin is an adipokine weighing 38 kilo Daltons which is mainly secreted by omental adipose tissue. At first, omentin was identified as lectin of solute binding galactofuranose (intelectin) (Yang et al., 2006). Research has shown that this adipokine enhances the uptake of insulin-stimulated glucose into human adipose tissue cells through the Akt signaling pathway. On the other hand, in cases of obesity, insulin resistance, and metabolic resistance, it reduces the expression of omentin in visceral adipose tissue (Buyukinan et al., 2018). An increase in insulin sensitivity and glucose metabolism are paracrine and endocrine effects of omentin in people with normal weight (Liu et al., 2015). The plasma concentrations of omentin-1, the main isoform in human blood circulation are reduced in obesity, and omentin-1 is inversely correlated with body mass index (BMI), leptin, insulin resistance, and waist circumference, and positively correlated with high-density lipoprotein cholesterol and adiponectin (de Souza Batista et al., 2007). Additionally, it plays a role in regulating energy expenditure in skeletal muscles (Liu et al., 2020). Yamawaki et al. were the first to describe the anti-inflammatory effect of omentin, demonstrating its preventive effect on TNF-α-induced cyclooxygenase-2 (COX-2) expression in vascular endothelial cells (Yamawaki et al., 2011).
Regular physical exercise is a widely recognized approach recommended as a solution for metabolic syndrome. However, the majority of exercise recommendations emphasize continuous training, which may be challenging to maintain due to the monotony of this type of training, making it difficult for individuals to transition from a sedentary lifestyle (Coquart et al., 2008). Consequently, alternative modes of training, such as interval training, have been suggested as a more feasible option for these individuals compared to continuous training (Coquart et al., 2008). Additionally, interval training has been shown to offer several benefits, including enhanced peak oxygen uptake, blood glucose, insulin levels, insulin resistance index, HbA1c, body mass, and BMI in obese patients (Coquart et al., 2008; Racil et al., 2016). Likewise, the aerobic interval mode of exercise training can be utilized as a time-efficient lifestyle intervention approach for individuals with metabolic syndrome (Mohammad Rahimi et al., 2020). On the other hand, resistance exercise training involves exercising a muscle or group of muscles against an external force to promote muscular fitness. The American Diabetes Association has recognized resistance training as a crucial component of a training regimen (Sigal et al., 2004). When conducted correctly, resistance training can offer numerous functional benefits such as improved glucose tolerance and insulin sensitivity in type 2 diabetes (Codella et al., 2018), as well as enhancement of metabolism and fitness in sedentary individuals (Shaw & Shaw, 2005).
Studies have demonstrated that aerobic interval and resistance training have different impact on inflammation, insulin sensitivity, and adipokine profile in patients with type 2 diabetes or metabolic syndrome. For example, aerobic interval exercise training has been shown to reduce high sensitivity CRP (hs-CRP) levels and increase adiponectin (Da Silva et al., 2020; Fedewa et al., 2017; Guadalupe-Grau et al., 2018; Mora-Rodriguez et al., 2018; Steckling et al., 2019), while resistance training has been found to decrease omentin-1 levels (AminiLari et al., 2017; Dianatinasab et al., 2020). These findings highlight the importance of tailoring exercise prescriptions to individual patient needs and goals. However, there is a lack of studies directly comparing the effects of these 2 different modes of exercise in sedentary patients with metabolic syndrome, particularly in relation to anti-inflammatory adipokines. Furthermore, previous studies have had shorter intervention periods, limiting the evaluation of longer-term effects. Therefore, the objective of our study was to investigate the effect of 12 weeks of aerobic interval training and resistance training on anti-inflammatory adipokines, hs-CRP, and clinical outcomes in sedentary men with metabolic syndrome.
Methods
Participants
Thirty-three men with metabolic syndrome (age: 46.2 ± 4.6 years; body mass index (BMI): 35.4 ± 1.9 kg/m2) participated in this study. Participants who met at least 3 of the 5 following criteria were classified as having metabolic syndrome: a waist circumference of ≥102 cm, a triglyceride level of ≥150 mg/dl, an high density lipoprotein cholesterol level of <40 mg/dl, a blood pressure of ≥130/80 mmHg, and a glucose level of ≥100 mg/dl (Grundy, 2004). Moreover, if the level of physical activity of participants was below 150 min per week, they were considered to be inactive (Füzéki & Banzer, 2018).
Recruitment for the study was conducted in Mashhad, Iran, through advertisements in local community centers. Potential participants were screened for eligibility based on pre-defined inclusion and exclusion criteria. Eligible participants were provided with written informed consent forms that included information on the study procedures, risks, and benefits. Participants were given adequate time to review the forms and ask any questions they had before deciding whether to participate. Those who agreed to participate provided written informed consent before enrollment. The informed consent process followed the ethical standards outlined in the Declaration of Helsinki and was approved by the Ethics Committee of the Ferdowsi University of Mashhad, Iran (project no. IR.MUMS.REC.1397.203).
The study enrollment and interventions took place in Mashhad, Iran, spanning from 1 June 2017 to 15 March 2018. Individuals who possessed the following characteristics were excluded from the study: cardiopulmonary disease, infection, physical restrictions or neuromuscular disability, a highly active lifestyle (defined as engaging in regular exercise more than once a week), past or present smoking habits, and consumption of weight loss medication.
