Abstract
Objectives
This study described the pharmacokinetics of bupivacaine after bilateral maxillary and caudal inferior alveolar nerve blocks in adult cats under general anaesthesia.
Methods
A total of 10 healthy adult cats (mean ± SD weight 4.8 ± 0.8 kg) were included in a randomised, prospective trial. The anaesthetic protocol consisted of acepromazine-methadone-propofol-isoflurane. Each cat randomly received 0.2 (BUPI2) or 0.3 ml (BUPI3) of bupivacaine 0.5% per site (4 and 6 mg per cat, respectively) (n = 5/group). Blood was collected before (time 0) and at 2, 7, 20, 30, 60, 120, 240, 360, 480 and 600 mins after all dental blocks. Plasma concentrations of bupivacaine were analysed using liquid chromatography-tandem mass spectrometry. The pharmacokinetics of bupivacaine were described using a non-compartmental analysis.
Results
Mean doses of bupivacaine were significantly different (BUPI2: 0.88 ± 0.14 mg/kg; BUPI3: 1.22 ± 0.21 mg/kg). For BUPI2 and BUPI3, mean maximum bupivacaine plasma concentrations (Cmax) were 825 ± 299 and 926 ± 197 ng/ml at 5.0 ± 2.7 and 9.6 ± 5.8 mins (time to peak concentration); mean area under the curve to the last measured concentration was 142 ± 36 and 180 ± 60 min*µg/ml; mean clearance was 5.4 ± 0.8 and 7 ± 5.7 ml/min/kg; mean elimination half-life was 245 ± 54 and 278 ± 90 mins; and mean residence time to the last measured concentration was 185 ± 13 and 182 ± 33 mins, respectively. Concentrations of bupivacaine were detected up to 600 mins (72 ± 22 ng/ml in BUPI2 and 104 ± 55 ng/ml in BUPI3).
Conclusions and relevance
Bilateral maxillary and caudal inferior alveolar nerve blocks using two volumes and doses of administration produced Cmax below those reported to cause toxicity in cats. Further studies are warranted to investigate the pharmacodynamics of dental blocks in cats.
Introduction
Loco regional anaesthetic techniques provide perioperative analgesia and reduce inhalant anaesthetic requirements. The maxillary, caudal inferior alveolar and infraorbital nerves are commonly desensitised using local anaesthetics to block transmission of nociceptive input during oral and maxillofacial surgery in cats and dogs.1 –5 These so-called dental blocks are cost-effective and can be considered a day 1 competency. 6 On the other hand, dosage regimens should be calculated to minimise local anaesthetic toxicosis in clinical practice. Before drug administration, negative aspiration of blood is confirmed, to avoid intravascular injection that could cause cardiovascular toxicity, particularly with local anaesthetics such as bupivacaine. 7 Resistance to injection should be avoided as it could indicate intraneural injection, potentially leading to nerve damage. 1
Bupivacaine is usually preferred over lidocaine for locoregional anaesthetic techniques of the oral cavity in small animals as it has a prolonged duration of action and may promote intra- and postoperative analgesia. 2 However, bupivacaine may produce neurotoxicity and cardiotoxicity when inappropriate doses are used or with intraneural or intravascular injections. 7 Despite its widespread use in veterinary medicine, studies on the pharmacokinetics of bupivacaine have rarely been conducted after dental blocks in cats or dogs. These studies are important to characterise the safety profile of a local anaesthetic technique and its clinical application, 8 especially when different injection volumes per site are used in feline dentistry. It is unknown, for example, if a dental block could be repeated if the first injection fails to produce local anaesthesia or if this would result in critically increased drug plasma levels leading to neurological, respiratory and/or cardiovascular signs of local anaesthetic toxicity.
The aim of this study was to determine the plasma concentrations and deriving pharmacokinetics of two volumes of bupivacaine 0.5% (0.2 or 0.3 ml per injection site) from concentration–time data plotting after bilateral maxillary and caudal inferior alveolar nerve blocks in adult cats under general anaesthesia.
