Abstract
Objectives
This exploratory study aimed to evaluate the methaemoglobin fraction in cats with sepsis, non-septic sick counterparts and healthy controls, and to assess the feasibility of using the methaemoglobin fraction as a biomarker for predicting clinically relevant outcomes such as duration of hospitalisation and mortality.
Methods
Medical records were retrospectively searched for all cases of confirmed sepsis presenting to the intensive care unit (ICU) of two referral hospitals between September 2016 and January 2019 in institution A and between March 2019 and October 2024 in institution B, with a methaemoglobin measurement taken on admission. Records were also searched for cats presenting to the ICU of institution B between March 2019 and October 2024 that met 2/4 septic inflammatory response criteria without evidence of infection. The methaemoglobin levels of these cases and those of previously collected data from healthy cats (from both institutions) were compared with those of the septic population. Data from each institution were analysed separately.
Results
A total of 47 cats with sepsis were enrolled (23 in institution A and 24 in institution B), as well as 100 non-septic sick cats from institution B and 53 healthy controls (42 in institution A and 11 in institution B). Data from each institution were analysed separately. The median methaemoglobin fraction was significantly higher in control cats compared with septic cats in institution A (1.7%, 95% confidence interval [CI] 1.6–1.9 vs 1.1%, 95% CI 0.7–1.5; P = 0.03). There was no significant difference in median methaemoglobin levels between combined septic survivors vs non-survivors (0.5%, 95% CI 0.4–0.5 vs 0.9%, 95% CI 0.4–0.5; P = 0.5).
Conclusions and relevance
This exploratory study was unable to demonstrate a significantly increased circulating methaemoglobin fraction in cats with sepsis or an association between methaemoglobin fraction and survival outcome. These findings support the feasibility of further research and highlight the need for adequately powered standardised multicentre studies to clarify the biomarker’s clinical utility.
Plain language summary
This study evaluated the levels of methaemoglobin (a form of haemoglobin that cannot carry oxygen) across three groups of cats: those diagnosed with sepsis (a severe infection causing whole-body inflammation), sick cats without evidence of infection and healthy control cats. The study also evaluated whether higher methaemoglobin levels were linked to a longer hospital stay or a greater risk of death in cats with sepsis. The study reviewed historical medical records from two veterinary referral hospitals of cats that presented to the intensive care unit. The results demonstrated that cats with sepsis did not have higher methaemoglobin levels compared with sick cats or healthy cats, and this was not linked to survival or length of hospitalisation.
Introduction
Methaemoglobin is formed when the iron in haemoglobin is oxidised from the ferrous (Fe2+) to the ferric state (Fe3+). 1 Although low levels are normally produced by auto-oxidation of haemoglobin, methaemoglobin is incapable of carrying oxygen and, consequently, increased levels are associated with cellular hypoxia. 2 Therefore, methaemoglobin levels in humans are usually kept below 1% through the counterregulatory cytochrome b5-methaemoglobin reductase pathway.3,4 This pathway reduces methaemoglobin to functional haemoglobin using nicotinamide adenine dinucleotide produced during glycolysis. Similar values have been reported in cats with mean methaemoglobin fractions of 0.35 ± 0.1%. 5 Various mechanisms for elevated methaemoglobin fraction exist, including increased oxidative stress (eg, endogenous versus exogenous), decreased clearance of oxidants (due to hepatic or renal dysfunction), congenital enzyme dysfunction and acquired enzyme dysfunction (such as acidaemia, mitochondrial dysfunction, renal failure or liver failure). In sepsis, many of these pathways can converge, alongside nitric oxide dysfunction, to cause methaemoglobinaemia. 3
Inflammation results in the production of reactive oxygen species such as nitric oxide (NO), which oxidises haemoglobin to methaemoglobin. 1 Increased methaemoglobin not only reduces oxygen-carrying capacity but also shifts the oxygen dissociation curve leftward, further impairing oxygen release to the tissues. 5
Large amounts of NO are produced at the endothelial lumen during sepsis, which in turn increases methaemoglobin concentration.6,7 It has been found that in humans with sepsis or septic shock, methaemoglobin levels are significantly higher than their non-septic counterparts.6,8 Methaemoglobin fraction is a useful marker of sepsis in neonatal children, and levels above 2% are associated with mortality. 7
Methaemoglobinaemia has been reported in the veterinary literature in association with toxin exposure and congenital enzyme deficiencies.9 –14 A recent study found that in dogs with sepsis, the methaemoglobin fraction is increased, and a methaemoglobin level exceeding 2.2% is associated with mortality. 15 Cats are particularly susceptible to methaemoglobinaemia owing to their chloride-dependent mechanism for oxygen release, increased number of sulfhydryl groups and inability to metabolise and conjugate some drugs.13,16 Despite this, to the authors’ knowledge, this is the first study examining the relationship between methaemoglobin concentrations and sepsis in feline patients.