After completing initial assessments, participants were randomly allocated to one of 3 groups in a 1:1:1 ratio: aerobic interval training (n = 12), resistance training (n = 10), or a non-exercise control (n = 11) group. Randomization was conducted using a random number generator. The intervention groups underwent training 3 sessions a week for 12 weeks, and the control group did not take part in any work out intervention for the same period. Patients who failed to attend over 20% of the exercise sessions were excluded from the study. For the aerobic interval training group, participants were expected to maintain their heart rate within a target range during the aerobic interval sessions, which was monitored using a heart rate monitor (Polar Electro, Espoo, Finland).
The exercise sessions in our study were conducted by qualified professionals who had received appropriate training and possessed relevant experience in exercise science. Prior to the commencement of the study, these supervisors underwent comprehensive training on the intervention protocol and were provided with detailed instructions on how to conduct the exercise sessions in a standardized manner. To ensure the fidelity of the intervention sessions, we implemented several quality control measures, such as regular observations of the exercise sessions by a quality control supervisor and random checks of the heart rate monitor data to verify that the participants were maintaining their heart rate within the target range. Any deviations from the established protocol were diligently recorded and promptly addressed.
Procedures
The study administrator, who was unaware of the group assignment, measured the participants' BMI and waist circumference at the beginning and end of the 12-week study period. Height (in centimeters) and body mass (in kilograms) of all patients were assessed using a digital stadiometer (Seca, Hamburg, Germany) and a digital scale (Beurer PS 07, Ulm, Germany), respectively, with a precision of 0.1 cm and 0.1 kg. The participants’ BMI was calculated as their body mass in kilograms divided by their height in meters squared (kg/m2). Waist circumference (in centimeters) was measured using a measuring tape (Seca 201, Southam, UK) at the midpoint between the last free rib and the iliac crest. The Inbody 720 Body Composition Analyser, manufactured by Biospace in Dogok-dong, South Korea, was employed to assess bioimpedance parameters including fat mass, percentage of body fat, muscle mass, and body water. After a brief 5-minute relaxation period in the supine position, the participants’ resting heart rate was assessed along with their blood pressure using a mercury sphygmomanometer.
Daily Nutritional Profile.
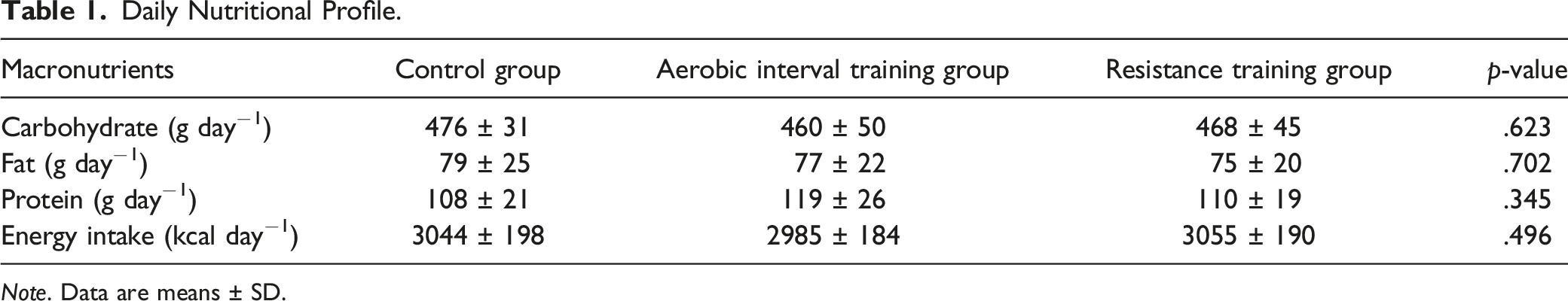
Note. Data are means ± SD.
To measure peak oxygen consumption (VO2peak; in ml/kg min) at baseline and after 12 weeks, participants underwent a Bruce treadmill walking test (Bruce, 1971) using a calibrated treadmill (Cosmos, Hp, Germany). The test was administered by a certified exercise physiologist with extensive experience in administering the Bruce protocol, and VO2 was evaluated continuously during exercise testing using breath-by-breath technology (Cortex, MetaLyzer 3B-R2, Leipzig, Germany). The Bruce protocol involves a series of incremental exercise stages on the treadmill and was chosen because of its established validity and reliability for evaluating maximal oxygen uptake.
Blood Assays
Blood samples were obtained between 8:00 and 10:00 in the morning following a 12-hour period of fasting overnight (after a 5 min rest in the supine position). Twenty milliliters of venous blood were extracted from the participants’ antecubital vein using a tube that contained K2EDTA. The samples were then allowed to coagulate at room temperature for a duration of 15 minutes. After centrifugation at 3000 g for 10 minutes, the samples were separated from blood cells. The plasma was then transferred into 0.5 mL plasma tubes using sterile pipettes. The aliquots were immediately stored at a temperature of −80°C in a refrigerator until the assay was performed. The levels of hs-CRP, adiponectin, and omentin-1 were measured using an ELISA Kit (ZellBio GmbH, Ulm, Germany) in accordance with the manufacturer’s instructions. The concentrations of glucose, triglyceride, total cholesterol, high- and low-density lipoproteins were determined using Pars-Azmun (Tehran, Iran) kits with an enzymatic process. The insulin levels were assessed through an ELISA process employing the Saman Tajhiz-Noor kit (Tehran, Iran). After measuring the plasma glucose and insulin, Homeostasis Model Assessment Insulin Resistance was calculated by the following formula to estimate insulin resistance (Salgado et al., 2010).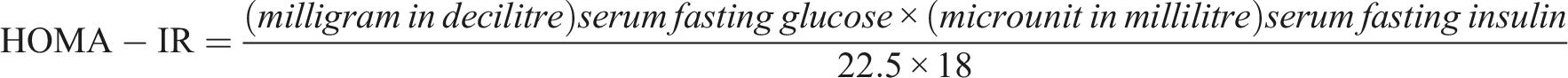
Training Protocol
The aerobic interval training program, as described by Stensvold et al. (2010), involved conducting exercise sessions on 3 days of the week (Monday, Wednesday, and Saturday). The program began with a 10-minute warm-up phase that was performed at 70% of the heart rate peak (HRpeak). This was followed by 4 sets of 4-minute intervals on a treadmill, during which the patients voluntarily walked or ran at 90% of HRpeak. The intervals were separated by 3 minutes of exercise at 70% of HRpeak. Finally, patients ended the session with a 5-minute cool-down phase. Overall, the total exercise time amounted to 43 minutes per session (Stensvold et al., 2010).
