Abstract
Objectives
This study evaluated the therapeutic effects of subconjunctival injection of mesenchymal stem cells (MSCs) for treating deep corneal ulcers and corneal perforation in cats.
Methods
Of the 24 cats identified with a deep corneal ulcer or perforation, 17 received MSCs and medical treatment; among them, six had deep corneal ulcers and 11 had corneal perforations. Of the seven cats that received only medical treatment, three cats had deep corneal ulcers and four cats had corneal perforations. All were monitored with fluorescein staining at 3–4 days and weekly assessments after MSC injection until fluorescein-negative status was achieved.
Results
Outcomes were compared between cats treated with subconjunctival MSC injections and MSC-untreated cats. Based on the results, 15/17 (88%) cats treated with MSCs became fluorescein-negative within 1–3 weeks, whereas 2/17 (12%) of those receiving subconjunctival MSC injections achieved this outcome within 5–6 weeks. During the mean follow-up period of 146 ± 138 days (range 15–398) after MSC injection, recurrence of corneal perforations was observed in four cats that were ultimately completely healed. All deep corneal ulcers healed uneventfully. In the MSC-untreated group, 4/7 (57%) achieved fluorescein-negative status within 3–7 weeks, while another 2/7 (29%) experienced recurrent corneal perforation on days 3 and 58, requiring conjunctival graft surgery. Postoperatively, fluorescein-negative status was achieved 25 and 72 days after the initial treatment, respectively. Of the seven cats, one (14%) had recurrent corneal perforation that had healed on day 103.
Conclusions and relevance
These findings highlight the potential of MSC therapy in promoting corneal ulcer and corneal perforation healing in cats.
Introduction
Corneal ulcers are a common and potentially severe condition in cats, often resulting from trauma, infection or underlying systemic diseases.1,2 If left untreated or poorly managed, corneal ulcers can progress to complications such as perforation, scarring or even loss of vision. 3 Deep corneal ulcers, which involve more than 50% of the corneal thickness, can lead to blepharospasm, keratitis, corneal oedema, ocular pain and anterior uveitis. On the other hand, corneal perforation, involving corneal defects with total corneal depth, can result in clinical signs such as blepharospasm, corneal oedema, shallow anterior chamber, hypotony, hyphaema and iris prolapse. 4 Corneal perforation may occur as a progression of complicated deep corneal ulcers or suddenly as a result of trauma. 5 Failure to treat deep corneal ulcers or corneal perforation can lead to vision loss, phthisis bulbi or even enucleation. 3 Typically, conventional treatments for corneal ulcers involve the application of topical antibiotics and systemic medications, including antibiotics and anti-inflammatory agents, along with protective measures such as Elizabethan collars. 5 However, these treatments may be limited by slow healing times and challenges in owner compliance. Corneal surgery, such as a conjunctival graft or amniotic membrane transplantation, has been suggested. However, these techniques are sometimes denied because of the risk associated with anaesthesia.
Recent advances in regenerative medicine have introduced mesenchymal stem cells (MSCs) as a promising therapeutic option for corneal ulcers in several species.6 –8 MSCs have unique properties, including anti-inflammatory effects, immunomodulation and the secretion of growth factors that promote tissue repair and regeneration. 6 These characteristics make MSCs an attractive alternative or adjunct to conventional therapies for accelerating corneal wound healing. 6
The current study aimed to evaluate the effectiveness of subconjunctival MSC injection in accelerating the healing of corneal ulcers in cats compared with conventional medical treatments. By analysing healing times, recurrence rates and complications, this research provides insights into the potential benefits and limitations of MSC therapy in real-world clinical settings, as well as offering a comprehensive understanding of MSC therapy’s role in managing corneal ulcers in feline patients.
Materials and methods
History and ophthalmic examination
A total of 24 cats were recruited into the study. Nine cats presented at the Ophthalmology Unit, Kasetsart University Veterinary Teaching Hospital, Bangkok, Thailand, with a unilateral deep corneal ulcer and 15 cats presented with a unilateral corneal perforation. The median age of the cats was 36 months (range 6–184). The signalment, history and ophthalmic findings are provided in Table 1.
