Abstract
Objective
A 6-month longitudinal surveillance study of asymptomatic healthcare providers (HCP) was carried out at a large urban academic medical center in the United States to assess whether their job occupation with higher exposure risks to SARS-CoV-2 would equate with higher risk of contracting COVID-19 at the beginning of the pandemic before COVID-19 vaccines were available.
Methods
A longitudinal cohort study design was used to collect and analyze immunological and virological monitoring data and self-report survey assessments of personal protective equipment (PPE) availability, adherence to infection control guidelines, and time spent on COVID-19 wards.
Results
Among 289 eligible participants, SARS-CoV-2 exposure risk was high with 48–69% participants working in COVID-19 units and more than 30% of them caring for COVID-19 patients. However, the seroconversion rate was low with only 2.1% of participants developing humoral or cellular immunity against SARS-CoV-2.
Conclusion
Our study findings suggest that, for this HCP cohort working at a large urban academic medical center, a low incidence of SARS-CoV-2 infection could be maintained under conditions of strict infection prevention protocols and reliable PPE availability.
Introduction
Healthcare providers (HCP) are at higher risk of exposure to SARS-CoV-2 than the general population (Houlihan et al., 2020; McCauley & Hayes, 2020; Nguyen et al., 2020). Moreover, because SARS-CoV-2 is a respiratory virus, aerosol-generating procedures increase the risk of transmission of SARS-CoV-2 to HCPs (Tran et al., 2012). Thus, HCPs working in COVID-19 units may be at greater risk of contracting SARS-CoV-2 compared to those who are not working in COVID-19 facing areas (Eyre et al., 2020; Houlihan et al., 2020; Ip et al., 2004; Kucharski & Nilles, 2020). As of July 20, 2022, almost 940,000 COVID-19 cases have been reported in healthcare personnel with close to 2300 deaths out of 527,243 cases for which death status is available (https://covid.cdc.gov/covid-data-tracker/#health-care-personnel). During the early stage of the pandemic (prior to July 2020), nurses constituted 30% of reported HCPs with COVID-19, making them the largest reported occupation with SARS-CoV-2 infection (Hughes et al., 2020). However, the relative risk of acquiring COVID-19 in a hospital setting versus community during the early phase of the pandemic remains poorly understood. This is further complicated by the fact that ∼70% of the infections are asymptomatic (Daria & Islam, 2022) and testing of asymptomatic workers was a workplace-dependent decision that did not get implemented until later in the pandemic.
The risk of SARS-CoV-2 infection among HCPs may be substantially reduced by infection control practices and policies in health care facilities. Early in the pandemic, guidelines were put in place to protect HCPs including diligent cleaning and disinfection, use of personal protective equipment (PPE), isolation, cohorting, and universal masking policy (Bielicki et al., 2020; Richterman et al., 2020). However, access to PPE in 2020 was challenging due to global supply shortages and increased demand, particularly during the early stages of the pandemic. PPE shortages were reflected in a nationwide May 2020 survey reporting 87% of nurses had to reuse a single-use disposable mask or N95 respirator and 27% were exposed to confirmed COVID-19 patients without wearing appropriate PPE (National Nurses United, 2020).
Objective
In this study to assess whether job occupation with higher exposure risks to SARS-CoV-2 would equate with higher risk of contracting COVID-19 at the beginning of the pandemic before COVID-19 vaccines were available, we carried out a 6-month longitudinal surveillance study in 2020 at the University of California Irvine Medical Center (UCIMC) in Orange County, CA, the sixth most populous county in the USA. The county experienced surges in COVID-19 hospitalizations during summer and fall 2020. For this study, asymptomatic HCPs were enrolled in late April 2020 and followed up until early November 2020, thus covering the summer surge. This study was initiated prior to the adoption of weekly asymptomatic screening at UCIMC. Nasopharyngeal swabs and blood collections were scheduled at four timepoints during this period for virological and serological monitoring. Blood samples were assessed for presence of SARS-CoV-2 specific binding and neutralizing antibody responses as well as SARS-CoV-2 specific T cell responses, while nasopharyngeal swabs were used to test for the presence of viral RNA. Immunological and virological data were then integrated with self-report survey assessments of PPE availability, adherence to infection control guidelines, and time spent on COVID-19 wards.
Materials and Methods
Study Setting and Population
This study was approved by the UC Irvine Human Research Protection committee. Data for this prospective observational study were collected between April 2020–October 2020 at UC Irvine Health’s Douglas Hospital, a 417-bed acute care academic medical center in southern California. The sample consisted of multidisciplinary clinicians practicing at the front lines caring for patients, including registered nurses, physicians, physical therapists, and non-licensed care facilitators.
Recruitment was achieved by visits of the designated units during 2 weeks in April 2020. Interested HCPs were emailed recruitment flyers, which included a link to an online REDCap survey. Listed exclusion criteria included: (1) signs and symptoms of COVID-19 (fever and shortness of breath), and (2) prior history of laboratory confirmed COVID-19 infection. Eligible HCPs were provided with informed consent and contact information for study investigators to ask questions. Consenting participants were asked to follow data collection procedures at 1–2-month intervals. Procedures at each time point included: (1) complete a 30-minute online questionnaire; and (2) schedule a visit to the study lab for specimen collection (blood draw and nasopharyngeal swab).