This exploratory study aimed to evaluate the methaemoglobin fraction in cats with sepsis, non-septic sick (NSS) counterparts and healthy controls. This study also aimed to assess the feasibility of using the methaemoglobin fraction as a biomarker for predicting clinically relevant outcomes, such as duration of hospitalisation and mortality, and to determine whether further prospective studies would be warranted.
We hypothesised that the circulating methaemoglobin fraction would be higher in cats with sepsis compared with their NSS counterparts and healthy controls, and that an increased methaemoglobin fraction would be associated with increased morbidity and mortality.
Materials and methods
This was a retrospective descriptive study. Ethical approval was granted by an ethical review committee (Social Science Research Ethical Review Board, reference URN SR2024–0172R2312).
Medical records from cats admitted to the intensive care unit of two veterinary referral hospitals were retrospectively searched for cases with confirmed sepsis and a methaemoglobin measurement taken at the time of admission. Cats were defined as septic based on meeting systemic inflammatory response (SIRS) criteria, as previously reported, 17 and a positive culture or intracellular bacteria noted on fluid cytology. Briefly this defined SIRS as two or more of four predetermined criteria, including a rectal temperature of 39.7°C or above (103.5°F) or under 37.8°C (100°F), heart rate greater than or equal to 225 beats/min or less than or equal to 140 beats/min, respiratory rate greater than or equal to 40 breaths/min or white blood cell count of 19,500/μl or greater or 5000/μl or less, or a band neutrophil fraction of 5% or above. Records were also searched for controls of NSS cats that met two or more of the four SIRS criteria without evidence of a bacterial infection or a negative bacterial culture. Records were searched from September 2016 to January 2019 in institution A and from March 2019 to October 2024 in institution B.
Cats were excluded from the study if they had a history of exposure to toxins or drugs that precipitated the development of methaemoglobinaemia, whether methaemoglobin was not measured on admission or whether medical records were incomplete. A detailed summary of septic case identification, exclusions and final enrolment is included in Figure 1. NSS patients were selected on a 2:1 basis, corresponding to approximately twice the number of enrolled septic cases.

Flow diagram for septic case selection/inclusion. MetHb = methaemoglobin fraction
Previously collected data from 42 healthy blood donor cats from institution A were chosen as our control group to determine normal methaemoglobin reference intervals (RIs), as there is no currently established RI for methaemoglobin in cats.18,19 In institution B, the control population consisted of 11 otherwise healthy cats presented for endoscopy because of foreign body ingestion/inhalation, as well as healthy blood donor cats.
Data collection
Patient parameters recorded included signalment, body weight, physical examination and point-of-care ultrasound findings. The duration of hospitalisation, the presence of any concurrent disease, the source of contamination, antibiotics administered in septic cases and the outcome were also included.
Venous blood gas analysis and the methaemoglobin fraction determined via CO-oximetry were measured using two venous blood gas analysers (ABL800 FLEX; Radiometer Medical ApS in institution A and Nova Stat Profile Prime Plus; Nova Biomedical in institution B). Venous blood samples for blood gas analysis and methaemoglobin measurement were collected in lyophilised lithium heparin syringes (A-Line 1 ml Critical Care Blood Collection Syringes; BD) from jugular or cephalic venipuncture and sealed anaerobically before analysis.
Additional diagnostic data included packed cell volume, total solids, haematology, serum biochemistry, plus culture and sensitivity results for confirmed septic patients.