Materials and methods
This study was approved by the institutional animal care committee of the Faculty of Veterinary Medicine (FMV; Comité d’éthique de l’utilisation des animaux), Université de Montréal (UdeM) (22-Rech-2209), and performed in December 2022.
Animals
A total of 10 healthy adult cats (six males, four females; mean ± SD weight 4.8 ± 0.8 kg) from the teaching colony of the FMV, UdeM, were included in a prospective, randomised, masked, experimental trial. Eligibility criteria included any healthy adult cat of any sex or breed. Cats were considered healthy according to their medical records, physical examination and values of haematocrit and total protein within the reference interval (28–47% and 59–81 g/l, respectively). Exclusion criteria included cats with a body weight below 2.0 kg, body condition score below 3/9 or above 6/9, those with anaemia (haematocrit <28%), hypoproteinaemia (total protein <59 g/l) or any signs of systemic disease. On day 1, cats were admitted, acclimated to the study site and personnel, and physical examinations were performed. They were housed individually in adjacent stainless-steel cages containing water and food bowls, a cardboard box for hiding and perching, a blanket, a litter box and a toy. On day 2, cats were anesthetised and the dental blocks were performed under general anaesthesia, followed by blood sampling. On day 3, cats were fed and a physical examination was performed; they were then transported back to their long-term accommodation. Cats were always manipulated using feline-friendly techniques. Synthetic pheromones were diffused at the study facilities from 2 h before their arrival until discharge.
General anaesthesia
Food, but not water, was withdrawn 8–10 h before anaesthesia. Each cat was premedicated with acepromazine (0.03 mg/kg, Atravet; Boehringer Ingelheim) and methadone (0.3 mg/kg, Comfortan; Dechra) combined in the same syringe and administered into the epaxial lumbar muscles. For each cat, areas around the cephalic veins were clipped and a local anaesthetic cream containing lidocaine 2.5% and prilocaine 2.5% (EMLA cream; Aspen Pharmacare Canada) was applied. Approximately 15 mins later, a cephalic catheter (22 G) was aseptically inserted into one of the cephalic veins and wrapped using a blue bandage (ie, for administration of medicines). Anaesthetic induction was achieved with intravenous propofol (Propoflo 28; Zoetis) administered until it was possible to place a supraglottic airway device (V-gel Advanced; DocsInnovent). Lidocaine was not used to desensitise the cats’ arytenoids before the introduction of the airway device. The device was then connected to a capnograph and a modified Mapleson D system. Cats were positioned in dorsal recumbency on a circulating warm-water blanket and their eyes were lubricated with ocular gel. Anaesthesia was maintained with isoflurane (Aerrane; Baxter) delivered in 100% oxygen for 1 h after the administration of the final injection of bupivacaine. Lactated Ringer’s solution (Lactated Ringer’s injection USP; Baxter) was administered intravenously at 5 ml/kg/h throughout the procedure, using the catheter with the blue bandage. A multiparametric monitor (LifeWindow 6,000V; Digicare Animal Health) was used to monitor arterial oxygen saturation, carbon dioxide end-tidal partial pressure, respiratory and heart rates, electrocardiogram, and systolic, mean and diastolic arterial pressure throughout the procedure. A second 22 G cephalic catheter was then inserted in the contralateral limb and wrapped using a red bandage (ie, for blood sampling). During general anaesthesia, dental nerve blocks were performed as described below. At the end of the procedure, cats recovered from anaesthesia in a quiet environment on top of a circulating warm-water blanket without any stimulation. The first cephalic catheter was removed after the removal of the airway device. Cats were fed 2 h after general anaesthesia with a commercial diet, provided twice daily, and ad libitum water.