The online questionnaire collected participant demographic information such as job title, job setting, personal characteristics, and recent history of potential interactions related to transmission risk. Survey items included: occupational role, time spent in the unit, exposure to COVID-19 patients, intubation and other procedures performed, use and availability of PPE, and travel history. For occupation, participants were asked “What is your clinical role?” and provided with a dropdown list of occupational roles with an option to specify "Other” and type in their role. For exposure to COVID-19 patients in the hospital, participants were asked the following questions: (1) "Have you actively been assigned to care for patients diagnosed with COVID-19 in the past 30 days?”; (2) “Were there patient(s) diagnosed with COVID-19 in the unit(s) you worked in the past 30 days?”; and (3) “Have you performed or assisted with a medical operation with a high exposure risk to COVID-19 in the past 30 days?” For symptom history, participants were asked, “Have you had any of the following symptoms in the past 30 days?” and were provided with a list of symptoms to select. A question about N95 respirator availability was added after the first round of data collection. Cohort IDs were de-identified and the 10 participants positive for antibody or T cell responses were attributed numbers 1-10 for this publication.
Binding Antibody Titers (ELISA)
Binding antibody titers against Nucleocapsid (NP) and receptor binding domain (RBD) were determined by an in-house enzyme-linked immunosorbent assay (ELISA). ELISA is a standard method for serological testing and is widely used for detecting and measuring antibodies against SARS-CoV-2. ELISA tests have a higher performance for detecting SARS-CoV-2 antibodies than lateral flow immunoassays but have lower sensitivity than chemiluminescence assays (Gong et al., 2021; Vengesai et al., 2021). Sensitivity and specificity of commercial ELISA tests for serological diagnosis of COVID-19 have been estimated to be greater than 83% and 97%, respectively (Vengesai et al., 2021). Anti-SARS-CoV-2 IgG positivity can be detected after an average of 12 days after exposure (Lynch et al., 2021). IgG titers decline over time but can still be detected among most patients after several months (He et al., 2021).
Plates were coated overnight at 4°C with 1 μg/mL SARS-CoV-2 Spike-protein Receptor-Binding Domain (RBD) or SARS-CoV-2 Nucleocapsid Protein (NP) (GenScript, Piscataway, NJ) in 1X PBS. Plates were then blocked using blocking buffer (0.05% Tween-PBS 1X with 5% non-fat dry milk) for 1 h at room temperature followed by 3 washes with 0.05% Tween-PBS 1X (wash buffer). Heat-inactivated plasma samples were subjected to serial 3-fold dilutions in duplicate with a starting dilution of 1:50. Plates were incubated for 1.5 h at room temperature. Plates were washed before detection with HRP-conjugated mouse anti-human IgG (dilution 1:4000, BD Biosciences, Franklin Lakes, NJ) and were incubated for 1.5 h at room temperature. Plates were washed and developed with o-phenylenediamine dihydrochloride (Sigma-Aldrich, St Louis, MI). The reaction was stopped by 1 M HCl. The optical density was measured at 490 nm using a Victor3 TM plate reader (PerkinElmer, Waltham, MA). End-point IgG titers were calculated using log-log transformation of the linear portion of the curve, and 0.1 OD units as cut-off. For each plate, a positive control sample was used to normalize ELISA titers among assays. IgG titer threshold was set at 2 standard deviations above the mean of plasma samples collected prior to SARS-CoV-2 pandemic.
Focus Reduction Neutralization Test (FRNT)
While ELISA tests are useful for measuring antibody titers in the sera, they do not provide information about the capacity of the antibodies to neutralize SARS-CoV-2. The FRNT is a neutralization assay that can estimate the functional capacity of anti-SARS-CoV-2 antibodies in the sera (Vanderheiden et al., 2020). FRNT is faster, has higher throughput, and is more accurate than other neutralization assays, such as plaque assays or cytopathic effect inhibition assays (Vanderheiden et al., 2020). Serially diluted heat inactivated plasma (1:3) were combined with 100 PFU of SARS-CoV-2 isolate USA-WA1/2020 (BEI resources) for 1 h before transferring to Vero E6 cells (ATCC C1008, ATCC, Manassas, VA) in 96-well plates. After 1 h, 1% methylcellulose (Sigma-Aldrich, St Louis, MI) at a 1:1 ratio was overlaid on the infected Vero cell layer and plates were incubated at 34°C for 24 h. After 24 h, the plates were fixed with 10% neutral buffered formalin (VWR, Radnor, PA) for 1 h before washing and addition of ice-cold methanol (Thermo Fisher Scientific, Hampton, NH) with 0.3% of hydrogen peroxide (Sigma-Aldrich, St. Louis, MO). Plates were incubated at −20°C for 10 min, then at room temperature for 20 min. Plates were then rinsed 3X with water and blocked for 1 h with 1X PBS plus 5% milk before adding anti-SARS nucleocapsid antibody (Novus Biologicals NB100-56576) at 1:1000 in 1X PBS plus 5% milk. Plates were developed by adding HRP anti-rabbit IgG antibody (Biolegend, San Diego, CA) and True Blue HRP substrate. Plates were imaged on an ELISpot reader (Autoimmun Diagnostika GmbH, Strassberg, Germany). Each plate contained a positive neutralization control and a negative control. FRNT 50 was calculated by non-linear regression analysis using normalized counted foci on Prism 7 (Graphpad Software, San Diego, CA).