Outcome was recorded as survival to hospital discharge, death or euthanasia. Cats euthanased for financial reasons were excluded from the study.
Statistical analysis
Statistical analysis was performed using freely available online software (https://www.statskingdom.com/). Data for each institution was analysed separately because of the different analysers used.
Data were expressed as standard descriptive statistics presented as median and range (min–max) based on their non-normal distribution. Normality was assessed graphically through visual inspection of the histogram and by using the Shapiro–Wilk test. A two-tailed Mann–Whitney U-test was used to evaluate differences between groups owing to the small sample sizes and non-parametric data. Box and whisker plots and calculations of Spearman’s correlation coefficients were used to assess correlations between continuous variables. Results were considered significant if P <0.05. A post hoc power analysis was performed using G*Power version 3.1 (two-tailed, alpha [α] = 0.05).
Results
A total of 47 cats (23 in institution A and 24 in institution B) with sepsis were enrolled, of which 28 (60%) were castrated males, 15 (32%) were spayed females, three (6%) were entire females and one (2%) was an entire male. The median age was 69 months (range 6–180) and the median weight was 3.9 kg (range 2–7.2). Median duration of hospitalisation was 6 days (range 1–16). The most common breeds included domestic shorthair (DSH) (n = 28), domestic longhair (DLH) (n = 6), Siamese (n = 3), Bengal (n = 2), Oriental Shorthair (n = 2), Abyssinian (n = 1), Birman (n = 1), British Shorthair (n = 1), Egyptian Mau (n = 1), Maine Coon (n = 1) and Russian Blue (n = 1). The causes of sepsis are summarised in Table 1. In the septic group, 30/47 (64%) cats survived to discharge.
Causes of sepsis
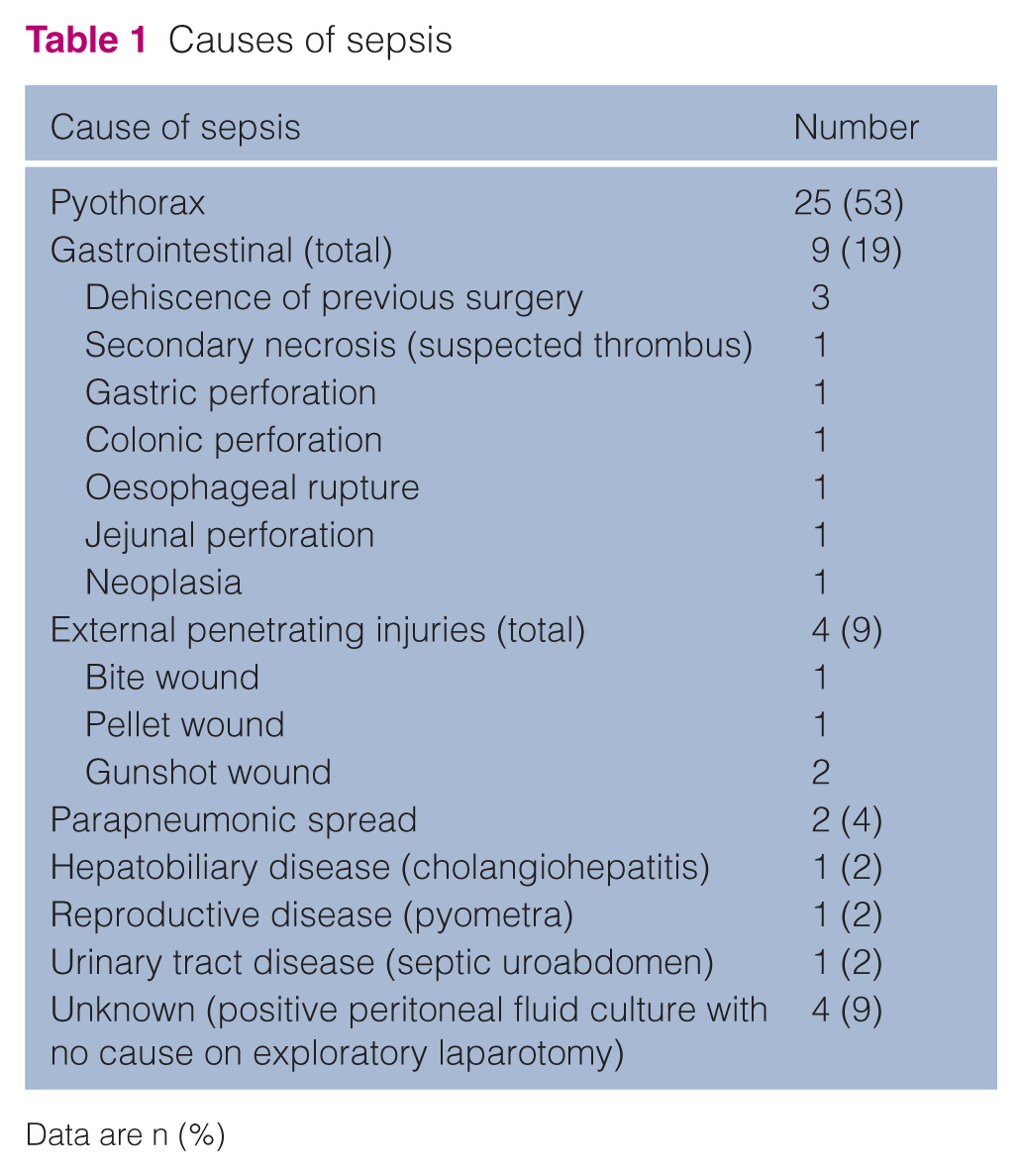
Data are n (%)
