Abstract
Objectives
The aim of this study was to characterise vaginal cytological patterns and standardise its interpretation in female cats. Vaginal cytology has a broadly recognised value for oestrous cycle staging because of the responsiveness of the vaginal epithelium to oestrogens causing changes in the cellular pattern. Unlike the bitch, vaginal cytology is not routinely used in queens.
Methods
A total of 39 vaginal smears were collected from 31 intact queens using a simple and quick procedure with little, if any, physical restraint, analysed for cellularity, clearing, epithelial cell types and the presence of red blood cells or neutrophils. Queens were classified in different oestrous phases based on reproductive history and serum progesterone levels.
Results
The 39 vaginal smears were classified as proestrus (n = 1), oestrus (n = 9), interoestrus (n = 10), dioestrus (n = 9) and anoestrus (n = 9). One vaginal smear was not allocated to any cycle phase because the queen was diagnosed with pyometra. Cellularity was poor in 21/39 smears, from which eight were from queens in oestrus. Cytological oestrus was characterised by 90% or more keratinised epithelial cells, unlike previously reported, and presence of clearing in a scarce to modestly cellular smear.
Conclusions and relevance
Progesterone concentration is needed to differentiate interoestrus from dioestrus because of the mixed-cell population on vaginal cytology. Keratinised vaginal cells are consistently found in the anoestrus smears of queens and should not lead clinicians to assume the queen is cycling. Neutrophils can be present in the different oestrous cycles in cats without being associated with clinical signs of disease, unlike red blood cells. These findings demonstrate that vaginal cytology can be a useful tool in feline practice for managing reproductive presenting complaints.
Plain language summary
This study looked at how vaginal cytology can be used to help determine what stage of the reproductive cycle a female cat is in. Although this technique is commonly used in dogs, it is not often used in cats. In total, 39 vaginal cell samples were collected from 31 healthy female cats to examine the types of cells present along with the clearing of the background and cell yield. These findings were compared with hormone levels and reproductive history to identify which stage of the cycle each cat was in. The study revealed that vaginal cell samples are mostly poorly cellular, especially in queens in oestrus. During the oestrus (heat) stage, keratinised epithelial cells with or without a nucleus amount to a minimum of 90% of all epithelial cells. Clearing (absence of debris in the slide’s background) is highly associated with the presence of oestrogen. Neutrophils were found in all cycle stages and did not always mean the cat had a reproductive problem. Hormonal tests (progesterone assay) are needed to distinguish some stages, namely interoestrus and dioestrus. Keratinised cells were found even when cats were not cycling. The origin of these cells in non-cycling cats is still unclear. In summary, vaginal cytology can be a helpful tool for veterinarians when evaluating the reproductive cycle of female cats, but it is better interpreted alongside hormone testing and clinical history.
Introduction
The feline reproductive tract is composed of two ovaries connected to the uterus (two uterine horns and a uterine body) through the oviducts. The uterus is then connected to the cervix, vagina, vestibule and vulva in the cranial to caudal direction.1,2 Contrary to the dog, the feline vagina is horizontal and short, measuring 4.0–5.0 cm from the vulva to the cervix. It features a vaginal fornix, which is an area just cranial and ventral to the cervical opening. The dorsal median fold is located in the upper part of the vaginal wall caudally to the cervix.3,4 The most caudal part of the vagina is the vestibule or urogenital sinus 5 of 1.0–2.0 cm length and greater diameter than the vagina itself (4 mm vs 1.0–2.0 mm). The fornix length increases and the vaginal height decreases during the follicular phase of the oestrous cycle, consistent with an increase in size of the dorsal median fold. 6 In contrast, the (cranial) vaginal diameter does not vary throughout the oestrous cycle, always being less than 2 mm. 6 The external urethral meatus is located on the floor of the vestibule. The clitoris resides in the clitoral fossa ventrally on the caudal vestibule, caudally to the external urethral meatus. 7
Vaginal cytology is an important tool in small animal reproduction as it allows veterinarians to stage the reproductive cycle, therefore playing an important role in breeding management together with serum progesterone assay. However, its importance differs between bitches and queens. The high sensitivity of the canine vaginal epithelium to oestrogens causes reliable cyclical changes in its cellular pattern, characterising each stage of the bitch’s oestrous cycle.8,9 The growing concentration of oestrogens during proestrus leads to an increase in the number of cell layers of the vaginal epithelium. During phases of prolonged oestrogen exposure, the cells in the upper layers of the canine vagina move progressively away from the blood supply, causing their death and the development of keratin precursors that determine vaginal epithelial keratinisation.10,11 The same is thought to happen in the cat, 12 although no study has showed an increased thickness of the feline vaginal mucosa during the follicular phase. The fact that ovulation is induced by coitus in a majority of queens makes vaginal cytology less frequently used in feline reproduction management. In addition, many veterinarians refrain from performing vaginal cytology in queens in oestrus because of a disputable 13 fear of inducing ovulation. Indeed, in other species, such as the rat, 14 the ferret or the polecat, 15 the technique of vaginal lavage as an alternative to vaginal swabbing has been employed for obtaining cells for vaginal cytology. In queens, this technique was employed in only one publication. 16 Vaginal lavage has been anecdotally reported by Dr Stabenfeldt to reduce vaginal stimulation in comparison with the use of the swab, potentially decreasing the risk of inducing ovulation. Moreover, there is a scarcity of studies featuring the clinical use of vaginal cytology in queens,13,16 –20 compared with the bitch. However, vaginal cytology can be a helpful tool in feline practice to manage queens that ovulate spontaneously,21 –24 exhibit silent or continuous heats16,25 and show oestrus after gonadectomy,26,27 as well as an important adjunct in the diagnosis of pyometra.28,29
The short duration of feline proestrus as well as a lack of uniformity on the definition of some of its phases may sometimes complicate the interpretation of feline vaginal smears. Some authors combine proestrus and oestrus in one single follicular phase,30,31 while others describe the cytological patterns of proestrus in detail.1,30
Further, the use of the word postoestrus differs among authors. Postoestrus is defined as the interoestrus period between one oestrus and the next in queens that have not been induced to ovulate. 32 Mills et al 20 employ it as a synonym of metoestrus. However, metoestrus generally refers to a luteal phase, and the cats in this study were not allowed to mate, which makes ovulation unlikely. 20 In contrast, Kanca et al, 13 use it as a synonym for interoestrus; however, interoestrus is the interval between two consecutive heats without ovulation.