Enzyme-Linked Immunosorbent Spot Assay (ELISpot)
EliSpot is a widely used method for measuring cellular immune response to antigens by detecting T cell activation. ELISpot allows for the measurement of individual cytokine-producing cells, which is not possible using ELISA (Schwarz et al., 2022). 300,000 PBMC/well were added to precoated anti-human IFN-γ mAb ELISpot PLUS plates, MABTECH kit (Cincinnati, OH). Cells were then stimulated with 1 μg/well of SARS-CoV-2 peptides (15-mer with 9-mer overlap, Thermo Fisher Scientific) for 18 h at 37°C. Plates were then washed with PBS and biotinylated anti-human IFN-γ mAb (1 μg/mL) was added for 2 h at room temperature. After washing with PBS, plates were incubated for 1 h at room temperature with streptavidin-ALP. Following washing with PBS, BCIP-NBT-plus substrate was added, and the plates were developed until spots appeared. The number of SARS-CoV-2 specific IFN-γ secreting spot forming cells were counted in ELISpot reader (Autoimmun Diagnostika GmbH, Strassberg, Germany) using AID ELISpot 7.0 Software.
Statistical Analysis
Survey data were exported from RedCap into R v4.0.2 for analysis. Chi-squared tests and Fisher’s Exact tests assessed changes in PPE availability and COVID-19 exposure over time. Two-sided p-values <0.05 were considered statistically significant. Spearman correlation analyses were performed with GraphPad Prism software.