The resistance training program was implemented 3 times a week on Tuesday, Thursday, and Sunday, and comprised of working out on 7 different exercise weight machines. These machines were the lateral pull-down, chest press, seated row, triceps push-down, knee flexion, knee extension, and leg press. During the initial week, participants performed 2 sets of 15–20 repetitions at an intensity of 40%–45% of their one-repetition maximum (1RM). Subsequently, in the second week, the number of sets and the intensity were raised to 3 and 45%–50% 1RM, respectively, while keeping the repetitions constant. Throughout the third to sixth week, the repetitions were reduced to 12–15, while the intensity was raised to 50%–60% 1RM. In weeks 7–10, the number of repetitions was further reduced to 8–12, and the intensity was increased to 65%–70% 1RM. In the final 2 weeks of the exercise program, the intensity of training was increased to 70%–80% 1RM, while the number of repetitions remained the same. The exercise sessions lasted approximately 45 minutes per session (Stensvold et al., 2010).
It is important to note that the entirety of the 2 intervention groups underwent their training during the afternoon. The training sessions were conducted post-lunch, approximately 3 hours later at 1600 h, and an hour later, the participants took their dinner. Moreover, before the start of the resistance training program, all participants underwent a familiarization period in the first week, during which they were introduced to the protocol. In the final session of this familiarization period, Brzycki’s formula was used to determine the participants’ 1RM strength for each exercise (Brzycki, 1993). The participants’ 1RM values for the bench press and leg press exercises were used as measures of strength for the upper and lower extremities, respectively. To ensure consistency, the same instructors oversaw all of the strength tests and made individual adjustments as needed.
Statistical Analysis
The statistical analyses were conducted using version 27.0 of the Statistical Package for Social Sciences (SPSS Inc., Chicago, USA) software. Prior to conducting the statistical analyses, all of the data underwent tests to assess homogeneity of variance using Levene’s test and normality using Shapiro-Wilk’s test. Considering the small sample size, the sample power for a post hoc F-test family was calculated for α level = 0.05; effect size = 0.4; 