The term ‘clearing’ was first introduced by Shille et al 30 as ‘the absence of non-cellular debris and of eosinophilic strings of mucus, as well as a lack of coalescence of cells into sheetlike aggregates’, being suggested as the more sensitive indicator of oestrogen activity in cats. Previously, ‘absence of cellular debris’ had been noticed by Mowrer et al 16 in cats during oestrus, and more recently, the term ‘clear background’ has been used as a synonym for the absence of cellular debris.13,31
The aim of this study was to characterise vaginal cytological patterns throughout the different cycle phases and to standardise the interpretation of vaginal cytology of the feline oestrous cycle, allowing for a broader and more appropriate use of the technique in queens.
Materials and methods
This descriptive study is based on vaginal smears from feline clinical cases selected retrospectively among the patients presented to the Veterinary Teaching Hospital of the University of Padova between March 2022 and March 2025 for reproduction-related reasons having undergone vaginal cytology during the visit. Queens included in the study were post-pubertal or at least 6 months old and could not be under treatment with any reproductive drug during the collection of the vaginal sample. Reproductive history was gathered, and serum progesterone (P4) concentration was retrieved when available. In case a P4 value was non-existent but blood was collected from the queen during the clinical appointment, the stored serum sample was retrieved and P4 assayed (Automated Immunoassay Analyser-360; Tosoh) after obtaining the owner’s consent. When P4 was unavailable, reproductive history was used to try and determine the cycle phase. When reproductive history was insufficient to identify the cycle phase, the vaginal smear was excluded from the present study. In accordance with Directive 2010/63/EU on the protection of animals used for scientific purposes and the European Commission Decision of 29 October 2012, extended by Protocol No. 02-2016, this study did not require formal approval from an ethics committee.
Vaginal cytology was performed by gently separating the vulvar labia with the thumb and index finger and inserting a water moistened cotton swab for approximately 1–2 cm, entering cranially from the dorsal commissure of the vulva, avoiding the clitoral fossa, after which it was removed. The swab was not rotated inside the vagina and the whole procedure lasted no more than 3 s (Video 1 in the supplementary material). The swab was then rolled twice onto a cytological glass slide, describing two lines, air-dried and stained with Diff-Quik. All cytological vaginal slides were identified and routinely stored.
Cytological smears were systematically analysed non-blindly under a light microscope (Eclipse Ci; Nikon) by two operators assessing the parameters indicated in Table 1. One operator (FC) first analysed all the slides, after which a second more experienced operator (MP) reassessed the slides confirming and, when necessary, correcting the previous analysis. When results differed greatly between the first and second operators, a third assessment was repeated blindly by the second operator. Percentages of each type of epithelial cells were considered different if the difference exceeded 10%. Vaginal smears were observed at × 100 and/or × 200 magnifications to detect the presence of debris, mucus, red blood cells (RBCs), score the background as clear or not and count epithelial cells. The degree of cellularity was established by counting the number of epithelial cells in five fields of × 100 magnification. A × 400 magnification was used whenever in doubt.
Parameters and respective scoring system used for vaginal cytology analysis

Parabasal (P) cells are identified as round cells with small cytoplasm and a large round nucleus taking up at least half of the cellular area. Intermediate (I) cells are characterised by large round cytoplasm with a round nucleus. Superficial (S) and anucleated (A) cells both have angular cytoplasm, sometimes with a folded aspect. S cells maintain the nucleus, although pyknotic to vestigial, whereas A cells are deprived of it. 33 P and I cells are classified as non-keratinised due to their roundly limited cytoplasm, different from S and A cells that are classified as keratinised owing to the polygon-shaped cytoplasm.
