Abstract
Case series summary
This retrospective case series describes seven diabetic cats treated with velagliflozin that were considered non-ideal candidates for this therapy. These more complicated diabetic feline cases were referred to the Small Animal Department, Faculty of Veterinary Medicine, Ghent University, Belgium. The sole inclusion criterion for this case series was treatment with velagliflozin (Senvelgo; Boehringer Ingelheim) after diagnosis of diabetes mellitus. Data on signalment, medical history, clinical findings, diagnostics, treatment, response and outcomes were available for all cats. The initial consultation for all cats took place between March 2024 and May 2025.
Relevance and novel information
Current literature on the use of sodium–glucose cotransporter-2 (SGLT2) inhibitors in feline patients remains limited, with most studies involving highly selected populations with strict exclusion criteria. This case series describes the use of velagliflozin in non-ideal candidates for SGLT2 inhibitor therapy, aiming to support clinicians managing such cases and to provide useful information for future prospective studies. In these non-ideal cases, the use of velagliflozin may be considered off label; its administration should comply with local ethical and legal regulations, with informed client consent obtained. Consultation with a veterinary specialist is recommended when clinical experience is limited. This case series includes diabetic cats with suspected and/or confirmed comorbidities that complicate diabetes management, such as hypersomatotropism and chronic kidney disease. In addition, it reports on the concurrent use of other treatments, including cabergoline and insulin. The potential interaction and possible synergistic effects of these combined therapies represent an area of interest for future research. To ensure optimal glycaemic control and enable individualised dosing, four of the presented cases were closely monitored using continuous glucose monitoring (Freestyle Libre Sensor 2; Abbott Laboratories). Furthermore, the availability of measurements of ketone concentrations in urine and/or blood provided valuable insight into the metabolic changes associated with this new treatment. The role of ketone monitoring in predicting treatment response and identifying potential adverse effects represents another important area for future research.
Plain language summary
Diabetes mellitus is a common disease in cats that causes high blood sugar levels and requires lifelong treatment. Traditionally, insulin injections have been the main treatment, but newer oral medications, such as velagliflozin, are now available. Velagliflozin helps lower blood sugar by allowing excess glucose to leave the body in the urine. However, there is limited information about how well this medicine works in cats with other health problems. This report describes seven diabetic cats treated with velagliflozin, even though they were non-ideal candidates for this therapy because they had additional diseases or were receiving other treatments. Some cats had conditions such as chronic kidney disease or hypersomatotropism (a hormone disorder that can make diabetes more difficult to control). The cats were monitored through regular check-ups, blood tests to measure ketones (substances that can indicate the most significant risk: diabetic ketoacidosis) and, in some cases, continuous glucose monitoring sensors that tracked their sugar levels throughout the day. Overall, this case series provides information about using velagliflozin in more complicated diabetic cats. It shows that, with close monitoring and careful selection of patients, this medication can be a useful option beyond the strict conditions of earlier studies. These observations can help veterinarians and researchers improve the safety and effectiveness of diabetes care in cats. In situations where standard treatment is not suitable, velagliflozin may be considered outside its approved indications. This should be done only in line with local laws and ethical guidelines, and after the client has given informed consent. If the clinician has limited experience with this treatment, consultation with a veterinary specialist is advised. The findings also show that more research is needed to better understand dietary strategies, the use of combined treatments and how to interpret ketone levels in diabetic cats.
Introduction
Diabetes mellitus (DM) is a common endocrine disorder in cats, characterised by persistent hyperglycaemia and glucosuria. Clinical signs include polyuria and polydipsia (PUPD), polyphagia and weight loss. Notably, approximately one-quarter of diabetic cats have underlying hypersomatotropism (HS), which complicates DM management owing to insulin resistance. 1 Effective management is essential to prevent complications such as clinical hypoglycaemia and diabetic ketoacidosis (DKA). Although insulin therapy combined with dietary modification remains the standard of care, it poses substantial challenges for both veterinarians and pet owners. These include the need for accurate insulin dosing, twice-daily injections, frequent glucose monitoring and the risk of clinical hypoglycaemia, as well as potential strain on the owner–pet relationship. The challenges of this treatment are substantiated by a study reporting that 30% of cats or dogs with DM are euthanased within the first year of diagnosis. 2 These limitations highlight the need for alternative treatment options that provide effective glycaemic control while minimising adverse effects. Bexagliflozin (Bexacat; Elanco) has been available in the USA since 2022, and velagliflozin (Senvelgo; Boehringer Ingelheim) in the EU since 2023, as novel treatments for feline diabetes.