Results
Study cohort characteristics
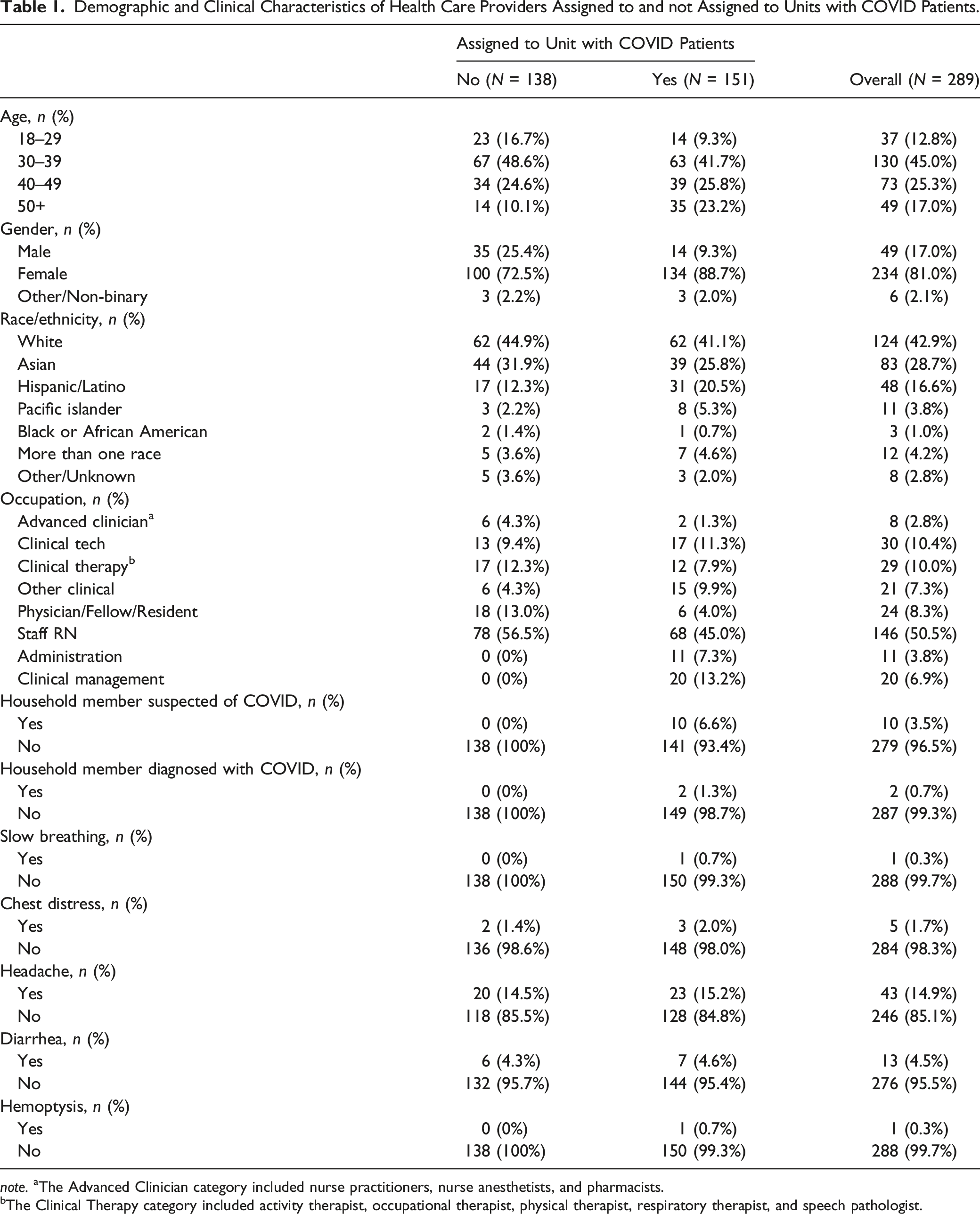
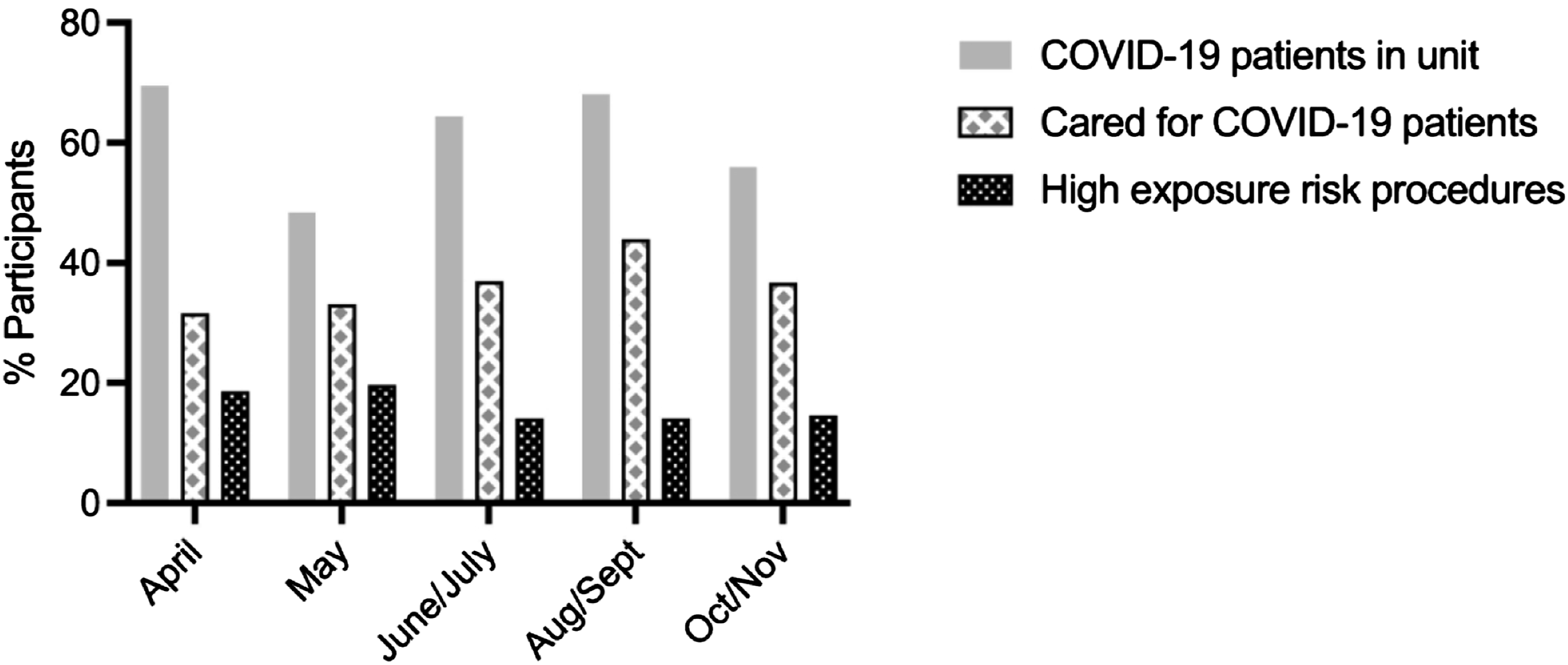
Overall, 289/299 study enrollees provided biological samples and were followed for up to 6 months. Most participants (84.1%; 243/289) made at least 2 sample collection visits, with 71.3% (206/289) providing samples at 3 or more visits (Figure 1). The majority of participants were female registered nurses (RN), aged 30–39. Participants were predominantly not of Hispanic or Latino ethnicity, and ∼43% identified as white (Table 1). At baseline, 3.5% individuals reported household members suspected of COVID-19 and 0.7% with household members diagnosed with COVID-19. Overall, 52.2% participants reported providing care for COVID-19 patients at any time during the survey period. Across follow-up time points, up to 43.8% of individuals reported that they provided hands-on care for diagnosed COVID-19 patients and up to 19.7% performed or assisted with a medical operation with a high exposure risk to COVID-19 (Figure 2). Among the participants working in COVID-19 units, COVID-19 symptom level was low (6% or less) except for headaches but those were also high in participants not