An oestrous cycle phase was attributed to each vaginal cytological slide after careful assessment of each smear and in accordance with the queen’s reproductive history and, if available, serum P4 concentration. Oestrous cycle phases were defined as follows: proestrus = early beginning of oestrus signs manifestation with basal P4 values (⩽2 ng/ml); oestrus = evident manifestation of oestrous signs with low (⩽2 ng/ml) or high (>2 ng/ml) P4 values; interoestrus = basal P4 concentrations (⩽2 ng/ml) with oestrus signs reported in the previous month; dioestrus = P4 concentration above 2.0 ng/ml in the absence of oestrus signs; and anoestrus = basal P4 concentrations (⩽2 ng/ml) without oestrus signs reported in the previous month.
Results
A total of 39 vaginal smears from 31 queens of nine different breeds were included in the study (one and six queens were smeared three and two times, respectively). The median age of the studied queens was 2.3 years (interquartile range [IQR] 3.5 years; range 4 months to 10.5 years). The identification of each queen along with her breed, age, date of vaginal cytology, reproductive history and P4 concentration are presented in Table 2. P4 concentrations were available for 30/39 smears.
Number, age and breed of the queen, reproductive history and serum progesterone (P4) concentration for each of the 39 vaginal smears

Cats have a letter if more than one vaginal cytology was performed on the same queen
mo = month; y = year
Studied queens underwent vaginal cytology in the course of reproductive appointments for a variety of presenting complaints, such as control of reproduction with low-dosage megestrol acetate (Estropill; MSD) 34 (n = 13), with 4.7 mg or 9.4 mg deslorelin implant (Suprelorin; Virbac) (n = 12), preoperative appointment for spaying (n = 8), pregnancy termination after mismating (n = 1), pregnancy diagnosis (n = 1), hypofertility (n = 1), confirmation of oestrus (n = 1), spontaneous ovulation (n = 1) and reproductive pathology (n = 1). The vaginal smears from queens treated with deslorelin or megestrol acetate were obtained before the beginning of treatment, ensuring that they were not under contraceptive effect during the appointment in which vaginal cytology was performed.
The 39 vaginal smears were classified as proestrus (n = 1), oestrus (n = 9), interoestrus (n = 10), dioestrus (n = 9) and anoestrus (n = 9). One vaginal smear (cat 7) was not allocated into any cycle phase because the queen in question had pyometra at the time of examination.
The median and IQR of each class of epithelial cells (P, I, S and A) found in each cycle stage are reported in Table 3 and illustrated in Figures 1–6.
