Abstract
Objectives
The goal of this study was to describe the incidence of surgical site infections (SSIs) in a large feline population undergoing gastrointestinal (GI) surgery and to identify factors associated with the development of SSIs specific to GI surgeries in cats, with a special focus on the usage of peri- and postoperative antibiotic treatment.
Methods
Medical records of cats undergoing GI surgeries were retrospectively analysed. Data collected included demographic data, type of surgery, indication, data from the anaesthesia protocol, antibiotic use and postoperative outcome. A multivariable logistic regression model was used to identify factors associated with SSI.
Results
Of the 188 included patients undergoing GI surgery, 20 (10.6%) developed an SSI. Of these, 11 (55%) were classified as superficial incisional infections, four (20%) as deep incisional infections and five (25%) as organ/space infections. The absence of perioperative prophylactic antibiotic (PPA) administration was associated with the highest SSI rate (52.4%, 11/21), whereas significantly lower rates were observed in patients receiving PPAs alone (6.7%, 7/105), extended PPAs (7.1%, 1/14) or PPAs followed by continued antibiotic treatment (2.1%, 1/48). Prolonged antibiotic use after surgery in otherwise healthy animals showed no additional benefit in reducing SSI rates.
Conclusions and relevance
We found that PPA administration was associated with a significantly lower incidence of SSI in cats undergoing GI surgery, while extended PPA use was not associated with an additional benefit. These findings provide important guidance for evidence-based antibiotic use in feline soft tissue surgery and support efforts to minimise unnecessary antibiotic administration.
Introduction
Gastrointestinal surgery (GI) is a common procedure in small animals.1 –3 A recent study reported that the most frequent indication in cats (75%) is removal of a foreign body (FB). 2 Other indications include mass removal (14.1%), intussusception (5.1%) and complications from previous surgeries, such as stricture or dehiscence (3.8%). 2
Surgical site infection (SSI) represents an important surgical complication that can arise after GI surgery, causing increased patient morbidity and prolonged hospitalisation leading to increased costs.4 –6 Interestingly, despite GI procedures being frequently performed, evidence on the frequency of SSIs after GI surgery and factors associated with their development in small animal surgery remains limited. In cats, this situation is even worse. Only three studies have examined SSI rates after GI surgery in cats.2,5,7 These studies report SSI rates in the range of 3.8–19% after enterotomies and enterectomies.2,5,7 Unfortunately, none of these studies examined associations between perioperative antimicrobial practices or other clinical factors and the occurrence of SSIs.
In contrast, human medicine provides more detailed data, showing that SSI rates vary substantially depending on the type of surgery 8 and the section of the GI tract involved.8,9 For example, rates can be as low as 2.1% for appendectomies and rise up to 18.2% in colonic surgeries.8,10
Although we lack GI specific data, several studies, including multiple different procedure types, have suggested potential risk factors for SSIs in small animals, including age, American Society of Anaesthesiologists (ASA) classification score, wound classification, anaesthesia and surgery time, use of implants, duration of hospitalisation, hypotension, low body temperature during anaesthesia, increased body weight, sex and underlying conditions such as endocrinological dysfunction and perioperative prophylactic antibiotics (PPAs).4,11 –19 Of these, PPAs are especially important and represent a highly controversial topic in veterinary medicine. Eugster et al, 13 Degen et al 12 and Vasseur et al 18 have confirmed PPAs as a protective factor for SSIs in some surgeries; however, Nicholson et al, 14 Vasseur et al 20 and Stetter et al 11 were not able to come to the same conclusion. A recent scoping review just emphasised a lack of information on the protective effect of PPAs, especially in soft tissue procedures, and raised concerns regarding this important gap in knowledge. 21
In a time where the misuse of antibiotics in both human and veterinary medicine is considered an important factor contributing to the development of antibiotic-resistant bacteria, further complicating treatment and increasing costs, it is mandatory to adhere to treatment guidelines and avoid the overuse of antibiotics.22 –24
Therefore, the goals of this study were to describe the incidence of SSI in a large feline population undergoing GI surgery and to retrospectively assess whether different preoperative antimicrobial protocols or other clinical variables were associated with differences in SSI rates in cats.