Signalment, history, ophthalmic lesion and outcome of 24 corneal ulcers and corneal perforations in cats treated with and without mesenchymal stem cell (MSC) injection

DSH = domestic shorthair; OD = oculus dexter (right eye); OS = oculus sinister (left eye)
Ophthalmic examination of the affected eyes revealed positive menace response and dazzle reflex. The other ophthalmic signs on the affected eyes were ocular discomfort, such as blepharospasm, third eyelid protrusion, positive fluorescein staining at the corneal ulcer, conjunctivitis and epiphora. Five cats (cats 6, 12, 15, 19 and 24) had anterior synechiae at 10, 6, 9, 6–7 and 8 o’clock on the affected eyes, respectively. Corneal neovascularisation was present in 18/24 eyes. For history taking, a pedicle conjunctival flap and amniotic graft were performed in one cat (cat 9), and a pedicle conjunctival flap was performed in one cat (cat 19), which finally became a graft failure. Four cats had health problems, including feline infectious peritonitis viral infection (cat 2), receiving diabetes mellitus treatment (cat 15), mediastinal lymphoma and hypertrophic cardiomyopathy stage B1 (cat 16) and feline leukaemia viral infection (cat 19). Conjunctival grafts were proposed for all cats. However, all owners denied surgery because of anaesthesia concerns, except for cats 9 and 19, which had unsuccessful surgeries. All cats were then offered subconjunctival MSC injections. After thorough discussion with each owner, 17 owners agreed to treatment involving MSC injection (cats 1–17), while the other seven owners denied treatment (cats 18–24). Therefore, these latter seven cats served as controls.
Cats were not randomly assigned to treatment groups. All owners were initially offered surgical intervention. After an informed discussion, owners selected either subconjunctival MSC injection combined with medical therapy or medical therapy alone. Therefore, group allocation was based on owner preference rather than randomisation.
This study was approved by the Institutional Animal Care and Use Committee of Kasetsart University, Bangkok, Thailand (reference number ACKU67-VET-054). This study was supervised for animal care and use for scientific research by the principal investigator, who was certified under the national licence No. U1-08938-2563.
MSC preparation
Cell isolation, expansion and cryopreservation of feline MSCs
Visceral adipose tissues were collected during ovariohysterectomy of a specific pathogen-free cat aged 3 years and free from feline foamy virus with owner consent. Then, 15 g of adipose tissue was preserved in Dulbecco’s phosphate-buffered saline (DPBS) and delivered to the laboratory under controlled temperature (4–8°C) within 2 h. The manufacturing of MSCs was executed under the standard facility for cell production of Precision Vet, and quality assurance was tested by an accredited ISO standard laboratory (ATGenes), as described elsewhere. 8 Briefly, the steps were as follows: (1) the tissue was washed with DPBS and digested with collagenase type I at 37°C for 30 mins; (2) after neutralising collagenase with inactivation medium, the tissue was filtered and centrifuged for stromal vascular fraction (SVF) separation; and (3) the supernatant was discarded and the SVF was resuspended and washed continuously with DPBS several times and plated in T-25 flasks with Dulbecco’s Modified Eagle Medium, 10% fetal calf serum and antibiotic.
Cells were then incubated at 37°C with 5% CO2. The culture medium was changed twice weekly to remove non-adherent cells. At 70–80% confluency, the cells were trypsinised for subculturing to the next passage. Then, the cells were trypsinised again for cryopreservation before being resuspended in cryoprotectants. The cells were divided into cryovials for cell cryopreservation after being evaluated for cell morphology, cell number and viability using a microscope, hemocytometer and trypan blue exclusion assay, respectively, as described elsewhere. 9 Cells were pre-cooled at –80°C overnight, then transferred to a nitrogen tank for cryopreservation.
Cell preparation
The MSCs were thawed at 37°C, washed with DPBS and randomly assessed for viability, as described elsewhere. 9 The MSCs were then resuspended in a sterile solution and kept at 4°C until MSC transplantation.
MSC characterisation
The MSC characterisation followed the process of the International Society for Cell & Gene Therapy, 10 involving cell morphology, plasticity, MSC markers and differentiation. Cultured cells were analysed using MSC markers with directly labelled commercial antibodies. Specifically, fluorescein isothiocyanate-labelled anti-mouse CD90 (Sigma Aldrich) and allophycocyanin-labelled anti-human CD105 antibody (Biolegend) were used as MSC-positive markers, while phycoerythrin (PE) Mouse Anti-Dog CD34 (BD Pharmingen) and PE-labelled anti-human HLA-DR antibody (Biolegend) were used as MSC-negative markers. Cells were stained with each antibody and analysed using FACSCalibur flow cytometry (Becton Dickinson); the data were analysed using CELLQuest Pro (BD Biosciences).