3 groups, and n = 30 through by the G-Power (Faul et al., 2007). It was shown that there was a 92.2% (actual power) for the analysis. These parameters were chosen based on previous studies that have examined the effects of exercise training on metabolic parameters in adults (Cauza et al., 2005; Tjønna et al., 2008). For comparisons of parametric variables between groups at baseline, a one-way analysis of variance (ANOVA) with post-hoc Tukey test was used. Changes in variables between groups were analyzed using a one-way ANOVA for repeated measures, while changes within groups were evaluated using paired-samples t-tests. Moreover, to reveal the effect size (ES) in repeated measures ANOVA test, partial eta-squared (
Results
Changes of Body Composition and Physiological Variables From Baseline to the End of the Study (Mean ± SD).
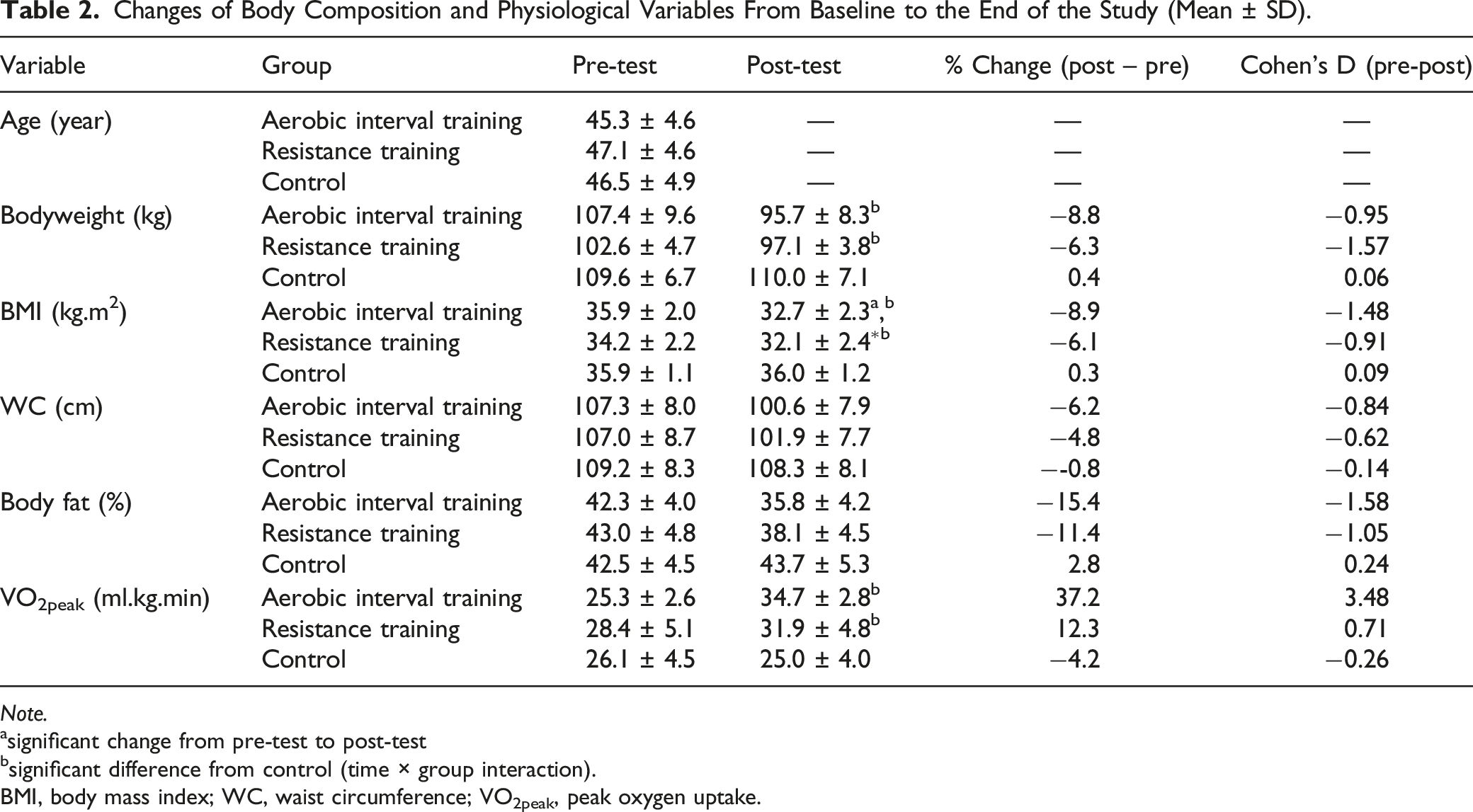
Note.
asignificant change from pre-test to post-test
bsignificant difference from control (time × group interaction).
BMI, body mass index; WC, waist circumference; VO2peak, peak oxygen uptake.
Changes in hs-CRP and Adipokines
Changes in hs-CRP and anti-inflammatory adipokines are given in Figure 1. The significant effects of time were observed for hs-CRP (p < .001, ηp2 = 0.505), omentin-1 (p < .001, ηp2 = 0.850), and adiponectin (p < .001, ηp2 = 0.870). Moreover, there were significant group-by-time interactions for hs-CRP ((aerobic interval training = 0.4 mg/L. d = −0.61) and resistance training = 0.2 mg/L, d =−0.33)), which significantly decreased (p < .001), while omentin-1 (aerobic interval training = 111.7 ng/ml, d = 7.74) and resistance training = 74.3 ng/ml, d = 3.54)) and adiponectin (aerobic interval training = 3.8 μg/ml, d = 5.06) and resistance training = 1.9 μg/ml, d = 1.98) significantly increased (p < .001 for both). Alterations (mean ± standard error) in hs-CRP, omentin-1, and adiponectin before and after 12 weeks of intervention. Hs-CRP: high sensitivity C-reactive protein. *Significant (p < .05) difference from baseline for each group, #significant difference (p < .05) between training and control groups (time × group interaction), $significant difference (p < .05) between aerobic interval training and resistance training groups (time × group interaction).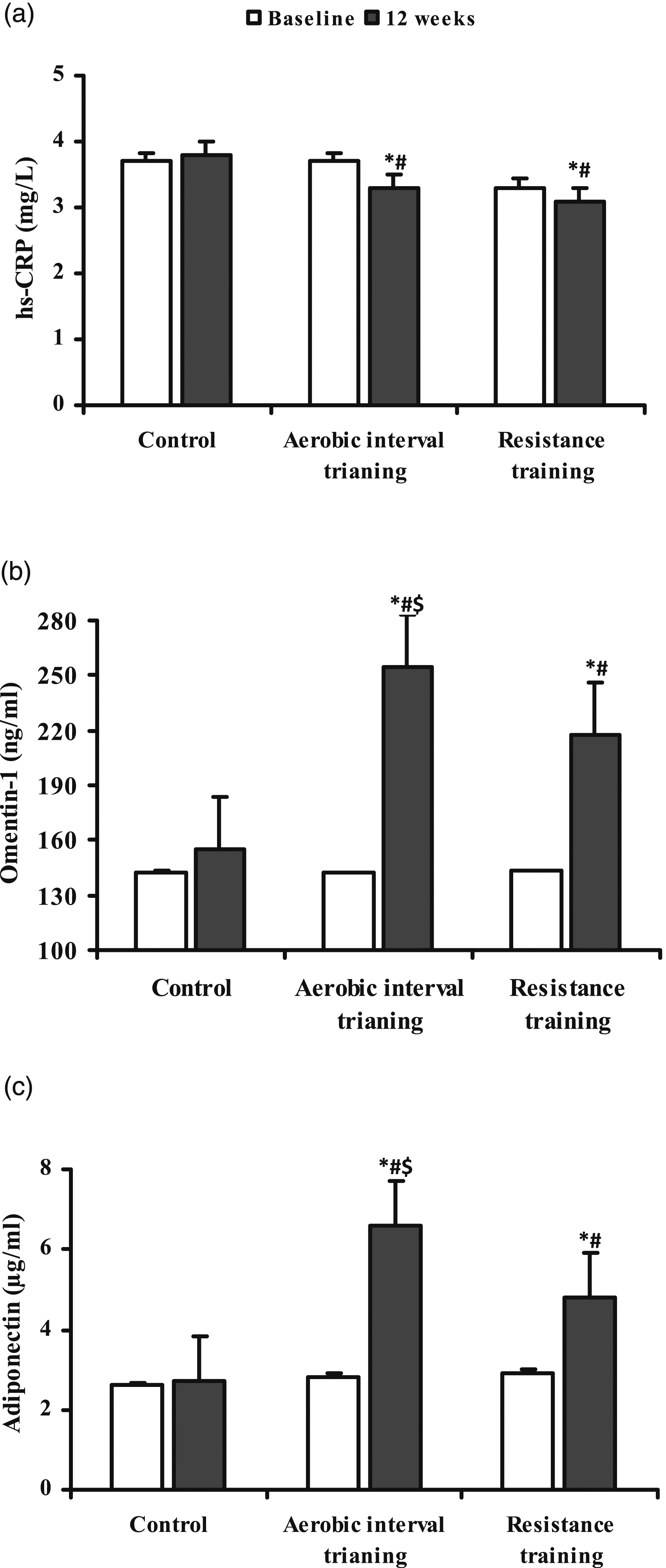
Changes in Lipids and Blood Pressure
Changes of Biochemical Variables From Baseline to the End of the Study (Mean ± SD).
