Abstract
Up to 1 in 3 youth in the United States have a childhood-onset chronic health condition (CHC), which can lead to neurodevelopmental disruptions in cognitive functioning and brain structure. However, the nature and extent of structural neurobiomarkers that may be consistent across a broad spectrum of CHCs are unknown. Thus, the purpose of this study was to identify potential differences in brain structure in youth with and without chronic physical health conditions (e.g., diabetes, hemophilia). Here, 49 T1 structural magnetic resonance imaging (MRI) images were obtained from youth with (n = 26) and without (n = 23) CHCs. Images were preprocessed using voxel-based morphometry (VBM) to generate whole-brain voxel-wise gray matter volume maps and whole-brain extracted estimates of cortical surface area and cortical thickness. Multi-scanner harmonization was implemented on surface-based estimates and linear models were used to estimate significant main effects of the group. We detected widespread decreases in brain structure in youth with CHCs as compared to controls in regions of the prefrontal, cingulate, and visual association areas. The insula exhibited the opposite effect, with cases having increased surface area as compared to controls. To our knowledge, these findings identify a novel structural biomarker of childhood-onset CHCs, with consistent alterations identified in gray matter of regions in the prefrontal cortex and insula involved in emotion regulation and executive function. These findings, while exploratory, may reflect an impact of chronic health stress in the adolescent brain, and suggest that more comprehensive assessment of stress and neurodevelopment in youth with CHCs may be appropriate.
Introduction
Childhood-onset chronic physical health conditions (CHC) requiring ongoing care are increasingly common in the United States (Perrin et al., 2007). Over 40% of children and adolescents have at least one CHC (https://www.cdc.gov/healthyschools/chronicconditions.htm). Chronic conditions are conditions that have lasted or are expected to last at least a year and require ongoing medical attention and/or limit activities of daily living (Ghandour et al., 2022; Goodman et al., 2013). Chronic physical health conditions, such as asthma and diabetes, differ from behavioral or developmental conditions (Goodman et al., 2013). Chronic physical health conditions are associated with functional impairments, impact day-to-day activities, and lead to higher medical costs, poor long-term health outcomes (Varni et al., 2007), and increased risk of behavioral and mental health problems in youth, compared to peers (Stefko et al., 2022). Adolescents with CHCs have fundamentally different life experiences and life course trajectories, compared to healthy peers. They are more likely to have a diagnosis of anxiety, depression, or PTSD (Brady et al., 2017), experience increased stress (Ersig and Williams, 2018), and be exposed to adverse childhood experiences and other traumatic stressors (Kerker et al., 2015). Throughout the lifespan, their condition-related experiences can lead to chronic and acute condition-related stress exposures. Despite recent work identifying relationships between exposure to adversity, internalizing disorders (e.g., anxiety, depression, PTSD), and structural brain development (Heyn et al., 2019; Merz et al., 2018), little research has examined the neurodevelopmental impacts of CHCs.
Chronic childhood stress may alter brain structure and behavior and cognition (Blakemore and Choudhury, 2006; Eiland and Romeo, 2013), suggesting a mechanism by which childhood-onset CHCs could impact neurodevelopment. For example, poor CHC management increases risks for negative mental and physical health outcomes in later life (Lozano and Houtrow, 2018), while the CHCs type 1 and type 2 diabetes also increase risks of cognitive disorders in children and adolescents (Marissal-Arvy and Moisan, 2022). To our knowledge, very few studies of biomarkers of health outcomes using neuroimaging have been done in youth with CHCs, although a few have specifically investigated youth with diabetes. Studies of brain structure in adolescents with type 1 diabetes identified relationships between diagnosis- and condition-specific factors and brain morphology (Jaser and Jordan, 2021). Analyses of gray matter volume (GMV) using voxel-based morphometry (VBM) in youth with type 1 diabetes and moderate/severe diabetic ketoacidosis (DKA) identified faster growth in total GMV and GMV in the prefrontal and cingulate cortices than those with no/mild episodes of DKA (Aye et al., 2018). However, studies have not yet compared brain morphology between adolescents with different CHCs and their healthy peers.
Brain morphology can be characterized using a variety of techniques, including structural magnetic resonance imaging (MRI). One useful technique to characterize whole-brain differences in gray matter volume using data from these scans is VBM. However, VBM is limited in its ability to identify specific morphological correlates of surface-based metrics like cortical thickness (CT) and cortical surface area (CSA). These surface metrics have distinct developmental and anatomical mechanisms (Rakic, 1995). Studies of volumetric impairments alone are also limited, supporting the inclusion of both volumetric and surface-based metrics in neuroimaging studies of distinct phenotypes (Winkler et al., 2010). This approach has yet to be implemented in the study of neurodevelopment in youth with CHCs.