The NSS population of cats included 100 cases, of which 53 (53%) were castrated males, 30 (30%) spayed females, 10 (10%) entire males and seven (7%) entire females. The median age was 109.5 months (range 2–240) and the median weight was 4.1 kg (range 1.1–8.9). Median duration of hospitalisation was 2 days (range 1–19). The most common breeds included DSH (n = 62), British Shorthair (n = 8), DLH (n = 6), Bengal (n = 3), Burmese (n = 3), Maine Coon (n = 3), Russian Blue (n = 3), Birman (n = 2), Ragdoll (n = 2), Abyssinian (n = 1), Bombay (n = 1), Norwegian Forest (n = 1), Oriental Shorthair (n = 1), Persian (n = 1), Siberian (n = 1) and Tiffany (n = 1). The underlying diseases are summarised in Table 2. In this group, 59/100 (59%) survived to discharge.
Underlying diseases in non-septic sick patients

Data are n (%)
The 53 healthy control cases (42 in institution A and 11 in institution B) included 30 (57%) castrated males, 14 (26%) spayed females, five (9%) entire males and four (8%) entire females. The median age was 9 years.1 –13 The most common breeds were DSH (n = 36), DLH (n = 5), Maine Coon (n = 6), British Shorthair (n = 3), British Longhair (n = 1), Russian Blue (n = 1) and Arabian Mau (n = 1). Of these, 49/53 were blood donors, and the remaining four cases included cats presenting for endoscopy for a witnessed acute foreign body ingestion (n = 2) or inhalation (n = 1) without any clinical signs, unlike the patients in the NSS category and an otherwise healthy staff pet that had pre-anaesthetic bloodwork performed before a routine dental scaling (n = 1).
The main clinicopathological findings in cats with sepsis, NSS cats and healthy controls are reported in Table 3.
Descriptive statistics of selected clinical and clinicopathological variables in cats with sepsis, non-septic sick (NSS) cats and healthy controls

Data are median (Q1–Q3)
MetHb (%) = methaemoglobin fraction; N/A = not applicable; RI = reference interval; WBCs = white blood cells
The median methaemoglobin fraction was significantly higher in control cats compared with septic cats in institution A (1.7%, 95% confidence interval [CI] 1.6–1.9 vs 1.1%, 95% CI 0.7–1.5; P = 0.03). However, there was no significant difference between control cats compared with septic cats in institution B (0.5%, 95% CI 0.3–1.9 vs 0.5%, 95% CI 0.4–0.5; P = 0.4). There was also no significant difference between septic cats and NSS cats in institution B (0.5%, 95% CI 0.4–0.5 vs 0.5%, 95% CI 0.4–0.5; P = 0.8). These results are summarised in Figures 2 and 3.