Each class of epithelial cells (parabasal, intermediate, superficial and anuclear) of the vaginal smears at different oestrous cycle stages

Data are median (interquartile range)
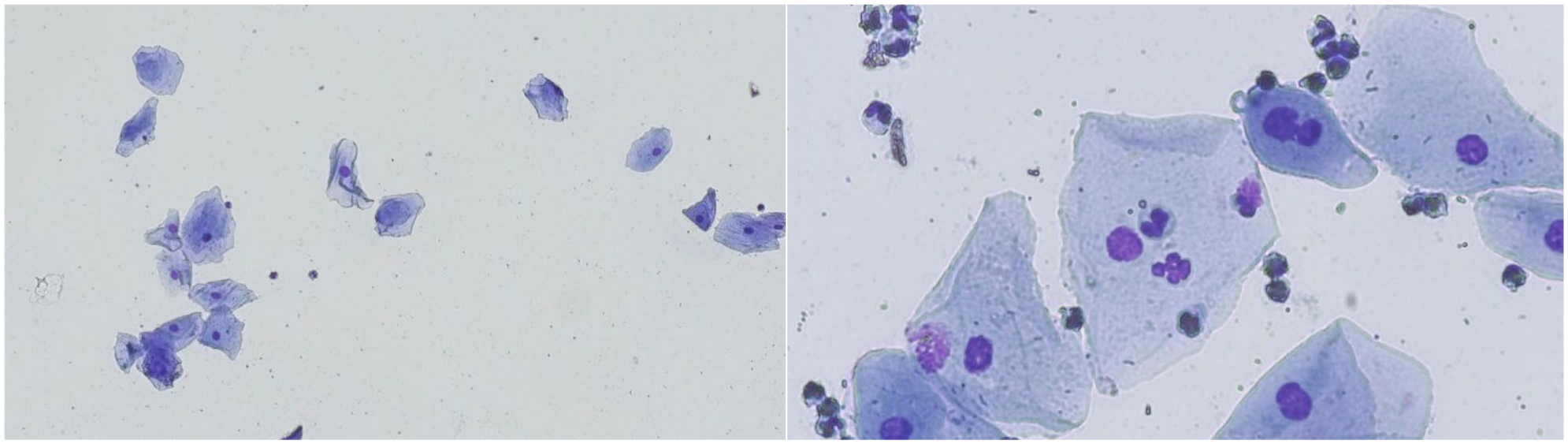
Feline vaginal smear indicative of proestrus (cat 31) characterised by 86% keratinised cells with or without a nucleus, with several neutrophils and metoestrus cells and absence of clearing (left: × 100 magnification, right: × 400 magnification)

Feline vaginal smear indicative of oestrus (cat 28A) characterised by 97% keratinised cells with or without a nucleus and clearing (absence of cellular and non-cellular debris or filaments) (× 100 magnification)

Feline vaginal smear indicative of interoestrus (cat 30) characterised by a mixed-cell population: 70% non-keratinised cells and 30% keratinised cells, with clearing (absence of cellular and non-cellular debris or filaments) (× 100 magnification)

Feline vaginal smear indicative of late interoestrus (cat 26) characterised by 95% keratinised cells with or without a nucleus, with clearing (absence of cellular and non-cellular debris or filaments) (× 100 magnification). This queen only showed behavioural signs of oestrus the next day
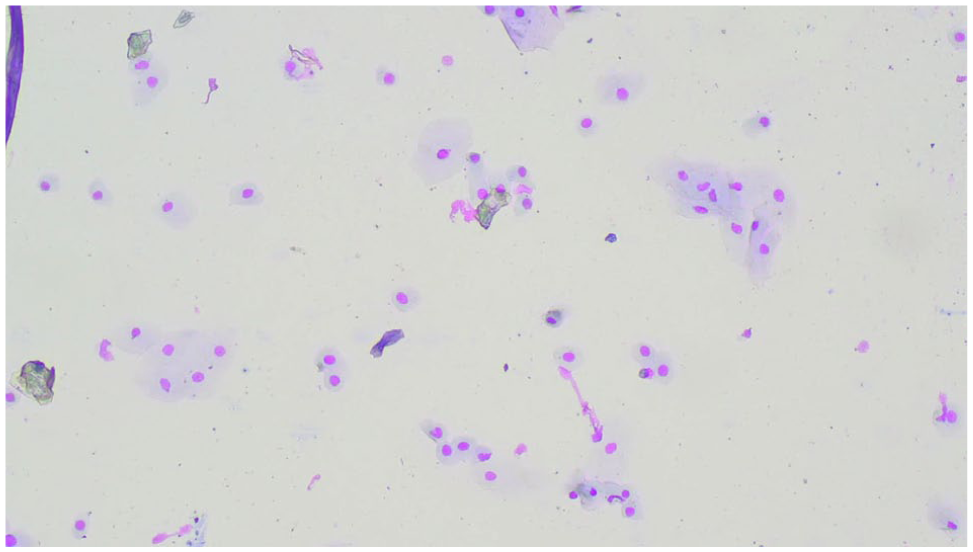
Feline vaginal smear indicative of dioestrus (cat 2C) characterised by a mixed-cell population: 75% non-keratinised cells and 25% keratinised cells, without clearing (× 100 magnification)
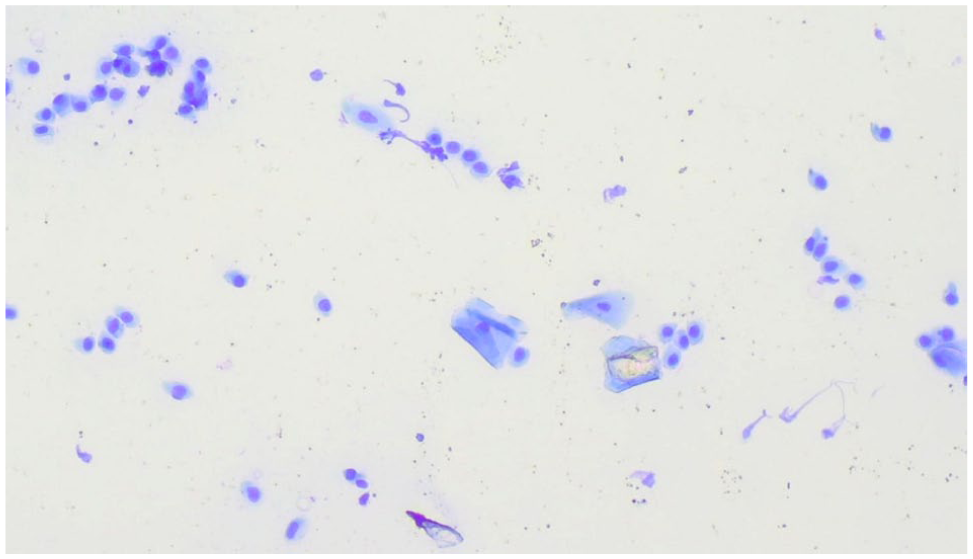
Feline vaginal smear indicative of anoestrus (cat 29) characterised by a majority of parabasal cells (78.5%) and 19% of keratinised cells, without clearing (× 100 magnification)
In the only smear of proestrus, the two predominant classes of epithelial cells were S (54.5%) and A (31.5%) cells (Figure 1). No P cells were observed, and 14% were I cells.
In oestrus, the two predominant classes of epithelial cells were keratinised, namely S (52 ± 27.5%; range 17.0–77.0%) and A (38.5 ± 8%; range 23.0–82.5%) cells, both keratinised (Figure 2). Two oestrus smears were classified as late oestrus, since oestrus signs had stopped or significantly decreased the day before, but the smear was characterised by a percentage of keratinised cells of 89.5% and 91.5%, respectively.
In interoestrus, the two predominant classes of epithelial cells were I (33.5 ± 19.3%; range 5.0–67.5%) and S (33 ± 13%; range 17.0–71.5%) cells (Figure 3). One interoestrus smear was subsequently classified as late interoestrus, since oestrus signs started the following day. Keratinised cells reached 95% and clearing was present (Figure 4).