Brain plasticity, or the act of changing and reorganizing neural circuitry in response to internal and external cues, is a hallmark of adolescent development (Giedd and Rapoport, 2010; Sydnor et al., 2023). Clinical evidence of disruptions to brain structure during this marked period of change in youth with CHCs may further impact disease management and short- and long-term health outcomes. For example, identifying the effects of CHCs on adolescents’ brains will provide data that could help identify adolescents at greater risk of poor health outcomes (Eiland & Romeo, Teicher & Samson, Halfon & Forrest 2018). Altogether, there is a crucial need for more research to characterize brain morphology in adolescents with CHCs. The current study sought to be one of the first studies to comprehensively compare brain structure between adolescents with and without CHCs using comprehensive whole-brain volumetric and cortical surface analyses in order to provide data-driven evidence on the impacts of CHCs on structural neurodevelopment as compared to normative development and open the door for future investigations. We hypothesize that youth with childhood-onset CHCs have atypical gray matter structure in brain regions previously associated with stress and adversity in adolescence, compared to age- and sex-matched typically developing youth. Identifying differences in brain structure between adolescents with and without CHCs would support further research to identify factors that may be contributing to these differences and develop novel therapeutic interventions.
Methods
Sample & Data
Data for this analysis included 49 structural magnetic resonance imaging (MRI) datasets from youth ages 13–16.9 in 2 groups: adolescents with childhood-onset chronic health conditions (CHC, n = 26) and typically developing youth (TD, n = 23). Inclusion criteria for youth with CHCs were a current diagnosis of a childhood chronic physical health condition, defined as Type 1 Diabetes, food allergy, Celiac Disease, Cystic Fibrosis, or Factor VIII or Factor IX deficiency, 13–16.9 years old, and with an MRI in the past 10 years. These CHCs were selected because they are lifelong, currently unable to be cured, and generate stress from ongoing management and the need to be prepared to respond to emergent situations (Ersig, 2019; Ersig and Williams, 2018). Minimal demographic information was available for CHC youth and only included biological sex and age at the time of MRI. The second group included imaging data from TD youth originally recruited into the Youth PTSD study (Heyn et al., 2019), in which youth without past or current psychiatric or other serious medical conditions completed a structural MRI at a UW Health Clinic. The TD youth were age- and sex-matched with CHC youth to avoid possible confounds.
All T1 imaging data were pooled from one of two groups at University of Wisconsin-Madison (UW). For the first group, medical record and imaging data for UW Health adolescent patients with childhood-onset CHCs were identified by the UW Madison’s Institute of Clinical and Translational Research (ICTR) Clinical Research Data Service (CRDS) through clinical data base queries. Based on patients and images identified by CRDS, MRI exams were pulled from clinical PACS by the Radius group in the UW Madison Dept. of Radiology. Those exams were then de-identified using the RSNA’s Clinical Trials Processor (https://mircwiki.rsna.org/index.php?title = Contact) and coded to match the EHR data pulled by the CRDS group. This analysis was reviewed and deemed exempt by the UW-Madison Health Sciences Minimal Risk Institutional Review Board.
Image Acquisition, Quality Control and Scanner Harmonization Methods
Participant Demographics and Scanning Parameters.
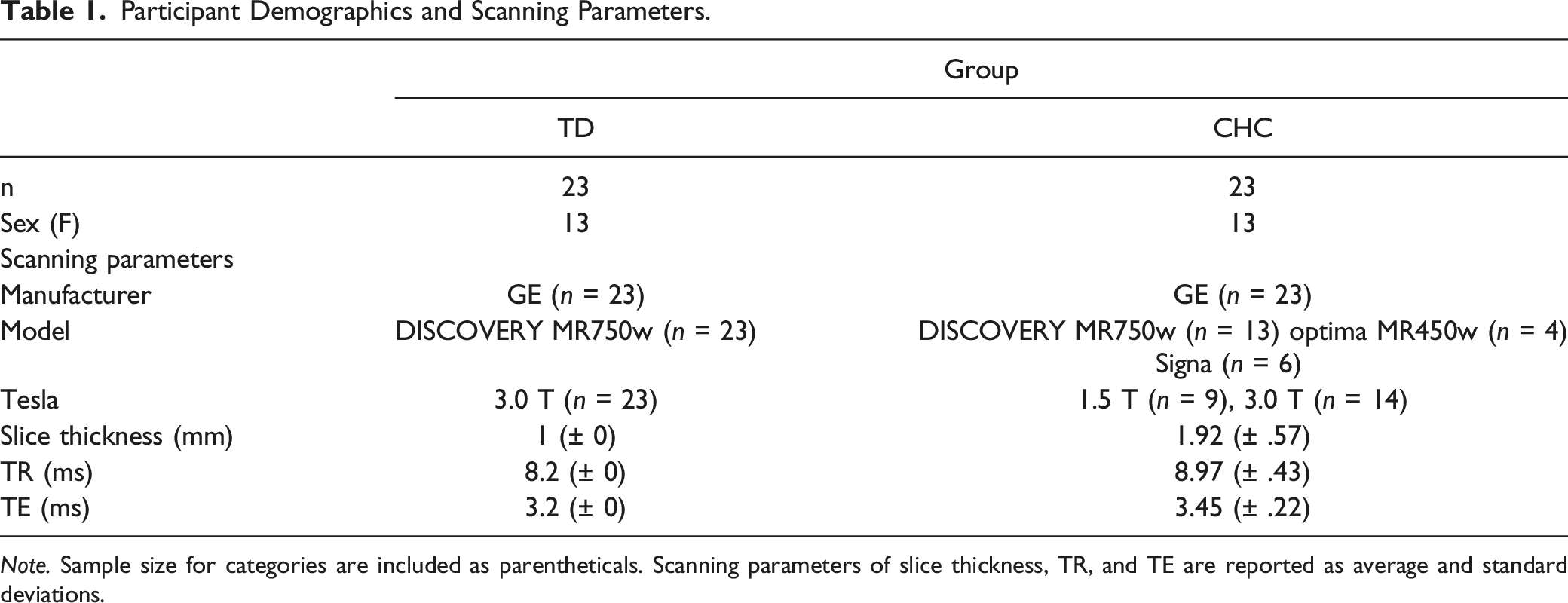
Note. Sample size for categories are included as parentheticals. Scanning parameters of slice thickness, TR, and TE are reported as average and standard deviations.