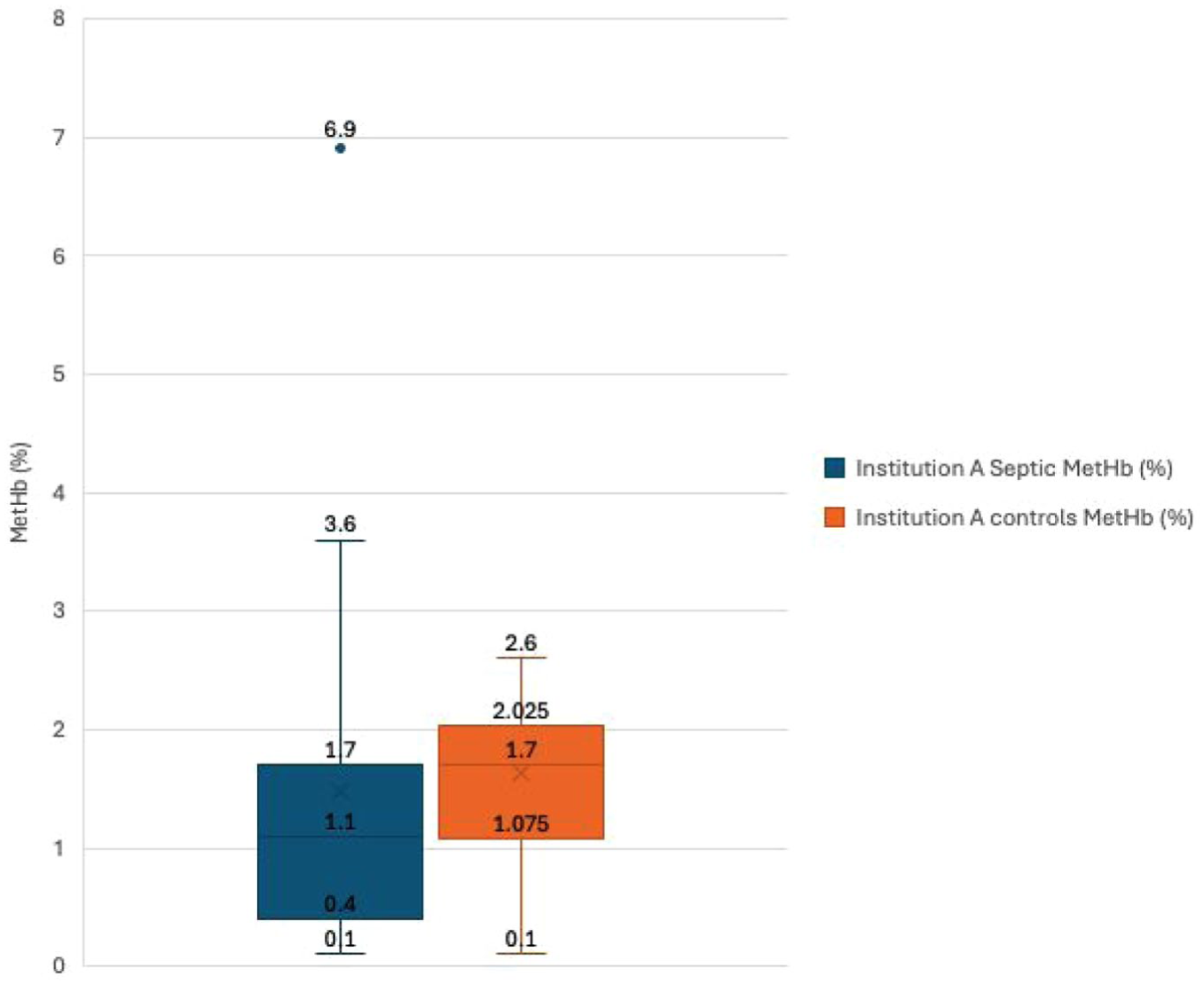
Box and whisker plot for methaemoglobin fraction (MetHb %) in septic vs control patients in institution A. Numerical values corresponding to the minimum, maximum, interquartile range and outliers are included on the figure

Box and whisker plot for methaemoglobin fraction (MetHb %) in septic vs non-septic sick (NSS) vs control patients in institution B. Numerical values corresponding to the minimum, maximum, interquartile range and outliers are included on the figure
There was no significant difference in methaemoglobin fraction between combined septic survivors vs combined non-survivors (0.5%, 95% CI 0.5–0.9 vs 0.9%, 95% CI 0.4–1.5; P = 0.5) (Figure 4).