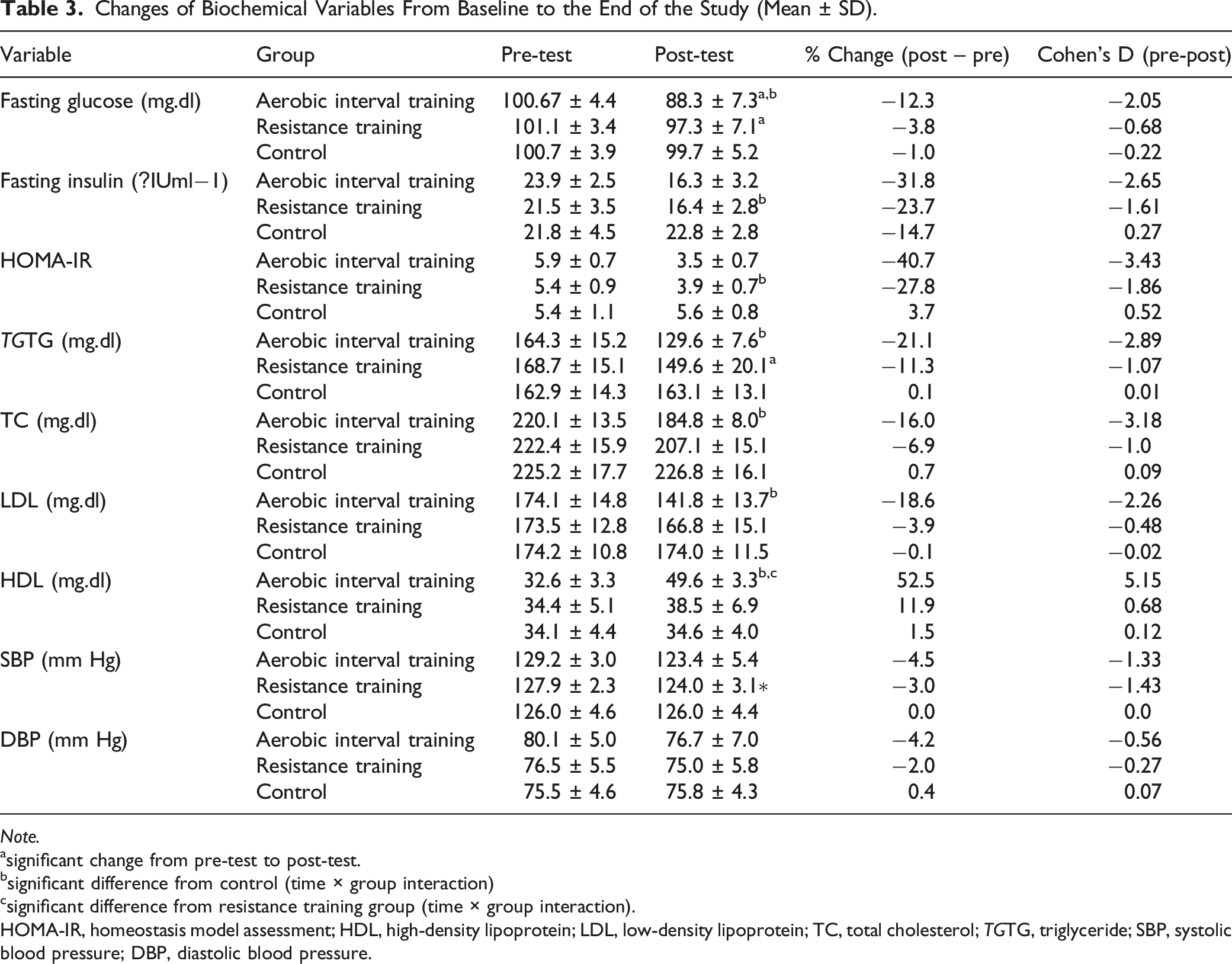
Note.
asignificant change from pre-test to post-test.
bsignificant difference from control (time × group interaction)
csignificant difference from resistance training group (time × group interaction).