Sodium–glucose cotransporter-2 (SGLT2) inhibitors represent a new class of oral antidiabetic drugs for cats. SGLT2 is a membrane protein in the proximal tubule of nephrons, responsible for reabsorbing approximately 90% of filtered glucose, while the remaining 10% is reabsorbed by sodium–glucose cotransporter-1 (SGLT1) further along the tubule. Inhibition of SGLT2 receptors reduces glucose reabsorption, promoting urinary glucose excretion.3 –6 In humans and mice, these drugs lower blood glucose concentrations without inducing clinical hypoglycemia. 3
In feline medicine, SGLT2 inhibitors, including bexagliflozin and velagliflozin, have been evaluated as potential alternatives to insulin therapy in clinically stable diabetic cats.3 –5 Previous studies applied strict exclusion and inclusion criteria, omitting cats with comorbidities, ketosis or ketonuria in the history or at presentation, and clinical signs such as vomiting, diarrhoea and anorexia. The study by Behrend et al 5 included cats previously treated with insulin and cats with a suspicion of HS. Reported success rates, defined by improvements in glycaemic control and/or improvement in clinical signs, range from 54% to as high as 88%. None of these studies reported episodes of clinical hypoglycaemia in cats, representing a significant advantage over insulin therapy.3 –5 The most common adverse effect, softer stools, was reported in 38–53% of treated cats; these episodes were generally mild and self-limiting within 2 weeks. 6 Persistent glucosuria may contribute to ongoing PUPD in some cats, with studies reporting persistent polyuria in 12–46% of cases, indicating that this clinical sign often does not resolve despite treatment.3 –5 The most serious, potentially life-threatening, adverse event associated with gliflozin therapy is DKA, which may occur even when blood glucose levels remain within normal or near-normal intervals, a condition referred to as euglycaemic diabetic ketoacidosis (eDKA). Overall, eDKA was observed in 5–7% of insulin-naive diabetic cats treated with gliflozins.3,5,6 Behrend et al 5 administered velagliflozin as monotherapy in both insulin-naive and previously insulin-treated diabetic cats. The incidence of DKA was higher in cats with prior insulin exposure (18%) compared with newly diagnosed cats (5%). Notably, 78% of cats diagnosed with DKA were euglycaemic and 86% of cases developed within the first 2 weeks of treatment. 5
Gliflozin therapy provides a convenient, once-daily, oral treatment option for managing stable diabetic cats, described as ‘happy diabetic cats’. These are patients that maintain normal appetite, hydration and activity levels, without clinical signs such as vomiting, diarrhoea, lethargy, cachexia or evidence of DKA, as confirmed by normal blood or urine ketone concentrations. In such cases, gliflozin has been associated with rapid glycaemic stabilisation, with measurable improvements typically seen within 7 days. In addition, the reduced need for frequent blood glucose monitoring and dose adjustments contributes to simplified long-term management. 5
However, because of the increased risk of DKA, particularly within the first 2 weeks of treatment, close monitoring is mandatory. Owners should watch for the typical clinical signs of DM, as well as signs of DKA including anorexia, lethargy, acute vomiting, dehydration, weight loss, respiratory distress or altered mentation. Current recommendations advocate, besides monitoring of clinical signs, the measurement of ketone levels on day 2 or 3 of treatment, with prompt veterinary evaluation recommended if ketonuria or marked ketonaemia is detected. As validated reference intervals (RIs) for blood or urine ketone concentrations in cats are currently lacking, any values exceeding the upper measurable interval of the device or showing a significant rise from baseline should be interpreted with caution and clinical context.
This case series describes second-line feline diabetes patients treated with off-label velagliflozin for various clinical indications. The purpose of this report is not to advocate off-label use of SGLT2 inhibitors, but to outline practical considerations, limitations and challenges associated with their use in complex clinical scenarios. The findings aim to contribute to a more informed and safe application of this therapeutic approach in the management of feline diabetes.
Case series description
Medical records of seven challenging second-line diabetic cats treated with velagliflozin were retrospectively reviewed. Data collected included signalment, medical history, comorbidities, diagnostic findings, treatment details, response and outcome. All cats were seen between March 2024 and May 2025. Follow-up was available for all and ongoing in some cases. Results of glucose and ketone monitoring are summarised in Table 1.