Nerve blocks
Cats were randomised (www.randomization.com; accessed 16 November 2022) to receive one of the two treatments, consisting of 0.2 ml (BUPI2) or 0.3 ml (BUPI3) bupivacaine 0.5% per injection site (total of 4 and 6 mg of bupivacaine per cat, respectively; n = 5/group). The maxillary nerve and caudal inferior alveolar nerve blocks were performed bilaterally, with the cat in dorsal recumbency, using a 25 G needle connected to a 1 ml syringe. Techniques are described in Figures 1 and 2. 9 In each block, the assigned volume of bupivacaine was administered after negative aspiration of blood.
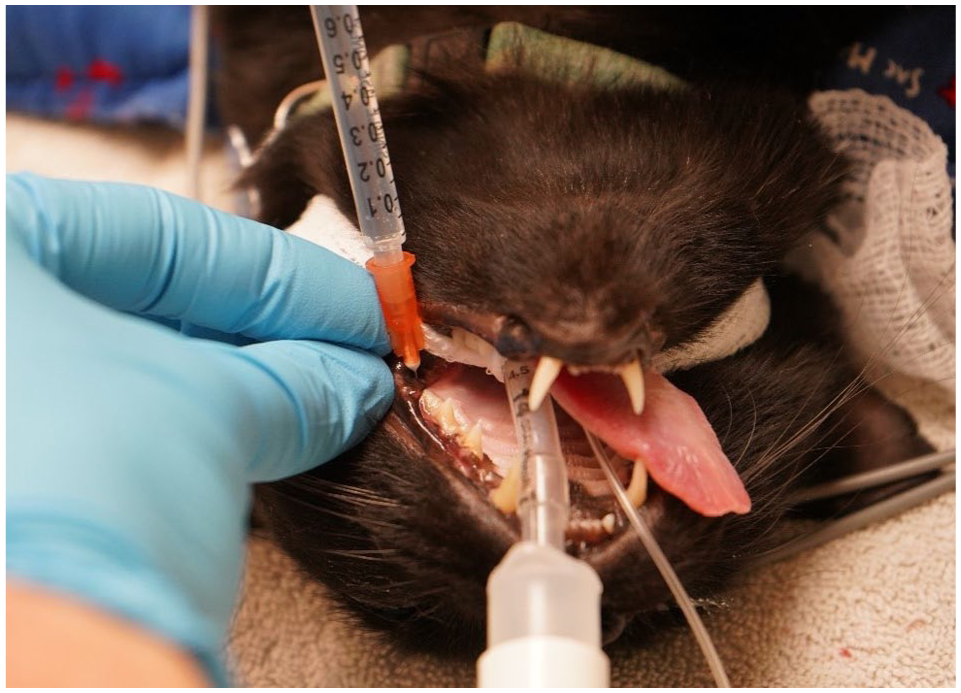
The maxillary nerve block was performed using an intraoral approach with the cat’s mouth opened. After palpation of the maxillary tuberosity caudal to the last molar tooth using the non-dominant hand, the needle was introduced behind the molar tooth and the maxillary tuberosity, almost perpendicular to the hard palate and at a depth of about 5 mm

An extraoral approach was used for the caudal inferior alveolar nerve block. The index finger of the non-dominant hand was placed intraorally and used to palpate the rostrocaudal margin of the last mandibular molar tooth and the rostral margin of the angular process of the mandible. The needle was inserted transcutaneously, perpendicular to the skin surface, rostral to the angular process of the mandible, and advanced medially to the mandible. The needle tip was positioned at the midway point of an imaginary line between the last mandibular molar tooth and the angular process (depth of approximately 5 mm), guided by the operator’s finger
Blood sampling
Venous blood sampling (1.0–1.5 ml) was performed under general anaesthesia at baseline (time 0) and at 2, 7, 20, 30, 60, 120, 240, 360, 480 and 600 mins after the last dental block. Blood samples at 2, 7, 20, 30 and 60 mins were collected under general anaesthesia. These time points were based on previous studies in cats.8,10,11 Haematocrit and total protein were re-evaluated after the last time point to exclude anaemia and hypoproteinaemia. The final volume of blood collected was less than 10% of each cat’s total blood volume. After each collection, the catheter was flushed with 0.5 ml of heparinised saline and the injection port was replaced to avoid contamination of subsequent samples. Blood was transferred to potassium EDTA acid tubes. Samples were kept on ice for no more than 30 mins and then centrifuged at 3500 ×g for 15 mins. Plasma was stored in a –80 °C freezer until analysis. The second venous cephalic catheter was removed after the last blood sample was collected.