Materials and methods
Data collection
Medical records of cats that had GI surgeries performed between October 2017 and March 2023 at the Clinic for Small Animal Surgery of the Vetsuisse Faculty Zurich were retrospectively reviewed. Cats that had GI surgery, including the stomach, duodenum, jejunum, ileum or colon, were included if they had a complete medical record, owner consent to use the data and a postoperative follow-up of at least 30 days. Surgical procedures in the oral cavity, oesophagus and rectum were excluded, as well as GI biopsies. Cases with evidence of a septic abdomen at the time of presentation due to abdominal effusion or systemic signs of infection were excluded. Demographic data retrieved from the medical records included age at presentation in months, breed, weight, sex and neuter status. In addition, the following data were recorded: type of surgery (enterotomy, enterectomy, gastrotomy and combinations), indication for surgery (FB, mass resection, intussusception, incarceration, macroscopically discoloured intestines, duodenotomy as part of biliary surgery and combinations), location in the GI tract (stomach, duodenum, jejunum, ileum, colon, caecum and combinations), surgeon’s expertise (intern under supervision of a diplomate or resident, resident, diplomate), surgery time (time from cut to completion of the last suture), anaesthesia time (time from induction to extubation), ASA score, lowest body temperature, lowest mean arterial pressure, lowest SpO2 level and duration of hospitalisation. In addition, we documented which antibiotic regimen was used. The decision to administer PPAs was made at the discretion of the attending surgeon and anaesthetist on a case-by-case basis. The antibiotic regimen was divided into four groups: (1) cats without PPAs (group A); (2) cats receiving PPAs during surgery (group B); (3) cats receiving PPAs during surgery and extended PPAs (ePPAs; continued for up to 24 h postoperatively) (group C); and (4) cats receiving PPAs followed by antibiotic treatment for more than 24 h postoperatively (group D).
Cats were allocated to group C if no medical reason for the continuation of antibiotics after surgery was documented in the medical records, suggesting prophylactic use. In contrast, cats were allocated to group D if postoperative antibiotic treatment was initiated because of clinical or diagnostic findings including systemic signs of severe inflammation, secondary underlying disease, suspected or confirmed aspiration pneumonia, or surgical technique-related issues. Secondary underlying disease was defined as the preoperative presence of concurrent systemic or localised conditions that justified postoperative antibiotic therapy. Examples in our study included pyuria, Giardia infection, pre-existing hepatopathy, infectious anaemia or pulmonary disease. Surgical technique-related issues were defined as intraoperative technical problems (eg, malfunctioning suction device) that, according to the surgeon’s judgement, warranted antibiotic coverage.
The type of antibiotic, the time between PPA application and incision, and reasons for continuing antibiotics postoperatively were recorded. Timing of PPA administration was categorised according to literature-based recommendations as follows: ‘too early’ (>60 mins before incision), ‘in time’ (30–60 mins before incision), ‘too late’ (<30 mins before incision) and after incision. The peri- as well as postoperative hygiene conducted according to the hospital protocol for preparation of the surgical site and the postoperative management were identical in all cases.
For cats that developed an SSI, the type (superficial incisional, deep incisional or organ/space, based on the Centers for Disease Control and Prevention [CDC] classifications 25 ) and severity of the SSI (grade 1–5 based on the Clavien–Dindo Score [CDS] 26 ) were determined.
A follow-up time of 30 days was chosen, as per the CDC recommendation. The outcome evaluation was based on the medical record if available. In cases in which this was not available, the owners were contacted through an online survey or by phone. Cases where the owners did not report any further problems postoperatively were considered to have no complications. In cases of reported complications or unreachable owners, the referring veterinarians were contacted to verify the outcome.
Statistical analysis
The statistical analysis was conducted using RStudio software, Version 2023.06.1+524. Logistic regression was used to analyse the relationship between continuous predictor variables and the occurrence of SSIs. The results were reported as odds ratios (ORs) with 95% confidence intervals (CIs), mean values and ranges. Categorical data were analysed using Fisher’s exact test, with ORs and 95% CIs calculated as well.
A multivariable logistic regression model was built using a stepwise, backward selection approach to examine the predictors of SSIs. Variables with P <0.2 in the univariable analysis and no signs of collinearity were included. Collinearity was assessed using the variance inflation factor, and variables showing high collinearity were excluded. Significant predictors were assessed and pairwise comparisons were conducted using the Tukey post-hoc test.