working in COVID-19 units (15.2% vs. 14.5%) (Table 1). Notably, during the entire study, none of the nasal swab samples collected during the scheduled visits were positive for SARS-CoV-2 by PCR virological testing (data not shown). Study cohort characteristics. Demographic and Clinical Characteristics of Health Care Providers Assigned to and not Assigned to Units with COVID Patients. note. aThe Advanced Clinician category included nurse practitioners, nurse anesthetists, and pharmacists. bThe Clinical Therapy category included activity therapist, occupational therapist, physical therapist, respiratory therapist, and speech pathologist. Self-assessment surveys reporting longitudinal COVID-19 exposure risks. Participants (%) who worked in units with COVID-19 patients, participants (%) who cared for COVID-19 patients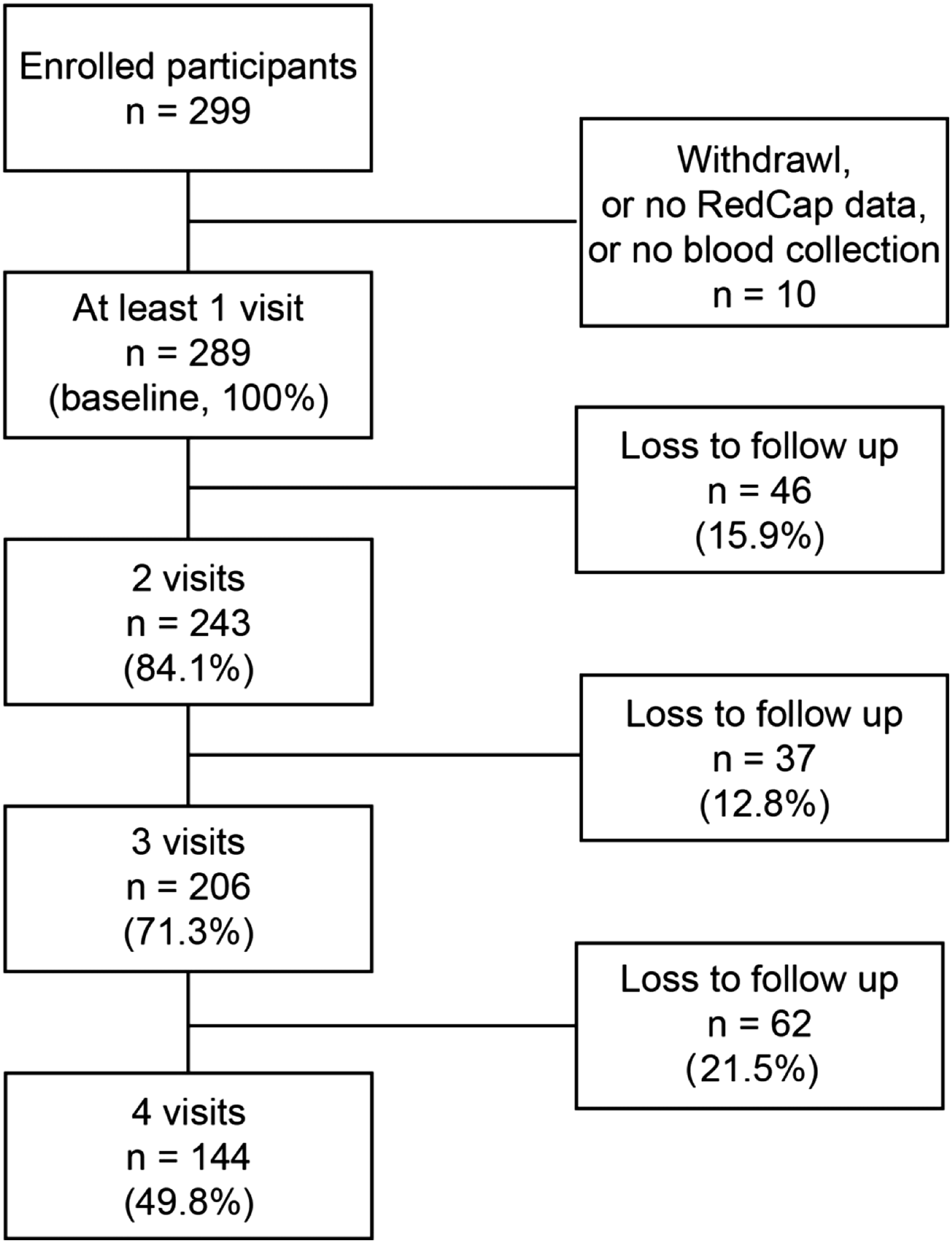
PPE Availability and Occupational Exposure Risk to SARS-CoV-2
SARS-CoV-2 being a respiratory virus transmitted by aerosols and small droplets, the survey focused on PPE components that help prevent transmission of such viruses, including face masks (N95 respirators and surgical masks) as well as face shields and goggles that protect mucosa. Figure 3 shows the percentage of participants who reported availability of PPE “most of the