Results of ketone and glucose measurements in seven challenging second-line diabetic cats treated with velagliflozin
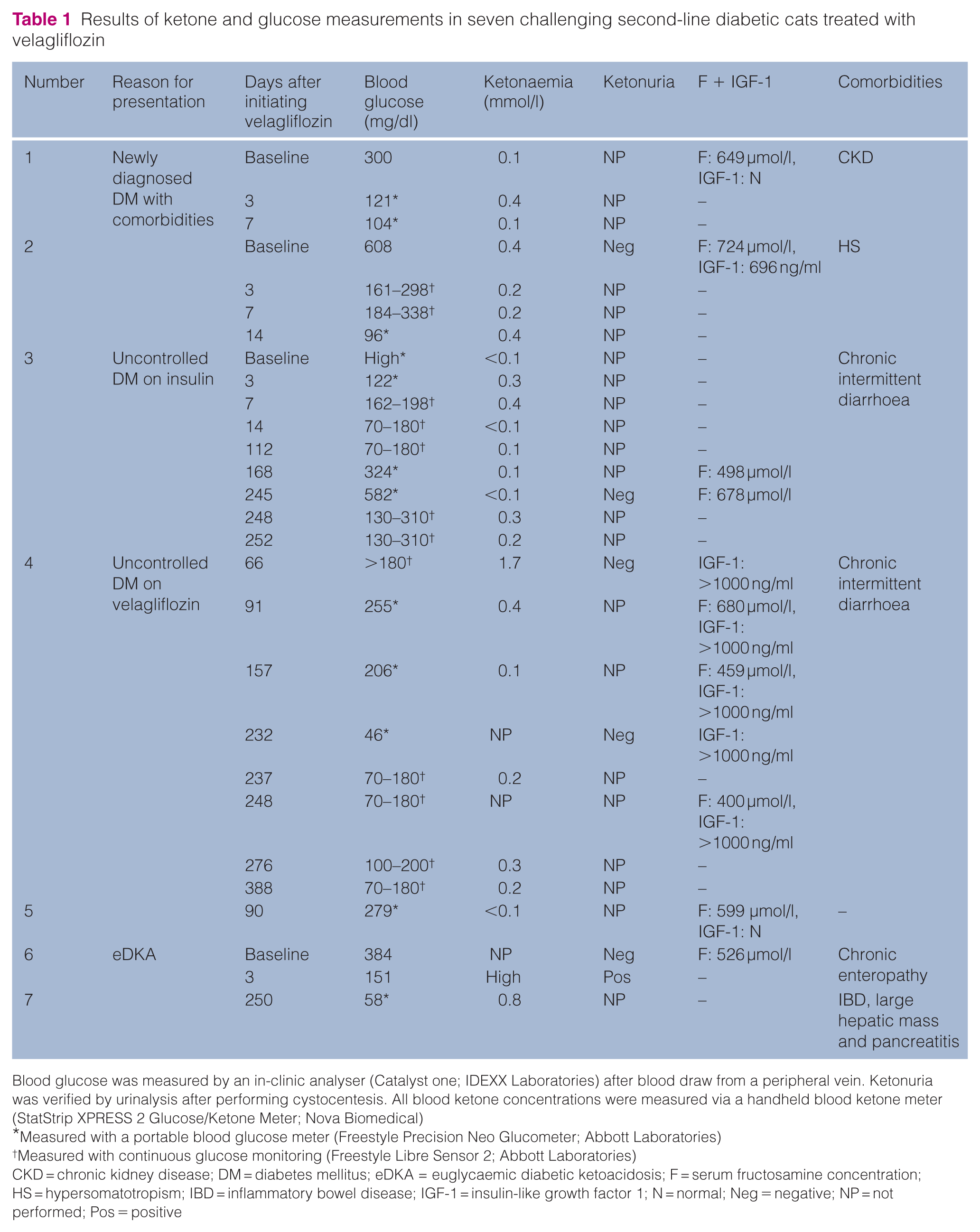
Blood glucose was measured by an in-clinic analyser (Catalyst one; IDEXX Laboratories) after blood draw from a peripheral vein. Ketonuria was verified by urinalysis after performing cystocentesis. All blood ketone concentrations were measured via a handheld blood ketone meter (StatStrip XPRESS 2 Glucose/Ketone Meter; Nova Biomedical)
Measured with a portable blood glucose meter (Freestyle Precision Neo Glucometer; Abbott Laboratories)
Measured with continuous glucose monitoring (Freestyle Libre Sensor 2; Abbott Laboratories)
CKD = chronic kidney disease; DM = diabetes mellitus; eDKA = euglycaemic diabetic ketoacidosis; F = serum fructosamine concentration; HS = hypersomatotropism; IBD = inflammatory bowel disease; IGF-1 = insulin-like growth factor 1; N = normal; Neg = negative; NP = not performed; Pos = positive
Cat 1 was a 13-year-old spayed female Somali with newly diagnosed DM and concurrent chronic kidney disease (CKD) (International Renal Interest Society [IRIS] stage 2) that was started on velagliflozin. Glycaemic control improved within days, with mild transient ketonaemia early in treatment. Velagliflozin was discontinued after 4 months at the owner’s initiative. The cat presented only 5 months after cessation of gliflozin therapy for a checkup and remission of DM was documented. Persistent clinical signs were attributed to CKD.