Surface-Based Morphometry
Surface morphometry extraction and subsequent cortical parcellation of all images was completed using Freesurfer Image Analysis Suite v6.0 using the cross-sectional recon-all default parameters (http://surfer.nmr.mgh.harvard.edu/). Technical details are available in previously published descriptions of this pipeline (Fischl et al., 2004). Here, we performed motion correction, skull stripping, subcortical segmentation, Talaraich transformations, and creation of final spherical surface maps for raw T1 images. Surface renderings were further parcellated into regions of interest using automated atlas-based parcellations using the Destrieux Cortical Atlas. Preprocessing resulted in whole-brain cortical surface area (CSA) and thickness (CT) estimates in 148 regions (74 per hemisphere) and estimated total intracranial volume (TIV) for each participant. While manual image inspection was not completed due to the preliminary nature of this study, images from two childhood-onset CHC participants failed reconstruction due to low image quality and were subsequently dropped, resulting in a total of 12% (n = 3) of the original CHC group who were removed from future analyses.
Voxel-Based Morphometry
Whole-brain VBM preprocessing was also performed using the Computational Anatomy (CAT12) toolbox (http://dbm.neuro.uni-jena.de/cat/) in SPM12 (Wellcome Department of Imaging Neuroscience, London, UK) in Matlab 8.3. All default parameters were used. Briefly, images were first spatially normalized to Montreal Neurological Institute (MNI) template space using a DARTEL algorithm, segmented into separate gray matter, white matter, and cerebrospinal fluid maps using tissue probability maps (TPM) included in SPM12, and smoothed using an 8 mm full-width half-maximum (FWHM) Gaussian filter. This preprocessing pipeline resulted in a smoothed gray matter volume (GMV) map with a final voxel resolution of 1.5 x 1.5 x 1.5 mm and an estimated total intracranial volume (TIV) for each participant.
Statistical Analyses
All statistical analyses of CT and CSA, as well as post-hoc testing and graphing, were completed in R (R Core Team 2016) and RStudio(R Studio Team 2021). Whole-brain rather than region-of-interest (ROI) analyses have been implemented across SBM and VBM analyses. To examine the relationship between surface morphometry and CHC status, group main effects on extracted CT and CSA estimates were estimated using linear models, controlling for age, sex, and scanner manufacturer. TIV was also included as a covariate in CSA analyses due to its association with total brain volume that may underlie any surface differences identified (https://surfer.nmr.mgh.harvard.edu/fswiki/eTIV). All estimates of CT and CSA, as well as other continuous predictors in the model (age, TIV), were rescaled prior to model estimations to ensure normal distribution. Due to the high volume of models run, multiple-comparison correction was employed using false discovery rate (FDR). In an effort to be conservative, all group main effects reported survived FDR correction across every effect and ROI model at pFDR < .01.
To examine the association between CHCs and gray matter volume, whole-brain VBM analyses were conducted to estimate the main effect of group using the 3dttest++ function in the Analysis of Functional Neuroimages (AFNI) software package. Age, sex, TIV, and scanner model were included as covariates. The scanner model was included as a covariate in whole-brain analyses and conservative whole-brain multiple comparison correction was applied. This approach uses cluster-level thresholds to control global false positive rates (FPR). An individual voxel-level threshold of p < .0001 was used, resulting in a whole-brain cluster threshold of 401 at a significance level of pFDR < .01. All reported group main effects survive whole-brain correction.
Results
Participant Demographics
Full participant demographic and scanner information can be found in Table 1. Following quality control measures, 46 youth were included in omnibus group analyses (TD, n = 23; childhood-onset CHC, n = 23). No group differences in age (t(35) = −.17, p = .86) or sex (X2 (1, N = 46) = 0, p = 1) were detected. In total, T1 images were collected across four GE MRI scanners: GE Discovery MR750w Site 1 (n = 23), GE Discovery MR750w Site 2 (n = 13), GE Optima MR450w Site 3 (n = 4), GE Signa Site 4 (n = 6). Acquisition parameters across scanners were similar: slice thickness (M = 1.88 mm; SD = 0.6 mm), TR (M = 8.9 ms; SD = .45 ms), and TE (M = 3.4 ms; SD = .22 ms). No group differences in image quality rating were identified.
CHC-related differences in cortical thickness and surface area
Main Effects of Group (TD Versus CHC) on Cortical Thickness, Cortical Surface Area, and Gray Matter Volume.