HOMA-IR, homeostasis model assessment; HDL, high-density lipoprotein; LDL, low-density lipoprotein; TC, total cholesterol; TGTG, triglyceride; SBP, systolic blood pressure; DBP, diastolic blood pressure.
Changes in Fasting Glucose, Insulin, and HOMA-IR
Changes in fasting glucose, insulin, and HOMA-IR are demonstrated in Table 3. There were significant main effects of time for fasting glucose (p < .001, ηp2 = 0.589), insulin (p < .001, ηp2 = 0.707), and HOMA-IR (p < .001, ηp2 = 0.805). Moreover, there were significant group by time interactions for all 3 variables (fasting glucose: p < 0.001, ηp2 = 0.520; insulin: p < .001, ηp2 = 0.679; and HOMA-IR: p < .001, ηp2 = 0.771). Fasting glucose values were significantly lower in the aerobic interval training group compared to the control during week 12, while insulin and HOMA-IR values were significantly lower in the resistance training group compared to the control.
Changes in Peak VO2 and Body Composition
Table 2 shows changes in VO2peak and body composition variables. Interestingly, we observed a considerable increase in VO2peak. Conversely, we noticed a considerable reduction in body weight and body mass index after aerobic interval and resistance training compared with the control group (p < .01). However, despite the fact that there were significant effects of time on body fat percentage (p < .001, ηp2 = 0.412) and waist circumference (p < .001, ηp2 = 0.578), no significant differences were observed between groups.
Correlations
Plasma level of hs-CRP was negatively associated with omentin-1 (r = −0.573; p < .001), adiponectin (r = −0.614; p < .001), high density lipoprotein (r = −0.584, p < .001), and VO2peak (r = −0.597; p < .001), and positively associated with glucose (r = 0.392; p = .024), LDL (r = 0.560; p = .001), triglyceride (r = 0.435; p = .011), total cholesterol (r = 0.467; p = .006), waist circumference (r = 0.360; p = .040), systolic blood pressure (r = 0.390; p = .025), and diastolic blood pressure (r = 0.418; p = .016).
Plasma level of omentin-1 was positively associated with adiponectin (r = 0.757; p < .001), VO2peak (r = 0.693; p < .001), and high density lipoprotein (r = 0.560; p = .001), and negatively associated with glucose (r = −0.577; p < .001), low density lipoprotein (r = −0.502; p = 0.003), triglyceride (r = −−0.531; p = 0.001), total cholesterol (r = −0.585; p < .001), waist circumference (r = −0.497; p = .003), and systolic blood pressure (r = −0.431; p = .012).
Plasma level of adiponectin was positively associated with VO2peak (r = 0.699; p < .001), and high-density lipoprotein (r = 0.796; p < .001). In addition, plasma concentration of adiponectin was negatively associated with glucose (r = −0.566; p = .001), low density lipoprotein (r = −0.664; p < .001), triglyceride (r = −0.670; p < .001), total cholesterol (r = −0.800; p < .001), waist circumference (r = −0.450; p = .009), systolic blood pressure (r = −0.509; p = .002), and diastolic blood pressure (r = −0.414; p = .017).
Discussion
The present study investigated the effects of aerobic interval and resistance exercise training on hs-CRP, anti-inflammatory adipokines, and clinical outcomes in patients with metabolic syndrome. The study found that both exercise modalities improved hs-CRP, omentin-1, and adiponectin levels. However, the analysis of the exercise modalities revealed that aerobic interval training resulted in the most significant improvements in plasma omentin-1 and adiponectin levels. Additionally, the study observed a significant decrease in plasma levels of glucose, triglyceride, low density lipoprotein, and total cholesterol and a substantial increase in high density lipoprotein after 12 weeks of aerobic interval training in patients with metabolic syndrome. Importantly, the changes in high-density lipoprotein were significantly greater in the aerobic interval training group compared to the resistance exercise training group. These findings suggest that aerobic interval training may be a promising intervention for improving metabolic and inflammatory biomarkers in patients with metabolic syndrome.
Elevated serum levels of hs-CRP, a biomarker with high sensitivity to systemic low-grade inflammation (Lakka et al., 2005), have been found to be significantly associated with obesity, insulin resistance, and metabolic syndrome (Festa et al., 2000). This study demonstrates that physical exercise is effective in decreasing plasma levels of hs-CRP in patients with metabolic syndrome. The findings suggest that the anti-inflammatory impact is not contingent upon the exercise modalities, which inconsistent with previous large trials in subjects with impaired glucose tolerance or patients with type 2 diabetes and the metabolic syndrome (Alizaei Yousefabadi et al., 2021; Balducci et al., 2010). In their systematic review and meta-analysis, Alizaei Yousefabadi et al., (2021 discovered that exercise was linked to a significant reduction in CRP levels in individuals with metabolic syndrome. Notably, when studies were categorized based on exercise training modalities, aerobic exercise regimens demonstrated a significant decrease in CRP levels, while studies focusing on resistance training showed a non-significant decline in CRP levels. Additionally, a further investigation pertaining to this matter indicated that Pilates and resistance exercise regimens reduced hs-CRP levels in elderly individuals. However, the decline in hs-CRP levels was more pronounced in the Pilates cohort (Pestana et al., 2016). Moreover, a systematic review and meta-analysis conducted by Fedewa et al., (2017) found that exercise training is linked to a reduction in CRP levels, regardless of the age or gender of the person; nevertheless, the greatest improvements in CRP levels occur when there is a decrease in body mass index or %fat (Fedewa et al., 2017). Our findings are consistent with the present study, as in addition to a decrease in hg-CRP levels, we observed significant concomitant reductions in both body weight and body mass index following both interventions.