Box and whisker plot for methaemoglobin fraction (MetHb %) in combined septic survivors compared with combined non-survivors (P = 0.5). Numerical values corresponding to the minimum, maximum, interquartile range and outliers are included on the figure
The data in Figures 2–4 overlap significantly between groups; in fact, the highest noted methaemoglobin fraction was noted in a survivor, indicating methaemoglobin lacks discriminatory ability in sepsis. However, given the limited statistical power of this study, it is not possible to determine if this overlap reflects a true absence of association or an inability to detect a small difference.
There was no correlation observed between methaemoglobin concentration and days of hospitalisation in institution A (Spearman’s rho [ρ] = 0.18, 95% CI –0.30 to 0.59; P = 0.43) or in institution B (Spearman’s ρ = −0.09, 95% CI –0.50 to 0.35; P = 0.67).
Post hoc and a priori power analyses were performed using G*Power (two-tailed, α = 0.05) to aid interpretation of the results, given the preliminary nature of the study. For our primary outcome of interest, whether methaemoglobin fraction was higher in septic cats compared with non-septic cats using a Mann-Whitney U-test comparison, a medium effect was observed for septic vs control cats from institution A (r ≈ 0.26; d ≈ 0.53). Based on this observed effect, approximately 60 animals per group would be required to achieve 80% power in future studies. The achieved post hoc power for the present comparison was approximately 50%. The achieved sample sizes were substantially smaller than the calculated requirements due to limited case availability, reinforcing the study’s exploratory nature and indicating that non-significant findings should not be interpreted as evidence of the absence of association.
Discussion
Sepsis is defined as life-threatening organ dysfunction caused by a dysregulated host immune response to infection.20,21 This dysregulation is characterised by overexpression of inflammatory mediators, including nitric oxide, which contributes to the hypotension and vascular hyporeactivity seen in septic shock.22,23 Higher nitric oxide levels are correlated with adverse outcomes in humans; however, measuring nitric oxide directly is challenging. Therefore, methaemoglobin has been utilised as a surrogate marker of nitric oxide. 24 Methaemoglobin is also thought to directly contribute to the pathogenesis of sepsis by increasing iron availability to pathogens, stimulating pro-inflammatory interleukins and increasing the expression of inducible nitric oxide synthase.1,25 Higher circulating methaemoglobin levels have been documented in septic people and dogs.6 –8,15 Methaemoglobin has not yet been investigated in cats with sepsis; therefore, in this small exploratory study, we aimed to evaluate the circulating methaemoglobin fraction in septic feline patients to determine its feasibility as a potential prognostic biomarker. However, in contrast to previous studies in people, we were unable to detect a significant difference in the methaemoglobin fraction between survivors and non-survivors.7,24 We were also unable to detect a correlation between a higher methaemoglobin fraction in septic cats and a longer duration of hospitalisation. However, this may be due to limited statistical power rather than a true lack of association. Interestingly, in contrast to our hypothesis, the methaemoglobin fraction was significantly higher in control cats compared with septic cats in institution A. Because of the study’s retrospective design, it is not possible to determine the exact cause. However, we hypothesise that this may be due to delays in sample processing, as sample processing is not standardised in these patients. Although samples are usually analysed immediately, processing may occasionally be postponed to prioritise more critically ill patients. This could have affected the methaemoglobin concentration, as once blood is sampled, autoxidation of haemoglobin into methaemoglobin can occur ex vivo.26,27 This unexpected finding suggests that the data from institution A may be unreliable, and therefore results should be interpreted cautiously. However, several other variables may alter methaemoglobin concentration, including haemolysis, which causes a true increase in methaemoglobin fraction, and lipaemia and icterus, which cause false elevations owing to measurement interference.28 –30 In addition, the co-oximeters used in this study cannot differentiate between methaemoglobinaemia and sulfhaemoglobinaemia; therefore, elevated levels cannot be excluded. Sulfhaemoglobin is another dyshaemoglobinaemia that can result from exposure to certain drugs, such as metoclopramide, or from endogenous hydrogen sulfide produced by intestinal bacteria.2,31