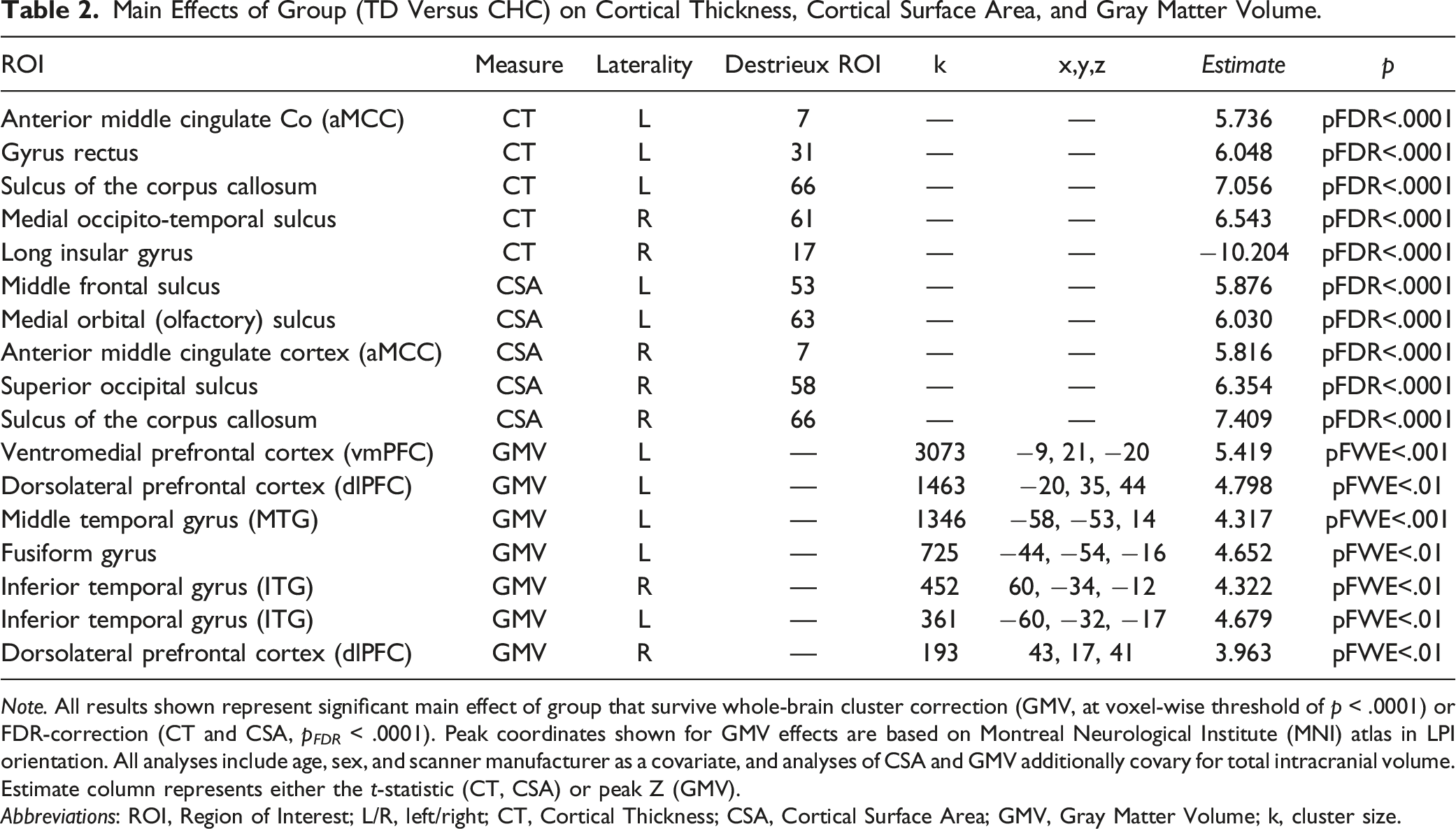
Note. All results shown represent significant main effect of group that survive whole-brain cluster correction (GMV, at voxel-wise threshold of p < .0001) or FDR-correction (CT and CSA, p FDR < .0001). Peak coordinates shown for GMV effects are based on Montreal Neurological Institute (MNI) atlas in LPI orientation. All analyses include age, sex, and scanner manufacturer as a covariate, and analyses of CSA and GMV additionally covary for total intracranial volume. Estimate column represents either the t-statistic (CT, CSA) or peak Z (GMV).
Abbreviations: ROI, Region of Interest; L/R, left/right; CT, Cortical Thickness; CSA, Cortical Surface Area; GMV, Gray Matter Volume; k, cluster size.
