Abstract
The management of diabetes mellitus (DM) in cats can benefit from an integration of medical and nutritional strategies, based on an understanding of the dynamic nature of the disease, together with an appreciation of the interrelationships between nutritional status and clinical status. In this context, a new classification system for feline DM is proposed, comprising three clinical status categories: those at risk of developing DM, those with clinical DM and those in diabetic remission. The influence of individual dietary components (carbohydrate, protein, fat and water fractions) on clinical outcomes is discussed, followed by overarching principles for the dietary management of diabetic cats, based on both the limited scientific evidence and the clinical experience of the authors. Key aspects of nutritional assessment, the principles of therapeutic weight reduction and feeding practices are covered. Using the classification framework, recommendations for nutritional management are proposed for cats at risk of development of DM, cats with clinical DM and those in clinical remission. Successful implementation of these recommendations can be achieved by using a relationship-centred approach, where owner concerns are addressed and goals for management are agreed in partnership. It is hoped that these perspectives will help guide veterinary professionals in their clinical management decisions, thereby improving health outcomes in cats in all three categories of DM.
Keywords
Introduction
In cats, the most common form of diabetes mellitus (DM) typically resembles type 2 DM in humans, in which both beta-cell (β-cell) dysfunction and insulin resistance contribute to disease onset and progression; there is also a strong association with obesity.1,2 Effective treatment strategies for feline DM now include both insulin3,4 and sodium-glucose co-transporter 2 (SGLT2) inhibitors.5,6 Nutrition plays a critical role in the management of feline DM, although there has been little clinical research on specific nutritional interventions and existing studies are difficult to compare.7 –9 Limited data also exist on the nutritional implications for cats treated with non-insulin medications, such as SGLT2 inhibitors, which promote urinary glucose excretion,5,6 and glucagon-like peptide-1 (GLP-1) receptor agonists, which influence appetite and insulin secretion. 10 In addition, the dietary management of cats at risk of developing DM and cats in diabetic remission remains inadequately studied.
By integrating existing knowledge and the clinical perspectives of the authors, this article aims to provide comprehensive and practical guidance for tailoring the nutritional management of individual diabetic cats, including those considered at risk of the disease and in remission. The recommendations are framed within a proposed classification system that accommodates different clinical states of DM and other nutritional and treatment factors (both conventional and emerging). Knowledge gaps informing future research directions on the nutritional management of diabetic cats are additionally highlighted.
The dynamic nature of DM in cats: integrating medical and nutritional considerations
DM in cats is a complex endocrine disease 11 that is dynamic in nature, with management needing to be tailored to the individual’s condition, taking account of disease status, nutritional status, treatment (insulin vs SGLT2 inhibitors), concurrent diseases and goals of treatment, together with the needs and preferences of the cat and owner. The metabolic consequences of DM are the result of relative or absolute deficiency in insulin activity, determined by the balance between insulin production, insulin sensitivity and glucose load. DM is characterised by an underlying disease process that causes progressive β-cell loss and dysfunction, exacerbated by the metabolic consequences of insulin deficiency, including glucotoxicity and impaired incretin effect.12,13 Insulin sensitivity is also dynamic, decreasing with increased adiposity, inflammation and stress, 14 while glucose load varies with the amount of gluconeogenesis, 15 intake of dietary digestible carbohydrates 16 and the amount of glucose lost in the urine. 17 Furthermore, insulin’s actions are typically anabolic in nature; therefore, beyond the hyperglycaemia that leads to glucosuria, polyuria and polydipsia, insulin deficiency can result in loss of fat and muscle mass, an increase in ketone production and eventually ketoacidosis. 18
Therapeutic goals for diabetic cats include controlling clinical signs, preventing complications (hypoglycaemia, diabetic ketoacidosis, peripheral neuropathy) and, when possible, achieving diabetic remission (a state of sufficient β-cell function that meets or exceeds insulin requirement as determined by insulin sensitivity and glucose load), with the overall aim to ensure a good quality of life for the cat and its owner. Additional goals of nutritional management include providing a nutritionally adequate diet and achieving and maintaining a healthy body condition. Both medication and diet influence the balance between insulin production, insulin sensitivity and glucose load, as well as their metabolic consequences. Consequently, achieving these therapeutic goals requires a combination of medical and nutritional assessments and interventions, and an understanding of the impact of these on one another, both acutely and over time. Management of DM is necessarily an ongoing and iterative process that must be adapted according to an individual cat’s response to interventions. The key clinical indicators of nutritional status are general wellbeing (physical and behavioural), body condition score (BCS), muscle condition score (MCS), body weight (BW) and appetite (food intake); current status must always be compared with previous status. These indicators are particularly important in the context of DM because of their metabolic implications; for example, obesity is a risk factor for feline DM and is itself a major cause of insulin resistance. 1 Obesity might also negatively impact incretin responses, leading to impaired postprandial insulin secretion. 19 Together, these effects can exacerbate DM severity or promote the transition from subclinical to clinical DM. Conversely, therapeutic weight reduction improves glucose sensitivity in healthy overweight cats, 20 and caloric restriction induces remission of DM in diabetic cats. 21 MCS is important in DM because muscle is normally the main ‘sink’ (ie, the location where glucose is taken up and stored or used) for circulating glucose. Therefore, loss of muscle mass reduces the body’s ability to clear glucose from the bloodstream.
A classification system for cats with DM to inform decisions on nutritional and medical interventions
There is a critical need to develop a tailored approach to the management of feline DM, and this is becoming increasingly feasible. To support this, veterinary teams would benefit from a framework for systematic classification that not only captures the current medical and nutritional status of a cat with DM but also considers the dynamic nature of DM with respect to health status and changes in BW and BCS. The authors propose a new classification system designed to provide a framework for precise but dynamic clinical and nutritional decision-making for individual cats, in addition to capturing the needs of subgroups with different clinical needs and therapeutic requirements (Figure 1). This system is intended to supplement and complement the Agreeing Language in Veterinary Endocrinology (ALIVE) diagnostic guidelines for feline DM, 22 which are validated by the European Society of Veterinary Endocrinology and Society of Comparative Endocrinology (Table 1).

Proposed classification system for cats with diabetes mellitus (DM) to inform decisions on nutritional and medical interventions. ALIVE = Agreeing Language in Veterinary Endocrinology; BCS = body condition score; GLP-1 = glucagon-like peptide-1; SGLT2i = sodium-glucose co-transporter 2 inhibitor
Key features of Agreeing Language in Veterinary Endocrinology criteria for the diagnosis of diabetes mellitus in cats 22

Bold emphasis in original
Similar to existing classification systems for chronic kidney disease (CKD) 23 and canine myxomatous mitral valve disease, 24 the proposed classification system accommodates both cats with a current or historic diagnosis of DM and those at risk of developing DM in the future. Existing diagnostic criteria do not consider the latter group, despite known associations between elevated measures of markers of glycaemia and the development of DM.13,25 In addition, since DM remission does not signify a return to the preclinical disease state, nutritional strategies for cats either at risk or in remission should be considered separately.
The classification system categorises cats across four dimensions, and a cat can move between categories as its clinical status changes, such as in response to treatment (Figure 1). The primary dimension is the diabetic status: A (‘at risk’), C (clinical diagnosis of DM according to ALIVE criteria) and R (remission from DM diagnosed with ALIVE criteria). The ALIVE definition of remission was established before SGLT2 inhibitors were available to treat cats and, consequently, only applies to cats who have had their insulin therapy successfully discontinued for 4 weeks without recurrence of DM. To accommodate cats who achieve remission after successful SGLT2 inhibitor therapy, here, the proposed DM-R category includes cats who are no longer dependent on exogenous medical treatment for maintenance of an acceptable blood glucose range.
The three subsidiary dimensions are body condition (underweight [U], healthy weight [H] or overweight [O]), BW trajectory (losing [L], stable [S] or gaining [G]) and current medical treatment to manage blood glucose (insulin [Ti], SGLT2 inhibitor [Ts], other treatment [Tx] or no treatment [Tn]). In the context of this classification system, a healthy body condition includes a BCS of 6/9 as a realistic and potentially beneficial target for an obesity-prone cat with DM, not least given that such a score is associated with increased longevity compared with a score of 5/9,26,27 while average lifespan is shorter in cats who have a BCS of 4/9. 27 BW trajectory is one of the classification’s sub-dimensions because dietary and treatment considerations differ for cats who are losing weight compared with those that are gaining weight.