Analysis and pharmacokinetics of bupivacaine
The plasma concentrations of bupivacaine were determined using a validated liquid chromatography combined with tandem mass spectrometry (LC-MS/MS) method.8,11,12 Cat plasma was used to prepare the calibrators with standards to enable limits of quantification in the range of 5–5000 ng/ml. Accuracy was in the range of 92.1–103.8% and the precision observed was better than 5.9%. Quality control concentrations of bupivacaine in cat plasma were determined in two batches using concentrations of 15, 250 and 3000 ng/ml. The following pharmacokinetic parameters were calculated using a non-compartmental method (PhoenixWinNonLin V.8.0; Pharsight): area under the plasma concentration–time curve from time zero to the last measured time point (AUC0-last; min*µg/ml); the maximum concentration of the drug in plasma (Cmax; ng/ml) and time to peak concentration (Tmax; mins); area under the first moment curve from zero to the last time point measured (AUMC0-last; min*µg/ml); terminal elimination rate half-life (T1/2; mins); and relative clearance indexed by bioavailability (CL/F; ml/min/kg). The mean residence time from zero to the last measured time point (MRT0-last; mins) was calculated using MRT0-last = AUMC0-last/AUC0-last.
Statistical analysis
This study did not include any sample-size calculations. The authors used the available number of cats to explore the pharmacokinetics of bupivacaine after dental blocks. Normality of the data was tested using the Shapiro–Wilk test. The data distribution was summarised using the mean ± SD for normally distributed variables or median (range) for non-normally distributed variables. Demographic data were analysed using independent sample t-tests or the Mann–Whitney U-test (P <0.05), depending on whether the variables were normally distributed or not. Statistical analyses were performed using the open-source software RStudio version 2022.07.1-554.
Results
Descriptive and pharmacokinetic data for each treatment group are available in Tables 1 and 2, respectively. Mean bupivacaine plasma concentrations are reported for each group in Figure 3. Doses of bupivacaine were significantly different between treatments (BUPI2: 0.88 ± 0.14 mg/kg; BUPI3: 1.22 ± 0.21 mg/kg). None of the other variables were significantly different between BUPI2 and BUPI3. Positive blood aspiration occurred in one male cat from BUPI3 before desensitisation of the right mandibular nerve. Compression was performed for 2 mins and the block was repeated afterwards. No signs of local anaesthetic toxicity were observed during the study.
Body weight, body condition score (BCS), haematocrit, total protein, the dose of propofol for anaesthetic induction, duration of anaesthesia and time to removal of the supraglottic airway device in cats after the administration of bupivacaine 4 mg/cat (BUPI2) or 6 mg/cat (BUPI3) by bilateral maxillary and caudal inferior alveolar nerve blocks in 10 cats

Data are mean ± SD or median (range)
Haematocrit reference interval 28–47%; total protein reference interval 59–81 g/l
Pharmacokinetics of bupivacaine after bilateral maxillary and caudal inferior alveolar nerve blocks using 4 mg/cat (BUPI2) or 6 mg/cat (BUPI3) in 10 cats (n = 5/group)

Data are mean ± SD
AUC0-last = area under the plasma concentration–time curve from zero to the last time point measured; AUMC0-last = area under the first moment curve from zero to the last time point measured; C600 = plasma concentrations of bupivacaine at 600 mins; Cmax = maximum bupivacaine plasma concentrations; CL/F = relative clearance indexed by bioavailability; MRT0-last = mean residence time from zero to the last time point measured; T1/2 = terminal elimination half-life; Tmax = time to maximum concentration.