In dioestrus, the two predominant classes of epithelial cells were I (49 ± 24.5%; range 5.0–67.5%) and S (22 ± 20%; range 4.0–41.5%) cells (Figure 5). Two dioestrus smears were classified as early dioestrus, since P4 was above 2.0 ng/ml, oestrus signs had disappeared 3 and 4 days previously, respectively, and the smear was represented by a mixed-cell population with predominance of I and S cells.
In anoestrus, the two predominant classes of epithelial cells were P (58.4 ± 24.4%; range 13.5–89%) and S (14 ± 5.5%; range 2.5–59.5%) cells (Figure 6).
Cellularity was classified as high in seven (interoestrus n = 2, dioestrus n = 2 and anoestrus n = 3), moderate in 10 (proestrus n = 1, oestrus n = 1, interoestrus n = 3, dioestrus n = 2 and anoestrus n = 3) and poor in 21 (oestrus n = 8, interoestrus n = 5, dioestrus n = 5 and anoestrus n = 3) smears. Of nine oestrus smears, eight were classified with poor cellularity. All other cycle phases were evenly distributed through the three cellularity classes.
Clearing was found in 12/39 smears, including all nine oestrus smears and three interoestrus (cats 26 [late interoestrus], 27 and 30) smears.
Neutrophils were found in 9/39 smears. Only 1/9 queens (cat 7) manifested clinical signs of reproductive infection and was later diagnosed with a pyometra (Figure 7). The remaining eight queens did not develop any subsequent reproductive problems. Two queens (cats 7 and 25 [interoestrus]) had more than 100 neutrophils per 100 epithelial cells. Three queens (cats 8 [dioestrus], 18 [interoestrus] and 31 [proestrus]) had 25–60 neutrophils per 100 epithelial cells. The remaining four queens (cats 9, 12A, 6B, 23) had fewer than 11 neutrophils per 100 epithelial cells; these queens were respectively in oestrus, interoestrus, dioestrus and anoestrus. Metoestrus cells (Figures 1 and 8) were found in 5/9 smears (cats 7, 12A, 18, 25 and 31, with >100, 10, 28, >100 and 52 neutrophils per 100 epithelial cells, respectively). No foam cells were identified in any smear. RBCs were only found in the smear of the queen with pyometra (cat 7).
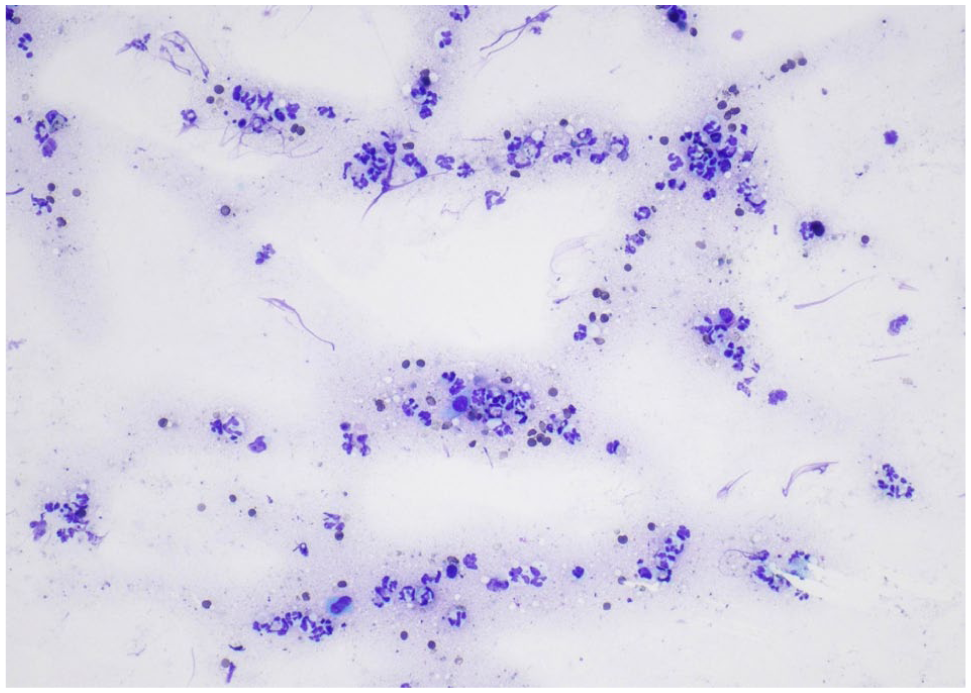
Feline vaginal smear of pyometra (cat 7) characterised by abundant neutrophils and red blood cells and few non-keratinised epithelial cells (× 100 magnification)

Feline vaginal smear indicative of dioestrus (cat 8) characterised by a majority of non-keratinised cells (85%), without clearing, abundant neutrophils (59 per 100 epithelial cells) and metoestrus cells (× 200 magnification) in a queen without clinical signs of reproductive pathology
Apart from vaginal keratinised epithelial cells, keratinised cutaneous cells were occasionally found in the vaginal smears (Figure 9).