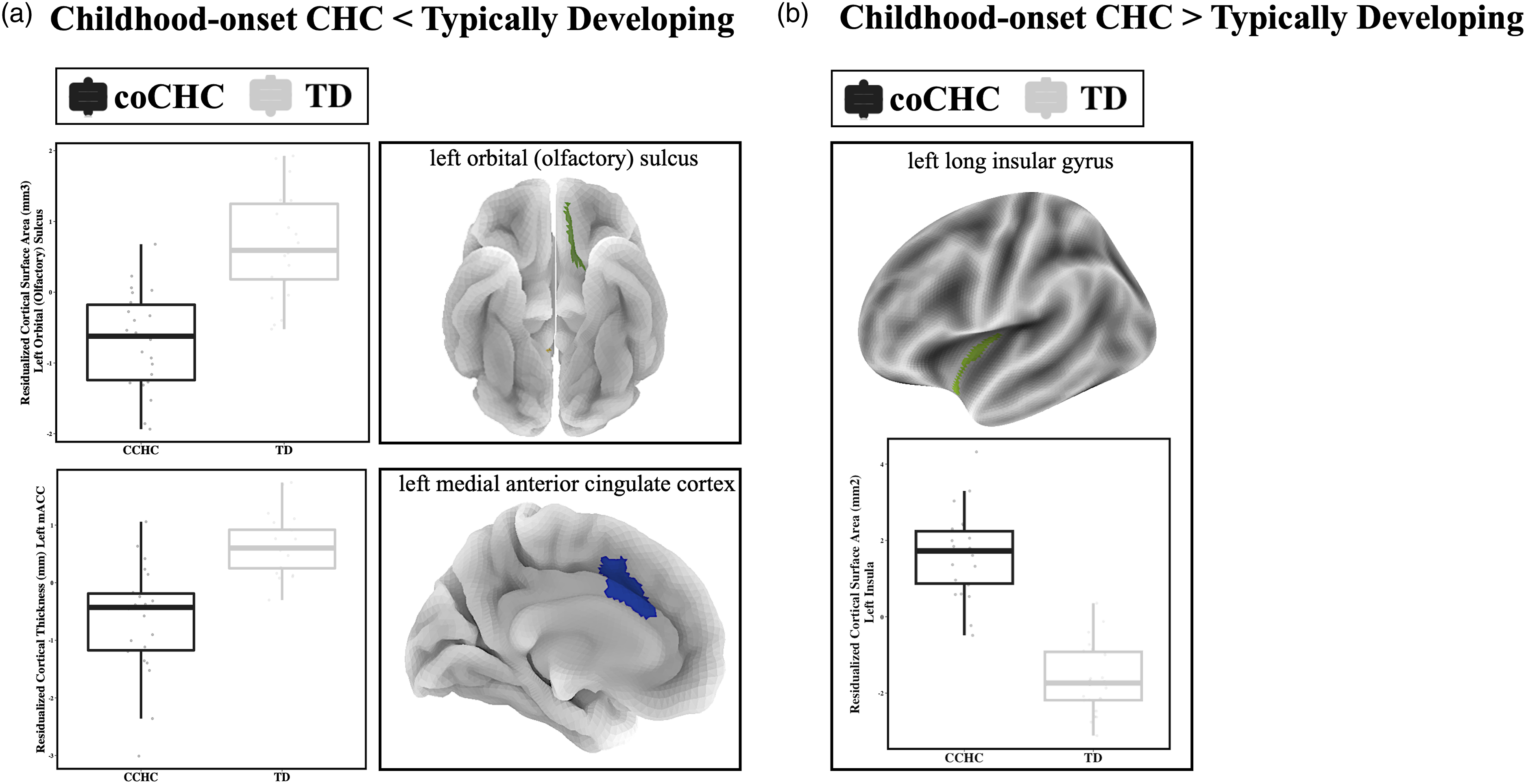
Differential cortical surface structure in adolescents with childhood chronic health conditions. Significant group main effects in cortical thickness and surface area when controlling for age, sex, and TIV (in CSA models) are displayed based on the direction of the effect (Panel A, CHC < TD; Panel B, CHC > TD). Boxplots represent residualized extracted ROI cluster estimates per participant, per group (TD = gray, childhood-onset CHC = black). ROIs are visually displayed on semi-translucent 3D-renderings of the left hemisphere. Abbreviations: CCHC, childhood chronic health condition; TD, typically developing; TIV, total intracranial volume, mACC, medial anterior cingulate cortex.
Following conservative site harmonization techniques, we detected five regions of CT and five regions of CSA that differ between youth with and without a childhood-onset CHC. Most brain regions, including bilateral portions of the cingulate cortex (Destrieux ROI 7, CT, t = 5.74, pFDR<.001; Destrieux ROI 7, CSA, t = 5.82, pFDR<.001), the orbital gyrus of the prefrontal cortex (Destrieux Atlas 63, t = 6.03, pFDR<.001), and bilateral sulci of the corpus callosum (Destrieux Atlas 66, left, t = 7.06, pFDR<.001; right, t = 7.41, pFDR<.001), exhibit patterns of decreased CT or CSA in childhood-onset CHC youth, compared to TD youth. Interestingly, three regions across all structural analyses showed the opposite pattern. The long gyrus and central sulcus of the insula (Destrieux ROI 17, t = −6.96, pFDR<.001) exhibits significantly increased CSA in childhood-onset CHC youth as compared to TD youth.