Cat 2 was a 15-year-old castrated male domestic shorthair (DSH) presented with newly diagnosed DM and elevated insulin-like growth factor-1 serum concentrations with pituitary microadenoma, consistent with HS. Velagliflozin was initiated, resulting in rapid glycaemic improvement confirmed by continuous glucose monitoring (CGM) (Figure 1). Cabergoline was concurrently added for HS management. Clinical control and stable glycaemia were achieved within 2 weeks.

Continuous blood glucose home monitoring (Freestyle Libre Sensor 2; Abbott Laboratories) from cat 2 diagnosed with both diabetes mellitus (DM) and hypersomatotropism. On day 1 (Thursday, 24 April 2025), the day of DM diagnosis, marked hyperglycaemia (beyond the measurable interval) was present. After administration of the first dose of velagliflozin (Senvelgo; Boehringer Ingelheim), a rapid decline in blood glucose occurred. On day 2 (Friday, 25 April 2025), the blood glucose values were just slightly above the target interval most of the time (70–180 mg/dl). Meals are represented by the apple icon
Cat 3 was a 10-year-old castrated male DSH, previously treated with insulin, that was referred for poorly controlled DM and chronic intermittent diarrhoea. The owner chose an easier treatment option and velagliflozin monotherapy was initiated after counselling on the increased risk of eDKA. After the first dose, a marked decrease in blood glucose occurred (Figure 2). Mild transient ketonaemia resolved within 2 weeks. After 3 months, remission of DM was confirmed, although relapse occurred after another 3 months. Reinitiation of velagliflozin restored glycaemic control.

Continuous blood glucose home monitoring (Freestyle Libre Sensor 2; Abbott Laboratories) from cat 3, whose owner opted for an easier treatment. On day 1 (Tuesday, 12 March 2024), marked hyperglycaemia (too high to measure) was present in the morning. After administration of velagliflozin (Senvelgo; Boehringer Ingelheim) at 10:30 am, a rapid decline in blood glucose occurred, with partial stabilisation later in the day. On day 2 (Wednesday, 13 March 2024), glycaemic variability was reduced, with most values within or slightly above the target interval (70–180 mg/dl). On day 3 (Thursday, 14 March 2024), a stable glucose profile was observed, with values largely within the target interval
Cat 4 was an 11-year-old castrated male DSH with HS and chronic intermittent diarrhoea that was referred for uncontrolled DM with persistent hyperglycaemia despite insulin and velagliflozin monotherapy. At the initial presentation, after 2 months of velagliflozin monotherapy, mild ketonaemia was observed in the absence of ketonuria or clinical evidence of eDKA. Combination therapy with insulin, velagliflozin and cabergoline, guided by CGM, showed good glycaemic control. Blood ketones could be repeated only after 1 month and were normalised. Five months later, the cat developed a severe hypoglycaemic episode with seizures and transient blindness while under pet sitter care. Therapy was adjusted by reducing the insulin dose and levetiracetam was temporarily introduced. Cabergoline was discontinued as no positive effect was observed and it is effective in only a subpopulation of cats. 7 Vision recovered within 7 weeks and the cat remained well controlled on the combined regimen with normal activity and glycaemia (Figure 3).