Macronutrients in the context of DM
Figure 2 proposes quantitative ranges for describing ‘low’, ‘moderate’ and ‘high’ carbohydrate, digestible carbohydrate, protein, fat and energy density, based on existing complete and balanced commercial feline diets. The proposed descriptors (Figure 2), although subjective, are intended to provide context for subsequent sections of the guidelines within the current article. It is recognised that the literature contains different and inconsistent interpretations of such descriptors that may depend on the disease under discussion28,29 or have been derived from original research or guidance publications.30 –33 Rather than making comparisons on a percentage dry matter or as-fed basis, the macronutrient content of different diets should be compared relative to energy (eg, percentage of metabolisable energy [ME] or grams per 1000 kcal), which enables accurate comparison of diets with varying energy density. Throughout these guidelines, the descriptors low, moderate and high for macronutrients are written in inverted commas, including when they refer to the descriptors used in an original research publication, which might not necessarily concur with our definitions. Descriptors for recommendations in these guidelines without inverted commas correspond to the definitions in Figure 2.

Descriptors for low, moderate and high content of carbohydrate, digestible carbohydrate, protein and fat, and corresponding energy density for commercial feline diets. *Proximate analyte, nitrogen-free extract, is determined indirectly, and therefore may be an inaccurate estimate of carbohydrate. It is calculated by subtracting measured percentages of crude protein, crude fat, crude fibre, moisture and ash from 100. #g/Mcal is calculated by dividing carbohydrate, protein and fat % metabolisable energy (ME) values by 3.5, 3.5 and 8.5, respectively, and multiplying by 10. †Digestible carbohydrate as represented by starch. ‡Proximate analyte, crude protein as % ME. §Proximate analyte, crude fat as % ME. ¶Energy density, kcal/kg, broadly applicable, independent of method of calculation
Carbohydrate
Carbohydrate is a chemical class of macronutrients that encompasses a range of saccharides (sugars including mono-, di- and oligosaccharides), starches and fibres of plant and animal origin. These are categorised by digestibility, fermentability by microbes in the gut and solubility.34,35 This discussion on carbohydrate places a greater focus on digestible carbohydrate and indigestible soluble/fermentable fibre than on insoluble fibre; the impact of insoluble fibre on glycaemia in cats appears to be negligible (Table 2).
Carbohydrate types that contribute to nitrogen-free extract and the declared carbohydrate content in pet food

Although the digestible carbohydrate content of a diet is often the primary concern when selecting diets for cats with DM, in practice it is difficult to ascertain accurately, not least because manufacturers are not required to declare carbohydrate content on pet food labels. Further, rather than being measured directly, carbohydrate content is instead estimated by calculating the nitrogen-free extract (NFE); this represents the difference between 100 and the sum of protein, fat, crude fibre, ash and water content (as a percentage of food as-fed) (Table 3). When the NFE estimation is reported by a manufacturer, it typically includes both digestible carbohydrate and indigestible soluble/fermentable fibre, such that it may overstate the content of digestible carbohydrate content. Indeed, relying only on the NFE, a diet with increased soluble/fermentable fibre content can be classed as moderate-carbohydrate despite containing a low level of digestible carbohydrate. Although still an approximation, the calculation of NFE using total dietary fibre instead of crude fibre is considered to be a closer estimation of digestible carbohydrate content of a diet (eg, NFE = 100 – (crude protein + crude fat + total dietary fibre + ash + moisture). 37
Using nutrient data in percentage as-fed (g/100 g of food) to estimate macronutrient content for an example commercial diet
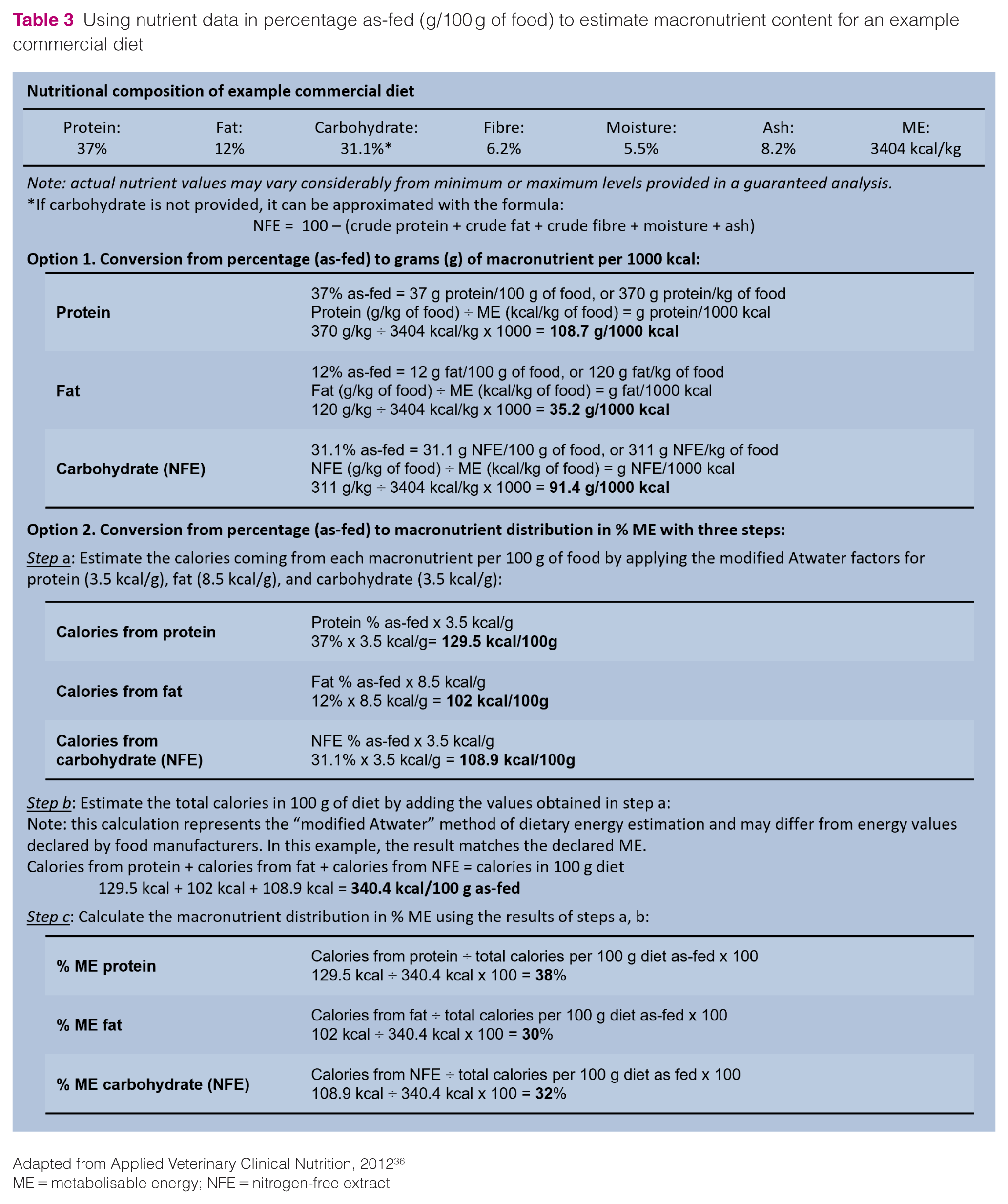
Adapted from Applied Veterinary Clinical Nutrition, 2012 36
ME = metabolisable energy; NFE = nitrogen-free extract
Digestible carbohydrates and glycaemic control in diabetic cats
Glucose is indispensable for certain tissues and in cats, as in other species, is produced during the breakdown of body glycogen or by gluconeogenesis. Although cats are classified as obligate carnivores, 38 they can readily digest and source glucose from digestible carbohydrates, which are not themselves nutritionally essential. Commercial feline dry and wet foods do not typically contain added sugars. Glucose derived from digestible carbohydrates (the most common in commercial pet foods being starch)39,40 has the potential to increase glycaemia directly.