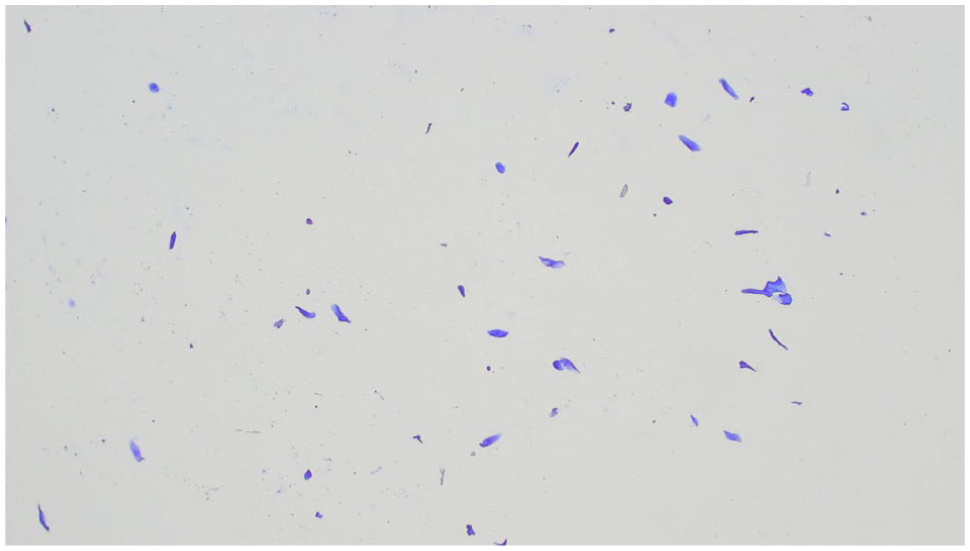
Feline vaginal smear of cat 28A, in oestrus at the moment of vaginal cytology, depicting an area characterised by cutaneous keratinised cells (× 100 magnification)
Discussion
This study analysed 39 vaginal cytologies from 31 queens at different stages of the oestrous cycle, encompassing the two seasonal periods (reproductive activity and inactivity) over 3 years. The sample of 31 queens is too limited to draw definitive conclusions; however, it encompasses female cats of a wide range of ages (4 months to 10 years) and a variety of nine breeds, including 45% of European Shorthair cats. A broader sample size would allow the study of potential breed differences regarding vaginal cytology.
The analysis of the vaginal slides focused on seven parameters: clearing, cellularity, epithelial cells, RBCs, neutrophils, foam cells and metoestrus cells.
Clearing, defined in our study as the absence of cellular and non-cellular debris or filaments, and thus a clear background deprived of any element, was present in all nine oestrus smears. Moreover, clearing was identified in three interoestrus smears: in one queen in which heat signs started the day after (cat 26 – late interoestrus), and in two queens in which heat signs had finished 6 days (cat 27) and 2–3 days (cat 30) before, respectively. These findings confirm the hypothesis formulated by Shille et al, 30 later supported by other authors,13,19 that clearing is a highly sensitive indicator of oestrogen activity in the queen as oestrogens liquefy the vaginal mucus in queens,2,30 allowing a clear visualisation of the background. In addition, clearing in three interoestrus smears indicates that its presence is also to be expected just before as well as after oestrus.19,30 Shille et al 30 observed early clearing in 13% of the cycles 2 days before the onset of the follicular phase and late clearing up to 5 days after its end, in 20% of the cycles. Queens 26 and 27 fall within this category. Queen 30, in which oestrus signs had finished 6 days before vaginal cytology, started treatment with megestrol acetate the next day and therefore did not cycle afterwards. As such, it is impossible to know whether clearing in this cat was a result of a potential upcoming follicular phase or the result of the previous one exceeding the maintenance of clearing up to 6 days from the end of oestrus. Based on the personal experience of the authors in analysing feline vaginal cytology, clearing should be assessed shortly after staining as the slide’s conservation or assembling procedure may contribute to the inadvertent inclusion of debris and non-vaginal material that may hinder the correct evaluation of the smear’s background.
Cellularity was assessed by counting epithelial cells in five × 100 magnification fields to standardise its classification for the purposes of this study. In the clinical environment, cellularity is a subjective parameter appraised simultaneously to the discrimination of epithelial cells and search for neutrophils or RBCs. More than 50% of the cytologies were characterised by having poor cellularity, meaning that fewer than 200 cells were present in five fields of × 100 magnification, giving strength to the anecdotal report that feline vaginal cytologies are much less cellular than canine ones. 35 In the bitch, high oestrogen concentrations during the follicular phase contribute to an increase in the number of cell layers of the vaginal epithelium. Therefore, proestrus and oestrus are associated with a high number of cells on vaginal cytology as more cells move away from the basal layers and the blood supply, and therefore exfoliate as they die. In our study, not only did we not find an association between high cellularity and the oestrus stage, but we uncovered a possible association between oestrus and poor cellularity. Out of nine smears, only one was not classified as having poor cellularity, but was moderate, with 239 epithelial cells per five fields of × 100 magnification (just above the cell number threshold for classification as moderate: 200). This potential opposite association in dogs and cats may be explained by the short duration of proestrus in cats (0.5–2 days).1,30 During this phase, an abrupt rise in oestrogen concentration, from below 15 pg/ml in anoestrus or interoestrus to a minimum of 20 pg/ml in the follicular phase, reaching more than 40 pg/ml in 24–48 h takes place. The two-fold increase in oestrogens observed over 24 h probably determines the absence of evident macroscopic alterations in the external genitalia of the queen during heat.2,30 Further research is needed to determine if the short duration of proestrus and the abrupt rise in oestrogens may limit the increase of the cell layers and in consequence the exfoliation of vaginal cells. Indeed, out of 39 smears, only one was classified as proestrus, reflecting the improbability of examining a queen in proestrus, as previously reported. 35
The percentage of epithelial cells varies throughout the cycle phase. Other publications have established different cut-offs for diagnosing oestrus: 80% or more superficial cells,13,36 more than 75% cornified cells 1 and more than 70% cornified cells. 34 Yet, in this study, all oestrus as well as late interoestrus smears were characterised by more than 90% of keratinised cells (range 90–100%). No progressive keratinisation in relation to the day of the oestrous cycle could be established, in agreement with previous observations.19,30 Interestingly, one queen (cat 4), with an oestrus smear with clearing and 91.5% of keratinised cells, had a serum P4 of 20.8 ng/ml. Queen 4 ovulated spontaneously, since she lived exclusively indoors and had no contact with any other cats during oestrus. This queen was still showing oestrus signs at the moment of vaginal smearing, which proves undoubtedly that the queen ovulated and did not cease oestrus behaviour, supporting other authors’ claim that ovulation does not shorten the duration of the oestrus phase. 30 The fact that the queen ovulated spontaneously raises the question of whether the suppression of oestrus signs occurs in response to a vaginal-stimulus-induced ovulation but not to a spontaneous one.