Results
Study design and demographic data
A total of 353 records of cats undergoing GI surgeries over a 5-year period were retrospectively reviewed. Of these, 146 were excluded because of missing data, duplicates, missing follow-up, or early death or euthanasia due to issues unrelated to the GI procedure. In addition, 19 cases were excluded because of a diagnosed septic abdomen at presentation time. Of the 188 included cats, 114 (60.6%) were male and 74 (39.4%) were female. Of them, 164 (87.2%) were neutered, 21 (11.2%) were intact and three (1.6%) had an unknown castration status. A total of 23 different breeds were included, the most common being the European Shorthair (n = 81, 43.1%), followed by the Maine Coon (n = 37, 19.7%) and the British Shorthair (n = 16, 8.5%). The mean age at presentation was 53.2 months and the mean body weight was 4.4 kg. A more detailed description of these parameters can be found in Table 1.
Univariable analysis of demographic data and their association with surgical site infection (SSI) occurrence in 188 cats undergoing gastrointestinal surgery

Data are n (%) or mean (range) unless otherwise indicated. Logistic regression was used for continuous variables and Fisher’s exact test was used for categorical variables
CI = confidence interval; OR = odds ratio; SSI+ = developed an SSI; SSI− = did not develop an SSI
Surgery type, indication and localisation
The most frequent procedure was enterotomy in 80/188 (42.6%) cats, followed by gastrotomy in 44/188 (23.4%), enterectomy in 41/188 (21.8%) and combinations of these procedures in 23/188 (12.2%). Of these combinations, 20 cats underwent both gastrotomy and enterotomy, while one cat each underwent gastrotomy and enterectomy, enterotomy and enterectomy, and gastrotomy, enterotomy and enterectomy. The primary indication for surgery was removal of a FB in 142/188 (75.5%) cats, followed by intussusception in 24/188 (12.8%) and GI masses in 11/188 (5.9%). A total of 44/188 (23.4%) cats had surgery on the stomach, 18/188 (9.6%) on the duodenum, 93/188 (49.5%) on the jejunum and two each (1.1%) on the ileum, colon and caecum, while 27/188 (14.4%) cats had surgery at multiple GI locations. Specific details regarding the surgical protocol (Table 2) and the anaesthesia protocol (Table 3) are provided.
Univariable analysis of the association between surgical protocol factors and the development of surgical site infection (SSI) in 188 cats undergoing gastrointestinal surgery, using Fisher’s exact test

Data are n (%) unless otherwise indicated
CI = confidence interval; OR = odds ratio; SSI+ = developed an SSI; SSI− = did not develop an SSI
Univariable analysis of the association between variables extracted from the anaesthesia protocol and the development of surgical site infection (SSI) in 188 cats undergoing gastrointestinal surgery

Data are n (%) or mean (range) unless otherwise indicated. Logistic regression was used for continuous variables and Fisher’s exact test was used for categorical variables
ASA = American Society of Anesthesiologists; CI = confidence interval; MAP = mean arterial pressure; OR = odds ratio; SSI+ = developed an SSI; SSI− = did not develop an SSI
Antibiotic regimen
Most cats received PPAs (group B: 105/188, 55.9%), followed by a combination of PPAs and postoperative antibiotic treatment (group D: 48/188, 25.5%), no antibiotics (group A: 21/188, 11.2%) and ePPAs (group C: 14/188, 7.4%) (Table 4). In group D, reasons for therapeutic antibiotic use included systemic signs of severe inflammation in 27/48 (56.3%) cats, secondary underlying disease in 15/48 (31.3%) cats, suspected or confirmed aspiration pneumonia in 8/48 (16.7%) cats or surgical technique-related issues in 2/48 (4.2%) cats. Of the 167 cats receiving PPAs, the timing of the administration was too early (>60 mins before incision) in 14 (8.4%), in time (30–60 mins before incision) in 29 (17.4%) and too late (<30 mins before incision) in 85 (50.9%). In 30/167 (18.0%) cats, antibiotics were administered after incision (Table 5). The most frequently chosen drug for PPAs was cefazolin in 85/167 (50.9%) cats, followed by amoxicillin/clavulanic acid in 70/167 (41.9%), ampicillin/sulbactam in 10/167 (6.0%), marbofloxacin in 1/167 (0.6%) and enrofloxacin in 1/167 (0.6%) (Table 5).