The differentiation potential of the cells into osteoblasts, adipocytes and chondrocytes was evaluated using Osteogenesis, Adipogenesis, and Chondrogenesis Differentiation Kits, following the manufacturers’ instructions and as described elsewhere. 11 After cell differentiation. Alizarin red staining, oil Red-O staining and Alcian blue staining were performed to verify osteogenic, adipogenic and chondrogenic lineage, respectively.
Sterility test
The sterility test was performed as described elsewhere. 8 The cultured medium was tested for bacteria, fungi and Mycoplasma contamination as well as the assessed level of endotoxin. All tests were performed following each manufacturer’s instructions.
Cell isolation, expansion and cryopreservation of feline MSCs
Attached spindle-like cells were observed on the surface of the flask. After each passage, cells expanded to 70–80% confluence during 5–7 days. The cultured medium showed no evidence of bacterial, fungal or Mycoplasma detection. The endotoxin level detected was below 0.5 EU/ml. MSCs were thawed and showed over 85% of cell viability before transplantation.
MSC characterisation
Surface marker expression revealed more than 90% positivity for CD90 and CD105, with less than 2% for CD34 and HLA-DR (MHC-II) (Figure 1).

Surface markers of feline mesenchymal stem cells, with CD90 and CD105 showing strong expression (>90%) in contrast to human lymphocyte antigen-DR (HLA-DR) and CD34 showing low expression (<2%). APC = allophycocyanin; FITC = fluorescein isothiocyanate; PE = phycoerythrin
After cell induction, each cell type was observed under a microscope and cell staining. In adipogenesis, lipid droplets were observed within the cytoplasm after the cells became rounded and larger. The droplets were stained with oil red-O. In osteogenesis, spindle-like cells changed into cuboidal and polygonal cells. Extracellular matrices were produced and stained with Alizarin red, which is specific to bone matrix. In chondrogenesis, an abundant extracellular matrix was produced from chondrocytes, with Alcian blue staining of the cell and matrix (Figure 2).
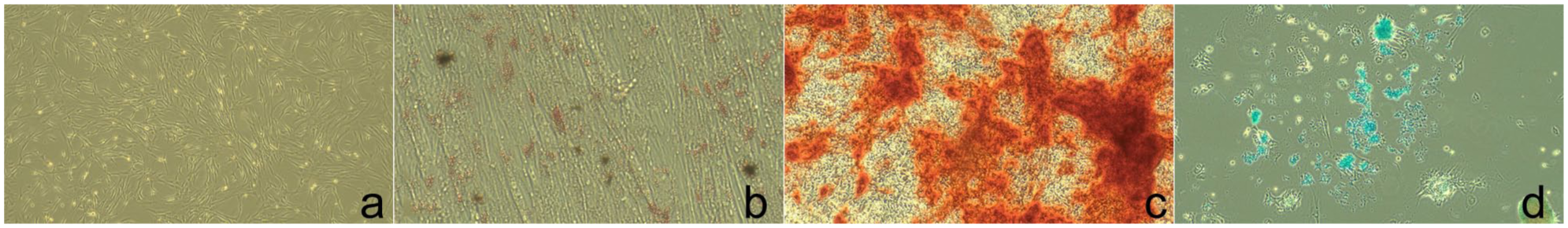
Adipogenic, osteogenic and chondrogenic differentiation of feline mesenchymal stem cells: (a) undifferentiated; (b) adipogenic differentiation; (c) osteogenic differentiation; and (d) chondrogenic differentiation
Treatment
Topical medications were prescribed in all cats: 0.5% moxifloxacin hydrochloride (q2h, Vigamox; Alcon-Couvreur NV), topical mydriatic with 1% atropine sulfate (q12h, Isopto; Alcon) and artificial tear with 0.3% sodium hyaluronate (q6h, Hialid 0.3%; Santen Pharmaceutical). Oral medications, amoxicillin-clavulanic acid (q12h, Clavaseptin; Vetoquinol) and tolfenamic acid (q24h, Tolfedine; Vetoquinol) were prescribed. All cats were recommended to wear an Elizabethan collar at all times.