Resistance training has been postulated to diminish hs-CRP levels through the augmentation of muscle mass, which in turn mitigates systemic inflammation and reduces adipose tissue (Libardi et al., 2012). In addition, research has shown that chronic exercise training can enhance adaptation in both skeletal muscle and immune cells, leading to lower levels of inflammatory factors, such as hs-CRP, in individuals who engage in regular physical activity (Nicklas & Brinkley, 2009). Another potential mechanism that could contribute to the reduction of hs-CRP levels following exercise training is a decrease in body fat mass, particularly abdominal obesity (Pestana et al., 2016). On the other hand, some studies have shown no considerable alteration in the hs-CRP plasma levels after combined exercise training (Libardi et al., 2012) and low, moderate, and high-intensity exercise training (Huffman et al., 2006) in sedentary overweight to mildly obese men and women. The differences in participants’ characteristics and the type of exercise training could explain the inconsistency with the outcomes of this study.
Among adipokines, omentin-1 is a useful marker for understanding the pathophysiology of obesity and insulin resistance (Hossein-Nezhad et al., 2012) by reflecting anti-inflammatory effects in obesity-related cardiometabolic disorders (Zouhal et al., 2021). It has been revealed that omentin-1 increases insulin action by activating protein kinase B to improve insulin signaling and glucose uptake by adipocytes (de Souza Batista et al., 2007). In the current paper, compared with the control group, we observed increases in omentin-1 in both intervention groups, and demonstrated larger increases in the aerobic interval training group compared to the resistance training group. Moreover, significant correlations were observed between adiponectin, VO2peak, high density lipoprotein, glucose, low density lipoprotein, triglyceride, total cholesterol, waist circumference, and systolic blood pressure and omentin-1 after exercise. Our results are consistent with findings of Saremi et al. (2010) and Atashak et al. (2022), but differ from Nikseresht et al. (2016) and AminiLari et al. (2017). The reasons for this discrepancy are not clear, but it is possible that the different exercise protocols or participant characteristics in these studies may have contributed to the different findings. As AminiLari et al. (2017) conducted their study in type 2 diabetic middle-aged women. Faramarzi et al. (2015) also demonstrated that 12 weeks of rhythmic aerobic exercise at an intensity of 55%–85% of maximum heart rate, combined with core stability training, did not result in significant changes in plasma levels of omentin, glucose, and insulin, or insulin resistance index (Faramarzi et al., 2015). It is possible that discrepancies in the findings can be attributed to differences in participants, intensity, mode and duration of exercise interventions.
The precise mechanism by which aerobic or resistance exercises increase omentin-1 levels is not yet fully understood. Nevertheless, it is possible that an increase in fat-free mass and a decrease in body fat percentage may contribute to this effect. Studies have demonstrated that weight loss is associated with an increase in circulating levels of omentin-1 (Conde et al., 2011). In this study, significant reductions in body weight and body fat percentage were observed in both intervention groups. Additionally, factors such as adipocyte size may play a role in the regulation of omentin-1 levels, as it has been suggested that adipokine production and secretion are influenced by adipocyte size (Skurk et al., 2007). Another possible mechanism underlying the increase in omentin-1 levels is the improvement in insulin sensitivity that occurs with weight loss, which has been shown to be a potent stimulator of omentin-1 production (Conde et al., 2011). Finally, increased levels of omentin-1 resulting from aerobic interval and resistance training have been shown to improve insulin sensitivity and glucose metabolism (Yan et al., 2011) by promoting phosphorylation of PKB and AMPK (Liu et al., 2020). Conversely, studies have found a negative correlation between omentin-1 and insulin, glucose, and IL-6 levels (Castro et al., 2019; de Souza Batista et al., 2007), but a positive correlation with adiponectin levels (Kataoka et al., 2014). Thus, the elevation of omentin-1 induced by exercise may play a role in regulating glucose homeostasis and promoting lipolysis. Taken together, evidence suggests that high and moderate-intensity aerobic training may be particularly effective in promoting increased production and secretion of omentin-1. While resistance training has also been shown to increase omentin levels, there is limited evidence regarding the effects of different intensities of resistance training on omentin plasma levels.
Adiponectin has been shown to exert numerous physiological actions that are beneficial in mitigating inflammation, insulin resistance, metabolic syndrome, obesity, cardiac fibrosis (Tore et al., 2007), fat accumulation in the liver (Adiyaman et al., 2020; Bouassida et al., 2010), and atherosclerosis (Okamoto et al., 2002). Our findings reveal that both intervention groups increased plasma levels of adiponectin compared to the control group and demonstrated larger increases in the aerobic interval training group compared to the resistance training group. These findings are consistent with previous studies that have investigated the effects of different exercise regimens on adiponectin. For instance, Levinger et al. (2011) demonstrated that both acute aerobic and power exercise resulted in a significant increase in adiponectin levels in obese individuals with type 2 diabetes. Similarly, Kelly et al. (2012) reported a significant increase in adiponectin levels in obese adults following a 7-day period of vigorous aerobic exercise. Nevertheless, other studies have reported no significant changes in adiponectin levels following exercise interventions. For example, a 12-week study by Ando et al. (2009) with prediabetic patients and a 9-month study by Guo et al. (2011) with individuals with metabolic syndrome found that neither resistance exercise nor aerobic exercise had any significant effect on adiponectin levels. There are several possible explanations for the observed differences in the effects of exercise regimens on serum levels of adiponectin among studies. One important factor may be related to the frequency of exercise, as the frequency used in the current study was 3 sessions per week, whereas the studies by Guo et al. and Ando et al. employed exercise frequencies of one and 2 sessions per week, respectively. Moreover, the type of exercise regimen used in each study may also contribute to the observed differences in adiponectin levels. While Guo et al. and Ando et al. employed continuous aerobic exercise and combined of resistance and continuous training, respectively, our study used aerobic interval and resistance training. It has been reported that adiponectin may play a role in regulating glycemic control (Fisher et al., 2005). In our study, we found that changes in adiponectin levels were significantly associated with changes in fasting glucose.