Association between perioperative antibiotic regimen and the occurrence of surgical site infections (SSIs) in 188 cats undergoing gastrointestinal surgery, showing the number of cats developing an SSI and statistical significance compared with the reference group (group A, no perioperative prophylactic antibiotics [PPAs]) in a logistic regression model
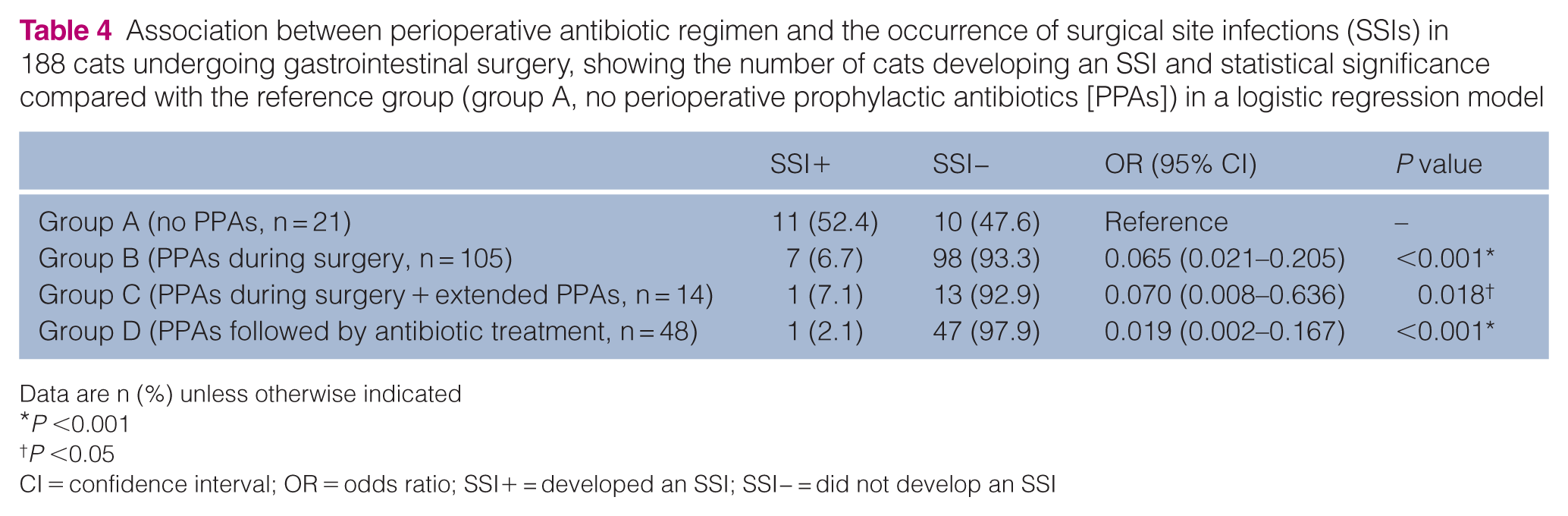
Data are n (%) unless otherwise indicated
P <0.001
P <0.05
CI = confidence interval; OR = odds ratio; SSI+ = developed an SSI; SSI− = did not develop an SSI
Overview of the timing and type of perioperative prophylactic antibiotic (PPA) administration and surgical site infection (SSI) development in 167 cats undergoing gastrointestinal surgery and receiving PPAs

Data are n (%) unless otherwise indicated. Cats without PPAs (n = 21) and unknown timing (n = 9) were excluded in the calculation of P values, ORs and 95% CIs
Too early = >60 mins before incision; in time = 30–60 mins before incision; too late = <30 mins before incision
CI = confidence interval; OR = odds ratio; SSI+ = developed an SSI; SSI− = did not develop an SSI
SSI rate and severity
The overall SSI rate was 20/188 (10.6%). According to CDC classification, SSIs were categorised as superficial in 11/20 (55.0%) cats, deep in 4/20 (20.0%) and organ/ space in 5/20 (25.0%) (Table 6). One cat was euthanased because of an SSI shortly after surgery, 7/20 (35.0%) cats were treated with local antiseptics, 18/20 (90.0%) received antibiotic therapy and 10/20 (50.0%) underwent revision surgery. One cat diagnosed with a superficial incisional SSI died because of issues unrelated to the SSI. The procedure-specific mortality rate was 1.1% (2/188).
Criteria, definition, treatment and Clavien–Dindo classification of surgical site infections (SSIs) in the 20 cats that developed an SSI after gastrointestinal surgery, according to the Centers for Disease Control and Prevention guidelines 26

CDS = Clavien–Dindo Score; SSI+ = developed an SSI
The highest SSI rate was observed in cats in group A (11/21, 52.4%), followed by group B (7/105, 6.7%), group C (1/14, 7.1%) and group D (1/48, 2.1%) (Table 4). When timing categories were further grouped, cats receiving PPAs after incision had a significantly higher SSI rate compared with those receiving PPAs before incision (4/30, 13.3% vs 4/130, 3.1%; P = 0.043).