Current evidence does not indicate that diets with ‘moderate’ to ‘high’ carbohydrate content are associated with DM development in cats.40–42 However, reducing digestible carbohydrate intake should be beneficial in managing feline DM through alleviation of the demand for insulin production via reduced postprandial glucose load. Reducing dietary carbohydrate intake might also improve regulation of blood glucose by modulating gluconeogenesis. 14 European legislation decrees that diets for cats with DM do not exceed more than 62 g of mono- and disaccharides per kg of food with a moisture level of 12%. 43 Although starch is the main contributor to digestible carbohydrate content, especially in dry diets, it is rarely quantified on pet food labels. Pet food manufacturers should indicate the starch content of therapeutic diets designed for cats with DM.
Dietary fibre
Dietary fibre is a functional description for carbohydrates and other substances that are indigestible by the animal in question. Dietary fibres are categorised on the basis of their viscosity, which refers to gel formation when hydrated, solubility in water and anaerobic fermentability by microbiota in the distal intestinal tract. 34 Dietary fibre is not essential for cats, but it can often provide them with specific health benefits derived from its physiochemical properties, 34 and when fermented by intestinal microbiota, provides important metabolites that support gastrointestinal function and possibly glycaemic control. 44
Depending on its type, fibre can also have an impact on glucose homeostasis and insulin requirement, as has been shown in humans.45,46 In general, soluble fibres delay gastric emptying and form viscous gels, effects that can result in slower digestion of starch and absorption of glucose. 47 In addition to a potential effect on intestinal microbiota, fibres may indirectly affect incretin hormone and adipokine secretion, which together may help to improve glucose homeostasis. 44 Insoluble fibres in particular can also reduce the caloric density of a diet and might also improve satiety, although evidence in cats to support the latter effect is mixed.48–51 However, excess inclusion of certain fibres can decrease the bioavailability of other essential nutrients and negatively affect stool consistency. 5
Protein
The amount of protein qualifying a diet as ‘high in protein’ has not been defined for cats but the minimum dietary protein requirement is empirically determined to be the smallest amount that will maintain muscle mass and prevent a net loss of body nitrogen. 53 Using this definition, the dietary protein requirement of cats is about twice that of dogs and humans. 54 The protein content of most over-the-counter feline diets comfortably exceeds this minimal dietary protein requirement.
Additional dietary protein beyond the minimal requirement is used principally for provision of metabolic energy rather than to replace body protein.55,56 Although excess protein may improve food palatability, catabolism of protein, unlike that of carbohydrate and fat, generates metabolic acid and nitrogenous waste, which might be undesirable in some clinical contexts, such as CKD. 57 Replacement of digestible carbohydrate with excess protein that provides equivalent ME might be beneficial when improvement in glycaemic control is sought.
The fasting and postprandial glycaemic responses of lean, healthy adult cats were moderately lower after 5 weeks of ingestion of a dry, ‘high-protein’ diet with ‘moderate’ carbohydrate content (ME carbohydrate/protein/fat: 27%/47%/26%) compared with a dry, ‘low-protein’ diet with ‘high’ carbohydrate content (ME carbohydrate/protein/fat: 48%/26%/27%). 16 The less marked glycaemic response might have been a consequence of increased protein intake, reduced intake of digestible carbohydrate or both. Glucose generated from the catabolism of amino acids through gluconeogenic pathways will be delayed in entering the circulation compared with glucose readily available from digestible carbohydrates. On a caloric equivalence basis, ingested protein should be less glucogenic than digestible carbohydrate because some amino-acid carbon is used to make ketones rather than glucose. 58 Moreover, amino acids from protein digestion might potentiate glucose-stimulated insulin secretion and stimulate secretion of enteroendocrine factors (ie, cholecystokinin [CCK], glucagon-like peptide 1 [GLP-1] and glucose-dependent insulinotropic peptide [GIP]) in cats more effectively than glucose, and that might slow digestion of food and increase satiety.12,59,60 However, the effect of dietary protein on satiety in cats is not entirely clear, with evidence that high-protein diets can both promote voluntary food intake or, conversely, reduce appetite.48,61,62
In humans, increasing dietary protein content in exchange for digestible carbohydrate might mitigate body protein wasting resulting from insulin insufficiency, caloric deficit caused by hyporexia observed in some cases or intentional caloric restriction to improve insulin sensitivity by reducing adipose mass.63,64 In addition, substituting dietary protein for digestible carbohydrates might be beneficial in helping to resolve hepatic lipidosis, 65 which may be coincident with feline DM. 66
Fat
Dietary fat is important for providing energy, as a palatant when derived from animal sources, 67 a source of nutritionally essential omega (ω)-3 and ω-6 fatty acids, and a matrix for absorption of the essential vitamins A, D, E and K. Dietary fat is, on average, 2.25 times more energy dense than either dietary carbohydrate or dietary protein (Figure 2).
The energy omitted when restricting carbohydrate content in a therapeutic diet for diabetic cats may be replaced by dietary fat rather than protein. Unlike dietary protein and digestible carbohydrate, dietary fat does not substantively generate glucose. Increased fat content is appropriate when a ‘low- but adequate-protein’ diet is desired – for example, for cats with concurrent DM and CKD. In humans, increasing dietary fat content slows gastric emptying 68 and increases the incretin response to feeding. 69 Fat is a potent stimulator of CCK release in cats 59 and promotes GIP secretion to a greater extent than proteins and glucose. 12 Indeed, GIP secretion occurs more widely along the gastrointestinal tract of cats compared with other species 70 and has been identified as a therapeutic target for the treatment of diabetes and obesity in humans. 71 The net effect of increasing fat ingestion is a reduced rate of glucose entry from the intestine, potentiation of insulin secretion and inhibition of glucagon release.
Dietary fat is less satiating than protein and carbohydrate, and its high potency in stimulating food reward may promote a preference for high-fat diets.72,73 An important consideration for cats with DM is that a ‘high’ dietary fat content can induce undesired weight gain if food intake is not controlled.74,75 In cats with or at risk of DM, excessive body fat promotes the development of insulin resistance, which effectively slows the rate of glucose disposal and increases the amount of insulin secretion needed to maintain euglycaemia. 76 Finally, feeding diabetic cats ‘very high-fat’ diets that are ‘very low’ in digestible carbohydrate content with an adequate protein content might theoretically heighten the risk of developing ketoacidosis, as reported in humans with DM. 77 Future research should focus on the ideal fat content in a diet for a cat with DM, taking into account the cat’s categorisation in the classification system (Figure 1).
Water
Most cats are maintained on commercially available dry (extruded) or wet (canned, pouch) diets. By weight, dry diets contain approximately 5–10% water and 2.7–5.0 kcal ME per gram whereas wet diets contain approximately 75–87% water but only 0.5–1.2 kcal ME per gram (Figure 2). When maintained on a wet diet, cats tend to ingest less energy than they would if given continuous access to dry diets. 78 This behaviour may result from caloric dilution by water or a decline in the palatability of wet diets after opening the package. Although wet diets typically have a high fat content, they also tend to have a high protein content, which is more satiating than fat. 72 Compared with wet diets, dry diets might delay gastric emptying which, in people, is associated with greater satiation; 79 however, the greater gastric distension from ingestion of wet food might also aid in post-ingestive satiation.80 –82 Therefore, feeding wet instead of dry food can facilitate energy restriction and promote weight reduction in cats, whether diabetic or not. Increasing dietary water content can also increase the energy expenditure per number of calories consumed.82,83 In humans with DM, exercise can improve insulin sensitivity; 84 similar benefits might potentially result from the greater physical exercise observed in cats fed a wet diet compared with a dry diet. 82 Increased physical activity might explain the greater probability of owner-reported DM remission in cats fed predominantly wet diets compared with a combination of dry and wet diets. 85 In addition, feeding cats a wet diet is associated with a higher total water intake,86–88 which can aid hydration that may be of benefit in DM.
Nutritional intervention studies in diabetic cats
Few nutritional intervention studies have been conducted in diabetic cats, with the majority focusing on the potential benefits of low carbohydrate content. However, these studies were either poorly controlled or not controlled at all. Interpretation of the results to help identify the ideal macronutrient composition for cats with DM is also confounded by the fact that a diet is the sum of its parts, and one macronutrient cannot be modified without impacting another. Therefore, it is difficult to ascertain if the clinical effects observed were due to the macronutrient that was intentionally decreased or the remaining macronutrient(s) that were consequently increased. To facilitate comparisons among diets in different studies, values for macronutrient composition are expressed as percentage ME, either taken directly from the cited research or calculated as detailed in Table S1 in the supplementary material.