Mean ± SD plasma concentrations of bupivacaine after bilateral maxillary and caudal inferior alveolar nerve blocks in 10 cats (n = 5/group) using 4 mg/cat (BUPI2; red) or 6 mg/cat (BUPI3; black)
Discussion
This study reported the plasma concentrations and pharmacokinetics of two volumes and doses of administration of bupivacaine 0.5% after bilateral maxillary and caudal inferior alveolar nerve blocks in cats. Clinical signs of local anaesthetic toxicity were not observed during general anaesthesia, even when plasma concentrations reached peak concentrations shortly (ie, 5–9 mins) after the administration of dental blocks. Doses administered (BUPI2: 0.88 ± 0.14 mg/kg; BUPI3: 1.22 ± 0.21 mg/kg) were significantly different. However, in both groups, plasma concentrations were below the reported range for toxicosis in cats.13 –15 In BUPI2 and BUPI3, peak plasma concentrations were approximately 0.825 and 0.92 µg/ml, respectively, lower than the concentrations reported to cause changes in the electroencephalogram associated with convulsive states (3.6 ± 0.7 µg/ml) 15 or to induce dysrhythmias (7.5–10.9 µg/ml), 14 hypotension (23 µg/ml), seizures (37 µg/ml) and cardiovascular collapse (110 µg/ml) in cats. 13 Both volumes and doses of administration of bupivacaine 0.5% can be used to desensitise the maxillary and caudal inferior alveolar nerves bilaterally in cats.
Dental blocks are an important component of multimodal analgesia in feline dentistry, when used in combination with opioids and non-steroidal anti-inflammatory drugs, as their use may block nerve conduction and nociceptive transmission in the oral cavity, for example, when multiple dental extractions are required. 5 The study did not evaluate any pharmacodynamic effects (eg, postoperative pain relief or isoflurane-sparing effects) of using different volume regimens. Therefore, it is difficult to know how increasing the volume and dose of bupivacaine (ie, BUPI3 vs BUPI2) would reduce the onset and prolong the duration of action of nerve blockade in the clinical setting. In a previous study, maxillary and/or inferior alveolar nerve blocks using a combination of bupivacaine (0.25 mg/kg) with lidocaine (0.25 mg/kg) reduced anaesthetic requirements, sympathetic stimulation and postoperative pain scores in cats undergoing dental extractions. 5 As much as this was not a dose-finding study, the authors commonly calculate the maximum dose of bupivacaine (2 mg/kg) before administration and then commonly inject 0.25–0.3 ml/site, if possible, even knowing that lower volumes may also provide adequate analgesia. The ideal dose regimens for dental blocks in cats are still a subject of further research, particularly with the combination of local anaesthetics and opioids to prolong the duration of analgesia.
Plasma concentrations of bupivacaine have been reported after intraperitoneal (IP), 11 orbital 16 or transversus abdominis plane (TAP) administration in cats. 8 Mean peak plasma concentrations were lower (825 and 926 ng/ml for BUPI2 and BUPI3, respectively) but peak concentrations were achieved faster (Tmax at 5 and 10 mins for BUPI2 and BUPI3, respectively) when compared with IP (1030 ng/ml) and TAP (1166 ng/ml) administration of bupivacaine 0.25% at 2 mg/kg, when both IP and TAP had a Tmax of approximately 30 mins. In addition, the median Cmax and Tmax were 1400 μg/ml at 17 mins and 1700 μg/ml at 28 mins for retrobulbar and peribulbar injections, respectively, when a total of 0.75 ml of bupivacaine 0.5% (3.75 mg) was administered. These data are difficult to compare as different doses, concentrations, injectate volumes and sites were used. However, they demonstrate that bupivacaine is rapidly absorbed after dental nerve blocks, reflecting the highly vascularised areas of the head. It is not known if this rapid absorption of bupivacaine is followed by a short and effective onset of local anaesthesia or if it would be safe to repeat a local block shortly after the first injection, given the fast drug absorption. Future pharmacokinetic–pharmacodynamic studies could provide further insight into these questions.