Exploratory CHC-related aberrations in gray matter volume as estimated by VBM
Finally, we characterized differences in GMV associated with childhood-onset CHC using whole-brain voxel-wise analyses and a conservative multiple-comparison correction. All results surviving correction are in Table 2 and visualized in Figure 2. Differential gray matter volume in adolescents with childhood chronic health conditions. Significant group main effects in gray matter volume when controlling for age, sex, scanner manufacturer, and TIV. Boxplots represent residualized extracted ROI or cluster estimates per participant, per group (TD = gray, CHC = black). Clusters are displayed on 2D renderings of the left hemisphere. Abbreviations: CHC, childhood chronic health condition; TD, typically developing; dlPFC, dorsolateral prefrontal cortex; vmPFC, ventromedial prefrontal cortex; TIV, total intracranial volume.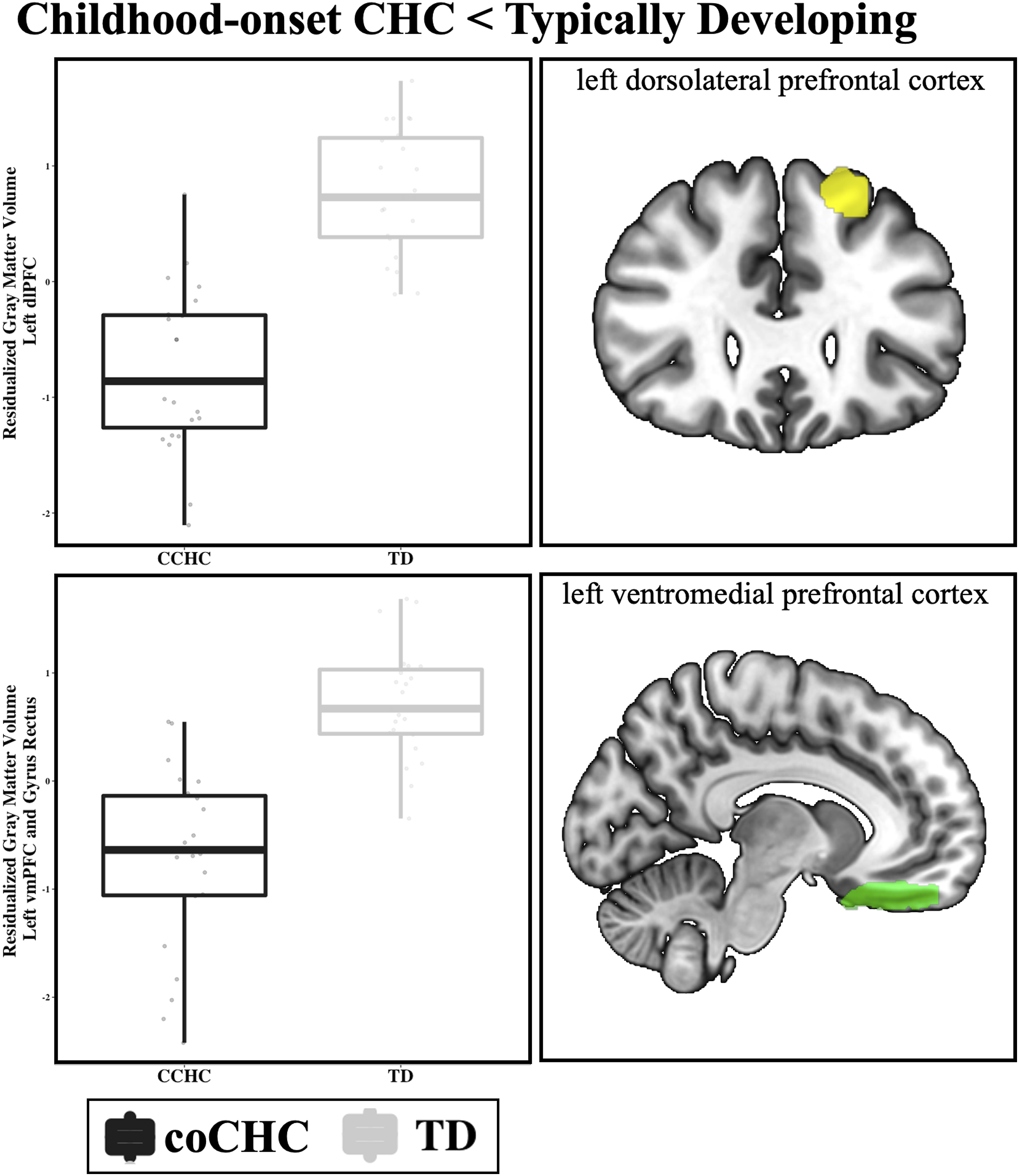
We identified significant group main effects in two critical prefrontal regions, the dorsolateral (dlPFC, k = 1338; x/y/z coordinates −20, 33, 44) and ventromedial (vmPFC; k = 1966; x/y/z coordinates −8, 20, −20) prefrontal cortex. In both regions, youth with childhood-onset CHC exhibit significantly decreased GMV as compared to TD youth. Similar patterns of decreased GMV in youth with CHCs were also identified in the bilateral middle temporal gyrus (MTG, L, k = 860, x/y/z coordinates −57, −54, 14; R, k = 431, x/y/z coordinates 61, −34, −12) and fusiform gyrus (k = 725, x/y/z coordinates −44, −55, 16).