Aerobic training has been demonstrated to induce weight loss and enhance lipolysis (Yamada et al., 2016), in part, through the upregulation of adiponectin secretion in both human and animal models (Sun et al., 2019; Zeng et al., 2007). As a result, adiponectin alters the concentration of high- and low-density lipoprotein and promotes glucose uptake in peripheral tissues (Jürimäe et al., 2006; Magherini et al., 2019). It is hypothesized that the stimulatory impacts on adiponectin secretion in both ovariectomized rat models of metabolic syndrome (Damirchi et al., 2010) and obese humans (Frankenberg et al., 2017; Lee et al., 2012) are due to lactate accumulation, increased adrenaline, glycogen depletion, and acidosis. To date, the precise impact of resistance training on adiponectin remains unclear. Nonetheless, there is evidence to suggest that resistance training can enhance muscle mass and reduce body fat percentage by elevating basal metabolic rate in individuals with metabolic syndrome (Strasser et al., 2010), augmenting blood circulation, and relocating adiponectin to the plasma (Gastebois et al., 2016).
The results of this study showed that 12 weeks of aerobic interval and resistance training resulted in a significant decrease in body weight and body mass index compared to the control condition. Waist circumference and body fat percentage decreased from pre-test to post-test. Consistent with our results, Ostman et al. (2017) reported a statistically significant change in body mass index with aerobic exercise. They also noted that while combined aerobic and resistance exercise is beneficial, it may provide a more limited range of benefits compared to aerobic exercise alone (Ostman et al., 2017). Our study revealed a significant improvement in peak VO2 in both the aerobic interval (greater than 1 MET; 3.5 mL kg−1 min−1) and resistance training (1 MET) programs after 12 weeks. This finding has clinical relevance and highlights the potential benefits of both exercise programs for improving cardiorespiratory fitness (Keteyian et al., 2012). A decline in age-predicted VO2peak has been linked to a higher risk of mortality in the general population (Lee et al., 2010) and in various chronic conditions (Sarullo et al., 2010; Sietsema et al., 2004), including diabetes (Reusch et al., 2013). As metabolic syndrome is frequently a precursor to diabetes, impaired VO2peak may be a concerning factor in individuals with metabolic syndrome. Moreover, our study also demonstrated that aerobic interval training had positive effects on both systolic and diastolic blood pressure, whereas resistance training only reduced diastolic blood pressure. Previous research has suggested that aerobic exercise may be more effective than resistance exercise in producing antihypertensive effects (Cornelissen et al., 2013; Cornelissen & Smart, 2013).
This study has several limitations that should be acknowledged. First, despite having a sample size with high actual power as described in the methods section, the small sample size may limit the generalizability of the findings. Second, only a 12-week intervention was analyzed, highlighting the need for longer-term, longitudinal studies in the future. Third, the study only included men, and therefore, the findings may not be generalizable to women. Fourth, while daily living activities and nutrient intake were assessed, the lack of precise monitoring of these factors during the 12-week intervention period is a limitation of the study. Therefore, we recommend that future exercise training programs be accompanied by rigorous monitoring of nutritional intake and daily living activities during the intervention period. Additionally, it would be valuable for future studies to include both men and women to confirm the generalizability of our findings.
Conclusion
In conclusion, our study demonstrated that both aerobic interval training and resistance training can improve the levels of anti-inflammatory adipokines and reduce inflammation in sedentary adults with metabolic syndrome. Additionally, exercise training led to significant improvements in lipid profiles, blood pressure, glucose metabolism, and body composition. These findings suggest that exercise training can be an effective non-pharmacological intervention for managing metabolic syndrome. However, our study has some limitations, such as the small sample size and the 12-week intervention period. Future studies with larger sample sizes and longer intervention periods are needed to confirm and extend our findings. Nonetheless, our study provides important insights into the potential benefits of aerobic interval and resistance training for the management of metabolic syndrome and highlights the importance of exercise as a key component of any comprehensive strategy for the prevention and treatment of metabolic disorders.
Footnotes
Acknowledgments
The authors acknowledge all the participants in this study who contributed their time to make the research possible.
Author Contributions
Kelian Gao contributed to acquisition, analysis, and interpretation drafted manuscript critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy Zhanguo Su contributed to conception and design critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy Junyan Meng contributed to acquisition drafted manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy Yuzhong Yao contributed to conception and design drafted manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy LiGuang Li contributed to acquisition and analysis critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy Yiping Su contributed to conception and design critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy Gholam Rasul Mohammad Rahimi contributed to conception and design critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the second batch of Industry-University Cooperative Education Project approved by Ministry of Education of China in 2022 (220800700292735); 2019 Yan ‘A University Graduate Teaching Change the Project (YDYIG2019035).