Continuous blood glucose home monitoring (Freestyle Libre Sensor 2; Abbott Laboratories) from cat 4, diagnosed with both diabetes mellitus and hypersomatotropism. Stable blood glucose concentrations were achieved with protamine zinc insulin (1 unit SC q12h, ProZinc; Boehringer Ingelheim), combined with oral velagliflozin (Senvelgo; Boehringer Ingelheim) based on ideal body weight. The green syringe symbol indicates insulin administration, the white box represents insulin dosing and the green box represents velagliflozin administration
Cat 5 was a 9-year-old castrated male DSH, earlier diagnosed with DM, that showed persistent PUPD after 3 months of velagliflozin therapy despite acceptable glycaemic parameters and absence of ketonaemia. The cat was otherwise clinically well, active and had regained weight. HS was excluded. Persistent PUPD was attributed to glucosuria-related osmotic diuresis and dietary adjustments were recommended to optimise weight and glycaemic control.
Cat 6 was a 10-year-old castrated male Devon Rex with DM and chronic enteropathy that developed lethargy and hyporexia 2 days after initiating velagliflozin therapy. Laboratory evaluation revealed eDKA. Stabilisation was achieved with intravenous fluids and short-acting insulin. Therapy was later changed to protamine zinc insulin. The cat recovered fully and velagliflozin was not reintroduced.
Cat 7 was an 11-year-old spayed female DSH known to have DM and inflammatory bowel disease that was presented with lethargy, anorexia and vomiting after treatment with velagliflozin for approximately 250 days. Although ketonaemia was only mild, DKA could not be entirely excluded given the coexistence of acidaemia, the absence of baseline beta (β)-hydroxybutyrate (BHB) data and the lack of established feline RIs. Abdominal ultrasonography revealed a large hepatic mass and pancreatitis. Given the poor prognosis, the owner elected euthanasia.
Discussion
This case series describes the clinical use, efficacy and safety of velagliflozin in cats with DM, including those with comorbidities, despite not fitting current label indications. Appropriate patient selection and clear communication with owners are and remain essential when initiating SGLT2 inhibitor therapy. Their favourable outcomes demonstrate that gliflozin therapy may be feasible beyond strictly defined candidate profiles, provided that owners are well informed and monitoring is close.
It also highlights practical aspects of glucose and ketone monitoring. Transient, mild ketonaemia was observed in several cats started on velagliflozin therapy, without clinical signs of DKA. Close observation during the first 2 weeks of treatment is essential, as eDKA can develop rapidly and may be missed owing to normal blood glucose levels, especially since this condition is still relatively new within the veterinary community. Early detection of anorexia, lethargy, vomiting or dehydration should prompt immediate ketone assessment.
Clinical hypoglycaemia is a known insulin-related adverse effect. 8 In contrast, studies on SGLT2 inhibitors in cats report hypoglycaemia as rare or absent when used as monotherapy.3 –6 However, cat 4 experienced a severe hypoglycaemic crisis during combined insulin and velagliflozin therapy. In other cases, clinical hypoglycaemia was not reported, suggesting that the combined therapy was most likely the cause. The episode occurred 2 months after achieving glycaemic control and coincided with reduced home monitoring when the owner was absent. In hindsight, subclinical hypoglycaemia should have triggered closer follow-up. Since gliflozins cause glucosuria regardless of glycaemia, its absence cannot confirm remission of DM. As cat 4 was hyperglycaemic and glucosuric 4 days after discontinuation of the combined therapy, we concluded that remission was not the underlying cause for the hypoglycaemic crisis.
In cats, the most significant risk associated with gliflozin therapy is eDKA. Successful treatment requires sufficient endogenous insulin production, as inadequate insulin levels promote ketogenesis and subsequent ketonaemia. Although there is no definitive method to assess β-cell function at diagnosis in diabetic cats, ketone concentrations at presentation can be used as a surrogate. Persistent insulin deficiency is associated with increased production of ketone bodies, particularly BHB.9 –11
Healthy cats show blood BHB concentrations below 0.1 mmol/l, while BHB in insulin-treated diabetic cats without ketosis may reach up to 1.0 mmol/l. 9 Thresholds above 2.4 mmol/l have been reported as highly sensitive for diagnosing DKA with handheld ketone meters.9,10 In this series, BHB measurement was used for ketone monitoring. Mild elevations (⩽1.7 mmol/l) occurred without clinical deterioration, suggesting that low-grade ketonaemia can be benign in treated cats. These findings highlight the need for further studies to define reliable RIs and diagnostic thresholds for blood ketones in diabetic cats treated with SGLT2 inhibitors. In terms of monitoring, there is temporal distinction illustrated between cases 6 and 7. During long-term therapy, attention should shift to detecting and managing comorbidities that may cause insulin resistance.