time” or “always”. Surgical masks were available for >88% of participants while N95 respirators were available for 84–88% of participants except in June/July when access decreased to 75%. Face shields and goggles became increasingly available with a gain of more than 20 points over the length of the survey. Similarly, protective clothing and round caps also became increasingly available, and around 50% of participants reported boot covers availability. In addition, gloves remained widely available over the entire study with more than 97% of participants stating that they had access to gloves most of the time or always (Figure 3). Self-assessment surveys reporting longitudinal availability of PPE. Participants (%) reporting PPE “always” or “most of the time” for surgical masks; N95 respirators (survey of N95 respirators was added after the first visit so no data are available for April); face shields; goggles; gloves; protective clothing; round caps and boot covers. Sample size of participants in each time period include: April (n = 124), May (n = 242), June/July (n = 157), August/September (n = 194), October/November (n = 161).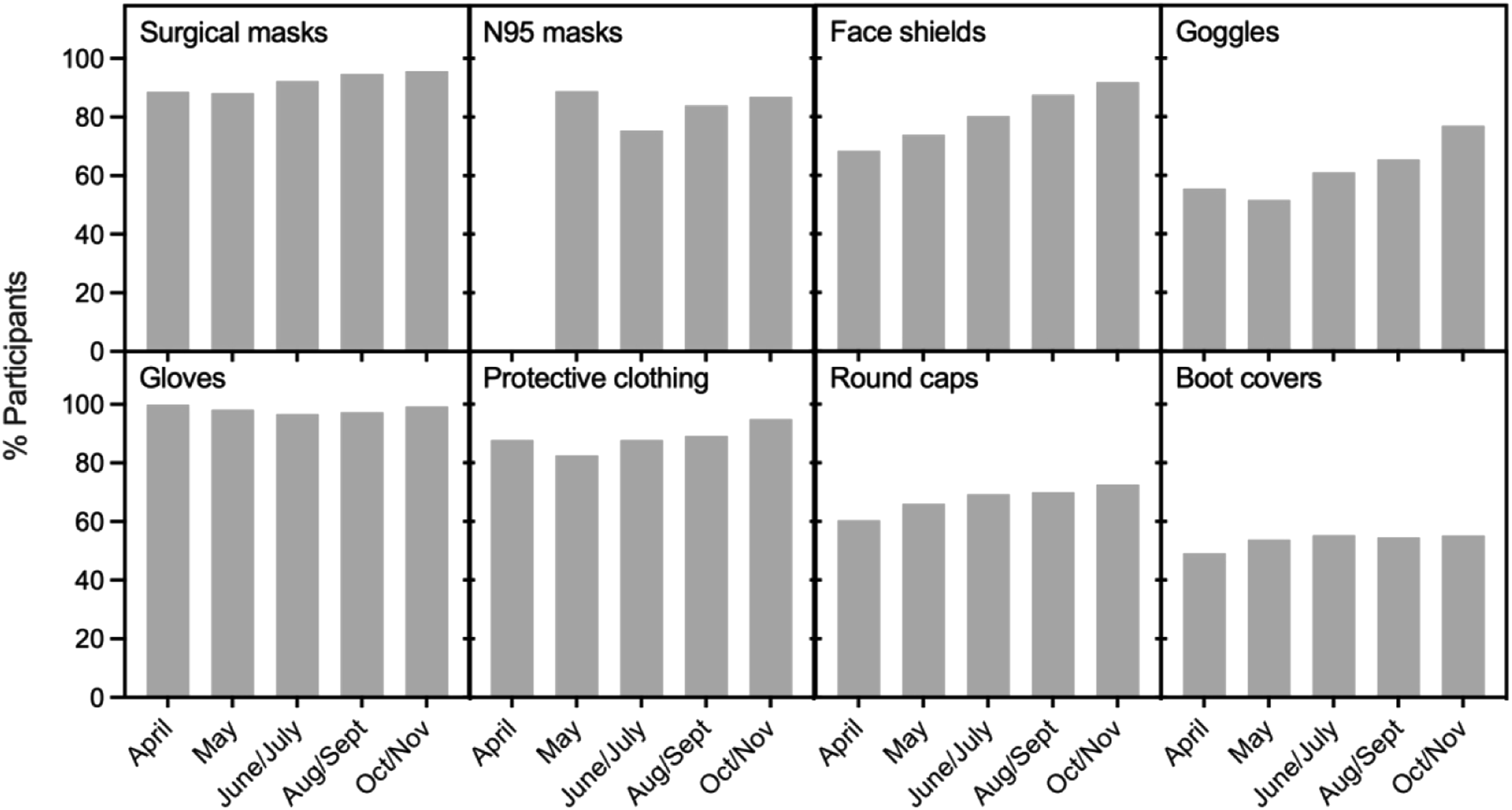
SARS-CoV-2-Specific Humoral and Cellular Responses in HCPs
Characterization of Biological Markers and Measurement Targets.