Univariable analyses (Tables 1–3) identified age, localisation, antibiotic regimen group and anaesthesia time as variables meeting the inclusion criterion for the multivariable model (all P <0.2). Timing was excluded from the model despite a low P value because of strong collinearity. In the final logistic regression model, antibiotic regimen was the only variable independently associated with SSIs, whereas age, localisation and anaesthesia time were not significant after adjustment. The final model (Table 4) indicated a protective effect of PPA use, but no benefit from extending antibiotic prescription up to 24 h or longer (Table 7). Using group A (cats without PPAs) as the reference, group B (cats receiving PPAs) had significantly lower odds of developing SSIs (OR 0.065, 95% CI 0.021–0.205; P <0.001). Cats receiving ePPA prophylactic or therapeutic antibiotics beyond the perioperative period (groups C and D) also had lower odds compared with the no antibiotics (group C: OR 0.070, 95% CI 0.008–0.636; P = 0.018; group D: OR 0.019, 95% CI 0.002–0. 167; P <0.001) (Table 4). When group B (cats with PPAs) was used as the reference, group A (cats without PPAs) had significantly higher odds of SSIs (OR 15.4, 95% CI 5.02–51.3; P <0.001), whereas ePPAs delivered prophylactically or therapeutically (groups C and D) did not confer additional significant protection (group C: OR 1.08, 95% CI 0.06–6.78; P = 0.95; group D: OR 0.30, 95% CI 0.016–1.74; P = 0.26).
Pairwise comparison of significant variables from the final logistic regression model using the Tukey post-hoc test for surgical site infections in 188 cats undergoing gastrointestinal surgery

SE = standard error
Discussion
The objective of this study was to evaluate the incidence and factors associated with the development of SSIs in cats undergoing GI surgeries with a special focus on the use of antibiotics.
Over a 5-year period, 20 SSIs were identified in a population of 188 cats, which concludes an SSI rate of 10.6%. Our SSI rate is higher compared with previous reports of 3.8% in cats, 2 but consistent with results of GI studies in mixed studies or dogs (7–17.2%).2,27,28
The antibiotic treatment regime (no antibiotics vs PPAs) showed the greatest impact on SSI development. According to the antibiotic stewardship guidelines for veterinarians,29,30 PPAs are not recommended in animals with an ASA score of 1–2 and surgeries lasting less than 90 mins in clean-contaminated procedures. Although these guidelines may apply to other clean-contaminated surgeries, our findings, along with those of a previous study, 12 challenge this assumption in the context of GI surgeries in cats. A study conducted by Degen et al 12 already indicated a protective effect of PPAs in GI surgery in a mixed data set including dogs and cats. Interestingly, the SSI rate without PPAs in that study (6/33 [18.2%] cases) was markedly lower than our current results (11/21 [52.4%]). This could indicate that cats are more susceptible to SSI when surgery is performed without PPAs. However, both studies were not randomised and the group sizes between patients receiving no antibiotics vs patients receiving PPAs vary greatly; this could also be because of selection bias. Consequently, our findings must be interpreted with caution. Further prospective randomised controlled studies are needed to validate these results.
Contrary to PPAs, ePPAs or continued antibiotic treatment after surgery did not exert any protective effect, which is consistent with the results of various previous studies4,12,28 and strengthens the recommendations of current antibiotic guidelines. 30
As research in human medicine indicates that unnecessary postoperative antimicrobial prophylaxis contributes to the development of antimicrobial resistance,16,31 reduction of antibiotic prescriptions in cases without clear indication is mandatory. Based on our results and the findings of previous studies,22 –24,32 neither ePPAs nor ongoing antibiotic treatment should be routinely initiated in cats undergoing GI surgery.