The next day, after informed consent was received from each owner, 1 million (five eyes), 2 million (two eyes) and 3 million (10 eyes) feline allogeneic adipose tissue-derived MSCs were freshly prepared in 0.15 ml. MSCs were injected subconjunctivally after applying local anaesthesia with 0.5% tetracaine hydrochloride (Alcon; Alcon-Couvreur) in 17 cats. All medications were continued after MSC injection (Table 2).
Duration of medical management of 24 eyes with corneal ulcerations and corneal perforations after treatment with or without mesenchymal stem cell (MSC injections

Data are mean ± SD (range)
Follow-up examinations, including fluorescein staining, were conducted at 2–4 days, 1 week and then weekly until a fluorescein-negative status was achieved. Complete corneal ulcer healing was determined by negative fluorescein staining on the corneal surface. 12 Cats receiving subconjunctival MSC injections (n = 17) and the MSC-untreated cats (n = 7) were monitored for comparison. Treatment outcomes, time to fluorescein-negative status, recurrence rates and complications were recorded.
Results
Among the MSC-treated cats, a fluorescein-negative status was achieved in 15/17 (88%) cats within 1–3 weeks and 2/17 (12%) cats within 5–6 weeks after the MSC injection, as shown in Table 1 and Figure 3. During the mean follow-up period of 146 ± 138 days (range 15–398) after MSC injection, recurrence of corneal perforation before healing was observed in four cats (cats 7, 8, 10 and 14).

Images of corneal ulcers and clinical improvement of four cats with subconjunctival mesenchymal stem cell (MSC) injection and two cats without subconjunctival MSC injection: (a–d) left eye of cat 2 with subconjunctival MSC injection for deep corneal ulcer on days 0, 16, 31 and 51, respectively, with (b) negative fluorescein staining on day 16; (e–h) left eye of cat 4 with deep corneal ulcer with subconjunctival MSC injection on days 0, 16, 28 and 55, respectively, with (f) negative fluorescein staining on day 16; (i–l) right eye of cat 6 with descemetocele and corneal perforation (arrow) with subconjunctival MSC injection on days 0, 14, 21 and 49, respectively, with (k) negative fluorescein staining on day 21; (m–p) left eye of cat 9 with corneal perforation (arrow) and anterior chamber collapse with subconjunctival MSC injection on days 0, 14, 34 and 62, respectively, with (n) negative fluorescein staining on day 14; (q–s) left eye of cat 23 with corneal perforation without subconjunctival MSC injection on days 0, 9 and 58, respectively, with (t) conjunctival graft surgery on day 114; (u–x) left eye of cat 24 with corneal perforation and anterior synechia without subconjunctival MSC injection on days 0, 12, 31 and 61, respectively, with (w,x) having recurrent corneal perforation on days 31 and 61, respectively
In the control group, 4/7 (57%) cats (cats 18–21) achieved fluorescein-negative status within 3–7 weeks. In one cat (cat 23), the corneal ulcer did not heal even though the owner kept applying topical medications for 49 days until recurrent corneal perforation occurred on day 58, requiring conjunctival graft surgery. The graft was in position and the ulcer healed on day 72 after initial treatment. One cat (cat 24) had recurrent corneal perforation and finally healed on day 103, as shown in Table 1 and Figure 3.
The non-parametric survival analysis log-rank test was used for a comparison of the days of negative fluorescein staining between deep/perforated corneal ulcers with and without MSC subconjunctival injection. A probability level of 0.0005 was considered statistically significant (Figure 4).

Survival plot comparing days of negative fluorescein staining between deep/perforated corneal ulcer with (stem cell) and without (control) subconjunctival mesenchymal stem cell injection
After subconjunctival MSC injection, all treated cats exhibited transient subconjunctival swelling, which was completely resolved within 3–4 days. No other complications related to subconjunctival MSC injection were observed. However, during the first week after injection, four cats experienced a recurrence of corneal perforation due to various challenges. Otitis externa in one cat led to head shaking and scratching. After advising the owners to consistently use an Elizabethan collar, the ulcers healed within 3 weeks. Collar detachment occurred in one cat, which resulted in recurrent perforation. Securing the collar more tightly resulted in ulcer healing within 5 weeks. Head shaking during oral medication administration was reported in one cat. Switching to injectable medication resolved the issue, with ulcer healing achieved within 3 weeks. One cat exhibited excessive salivation due to atropine eye drops, leading to head rubbing and recurrent corneal ulcer. Substituting atropine with a topical combination of 0.8% tropicamide and 5.0% phenylephrine hydrochloride eliminated the issue, with ulcer healing within 2 weeks. Visual assessments were conducted on all cats after treatment. Vision was preserved in all cases.