In one study, changing diabetic cats (n = 9) from a ‘high-fibre, moderate-fat’ wet diet (ME carbohydrate/protein/fat: 23%/39%/38%; 2.7% crude fibre as-fed) to a ‘high-protein, low-carbohydrate’ wet diet (ME carbohydrate/protein/fat: 7%/46%/47%; 1.0% crude fibre as-fed) resulted in a decrease in daily insulin dose in 8/9 cats and discontinuation of insulin treatment in 3/9 cats. 8 Furthermore, in an uncontrolled study, improved glycaemic control occurred in 18 diabetic cats fed a ‘low-carbohydrate’ wet diet (ME carbohydrate/protein/fat: 5%/34%/61%) containing acarbose (an intestinal glucose absorption inhibitor) for 4 months, with exogenous insulin being discontinued in 11 cats. 89 In a randomised trial, exogenous insulin was discontinued in more diabetic cats fed a ‘low-carbohydrate, low-fibre’ wet diet (ME carbohydrate/protein/fat: 12%/37%/51%; fibre 0.1 g/100 kcal [fibre type not specified]) compared with those fed a ‘moderate-carbohydrate, high-fibre’ wet diet (ME carbohydrate/protein/fat: 26%/40%/41%; fibre 3.1 g/100 kcal [fibre type not specified]) (22/31 vs 13/32); however, glycaemic control improved in both groups. 7 As protein % ME was relatively similar between the diets used in this study, it could be postulated that the effects observed were due to lower carbohydrate, higher fat or a combination of both.
In another study, likely underpowered, there was no difference in glycaemic control between diabetic cats fed a ‘low-carbohydrate, high-protein’ diet (dry-format option, ME carbohydrate/protein/fat: 12%/49%/40%; wet-format option, ME carbohydrate/protein/fat: 9%/44%/47%) (n = 6) and those fed a ‘moderate-carbohydrate, high-protein diet’ (dry-format option, ME carbohydrate/protein/fat: 28%/32%/40%; wet-format option, ME carbohydrate/protein/fat: 4%/37%/59%) (n = 6); despite the large differences in macronutrient composition across diet formats, cats in both groups were stated to have consumed relatively similar proportions of wet and dry. 9 A crossover study compared the impact of feeding a wet diet with ‘low’ insoluble fibre content for 24 weeks with a wet diet with ‘high’ insoluble fibre content. The ‘low’ insoluble fibre diet was achieved by adding corn starch to the base diet (total insoluble fibre: 11.3 g/Mcal; ME carbohydrate/protein/fat: 37%/43%/20%), while the ‘high’ insoluble fibre diet was achieved by adding cellulose to the base diet (total insoluble fibre: 58.4 g/Mcal; ME carbohydrate/protein/fat: 29%/48%/23%). 47 The ‘high’ insoluble fibre diet resulted in decreased serum glucose concentration compared with the ‘low’ insoluble fibre diet. Finally, in a randomised study that evaluated the effect of caloric restriction on achievement of remission, overweight diabetic cats in both the caloric restriction and control group were fed a wet diet (ME carbohydrate/protein/fat: 11%/64%/25%), a dry diet (ME carbohydrate/protein/fat: 26%/49%/24%) or a combination of the two. 21 A remission rate of 50% was achieved in the caloric-restriction group compared with 30% in the control group. Even in cats not achieving remission, caloric restriction resulted in significant improvements in glycaemic control (as measured by glycaemic variability and reductions in exogenous insulin requirements) compared with the control group. Although this study does not permit interpretation of benefits of different macronutrient contents, it does highlight that diets with such a macronutrient profile can be used to achieve diabetic remission in overweight diabetic cats when fed to caloric restriction or to weight maintenance.
It is noteworthy that 4/67,8,47,89 of the aforementioned nutritional intervention studies in cats with DM fed exclusively wet diets, while the remaining two used a mixed-feeding regimen.9,21 A comparison of the effectiveness of wet diets vs dry diets in cats with DM has not been studied, although in one study where mixed feeding was applied, the odds of achieving diabetic remission were not impacted by the proportion of wet/dry diet consumed. 21
In a retrospective questionnaire study in diabetic cats (n = 405), based on the recall of the cat owners, the chance of achieving independence from exogenous insulin was three-fold higher when the cats were predominantly fed (⩾75%) a commercial ‘low carbohydrate’ wet diet compared with either dry or wet therapeutic diets. 85 The composition of these diets was not described in the study, and the definition of diabetes and diabetic remission differed from internationally agreed definitions. 85
Interpretation of the results of these studies is confounded by multiple factors. Diets differed not only in carbohydrate content but also in energy density, and protein, fibre and fat content. The carbohydrate source and proportions that were digestible also varied and were often poorly defined. The effect of weight loss (either intentional, eg, through deliberate caloric restriction, or unintentional, eg, due to poor diabetic control) on glycaemic control is also a potential confounder when attempting to compare studies. For example, in one study of cats with concurrent DM and obesity, improved response to a ‘low carbohydrate’ diet was associated with reductions in body fat percentage and an increase in lean body mass. 89 With these limitations in mind, diets with a low carbohydrate content appear to be most effective in improving glycaemic control and inducing diabetic remission, although improvements in these outcomes were also found with moderate carbohydrate diets containing a low-digestible carbohydrate (ie, starch) content when provided as part of a weight reduction programme. Much research remains to be conducted to determine the ideal macronutrient profile for cats with DM.
Overarching principles in dietary management for diabetic cats
Recommendations in this section apply to all categories and subsidiary dimensions of the classification framework. For guidance specific to each DM category of cats, please see the corresponding sections that follow.
Several diet types are available for feeding healthy pet cats, including commercially manufactured and home-prepared diets presented in various formats (eg, ‘fresh’, dry extruded kibble, wet [can or pouch] and freeze-dried) and designed to offer various attributes. In all cases, the primary diet (ie, the food from which a cat derives ⩾90% of its energy and essential nutrients) should be nutritionally complete and balanced for their life stage, most likely adulthood in the context of DM.
Although lacking some of these advantages (see Box 1), some commercial non-therapeutic diets (also known as ‘over-the-counter’ or ‘retail’ diets), particularly wet formulas, will have macronutrient profiles that align with nutritional recommendations for diabetic cats. These diets might be preferred because they are more available or affordable than therapeutic diets. Dietary macronutrient content relative to energy (eg, percentage of ME or grams per 1000 kcal) is not required on a pet food label, although the information may be provided on manufacturer websites, in product guides or on request by contacting the manufacturer. Using the available information from the guaranteed or typical analysis, veterinarians can also perform calculations to convert nutrients from percentage as-fed to an energy basis (Table 3).
Purpose-formulated diets for diabetic cats

Macronutrient composition is not the only consideration in diet selection for diabetic cats. Over-the-counter diet formulations and many purpose-formulated diets are usually designed to provide adequate concentrations of essential nutrients when fed at an average daily energy allowance for weight maintenance (ie, 75–100 kcal/kg0.67). 90 Cats with below-average calorie requirements are at risk of unwanted weight gain if fed according to average calorie needs. In these cases, restricting calories below a manufacturer’s recommendations, to maintain a desirable BCS, might lead to inadequate essential nutrient intake. Therefore, over-the-counter or purpose-formulated diets designed to be fed for weight maintenance could be nutritionally inadequate for cats with low energy needs or cats requiring calorie restriction for therapeutic weight reduction. If a home-prepared diet is desired for a diabetic cat, consultation with an appropriately qualified veterinary clinical nutritionist is advised.
The nutritional approach should always be tailored for an individual cat’s needs (see Box 2 for general dietary recommendations), guided by both the DM category and findings from nutritional and medical assessment (see later sections). Although there is some evidence regarding the optimal dietary macronutrient profile for DM-C cats, minimal evidence is available on which to base recommendations for DM-A and DM-R cats.
General dietary recommendations for diabetic cats

Nutritional assessment and monitoring
Nutritional status is the fifth vital assessment, and at every visit, a nutritional assessment should be conducted, whereby patient, diet, environmental and feeding management factors are evaluated (Table 4). This guide gives the most relevant considerations for nutritional assessment of DM-A, DM-C and DM-R cats. For more comprehensive guidance about performing and using a nutritional assessment, readers are referred to Freeman et al. 92
Key factors evaluated in the nutritional assessment of cats with diabetes mellitus

Adapted from Freeman et al 92
BCS = body condition score; BW = body weight; CKD = chronic kidney disease; MCS = muscle condition score
Information gathered in the above nutritional assessment must be analysed alongside relevant medical information to determine the nutritional goals and plan for the cat. In formulating a nutrition plan, veterinarians should evaluate whether the current diet composition meets the defined nutritional goals or, if diet change is warranted, recommend an alternative feeding (diet type, quantity, frequency of feeding, guidance on permissible treats or snacks and advice on feeding management) and monitoring plan.