Despite differences in Cmax and Tmax, the elimination half-life and relative clearance indexed by bioavailability were similar between BUPI2 (5.4 ± 0.8 h and 5.2 [2] ml/min/kg), BUPI3 (7.0 ± 5.7 h and 4.6 [13] ml/min/kg), and previous studies using the IP (4.8 ± 2.70 h and 5.8 ± 3.0 ml/min/kg) and TAP (4.22 ± 0.92 h and 5.3 ± 1.8 ml/min/kg) routes of administration. Therefore, bupivacaine could have similar elimination times and clearance rates, even when using different dose regimens and routes of administration. In addition, plasma concentrations of bupivacaine have been detected up to 480 mins after IP and TAP administration, and up to 600 mins in BUPI2 and BUPI3. This may reflect the high lipophilicity and extended absorption/distribution phases of the drug (ie, mean residence times longer than 180 mins) in all of the above pharmacokinetic studies in cats. IP and TAP administrations have produced postoperative analgesia up to 10 h after ovariohysterectomy.10,17,18 Pain scores were decreased up to 4 h (ie, the last time point evaluated) postoperatively after maxillary and/or inferior alveolar nerve blocks with lidocaine and bupivacaine in cats undergoing dental extractions. 5 It would be interesting to know whether these dental blocks with bupivacaine provide an extended duration of postoperative pain management than previously reported in dentistry.
The administration of two volumes of levobupivacaine 0.5% has been described in dogs after infraorbital (0.11 ml/kg2/3) and/or inferior alveolar nerve blocks (0.18 ml/kg2/3). Levobupivacaine is the active l-isomer of bupivacaine and has similar effects to racemic bupivacaine. However, it has the advantage of being less cardiotoxic than its racemic mixture. 19 Plasma concentrations of levobupivacaine were measured up to 2 h after administration. 20 The median Cmax of levobupivacaine was 1335 ng/ml, slightly larger than BUPI2 and BUPI3 in this study but with a similar Tmax (7 mins), demonstrating some pharmacokinetic similarities between the canine and feline species.
The present study has some limitations. The authors did not attempt to analyse the active metabolites of bupivacaine as their clinical relevance is unknown. This study involved a small sample size of five healthy cats per group with some individual variability. A pharmacokinetic study in a large sample size involving the target population (eg, cats undergoing dental extractions) would be of interest. However, challenges with this approach would include different comorbidities, anaesthetic regimens and durations, and invasiveness of oral procedures. It is not known how pharmacokinetic parameters in this study can be extrapolated to, for example, cats with hypotension, hypovolaemia and/or hypercapnia using different anaesthetic protocols or with different underlying diseases. The aforementioned conditions may impact the absorption, distribution, metabolism and excretion of anaesthetics, including bupivacaine.
Conclusions
Bilateral maxillary and caudal inferior alveolar nerve blocks using two volumes and doses of administration produced Cmax below those reported to cause toxicity in cats. Further studies are warranted to investigate the pharmacodynamics of dental blocks in cats.
Footnotes
Acknowledgements
The authors thank Fleur Gaudette from the Pharmacokinetics core facility of the Centre de Recherche, Centre Hospitalier de l’Université de Montréal (CRCHUM), for carrying out LC-MS/MS.
Author note
An abstract of this study was presented at the 2023 European Association of Veterinary Pharmacology and Toxicology, Bruges, Belgium. Data are available from the authors upon reasonable request.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of experimental animals and the study therefore had prior ethical approval from an established (or ad hoc) committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