Discussion
Substantial physical, emotional, psychological, and social change during adolescence, combined with increased brain plasticity, makes it a period of heightened vulnerability (Bick and Nelson, 2017). Disruption of these changes may affect short-term outcomes as well as adult biological and behavioral phenotypes (Lo Iacono and Carola, 2018). While many factors may contribute to these disruptions, including stress, hormones, and cognitive and social factors, one understudied factor may be living with a childhood-onset CHC. The current study compared brain structure in youth with a range of CHCs (n = 23) to TD youth (n = 23) using whole-brain voxel-based and surface-based morphometry. To our knowledge, this study represents a unique, trans-diagnostic characterization of neurodevelopmental markers of childhood-onset CHCs. Results are relevant to nursing practice and research and support a holistic approach to health and well-being in youth with CHCs (Atalla et al., 2020; Figuracion et al., 2023).
Following rigorous multiple-comparison correction and multi-site scanner harmonization techniques, we identified group differences in higher-order cortical regions associated with emotion regulation, memory, and decision-making, such as the prefrontal cortex, anterior cingulate cortex, and middle temporal gyrus, where CHC youth show lower gray matter structure as compared to TD youth. Conversely, in the insular gyrus, youth with CHCs show increased cortical surface area.
Our results indicate that youth with CHCs show extensive reductions in widespread cortical regions as compared to their typically developing peers. Specifically, we detected significant reductions in CSA in the lateral prefrontal cortex. Exploratory analyses of GMV identified reductions in the ventromedial and dorsolateral prefrontal cortex (vmPFC; dlPFC). The PFC exerts top-down control of cognitive and emotional regulation (Menon & D’Esposito, 2022). Subregions of the PFC have also been associated with distinct functions: the vmPFC is involved with implicit emotion and reward processing (Buades-Rotger et al., 2021), while the dlPFC is primarily involved with explicit cognitive control, decision-making, and working memory (Buhle et al., 2014). Impairments in PFC structure and function have been identified in studies of individual childhood-onset CHCs, including Type 1 diabetes (Foland-Ross et al., 2019) and cystic fibrosis (Roy et al., 2021), along with preliminary evidence of variability in global GMV in hemophilia A (Al-Huniti et al., 2019). Specific CHCs may induce structural abnormalities due to disease-specific states or symptoms; however, these could be related to shared factors, such as the potential for increased cumulative stress (Ersig and Williams, 2018). Although similar regions are identified across conditions, the direction of the effect is not consistent, which may be due to the relative infancy of this domain of research and small sample sizes. Nurse researchers could advance knowledge by developing larger, prospective studies of individual, social, and neurodevelopmental factors related to CHCs in adolescents, to explore regional similarities identified across diagnoses. Findings could be used to address the impact of living with a CHC on neurodevelopment.
While the PFC is a critical node for typical and atypical adolescent neurodevelopment, plasticity was evident in other significant regions. These include increased CSA in the insular cortex in CHC youth as compared to TD youth. The insula is associated with diverse physiological functions, including interoception, the innate sense of the physiological condition of one’s body states (Craig, 2002), and may also play a role in the pathology of psychiatric disorders and cognitive deficits (Goodkind et al., 2015). While insula structure has been associated with childhood internalizing disorders and traumatic experiences (Klabunde et al., 2017), this is one of few studies of childhood-onset CHCs specifically implicating insula structure (Naegel et al., 2014; S. Zhang et al., 2022, 2023; X.). Regional differences may represent an underlying common dimension of self-valuation of physiology across chronic illness diagnoses.
While the mechanisms of these neurobiomarkers of CHCs in adolescents are still unknown, chronic stress may be one possible mechanism linking CHCs and aberrations in brain structure, perhaps through an inflammation pathway. Compared to their peers, adolescents with CHCs experience increased chronic stress (Ersig and Williams, 2018). Chronic stress has previously been operationalized as an “environmental event or chronic conditions that objectively threaten the physical and/or psychological well-being of an individual” (Grant et al., 2004), which inherently encompasses childhood-onset CHCs. Research identifying biomarkers of childhood chronic stress has converged upon the PFC and ACC as critical nodes for differential structure and function (Sheth et al., 2017), changes that may be detectable into adulthood (Moreno-López et al., 2020). The PFC may be particularly vulnerable to uncontrollable stress during adolescence, during which powerful signaling pathways act to take the PFC “offline” as a compensatory response to focus energy on critical survival functions rather than top-down cognitive and emotional regulation (Datta and Arnsten, 2019). Youth with CHCs are continuously managing and dealing with a potentially dangerous chronic disease that is out of their control, which may lead to chronically deprioritizing and weakening the PFC’s response and normative connectivity, and detectably lower gray matter structure. This potential mechanism is further supported by the relationship between chronic stress exposure and susceptibility to psychopathology and impaired cognitive functioning (Hanson et al., 2012), patterns repeatedly identified in youth with CHCs (Compas et al., 2017). Neuroimaging of adolescents with CHCs can facilitate studies examining associations between factors such as chronic stress exposure and health outcomes (Figuracion et al., 2023).