Although reperforations were observed more frequently in the MSC-treated group, all such cases ultimately healed without surgical intervention. These events were associated with external factors such as collar detachment, excessive head shaking and medication-related adverse effects rather than clear evidence of treatment failure.
No such complications, including otitis externa, medication-related salivation or collar detachment, were reported in the control group. The owners consistently followed treatment protocols, including the use of collars.
Discussion
This study demonstrated that subconjunctival MSC injection has the potential to reduce the healing time for corneal ulcers compared with conventional topical medication alone. Of the cats in the study, 15/17 MSC-treated cats achieved fluorescein-negative status within 2–3 weeks, while 4/7 cats in the control group required 3–7 weeks. In the control group, two cats required surgery with a pedicle conjunctival flap for healing, while another cat had recurrent corneal perforation before the corneal ulcer finally healed.
The treatment of deep corneal ulcers in veterinary patients is multifaceted, involving both intensive medical management and surgical interventions. 13 Surgical options requiring physical protection range from conjunctival grafts3,5 and corneoconjunctival transpositions 14 to keratoplasties utilising biomaterials, such as porcine small intestinal submucosa (SIS),15 –17 amniotic membranes18,19 and bovine pericardium. 20 Conjunctival grafts have been widely used as a mechanical support for thin or perforated corneas, offering an immediate source of fibroblasts for stromal collagen regeneration and a conduit for systemic antibiotics. 21 However, the resultant permanent scarring can greatly impair vision, particularly when the scar is large or centrally located. 22 Similarly, biomaterial grafts, such as SIS, amniotic membranes and bovine pericardium, have been associated with reduced corneal scarring compared with conventional techniques, but all these techniques require general anesthesia, 17 microsurgical instruments and trained personnel.2,5 Complications such as aqueous leakage, graft dehiscence and axial corneal leukoma remain a concern.15,17 In a study on corneal perforations in 45 cats treated with conjunctival flap and corneal suturing, vision was saved in 29 (64%) eyes and lost in 16 (36%). 3 The failure of the graft was attributed to large defects and devitalised corneal tissue. 3 By comparison, all MSC-treated eyes in the present study retained their vision.
Subconjunctival MSC injection has emerged as a minimally invasive alternative treatment that is particularly advantageous for patients where anaesthesia poses serious risks. Unlike surgical interventions, MSC injections avoid the need for general anaesthesia, reducing perioperative risks and resource requirements. Furthermore, its non-invasive nature makes it a viable option for cases where surgical management is contraindicated or inaccessible.
Subconjunctival injection has been identified as an effective route for intraocular drug delivery, particularly for targeted therapies in ocular diseases. 12 In the present study, subconjunctival injection was utilised as the route for MSC administration because of its ability to deliver higher concentrations of cells directly to the eye and sustain their therapeutic effects over an extended period. This method allows for rapid cell delivery (within 12 h) and ensures cell survival for 3–5 weeks, providing sufficient time to support wound healing and tissue regeneration. 7 Furthermore, subconjunctival injection offers distinct advantages over systemic MSC administration by minimising the risk of severe systemic immune responses and thus making it a safer and more convenient route for ocular disease treatment. 12 The present study highlighted the utility of subconjunctival injection as a practical and efficient method for MSC transplantation in ocular therapy by supporting its potential as a preferred delivery route for future regenerative medicine applications in ophthalmology.
Regenerative medicine has gained considerable attention in recent years. The primary goal of regenerative medicine is to replace or regenerate damaged cells, tissues or organs, and restore normal function. 23 MSC therapy is a prominent approach within regenerative medicine, offering promising results in diverse conditions. In human ophthalmology, MSCs have been studied for their potential in treating corneal injuries, corneal dystrophies, limbal stem cell deficiencies, epithelial corneal ulcers, persistent corneal epithelial defects and corneal endothelial diseases.6,24,25 Similarly, in veterinary ophthalmology, in vivo studies across species, including dogs, horses and cats, have demonstrated the therapeutic potential of MSCs in managing ocular conditions. In dogs, MSC therapy has been evaluated for corneal ulcers, chronic superficial keratitis and keratoconjunctivitis sicca.7,8,26 –31 In horses, MSCs have been studied for immune-mediated keratitis, corneal ulcers, uveitis and retinal detachment.32,33 In cats, eosinophilic keratitis has been successfully treated with MSCs. 34 These findings underscore the potential of MSC-based therapies as an effective and versatile treatment modality in both human and veterinary ophthalmology, warranting further investigation to optimise their clinical applications.