The other cycle phases, mainly interoestrus and dioestrus, were characterised by a mixed-cell population in which I and S cells were the most prevalent types. Keratinised and non-keratinised cells were more evenly distributed in interoestrus than in dioestrus. In the latter, the average percentages of non-keratinised (mainly I) cells were higher than the percentages of keratinised (mainly S) cells. P and A cell percentages are fairly constant between dioestrus and interoestrus. Indeed, a study found a significantly higher percentage of I cells in dioestrus (21.4%) compared with interoestrus (10.8%), 13 although those percentages are lower compared with the ones in this study (50.3% and 39.5%, respectively). The same study also found a significantly higher cell and cytoplasmatic area, as well as cell diameter and perimeter, in interoestrus compared with dioestrus. Although differences in cellular area and percentages of I and S cells may exist between interoestrus and dioestrus, these are not noticeable in the routine assessment of a cytological slide in a clinical setting. 30 Vaginal cytology is a non-invasive procedure that allows us to obtain a fast result. Therefore, epithelial cell counting and cell measurements are not performed by the clinician. To be of use, vaginal cytology should provide a quick and visually intuitive answer, which does not happen when discriminating interoestrus and dioestrus smears. This did not come as a surprise, as vaginal cytology indirectly reflects the fluctuation in oestrogen concentration but not in progesterone, and the differentiating factor between interoestrus and dioestrus is the occurrence of ovulation and consequent progesterone secretion. 1
Keratinised cells (S and A) were consistently present in all nine anoestrus smears analysed, from a minimum of 1.5% (cat 24) to a maximum of 48.5% (cat 17), with a median of 16 ± 11.5%. The origin of these keratinised cells in queens is still unknown; however, it is possible that it may reflect oestrogen activity/fluctuation even at basal levels during periods of reproductive inactivity. Another explanation may be the fact that the presently described procedure for vaginal cytology in queens favours more the collection of cells from the vestibule rather than from the vagina itself. The embryonic origin of these two structures differ; the vestibule (and caudal vagina) develop from the urogenital sinus (ectoderm-derived), whereas the cranial part of the vagina derives from the Müllerian ducts (mesoderm-derived), 37 which may also determine a different sensitivity to oestrogens. It would be interesting to look for the presence of these keratinised cells in both the vaginal smears of queens in anoestrus induced by contraceptive treatment (progestogens or deslorelin) and the smears collected from the more cranial vagina and not the vestibule or caudal vagina. The latter would require the use of a speculum and sedation of the queen. Only one other publication has mentioned the presence of (few) cornified cells in anoestrus smears. 16 Although the terminology employed is anoestrus, it is not clear in the publication if anoestrus is interpreted as the period of seasonal reproductive inactivity or rather as the period between two oestruses without mating (in this study defined as interoestrus) as the reported duration was from 10 to 18 days. 16 Interestingly, in this study the vaginal lavage technique was employed rather than vaginal swabbing.
Vaginal keratinised cells are not to be mistaken for cutaneous cells, also referred to as keratinocytes, which are keratinised squamous epithelial cells38,39 (Figure 9), flatter and narrower in comparison with S and A cells. The presence of these cells in vaginal cytology may be due to the inadvertent touch of the slide by the operator’s finger or of the vulvar labia with the cotton swab while inserting it. These cells should not be considered when assessing feline vaginal cytology. The authors have also observed these cells when performing preputial cytology if the outer part of the prepuce was invertedly touched when entering the preputial orifice.