Urine dipsticks, which detect only acetoacetate (⩾0.5 mmol/l), were not used in this series as BHB is the predominant ketone with ketosis, but they remain common in primary care. 12 They may lag behind blood values, remain positive after resolution and are semi-quantitative, introducing subjectivity. 13 Up to 30% of healthy cats may test positive despite being non-diabetic 14 and only 55% of owners can reliably collect samples at home. 15 These limitations reduce diagnostic accuracy. In contrast, handheld BHB meters provide more reliable and practical monitoring, especially in cats treated with SGLT2 inhibitors.
Conventional DKA is mostly managed with initial fluid resuscitation before starting short-acting insulin and dextrose therapy, whereas eDKA requires their simultaneous administration to suppress ketogenesis and prevent hypoglycaemia, as blood glucose levels are often normal or only mildly elevated. 16
Besides DKA, other side effects associated with gliflozin treatment were observed in this series. Persistent PUPD, as seen in cat 5, may reflect ongoing glucosuria rather than poor control and should not automatically be interpreted as treatment failure. Owners should be advised that this clinical sign may persist despite stable glyacemia. Mild gastrointestinal side effects, such as softer stools, were also observed and may be due to cross-inhibition of intestinal SGLT1 transporters, leading to increased faecal water content and mild osmotic diarrhoea secondary to incomplete glucose absorption. 6 However, given the non-specific nature of this sign and the transient course, alternative causes could not be excluded.
Comorbidities appear to influence both the safety and efficacy of velagliflozin. Current guidelines advise against gliflozin use in cats with reduced renal function. 3 Cat 1 tolerated therapy well despite IRIS stage 2 CKD, although dehydration remains a theoretical risk and reduced renal function may diminish glucose excretion. In humans, SGLT2 inhibitors may confer renal benefits, but such effects have not yet been demonstrated in cats. 17 HS may contribute to insulin resistance while sparing β-cell function, potentially explaining the usability of gliflozin therapy. Combination therapy with cabergoline may improve glycaemic control by reducing growth hormone secretion, but the response is inconsistent, as shown in cats 2 and 4. 7
Nutritional management may affect treatment safety. Cat 5 was recommended a conventional diet. However, in humans, low-carbohydrate diets combined with SGLT2 inhibitors can enhance ketogenesis. 16 Because eDKA is associated with carbohydrate deficit, adequate caloric and carbohydrate intake should be maintained in treated cats. Most published feline studies kept cats on maintenance diets, suggesting that strict low-carbohydrate formulations may not always be required.3 –5 More research is needed to guide dietary recommendations.
Two cats in this case series achieved diabetic remission. 18 Although data with insulin therapy are heterogeneous, there is a general association between tighter glycaemic control, particularly early in the disease course, and an increased likelihood of remission.8,19,20 As glycaemic control with SGLT2 inhibitors is generally good, remission may be expected in a subpopulation of treated cats, although current studies did not assess this.3 –5 These cases suggest that improved glycaemic control with SGLT2 inhibitors may support β-cell recovery and remission in selected cats, although long-term monitoring and further research are needed to clarify its role in achieving and sustaining remission.
Overall, this case series suggests that velagliflozin can be effective in managing complex feline diabetic cases, with appropriate monitoring. Although eDKA is the most serious potential complication, its risk can be minimised through owner education, early ketone testing and careful patient selection. As this was a retrospective case series, cases were not managed uniformly and some clinical findings, including chronic diarrhoea observed in several cats, were incompletely explored, although these did not appear to worsen during treatment. Other potential causes of metabolic acidosis, such as severe azotaemia, were not investigated. Moreover, the number of cases is small and heterogeneous, reflecting the off-label and exploratory use of a novel medication. These limitations indicate a need for prospective, controlled studies to further assess the safety and efficacy of velagliflozin and to identify which non-ideal candidates may be appropriate for its use. Off-label use of velagliflozin should always adhere to regional legal and ethical frameworks with informed client consent. Practitioners are encouraged to consult a specialist when considering such treatment in cases where clinical experience is still emerging.
Conclusions
This case series highlights the potential of velagliflozin in managing feline DM in cats with significant comorbidities. Glycaemic control was achieved with velaglifozin therapy in all cases. Remission of DM was achieved in two cases and one case relapsed after 3 months. The occurrence of eDKA underscores the need for careful patient selection, early monitoring and proactive owner communication. The findings also emphasise the need for further research into nutritional strategies, combination therapies and interpretation of ketones.
Footnotes
Conflict of interest
One author, SD, is a member of the European Advisory Board of Boehringer Ingelheim, which is the company selling the investigated drug.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