Unintended and significant weight loss (eg, acute loss of ⩾5% BW over 1 week or chronic loss of ⩾10% BW over 1 month) suggests uncontrolled diabetes, concurrent disease processes or inadequate nutritional management, necessitating further medical work-up and possible dietary re-evaluation. In addition, hypo- or anorexia (⩾3-day duration of significantly reduced intake, counting days before presentation) that is not resolved by patient response to initial medical interventions warrant prompt consideration of diabetic complication (eg, ketosis, pancreatitis) and interventions to support adequate food intake, which might include assisted feeding. 93
The energy needs of cats vary widely, but can be informed by evaluating the current calorie intake, BW and BCS, in conjunction with the desired trajectory of BW and BCS (targeting weight gain, maintenance or reduction) (Figure 1). If it is not possible to determine an appropriate daily energy requirement from evaluation of the diet history, this can be estimated using an appropriate formula (see Box 3). 90
Average daily energy requirements of adult cats

To avoid overestimation when calculating energy allowances for cats who are overweight or have obesity, calculations should be based on the ideal, rather than the current, BW. If the ideal BW cannot be determined from sources, such as historical weights in the cat’s medical records, it can be calculated from the current BW and BCS (Figure 3).

Calculation of daily ration for a cat commencing therapeutic weight reduction. *Ideal body weight (IBW) is probably best determined from historical medical records, but if this is not possible, it can be estimated from the current weight and body condition score (BCS). Estimation of IBW is a guide and, because of individual variability, it may either over- or under-estimate IBW. †Alternative equations exist for the calculation of energy requirement. 94 The equation proposed here has been previously used for therapeutic weight reduction studies in both overweight 49 and overweight and diabetic cats.21 ‡An allowance for 10% of calories from treats may be incorporated but these should be deducted from the daily calorie allocation for the main meal
For cats in the DM-C and DM-R categories, ongoing monitoring of their nutritional status should continue alongside monitoring of glycaemic control. The monitoring should include a review of diet acceptance and tolerance, based on verbal reports from the owner’s recollection or from a food diary. Adherence to dietary recommendations should be evaluated, while current energy intake should be compared with expected or estimated requirements. The veterinarian should also evaluate whether the degree and rate of any desired changes in BW and BCS are appropriate. If therapeutic weight reduction has been implemented (see section ‘Principles and practicalities of therapeutic weight reduction’), changes in BW should be used to determine progress. Over the longer term, the feeding plan should be adjusted to ensure that goals for BW and BCS are met, with food intake being incrementally increased or reduced by small amounts (typically 5–15% at a time), with re-evaluation in 2–4 weeks until the target BW has been achieved. Beyond therapeutic weight reduction, daily energy intake might need to be adjusted according to changes in glycaemic status and insulin sensitivity because of medical treatment.
Principles and practicalities of therapeutic weight reduction
Obesity is an important medical condition in domestic cats; among adults in the 7–12-year-old range, 21.7% have an obese body condition and 44.8% are considered overweight. 95 The association between obesity and DM has been demonstrated in many studies;96 -99 the likelihood of DM is up to four times greater in cats with obesity, 35 and the risk increases with increasing severity of obesity.97,99 Obesity causes insulin resistance in cats, 1 and in other species, this leads to an increase in insulin production to maintain normoglycaemia.100,101 Individuals with underlying β-cell dysfunction, β-cell loss or both cannot compensate completely for this increased insulin requirement, resulting in relative and, eventually, absolute insulin deficiency and hyperglycaemia, 101 which can then progress to DM. Depending on the stage of disease (eg, severity of β-cell dysfunction), therapeutic weight reduction can improve insulin sensitivity to a manageable level, sometimes inducing remission. 21 Attaining and maintaining a healthy BW is, therefore, a critical aspect of management in all three DM categories. Change in BW over time is a more sensitive and precise marker than BCS for changes in energy balance in the individual, is easy for the owner to measure regularly at home and is an excellent tool for monitoring change over time. Dedicated digital scales may assist the pet owner in this respect.
Therapeutic weight reduction protocol for diabetic cats
For any cat with obesity, veterinary professionals need to take time to develop a therapeutic weight reduction protocol that is tailored to the individual (see Box 4). During initial planning, both an ‘ideal’ and a ‘target’ BW need to be set. The ideal weight is a weight within the body fat range considered to be optimal for health, where calculations best reflect energy requirements. Although the healthy BW range arguably includes BWs covering both BCS 5/9 and 6/9, depending upon priority (eg, minimising disease risk or maximising longevity), energy estimates based on a BW corresponding to BCS 5/9 are most appropriate when setting a therapeutic weight reduction protocol.104 –108 The target weight is defined as the endpoint of therapeutic weight reduction decided based on the clinical priorities for the cat. In many cases, the target weight and ideal weight do not match – for example, when only a modest reduction in BW is necessary to improve health.
Dietary strategy for therapeutic weight reduction in diabetic cats with obesity

There is little research on the energy requirements for therapeutic weight reduction in cats with DM but, from the experiences of some of the authors, the same calculations can be used as those for non-diabetic cats on a weight reduction plan. Figure 3 presents one method for calculating this starting energy requirement, while recognising that others exist. Although starting energy allocations may differ, depending upon the calculation method, in practice the actual daily energy intake tends to converge because allocations are adjusted incrementally over time according to the progress of weight reduction. Using this approach, a realistic rate of weight reduction is typically in the range of 0.5–1.0% per week,104,108 a rate at which lean tissue loss should be minimised. 107
Weight rebound is a common phenomenon affecting approximately half of cats who have successfully reached their target weight; cats aged under 9 years are at greater risk. 106 Although the benefits of feeding weight reduction diets to avoid weight rebound after a therapeutic weight reduction programme in cats has not yet been shown, data do exist for dogs. 109 In any case, BW should be monitored regularly even after the completion of a weight reduction programme. Monitoring can coincide with medical checks for DM and modifications to food allocation should then be made if necessary.
How to feed a cat with DM and make dietary transitions
A plan for nutritional management is always best addressed during a bespoke consultation, which provides the time and opportunity to discuss management in detail and address owner concerns and questions. Besides the technicalities of recommending a specific diet(s) and daily feeding quantity, the plan needs to give owners practical guidance on the cat’s feeding regimen (Table 5). The owner may not fully appreciate the amount of variation in energy density among cat foods and that starting a recommended diet may require a substantial change in portion size compared with the previous diet. The owner should be clear about exactly how much to feed and ideally use electronic food scales rather than volumetric measurements (eg, allocations using a measuring cup) to ensure accurate rationing. Owners of cats with polyphagia or a history of obesity may need to be counselled on how much food a healthy cat would be expected to eat and on strategies to address food-seeking behaviour (see Table 5).
Summary of recommendations on how to feed a cat with diabetes mellitus

Basal insulin: an insulin that mimics the physiologic basal secretion of insulin (ie, insulin secretion between meals). An insulin formulation is considered to be ‘basal’ if its time-action profile is relatively flat and preferably ‘peakless’; namely, having no distinct peak throughout and even beyond a 24-h period 3
Having regularly spaced meals of equal size might improve diabetic control in cats with DM by decreasing variation in postprandial glycaemic load. However, there is more flexibility regarding the timing of food consumption with insulin-treated cats compared with diabetic dogs and the authors do not consider it critical to time insulin injections to coincide with feeding. In addition, strictly feeding at set times is neither feasible nor desirable for some cats, including those that are habitual grazers or have a picky appetite. In such cases, food can be provided throughout the day, allowing the cat to graze at will. Nonetheless, it is still important to regulate the overall daily food intake for all cats.
Nutritional guidance by proposed DM classification
The following sections provide specific nutritional recommendations for each of the proposed clinical categories (Figure 1). Please refer to Figure 2 for the descriptors high, moderate and low in the context of the recommendations that follow.