Neutrophils were found in more than 20% of the smears in different quantities (from 1 to >100 neutrophils per 100 epithelial cells). Neutrophils were found in at least one smear of each cycle phase. Neutrophils were scarce in the single anoestrus and oestrus smears in which they were detected; 1 and 1.5 neutrophils per 100 epithelial cells, respectively. Abundant neutrophils were found in three non-pathological smears of interoestrus (>100 neutrophils), dioestrus (59 neutrophils) and proestrus (51.5 neutrophils), while they were very few in other interoestrus and dioestrus smears. No significant difference had been found between the number of neutrophils in dioestrus and interoestrus (postoestrus) in a previous study. 13 Thus, a difference in the number of neutrophils associated with the shift from one cycle stage to the other (similar to the bitch), as previously suggested, 30 cannot be supported by the present results. Serial vaginal cytologies from the end of oestrus and throughout interoestrus and dioestrus would be necessary to establish whether such a correlation exists. Neutrophils, even in large quantities (>50 neutrophils per 100 epithelial cells), are not an indicator of reproductive pathology unless accompanied by clinical signs. The same does not stand for RBCs.11,31,40 RBCs are observed during proestrus in the bitch because of the role of oestrogens in increasing vascular permeability, and therefore allowing the passage of RBCs into the uterine and vaginal lumen through the open cervix.11,41,42 However, RBCs are not observed in cats16,17 unless under pathological situations, 20 as confirmed by our current study or in case of mucosal irritation induced by the swab. 1 A study assessing vaginal smears from seven cats throughout different cycle stages found RBCs only in the two queens that presented with vaginal discharge and bacterial growth on vaginal culture (haemolytic and non-haemolytic Staphylococci). 20 The reasons for the lack of RBCs from the feline vaginal smears are not clear and might have to do with the short proestrus phase and rapid rise in oestrogens. Only Shille et al 30 reported to have rarely observed RBCs in the early follicular phase. 30
An interesting finding of this study is the identification of keratinised S (with nucleus) metoestrus cells (Figure 1). Metoestrus cells are defined as vaginal epithelial cells containing one or more neutrophils on the cytoplasm. 33 Some authors classify these cells as non-keratinised or intermediate.10,11,31,35 Their presence demonstrates the phagocytic capabilities of the vaginal epithelium. In the bitch, these cells are mostly observed in early dioestrus but can actually be found whenever neutrophils are present on vaginal cytology, as in early proestrus or in pathological situations like vaginitis or pyometra, as well as in preputial cytologies.10,11 When the vaginal epithelium becomes thicker and further keratinised, neutrophils become less visible or absent on vaginal cytology because of the increased distance between the capillaries in the basement membrane and the vaginal lumen, preventing neutrophils from moving through the more numerous cell layers during diapedesis.11,42 Neutrophils are observed in early proestrus in bitches, when the cervix opens and blood passes into the vagina, favouring bacterial growth, but are no longer present in late proestrus. 35 Therefore, neutrophils on canine vaginal smears are not concomitantly observed with keratinised cells (in high percentages).11,42 As an increase in thickness of the feline vaginal epithelium during the follicular phase has not been established, and may not exist, it is hypothesised that neutrophils may reach the vaginal lumen of queens in heat and thus be phagocytised by keratinised nucleated cells.
The presence of bacteria was not checked on vaginal smears and vaginal cultures were not available for the studied queens. Vaginal culture results may have helped to better interpret the presence and abundance of neutrophils, similar to that in dogs. 41 In addition, oestrogens were not assayed, which might have allowed a more precise attribution of cycle phases. Furthermore, the authors’ choice not to use image analysis software to establish cell parameters 13 constitutes a limitation, as this would have allowed us to obtain detailed information on cell morphology and characteristics. On the other hand, we decided not to make use of sophisticated equipment that is both time- and money-consuming, precluding the expeditious character of a routine vaginal cytology assessment.
Conclusions
Vaginal cytology remains a simple, non-invasive and cost-effective tool for reproductive assessment in both dogs and cats. In this study, we aimed to provide information on feline vaginal cytological patterns that can be of use for both reproductive specialists and general practitioners. Although vaginal cytological patterns are not clearly defined in queens, this study enhances its clinical applicability by supporting and completing previously reported evidence, as well as introducing new insights into its interpretation. Cytological oestrus was reliably characterised by clearing – often appearing before or lasting beyond behavioural signs of heat – together with sparse to moderate cellularity and keratinisation of 90% or above. Cytological identification of proestrus remains challenging because this phase is brief. Interoestrus and dioestrus are cytologically indistinguishable; therefore, serum P4 measurement is required when confirmation or exclusion of ovulation is necessary. Anoestrus is characterised by a predominance of non-keratinised P cells, although keratinised cells may still be present in varying proportions, and their presence should not be misinterpreted as evidence of oestrus. Except for oestrus, cell yield is independent of the cycle phase. Neutrophils can be found in all stages and are not indicative of pathology, whereas RBCs are rare and suggest a pathological condition in queens. Further studies are warranted to explore the physiological basis of keratinisation during anoestrus and to better understand the influence of rising oestrogen on feline vaginal cytology and physical changes during the follicular phase. This work contributes to the standardisation of feline vaginal cytology interpretation and supports its broader clinical use in feline practice.
Footnotes
Acknowledgements
The authors would like to thank Dr Sebastian Artl for his insight on the presence of keratinised cells in feline anoestrous smears.
Supplementary material
The following files are available as supplementary material:
Video 1. Vaginal cytology performed on a queen using a moistened cotton swab.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