In cats, adipose tissue-derived MSCs (AD-MSCs) are easier to collect and exhibit superior proliferative potential compared with bone marrow-derived MSCs, making the former the preferred source for therapeutic applications. 35 MSCs have been evaluated as therapeutic agents for inflammatory, degenerative and immune-mediated diseases in cats, such as chronic gingivostomatitis, kidney disease, inflammatory bowel disease, pancreatitis, idiopathic cystitis, asthma, osteoarthritis and cardiomyopathy. 36 AD-MSCs can be characterised by their ability to differentiate into multiple cell lineages, immunomodulatory properties and paracrine-mediated promotion of tissue regeneration. 37 AD-MSCs secrete extracellular vesicles containing trophic growth factors such as vascular endothelial growth factor and transforming growth factor-beta. These trophic factors inhibit apoptosis and scarring, stimulate angiogenesis and promote mitosis of tissue-specific progenitor cells, as well as facilitating the synthesis of new, organised collagen within the stroma, 37 alleviating corneal scars and eventually enhancing corneal transparency and promoting corneal wound healing. 37 Their immunomodulatory activity suppresses lymphocytic surveillance, reducing the risk of autoimmunity and enabling the use of allogenic MSCs in a variety of clinical settings in animals.38,39
This study highlighted the challenges of real-world implementation, particularly regarding owner compliance and post-treatment care in the first week. Complications, such as scratching, head shaking or collar detachment, increased the risk of recurrent corneal perforation. After MSC injection, recurrence of corneal perforations was observed in four cats. Recurrence of corneal perforation was not associated with surgical procedures but was observed after MSC injection. This finding emphasised the necessity of meticulous post-injection monitoring to promptly identify and manage potential complications, after the affected cases had been appropriately treated based on the underlying aetiology. All the corneal perforations were ultimately completely healed, and all deep corneal ulcers were healed uneventfully. Proper adjustments, such as consistent collar use and appropriate medication administration, can support successful outcomes regarding corneal ulcer healing in cats. However, a study limitation was that 3/24 cats did not receive pain medication after referral, as the owners declined further analgesia after prior administration of non-steroidal anti-inflammatory drugs before referral. According to the standard of care, these cats could have been considered for alternative forms of analgesia, such as opioids.
Two cats with systemic health conditions were included in the MSC-treated group. One diabetic cat achieved a fluorescein-negative corneal surface within 2 weeks after treatment. Another cat, which had an underlying cardiac disease unlikely to interfere directly with corneal repair also exhibited favourable healing outcomes. However, the number of MSC-treated cats with concurrent systemic diseases was limited in this study. Therefore, these findings should not be overinterpreted as evidence that MSC therapy can overcome systemic impediments to corneal ulcer healing; further investigation with a larger sample size is warranted.
A major limitation of this retrospective study is the lack of randomisation and matching between treatment groups. The MSC-treated group included a higher proportion of severe cases and cats with previous failed surgical interventions, whereas the control group included more moderate cases. These differences limit direct comparison between groups and preclude definitive conclusions regarding the superiority of one treatment over another. An additional limitation of this retrospective study was the small number of cats in the control (MSC-untreated) group. A number of deep corneal ulcers and corneal perforations were excluded, as most owners of such cases opted for surgical intervention at the first presentation, except for two cats with a prior history of failed corneal surgery whose owners declined repeated surgical treatment.
Conclusions
Subconjunctival MSC injection appears to be a promising adjunctive or alternative treatment option for feline corneal ulcers and corneal perforations, particularly in patients where surgical intervention is declined or contraindicated. However, given the retrospective design and lack of group matching, these findings should be interpreted cautiously, and prospective randomised controlled studies are warranted.
Footnotes
Acknowledgements
We acknowledge all pet owners for their cooperation and for providing information on their pets. Associate Professor Suwicha Kasemsuwan assisted with the statistical analysis in this work. The Precision Vet Co (Nonthaburi, Thailand) provided the stem cells for this work.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Financial support was received from the Faculty of Veterinary Medicine, Kasetsart University.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