Apart from the total rate of SSIs, it seems also reasonable to take the severity and consequence of an SSI for the affected cat into consideration. Although a superficial infection that is easily treated with local wound care and/or antibiotics (CDS 1 and 2) might not warrant systematic protective usage of antibiotics, deep and organ/space infections that result in revision or worse consequences do. We detected a relatively high proportion of severe (CDS >3) SSIs in all groups. In group A, the proportion of severe SSIs was 7/11. Two out of these cases were associated with a dehiscence. In comparison, the proportion of severe SSIs in cats that did receive any type of antibiotic treatment (groups B, C and D) was 6/9, with three of them related to a dehiscence. This could indicate that the usage of antibiotics does not impact the proportion of severe complications. However, this finding also needs to be verified by bigger prospective randomised controlled studies. Every cat that developed an organ/space SSI in our study also showed signs of dehiscence, which will result in infection irrespective of the chosen antibiotic regimen. The overall dehiscence rate was 2.7% (5/188), which is comparable to previous studies that documented dehiscence rates in the range of 0–2.3%.5,33 –35 Larger studies in dogs report dehiscence rates up to 11%. 36 This difference could be due to actual species differences or simply be the effect of different sample sizes and underreporting in the relatively small feline studies.
Apart from antibiotic regimen, no other tested parameter showed a significant effect on SSI development. Contrary to findings in human medicine,8,9 no correlation was identified between SSI incidence and location in the GI tract or specific surgical procedure. This could be due to the relatively small sample size (n = 188) compared with larger human studies with over 2000 patients. Despite being the largest feline GI surgery data set to date, our sample size remains relatively small for investigating multiple factors associated with the development of SSIs.
In addition, neither surgery4,11,13,14,23,37 nor anaesthesia time14,19,38,39 was significantly associated with the development of SSIs, in contrast to what has been reported previously. This finding could be attributed to the wide variance of surgery (18–202 mins) and anaesthesia times (50–310 mins).
As this is a retrospective study, several important limitations must be listed. First, because of the retrospective nature of the study, the outcome of all cases might not be accurate and some minor, self-limiting SSIs may have been missed. A further limitation is that the decision to administer PPAs was determined by the attending surgeon and anaesthetist, which resulted in unequal sample sizes between groups. This clinician-dependent allocation also means that potential confounding by clinical-related factors cannot be excluded. Because of the retrospective nature of this study, the timing of PPA administration was not standardised. It should be noted that most cats received PPAs less than 30 mins before or after incision, which is considered too late according to current guidelines. 29 Although no significant association between SSI occurrence and the timing categories was found, cats receiving antibiotics after incision showed a significantly higher SSI rate compared with those receiving PPAs before incision. We hypothesise that in GI surgeries, the exact timing of PPA administration may be less critical than in other surgical procedures. In contrast to other surgeries, where the target site is exposed immediately upon incision, in GI surgery the stomach or intestines are typically opened later during the procedure. Therefore, even a slight delay in administration might still result in adequate antibiotic concentrations at the time of luminal exposure. However, this hypothesis cannot be proven within the scope of this study and may not apply uniformly to all cases.
Therefore, the difference in SSI rates must be interpreted with care. In addition, although this is the largest data set on feline GI surgery to date, the sample size is still small, considering the magnitude of factors that have been investigated.
A further limitation is that the prevalence of SSIs is potentially overestimated. For many cases with superficial SSIs, the diagnosis of an SSI was confirmed by a private vet, and according to CDC guidelines, any case deemed an SSI by a clinician must be reported as such. However, it remains uncertain how many of the superficial SSIs in this study would meet the criteria if assessed by a surgeon specifically trained in CDC standards. As part of the limitations of a retrospective study, validation and grading of SSI, even based on CDC criteria, is difficult, especially regarding the rate of superficial infections. To allow a more accurate classification of SSI and to reliably evaluate associations in a multivariable model, a prospective randomised controlled study is needed.
The statistical limitations of our study should also be acknowledged. The selection of variables for the multivariable model was based on univariable analysis, including only those with P <0.2. Although this approach provides a clear and reproducible selection strategy, it may exclude other clinically relevant factors and could limit the generalisability of the findings. The use of a stepwise backward selection procedure could introduce bias in predictor selection.
In summary, the findings of this study must be re-evaluated in a prospective, controlled study to verify their true impact in feline patients undergoing GI surgery.
Conclusions
This retrospective study demonstrates that PPA administration was significantly associated with a lower rate of SSIs in cats undergoing GI surgery. Conversely, extended postoperative antibiotic use does not confer additional benefits in otherwise healthy patients. These findings support the prudent use of antibiotics to optimise patient outcomes and minimise unnecessary antimicrobial exposure.
Footnotes
Acknowledgements
The authors extend special thanks to Dr med vet Simon Rüegg for statistical assistance and to med vet Madeleine Degen for contributing to data collection.
Author note
Data will be made available upon request.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