One possible pathway leading to differences in brain structure may be a neurotoxic effect of inflammation. Chronic stress may induce increased inflammatory responses (Kim and Won, 2017), resulting in excitotoxicity, decreased synaptic plasticity, apoptosis, and neuron death (Doney et al., 2022; Lanser et al., 2020). Some neurotoxic effects have been hypothesized to specifically impact brain regions involved in emotion regulation, such as the PFC, amygdala, and hippocampus (Han and Ham, 2021; Kim and Won, 2017). Alternatively, many CHCs, including diabetes, asthma, and arthritis, are related to inflammation, which may lead to decreased brain structure regardless of the influence or severity of chronic stress. Further research is needed to identify whether the neurobiomarkers identified in the current study are unique to the experience of a childhood-onset CHCs or a by-product of generalized chronic stress, and what role the inflammatory response plays in either the CHCs themselves or via a stress response pathway. Future research could explore integrating stress measurement and interventions to decrease chronic stress into clinical care of youth with CHCs.
The current study has several limitations. We were unable to collect specific data on demographics or illness characteristics that may play an important role in the morphological effects identified, including diagnosis, age of onset, the extent of disease, or success of disease management. Also unknown are youth experiences of stress or any other social determinants of health (e.g., household income, parent education). These key factors help explain the extent of atypical neurodevelopment function in child-onset chronic illness (Armstrong, 2006) and should be included in future investigations. While we included all age- and sex-matched CHC participants with usable data to the best of our ability, there is a critical need for larger sample sizes. MRI scans at different locations with different scanners may also limit our analyses. This limitation was addressed by using only comparable scanning sequences for the entire cohort. We also employed scanner harmonization techniques in cortical surface analyses and included scanner as a covariate in whole-brain omnibus VBM analyses to remove as much variance due to scanner manufacturer as possible.
Conclusion
This study represents a novel investigation of brain structure aberrations within a trans-diagnostic cohort of adolescents with childhood-onset CHCs. Following conservative correction and scanner harmonization, we identified unique structural variations in the PFC, cingulate, and insular cortices in CHC youth as compared to TD youth. Conversely, in the insular gyrus, youth with CHCs show increased CSA. This exploratory report highlights the need for more comprehensive investigations. Childhood-onset CHCs are already associated with poor childhood and adulthood outcomes, and differential brain structure and function are linked to these outcomes, representing a malleable biomarker for novel therapeutic interventions, especially during the critical period of brain plasticity throughout adolescence.
Implications for Nursing
Based on the results of this study, nurse clinicians could incorporate assessment of risk behaviors in youth with CHCs, as changes in brain structure may be associated with changes in behavior. Although this study did not directly examine the potential connection between increased chronic stress in youth with CHCs and changes in brain structure, nurse clinicians could also integrate stress assessment and use of interventions designed to mitigate stress and improve resilience among youth with CHCs.
Nurse researchers are optimally positioned to integrate neurological biomarkers into future studies (Atalla et al., 2020; Figuracion et al., 2023) to identify contributing factors and develop nursing interventions designed to address them. In populations at increased risk of poor health outcomes, such as youth with CHCs, neuroimaging can serve as a biomarker of the biological impact of various exposures over time. Measures could be used to monitor response to interventions designed to alter those exposures. In this study, changes in brain structure in youth with CHCs were located in brain regions commonly associated with the stress response. . Nurse researchers should examine the relationship between stress-related variables (e.g., stress exposure, perceived stress) and brain structure in youth with CHCs. This will help determine whether there is a biological impact of chronic stress on the developing brain in youth with CHCs. Exploring relationships between other factors and brain structure could also identify protective factors, which could then be assessed as the basis for future interventions.
Footnotes
Author Contributions
Heyn, S: Conception, Design, Data Acquisition, Data Analysis, Interpretation, Drafted Manuscript, Critically Revised Manuscript, Gave Final Approval, Agrees to be held accountable for all aspects of work, ensuring integrity and accuracy; Herringa, R: Conception, Design, Data Acquisition, Data Analysis, Interpretation, Critically Revised Manuscript, Gave Final Approval, Agrees to be held accountable for all aspects of work, ensuring integrity and accuracy; Ersig, A: Conception, Design, Data Acquisition, Interpretation, Critically Revised Manuscript, Gave Final Approval, Agrees to be held accountable for all aspects of work, ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding for this study was provided by the National Institute of Mental Health Career Development Award (K08 MH100267, to RJH; R01 MH117141, to RJH; R01 MH115910, to RJH), American Academy of Child and Adolescent Psychiatry Junior Investigator Award (to RJH), NARSAD Young Investigator Grant (to RJH), University of Wisconsin Institute for Clinical and Translational Research Translational Pilot Grant Award (NIH/NCATS UL1TR000427, to RJH). The project described was also supported by the Clinical and Translational Science Award (CTSA) program, through the NIH National Center for Advancing Translational Sciences (NCATS), grant UL1TR002373. Support for this research was provided by the University of Wisconsin-Madison, Office of the Vice Chancellor for Research and Graduate Education with funding from the Wisconsin Alumni Research Foundation. None of these funding sources had a direct effect in the design, analysis, or interpretation of the study results, nor in preparation of the manuscript. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