Nutritional guidance for DM-A cats
The optimal nutritional plan for a DM-A cat will take into consideration whether nutrition has the potential to modify the individual’s risk factors for DM, the presence of a nutritionally responsive comorbidity and any need to adjust energy intake to achieve or maintain an acceptable BCS. There are many risk factors for feline DM,11,110 some of the most significant of which are shown in Figure 4, along with a decision tree to help aid whether a specific nutritional approach is warranted.
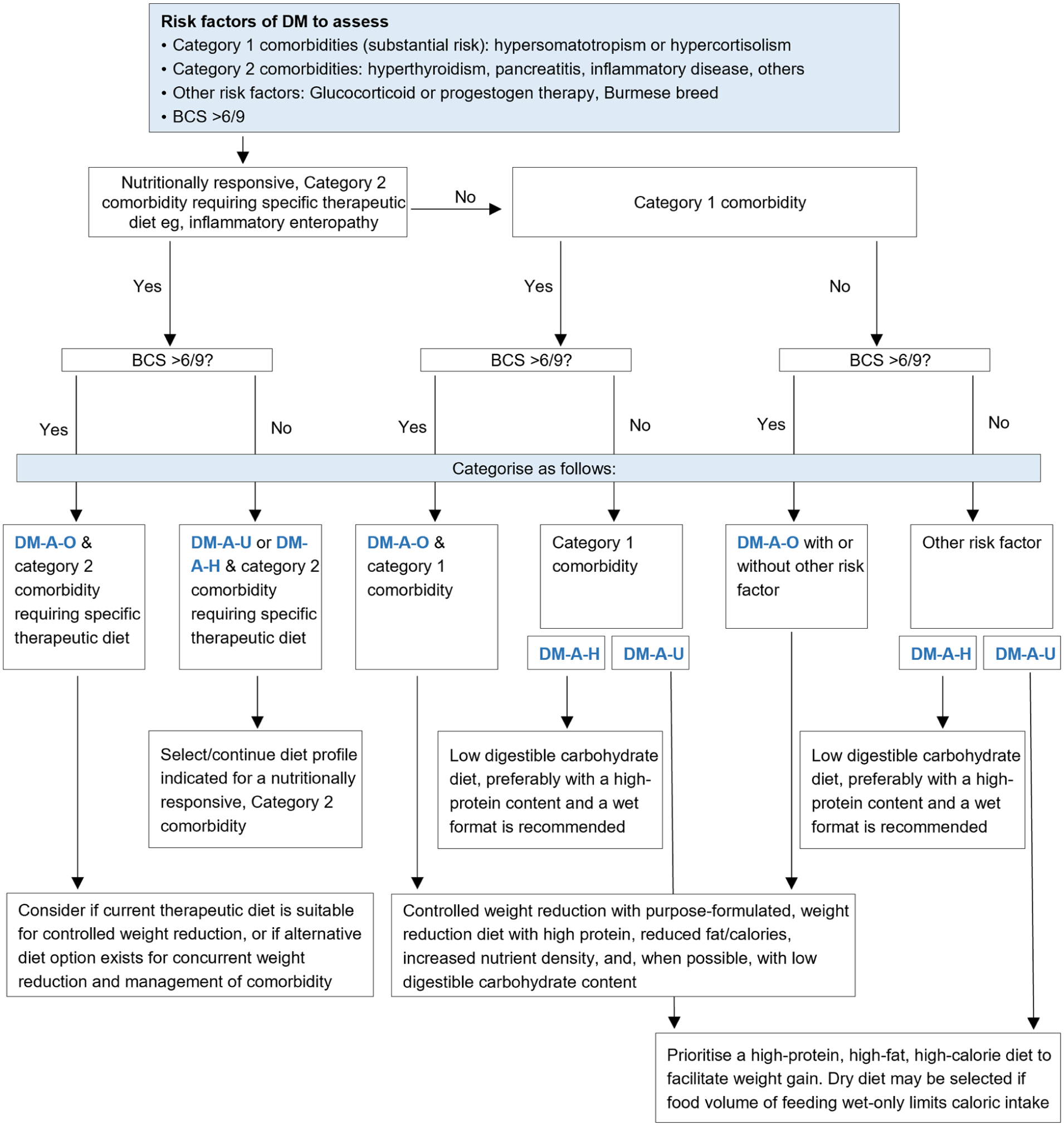
Decision tree to determine whether a specific diet composition is warranted for a DM-A cat. BCS = body condition score; DM = diabetes mellitus; DM-A = at risk of DM; DM-A-H = at risk of DM with a healthy body condition; DM-A-O = at risk of DM with an overweight body condition; DM-A-U = at risk of DM with an underweight body condition
Risk factors for diabetes can influence the diet composition selected for a DM-A cat in different ways (Figure 4). In general, if there is no medical indication other than DM that might affect the appropriate dietary carbohydrate content to target, decreasing digestible carbohydrate intake can be considered in order to minimise postprandial glucose and insulin responses, and avoid glucotoxicity in cats with suspected abnormal glucose metabolism or subclinical DM. 16 That said, no reliable methods currently exist to detect subclinical DM or identify which DM-A cats will progress to clinical DM; 13 in addition, while current evidence does not indicate an association between increased carbohydrate intake and the development of DM in cats, the impact of reduced carbohydrate intake on risk of DM development in at-risk cats remains to be investigated. However, given what is known about the nutritional management of DM-C cats, it would be reasonable to expect a beneficial effect of reduced digestible carbohydrate intake in DM-A cats with abnormal glucose metabolism. Owners of cats at significant risk of DM should be made aware of the clinical signs to monitor for in their pet.
Obesity is arguably the DM risk factor most amenable to nutritional intervention and a DM-A-O classification is probably the most common reason for recommending dietary modification in a DM-A cat. In the authors’ opinion, cats with hypersomatotropism or hypercortisolism, but without clinical DM, are also likely to benefit from dietary modification (Figure 4). This scenario is uncommon. However, the fact that most cats with hypersomatotropism or hypercortisolism are clinically diabetic at the time of diagnosis suggests that any non-diabetic cat with these conditions should be considered at substantial risk of developing clinical DM in the near future.111,112
Figure 5 proposes nutritional recommendations for DM-A cats according to BCS, BW trajectory and the development of comorbidities.