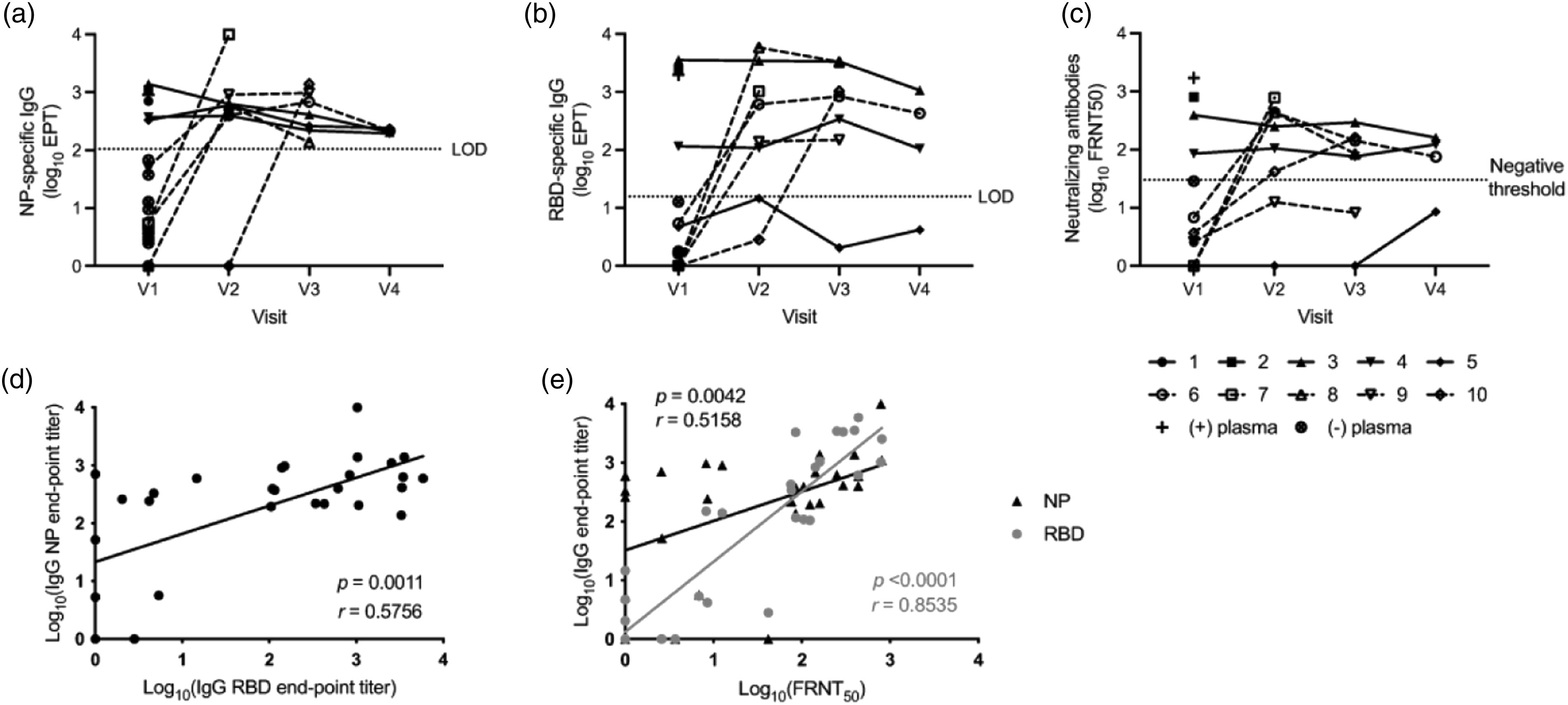
Humoral responses in healthcare workers who seroconverted. (A and B) Longitudinal IgG endpoint antibody titers directed against NP (A) or RBD (B) from HCP who seroconverted. (C) Longitudinal neutralizing antibody titers from HCP who developed binding antibody responses. Dashed lines in panels (A–C) are the limit of detection thresholds that were defined using pre-pandemic plasma samples. (D) Correlation between NP-specific and RBD-specific IgG antibody end-point binding antibody titers. (E) Correlation between antibody neutralization titers and IgG specific end-point binding antibody titers. P and r values in grey for RBD and in black for NP datasets.
We then assessed if HCPs with detectable antibody responses also generated functional SARS-CoV-2 specific T cell responses (Table 2). PBMC from the 10 participants of interest were stimulated ex vivo with overlapping peptide pools covering the entire viral proteome and the number of SARS-CoV-2-specific T cells producing IFN-γ was determined by INF-γ ELISpot. All 10 participants with measurable antibody titers also developed SARS-CoV-2-specific T cell responses which varied between subjects, visit timepoints and viral open reading frames (Figure 5). In addition, the kinetics of the response was subject-dependent with four participants (participants 3, 6, and 7) showing a decrease in T cell response over time while 2 participants (9 and 10) showed increasing response over time and one participant (8) showed an increase followed by a decrease in T cell response (Figure 5). In all 10 participants IFN-γ T cell responses targeted structural proteins M, N and E, which are among the main targets of CD4 and CD8 T cell SARS-CoV-2 responses (Grifoni et al., 2021). S protein is also a main target and T cell responses in 8 participants targeted it while 5 and 8 did not at the timepoints tested. Non-structural proteins Nsp4-Nsp5 were targeted in all 10 participants. The 3 strongest responders (2, 3, and 6) targeted Nsp6-13, S part 2, ORF3a-E-M, Nsp14/ORF6-7a-7b-8-10 and N. Interestingly, while participants 6, 7, and 8 had binding antibody titers below the assay detection limit at Visit 1 (Figure 4(A)-(B)), they had measurable T cells responses at that timepoint. Furthermore, participant 6 had very strong responses against all tested peptide pools (Figure 5). T cell responses in healthcare workers who seroconverted. Heatmap representing the frequency of antigen-specific T cell IFN-γ production determined by ELISPOT assay. Color scale is based on the lowest and highest value of the data included. Each row represents a participant’s visit and each column a specific pool of overlapping peptides.