Multiple scoring systems have been suggested as diagnostic criteria for sepsis.21,32 In the present study, we opted to use the guidelines of criteria previously described by Babyak and Sharpe of two or more of four SIRS criteria 17 as opposed to three or more of SIRS criteria. 33 This was chosen because the previously proposed guidelines come from a single observational study of cats with severe sepsis diagnosed on post-mortem examination. 33 These guidelines are therefore highly specific for severe sepsis but may not be representative of all cats with SIRS or cats with less severe sepsis. This would likely have resulted in us missing cases in our population of interest; therefore, we used the adjusted definition of two or more of four SIRS criteria, similar to the guidelines used in dogs.34,35 Notably, in the septic population, only 18/47 (38%) cases met the definition of three or more of four SIRS criteria. Currently, the Sepsis-3 criteria have abandoned the SIRS criteria in favour of sequential organ failure scoring. 20 However, because of the retrospective nature of our data collection and consequential missing data, this would have been difficult to perform in our data set. Furthermore, the current veterinary definition of sepsis has not been updated in line with human guidelines at this stage. 21 This study provides preliminary insight into the use of methaemoglobin fraction as a potential biomarker in cats with sepsis; however, important limitations must be acknowledged. As a result of the retrospective nature of the study design, inevitably, some variables of interest were unavailable for all patients, and the study population was restricted to cases of sepsis presenting during the study period at the two participating institutions. Furthermore, because of incomplete or missing clinical variables in this data set, we were unable to apply illness-severity adjustment scores, such as the acute patient physiologic and laboratory evaluation (APPLE) scoring system. 36 Moreover, a major limitation of this study was the interinstitutional inconsistency observed between the two data sets. Owing to the different analysers used in each institution, we decided to analyse the data separately despite there being precedent for analysing data combined from different analysers. Furthermore, healthy blood donors were selected as the control population; however, the donor programme is less well established at institution B, resulting in fewer available donors, requiring additional healthy animals to be recruited, resulting in a more heterogeneous control group. Consequently, the sample sizes were relatively small, particularly in the control group at institution B, resulting in inadequate statistical power to detect true differences in our primary comparisons. Methaemoglobin was recorded at admission and at the highest point within 24 h; however, these values capture only a limited snapshot of what is a dynamic condition. Therefore, serial methaemoglobin measurements during hospitalisation may have yielded different results. Furthermore, given the heterogeneous nature of sepsis, and to examine the potential role of methaemoglobin as a biomarker of critical illness, we evaluated a cohort of critically ill patients without sepsis in institution B. However, because of the retrospective design of this study, a comparable cohort was not available at institution A. Patients with a known toxin exposure were excluded to minimise the risk of confounding variables. However, as a result of the retrospective nature of this study, we cannot exclude the presence of other factors that may affect methaemoglobin levels, such as lipemia. 37 These findings nevertheless demonstrate the feasibility of future research in this area. Ideally, subsequent prospective studies should incorporate a priori power analysis, use standardised protocols, adjust for disease severity and ensure appropriately sized control groups to better assess the clinical utility of this biomarker in feline sepsis.
Conclusions
This exploratory study did not demonstrate an increased methaemoglobin fraction in septic cats, and nor did it identify any association between methaemoglobin levels and outcome. Therefore, it is possible that methaemoglobin may not behave in cats the same way as in other species. However, as this study was likely underpowered to detect clinically significant differences, adequately powered studies are required to determine whether this is true and to clarify the prognostic significance of methaemoglobinaemia in cats with sepsis.
Footnotes
Author note
The data underlying this study is available from the corresponding author upon reasonable request.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore, additional informed consent for publication was not required.