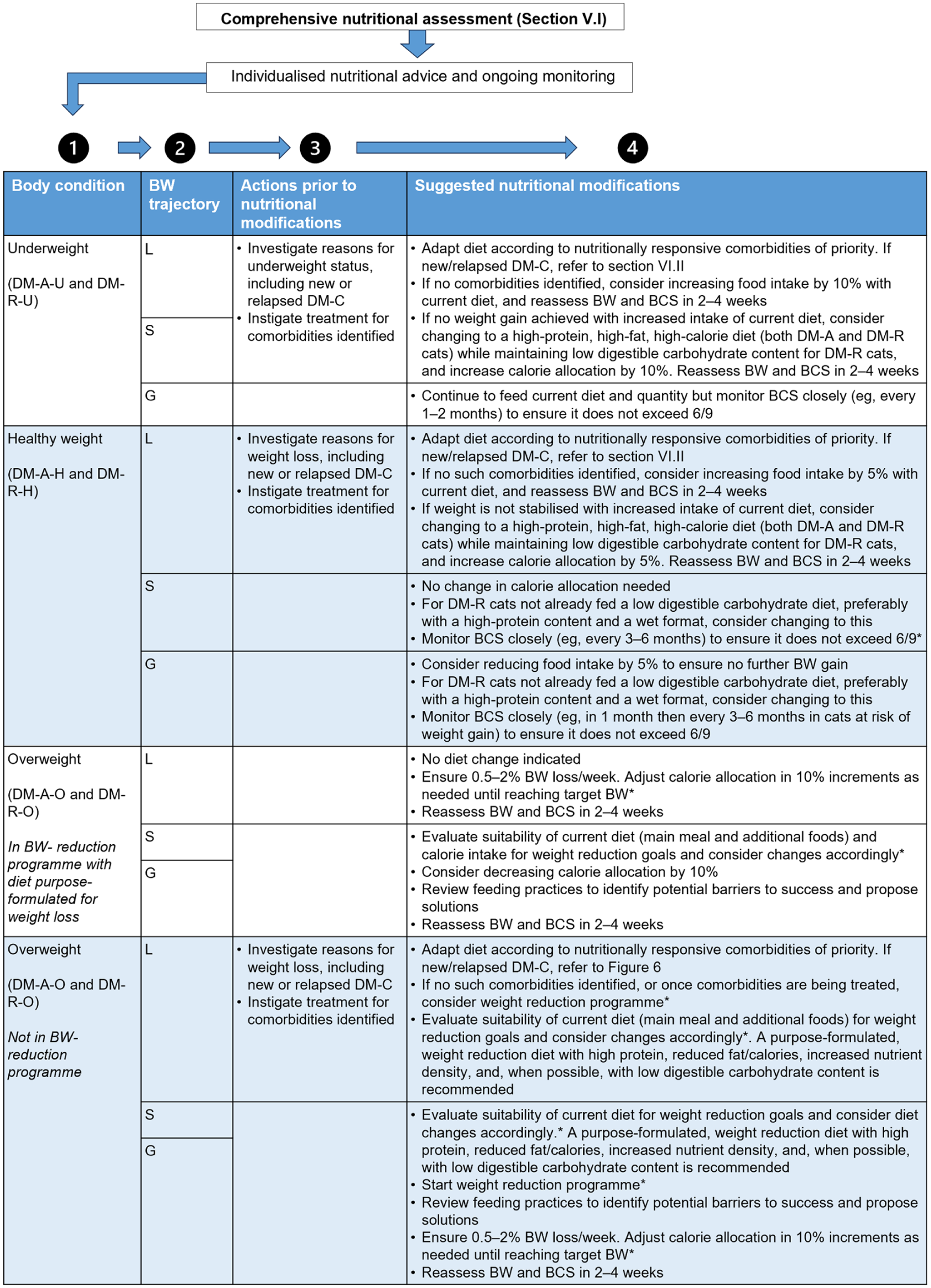
Proposed nutritional guidance for cats at risk of diabetes mellitus (DM-A) and cats in remission from DM (DM-R). *Please refer to section ‘Principles and practicalities of therapeutic weight reduction’. Note that therapeutic weight reduction is most likely to succeed when the veterinarian and owner can fully focus on the weight reduction programme as a single intervention rather than one introduced alongside management of another comorbidity. BCS = body condition score; BW = body weight; DM-A-H = at risk of DM with a healthy body condition; DM-A-O = at risk of DM with an overweight body condition; DM-A-U = at risk of DM with an underweight body condition; DM-R-A = in remission from DM with a healthy body condition; DM-R-O = in remission from DM with an overweight body condition; DM-R-U = in remission from DM with an underweight body condition; G = gaining; L = losing; S = stable
Nutritional guidance for DM-C cats
Selecting a nutritional strategy to complement medical management in DM-C cats
Once pathological weight loss has been arrested and the owner and cat are ready to address the potential benefit of dietary changes (see Box 5), the veterinarian should establish if there is a rationale for specific nutritional modifications (eg, if a cat’s current diet does not align with the recommendations, there would be reason to consider changing it). Factors to consider when making such decisions include assessment of current glycaemic control, nutritional assessment (especially BCS, the current BW and any changes relative to diagnosis, evaluation of feeding practices and diary; see section ‘Nutritional assessment and monitoring’) and, ultimately, the treatment goals for the individual patient.
Initial nutritional considerations at diagnosis of DM or relapse

The nutritional approach for a DM-C cat should then be guided by the cat’s current BCS, recent BW changes, the desired BW trajectory and owner-provided dietary history (Figure 6). Cats with historical obesity will often present as DM-C-H or DM-C-U due to DM-induced weight loss. Information on prior obesity can provide insights into the patient’s risk of obesity recurrence and inform the nutrition and monitoring plan. As for cats in all DM categories, adjustments to the initial nutritional plan, such as diet type and caloric allocation, should be based on ongoing veterinary reassessment of the cat’s response to therapy. In DM-C cats, the clinician should always be conscious of the potential effects of dietary modification on glycaemic control.

Proposed nutritional guidance for cats with clinical diabetes mellitus (DM-C). *Please refer to section ‘Principles and practicalities of therapeutic weight reduction’. Note that therapeutic weight reduction is most likely to succeed when the veterinarian and owner can fully focus on the weight reduction programme as a single intervention rather than one introduced alongside management of another comorbidity. BCS = body condition score; BW = body weight; DM-C-H = clinical DM with a healthy body condition; DM-C-O = clinical DM with an overweight body condition; DM-C-U = clinical DM with an underweight body condition; G = gaining; IGF-1 = insulin-like growth factor-1; L = losing; S = stable; SGLT2, sodium glucose co-transporter 2
A low-digestible carbohydrate diet, preferably with a high-protein content and wet format, is usually recommended for a DM-C cat (Figure 6), including for DM-C-H cats with good glycaemic control, because it might increase the likelihood of achieving remission. It is important to highlight that diets containing moderate carbohydrate content might be acceptable for cats with DM, but only if the digestible carbohydrate fraction remains low.
In DM-C-U and DM-C-O cats, other dietary factors are prioritised to ensure that the overall diet profile is best adapted to support goals relating to BW and BCS (Figure 6). In DM-C-U cats, a low-digestible carbohydrate, high-fat, high-protein, energy-dense diet is recommended to facilitate weight gain. In DM-C-O cats with stable signs of DM and fair-to-good glycaemic control, therapeutic weight reduction is recommended by feeding a purpose-formulated high-protein, reduced fat and energy-dense diet with an increased essential nutrient density relative to energy content. When available, a low-digestible carbohydrate wet formula that meets these criteria is the preferred choice (see Box 2), although a combination of wet and dry food can be used. Ideally, this diet should be continued even after the target weight has been achieved, as a means to help prevent weight regain and ensure adequate nutrition, given that cats have relatively low maintenance energy requirements after weight reduction. 113
Unintended weight loss in a DM-C cat after starting treatment usually indicates that the treatment is inadequate or not optimised to achieve good glycaemic control. In such cases, veterinarians should investigate treatment compliance, confirm adequate caloric allocation and intake, and re-evaluate medication type and dose. If uncontrolled weight loss continues despite good glycaemic control practices and appropriate food intake, the cat should be rechecked for comorbidities contributing to weight loss, such as exocrine pancreatic insufficiency, chronic enteropathies, pancreatic or other type of tumour, CKD, hyperthyroidism and pancreatitis. A dietary change might be necessary if there is a comorbidity that requires a different nutritional approach from that of DM (eg, CKD); otherwise, the nutritional plan should be guided by weight status.
Integration of dietary and medical strategy
Our proposed classification system for cats with DM includes a subsidiary dimension that indicates current medical treatment: insulin (Ti), oral SGLT2 inhibitor (Ts), other treatment (Tx) and no specific therapy for glycaemic control (Tn) (Figure 1). Both medical and nutritional management will have an impact on glycaemic control and the overall demand for endogenous insulin with exogenous insulin or SGLT2 inhibitors treatments predicted to alleviate glucotoxicity; the reduction of insulin demand and alleviation of glucotoxicity will increase the likelihood that residual β-cell capacity will become sufficient to achieve and maintain diabetic remission.
The restoration of adequate glycaemic control in DM-C cats should also lead to improvements in clinical signs such as weight loss, polyuria, polydipsia and polyphagia. However, a substantial proportion of cats cannot self-regulate their food intake to maintain ideal body condition and will overconsume when food is available; this might be incorrectly perceived as ongoing polyphagia and can lead to unwanted weight gain.
The effects of any dietary change on medication requirement must be closely monitored and considered. Typically, transitioning from a dry diet to a wet diet will substantially reduce intake of digestible carbohydrate, causing a substantial and rapid reduction in insulin requirements that might lead to hypoglycaemia in insulin-treated cats. The profiles of the specific diets being transitioned from and to should be compared to assess the potential impact on medication requirement; some dry diets have a similar digestible carbohydrate content to some wet diets, and not all wet diets are equally low in digestible carbohydrates. Furthermore, since total carbohydrate intake is determined defined by a combination of dietary carbohydrate content and the quantity of diet consumed, the impact of changing caloric allocation on carbohydrate intake should also be considered.
In summary, decisions on the management of cats with DM must always take account of the dynamic interactions between diet type, food intake, medication, BW, body composition and the functional capacity of the endocrine pancreas. When possible, it is advised to refrain from simultaneous changes in nutritional and medical management.
A range of insulin formulations are prescribed for diabetic cats, dependent on licensing and availability in different countries; the modalities of insulin treatment in cats have recently been reviewed. 114 It should be emphasised that our current understanding of specific nutritional strategies for diabetic cats that informs the guidance in Figure 6 is largely derived from evidence in cats treated with exogenous insulin.