Occupational & Community Exposure History among Health Care Providers with SARS-CoV-2 Seroconversion.

Discussion
It is widely believed that, due to their occupation, health care workers are at a significantly increased risk of SARS-CoV-2 exposure and infection. However, this risk is difficult to assess given that most infections are asymptomatic. In this study, we assessed the rate of asymptomatic, subclinical SARS-CoV-2 infection of HCPs during the summer 2020 surge at a large American urban medical center by integrating serological responses and self-reported risk exposure surveys to better understand the association between risk exposure and infection.
Overall, the participants’ exposure risk at UCI Medical Center was high as 48–69% participants worked in COVID-19 units and more than 30% participants cared for COVID-19 patients. Nevertheless, infection rate was low with only 2.1% seroconversion rate (5/243) during the study timeframe, likely due to good adherence to infection prevention measures and high PPE use as surgical masks were widely available throughout the study and N95 respirator availability increased over time. This low seroconversion rate is consistent with a study that was also conducted in Orange County during May-June 2020 which observed similar findings of a low seroprevalence of 1.13% (Brant-Zawadzki et al., 2020). Another study performed in April-May 2020 at an integrated healthcare system in Indiana also observed a low SARS-CoV-2 prevalence of 1.6%. Notably there was no statistically significant difference in seroconversion level between employees from high-risk settings and those in low-risk settings (Brant-Zawadzki et al., 2020; Hunter et al., 2020). In addition, both studies were conducted around May 2020 when their respective institutions also had guidelines already in place for PPE use and other infection prevention measures. In contrast, in a study conducted in London, England in March 2020, 25% of participants were seropositive at study entry and 20% of HCPs seroconverted during the study; notably, strict PPE guidelines were not introduced until April 1, 2020 in England (Houlihan et al., 2020).
Despite the occupational risk of exposure for HCP, because only 5 out of 243 participants seroconverted during this longitudinal study, we propose that the occupational risk does not appear to be higher than the community risk in surrounding Orange County at the time. Indeed, the infection level for HCP in other studies was no different from the concurrent infection rate in the general population (Brant-Zawadzki et al., 2020; Hunter et al., 2020). This would also explain that while we collected extensive survey data on seroconverted HCPs in terms of their occupational exposure risks during the study time frame, no reliable patterns emerged (see Table 3). Binding and neutralizing antibody levels in seroconverted HCP remained fairly stable during the length of the study. Interestingly, participants 1 and 5 were positive for N-specific binding antibodies but negative for RBD-specific antibodies. RBD is located in the S1 subunit of the spike protein and investigations of kinetics of SARS-CoV-2 antibodies revealed discrepancies with some studies showing that spike-specific antibody levels were higher than S1-specific antibodies (Fenwick et al., 2021) while others revealed earlier detection and higher RBD-specific antibody levels compared to S1-specific antibodies (Brochot et al., 2020), thus measuring anti-spike antibodies in our cohort could have yielded different results. In addition, in asymptomatic subjects, N-specific antibody levels were higher than S1- and S2-specific antibody levels and in symptomatic patients N-specific antibody titers reached higher levels earlier after symptom onset than RBD-specific and spike-specific titers (Brochot et al., 2020; Burbelo et al., 2020). The kinetics of functional T cell responses was more dynamic than the antibody responses and T cell response levels were also participant- and timepoint-dependent as observed by Kim et al. who noted peak T cell responses specific for S1, S2 and N antigens 1 month post symptom onset (Kim et al., 2021).
Limitations and Conclusion
Although data were collected rigorously, study limitations include collection from a single urban academic medical center and a small sample size that arguably did not represent the HCP population at large. Potential biases include voluntary participation of study population that conceivably was already aware of risk and therefore enrolled because of the regular testing provided. We also recognize a potential bias towards nurses, who constituted 50% of the sample, and the non-representation of HCPs involved with risky procedures, such as interventional pulmonologists. Despite these limitations, our study findings indicate that working as a HCP at a large urban academic medical center does not necessarily equate to higher risk when PPE is widely available.
Footnotes
Acknowledgments
We thank all HCPs who participated in this study. We would like to thank Dr. Izabela Coimbra Ibraim and Mr. Allen Jankeel for technical assistance.
Author Contributions
SS: Conceptualization, obtained funding, managed data and specimen collection.
MB: Conceptualization, obtained funding, participant recruitment.
DCM: Figure preparation, wrote first draft.
HV: Data analysis, co-wrote first draft.
BMD: Laboratory experiments, data analysis.
IM: Conceptualization, obtained funding, managed laboratory experiments.
All authors contributed to editing the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by COVID-19 Basic, Translational and Clinical Research Fund from UC Irvine Office of the Vice Chancellor for Research and UL1TR001414–06 (NIH/NCATS).