Although further research is required to understand the optimal nutritional strategy to complement treatment with the recently available SGLT2 inhibitors, it is likely that adapted nutrition will have a role in the management of these cases. Gastrointestinal side effects of loose faeces and diarrhoea were observed in some cats treated with velagliflozin6,115 and bexagliflozin, 5 and are likely due to off-target inhibition of SGLT1 receptors in the gut, 116 leading to incomplete absorption of glucose and secondary osmotic diarrhoea. 117 These side effects were self-limiting and resolved within a week in most cats treated with velagliflozin. 115 Humans treated with SGLT2 inhibitors typically experience modest weight loss, but the effects of these medications on the BW and composition of cats is currently unclear. 118 Weight gain has been reported with both velagliflozin 6 and bexagliflozin 5 treatment in diabetic cats, consistent with findings from a randomised controlled clinical trial in which there were fewer cats in underweight body condition after velagliflozin treatment compared with before. 115 However, the proportion of cats with an overweight BCS also decreased in the study, and it is not clear whether these changes in BCS were directly attributable to medication because dietary factors were not controlled. In clinical trials of SGLT2 inhibitors in diabetic cats, polyphagia was the clinical sign least likely to show improvement,5,6,115 a finding that could have clinical implications for BW management. The polyphagia observed was most likely multifactorial; it is possibly explained by compensatory hyperphagia linked with the energy losses from glucosuria, a hypothesised mechanism in people, 118 but it might also be a manifestation of poor self-regulation of food intake observed in some cats. In humans treated with SGLT2 inhibitors, diets with less than 26% ME carbohydrate are not recommended owing to the risk of euglycaemic ketoacidosis. 119 As the metabolic processes of carnivores are adapted to low carbohydrate consumption, and cats readily utilise dietary protein to maintain blood glucose via gluconeogenesis, it seems unlikely that this feeding strategy carries the same risk for cats treated with these medications. 120
Nutritional guidance for DM-R cats
Many DM-R cats are likely to have ongoing subnormal glucose tolerance.121,122 A major aim in their care is prevention of relapse to clinical DM, which is commonly observed. 123 There is no dedicated research on the optimum dietary composition for DM-R cats. However, it seems logical that a nutritional plan that decreases endogenous insulin requirement will help a cat’s β-cell function remain sufficient to maintain remission.
At remission, many DM-R cats are likely to receive a low-digestible carbohydrate diet that was instituted for management of their DM and which might be a therapeutic DM diet. In these cases, the authors advise continued feeding of a low-digestible carbohydrate diet if comorbidities allow. In time, an over-the-counter diet low in digestible carbohydrates might be considered a reasonable alternative to a therapeutic DM diet for reasons of accessibility and affordability. However, the suitability of the diet for achieving weight-management goals while ensuring nutritional adequacy must be evaluated, particularly in cats with below average energy requirements (see section ‘Overarching principles in dietary management for diabetic cats’).
Remission can sometimes occur even when a cat has not been fed a low-digestible carbohydrate diet. This might be the case for a cat fed a specific diet for management of a comorbidity and, in this instance, that diet should be continued. If this is not the case, transitioning to a low-digestible carbohydrate diet could still be considered. Although there is no evidence that reduced digestible carbohydrate intake can prevent relapse, implementing this dietary change is rational because of its potential benefits.
Feeding of DM-R cats can be adjusted over time according to changes in BW and BCS and the development of any new comorbidities, in which case guidance similar to DM-A cats (Figure 5) is recommended. For DM-R-O cats, therapeutic weight reduction programmes should either be continued or introduced because this should reduce obesity-associated insulin resistance and, therefore, decrease the likelihood of relapse of DM.
Effective communication of nutritional guidelines: a relationship-centred approach
The best advice on nutritional and disease management cannot be successful unless the carer understands and agrees with short- and long-term management goals and the recommended actions to achieve these. A relationship-centred approach is a collaborative partnership between the veterinarian and owner in which there is effective communication of clear recommendations and their rationale. This approach has a positive influence on owners’ implementation and adherence to those recommendations. 124 With little evidence-based information to guide effective relationship-centred communications during the monitoring of cats with DM, we suggest three key elements based on learnings from the field of obesity: trust, education and reinforcement (see Box 6).
Key elements of effective communication with owners of diabetic cats


Diabetes mellitus monitoring and communication plan. *ALIVE Diabetic Clinical Score: https://www.esve.org/alive/search.aspx. ALIVE = Agreeing Language in Veterinary Endocrinology; BCS = body condition score; BW = body weight
Conclusions
The clinical status of cats with DM evolves over time, depending upon individual, nutritional, therapeutic and management factors. The aim of the classification system proposed in this article is to help guide veterinary professionals towards better individualised nutritional recommendations for diabetic cats under their care. The provision of appropriate diets with low-digestible carbohydrate content, fed in an appropriate manner, can improve long-term health outcomes in diabetic cats, potentially preventing cats at risk from developing clinical DM (DM-A), helping DM-C cats to achieve improved glycaemic control and remission, and maintaining remission in DM-R cats. Nutritional management should be integrated with classical or newly developed medical management and take account of the dynamics of the cat’s nutritional status. Ongoing monitoring of BCS, BW and food intake, combined with monitoring of glycaemic status and any medical treatment, can improve clinical outcomes. Relationship-centred care built on effective communication might also contribute to successful outcomes. Overall, it is hoped that implementation of the advice offered herein can result in reduced rates of euthanasia, improved quality of life and extended longevity for diabetic cats. The classification system has additional potential to frame future research on the optimal combination of nutritional and medical interventions for cats at different stages of disease. In the future, advances in the diagnosis of subclinical DM may be incorporated into the classification system. Clinical experience with the use of this classification system and research into outcomes with its implementation are now needed.
Supplemental Material
Table S1
Macronutrient distribution calculations.
Footnotes
Acknowledgements
The authors wholeheartedly thank Susanna Ryan of Ryter Ltd for her meticulous and skilful editorial assistance, which was funded by Royal Canin SAS.
Supplementary material
The following file is available as supplementary material:Table S1: Macronutrient distribution calculations.
Conflict of interest
Tabitha Hookey, Tanya Schoeman and John Flanagan are employees of Royal Canin SAS. Charlotte R Bjørnvad has research, advisory and speaking contracts with Royal Canin and Boehringer Ingelheim, and speaker contracts with Hills and Vuffeli, but with no conflict of interest associated with the current manuscript. Robert C Backus served as director of the Nestlé Purina Endowed Program in Small Animal Nutrition at the University of Missouri, Columbia, during the period of manuscript preparation. Lucy J Davison is in receipt of research funding from the UK Medical Research Council, PetPlan Charitable Trust, American Kennel Club Canine Health Foundation, Dechra Veterinary Products and Evetts-Luff Charitable Trust, but with no conflict of interest associated with this manuscript. Linda Fleeman has received honoraria for educational seminars and manuscripts for Boehringer-Ingelheim, MSD Animal Health, Zoetis, Royal Canin and Nestle Purina, consulting fees from Dechra Boehringer-Ingelheim and Amacas, and research support from Royal Canin. Federico Fracassi has received honoraria for educational seminars, financial research support or consulting fees from MSD Animal Health, Dechra, Boehringer-Ingelheim, Royal Canin, Nestlé Purina, Hill’s, MyLav and Forza10. Alexander J German is an employee of the University of Liverpool but his position is funded by Royal Canin. He has also received financial remuneration and gifts for providing educational material, speaking at conferences and consultancy work. Chen Gilor had research, advisory and speaking contracts with Dechra, Edge Animal Health, CEVA, Okava, Merck Animal Health, BiomEdit, Boehringer-Ingelheim and Baycom. Ruth Gostelow has research, advisory and speaking contracts with Royal Canin. She is a member of the Boehringer Ingelheim Global Diabetes Advisory Committee.
Funding
The authors attended an Advisory Board meeting convened to discuss the nutritional management of cats with diabetes mellitus, funded by Royal Canin SAS, and were compensated for their time and for the preparation of this manuscript by Royal Canin SAS.
Ethical approval
This work did not involve the use of animals and therefore ethical approval was not specifically required for publication in JFMS.
Informed consent
This work did not involve the use of animals (including cadavers) and therefore informed consent was not required. No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

