Abstract
Objectives
The aim of this study was to characterise cervical ventroflexion in cats, investigate which diseases are associated with its occurrence and evaluate treatment outcome.
Methods
This retrospective, two-centre study spans the period from 2003 to 2024. The inclusion criteria consisted of complete medical records, presence of cervical ventroflexion, baseline clinicopathological testing and a diagnosis. A descriptive statistical analysis was performed for all cats. Univariable and multivariable analyses were used to compare the feline hypokalaemic myopathy (FHM) group vs all other diagnoses.
Results
A total of 86 cats met the inclusion criteria. The most common diagnoses associated with cervical ventroflexion were FHM (42/86, 48.8%), hyperthyroidism (10/86, 11.6%), thiamine deficiency (9/86, 10.5%), immune-mediated polyneuropathy (6/86, 7%), cervical ischaemic myelopathy (5/86, 5.8%), acquired myasthenia gravis (3/86, 3.5%) and feline infectious peritonitis (FIP) meningoencephalomyelitis (3/86, 3.5%). Absence of additional neurological deficits (P = 0.020) was significantly associated with a diagnosis of FHM, whereas lateralisation of neurological signs (P = 0.004) was negatively associated with FHM. In most of the cats (65/86, 75.6%) that received treatment depending on the diagnosis, cervical ventroflexion had been reversible upon discharge.
Conclusions and relevance
Cervical ventroflexion is a neurological sign associated with either FHM when present as the sole sign, or another diagnosis when accompanied by additional neurological signs. It is usually reversible with treatment. This information may further assist clinicians in decision-making, especially when time or financial constraints exist.
Keywords
Introduction
Cervical ventroflexion is defined by paresis of the paraspinal cervical muscles leading to reduced muscle tone and an inability to resist passive movement (Figure 1; see video in the supplementary material).1,2 In affected cats, the neck remains flexed with the chin tipping in the thoracic inlet, while maintaining a forward-directed gaze, resembling a ‘dropped head’. 1 Cats lack a nuchal ligament, predisposing them to this more dramatic cervical posture when compared with other species. 3 Cervical ventroflexion can be spastic or flaccid.4,5 The latter is most common and is neuroanatomically localised to the peripheral nervous system (spinal nerves, neuromuscular junction or skeletal muscles) or, less commonly, the central nervous system (lower motor neurons within the cervical spinal cord grey matter).1,4 Spastic cervical ventroflexion is rarely reported in cats with thiamine deficiency and has been associated with seizure-like activity.6 –8 This may represent cervical dystonia (anterocollis), which could be attributed to an upper motor neuron disorder including basal nuclei. 9
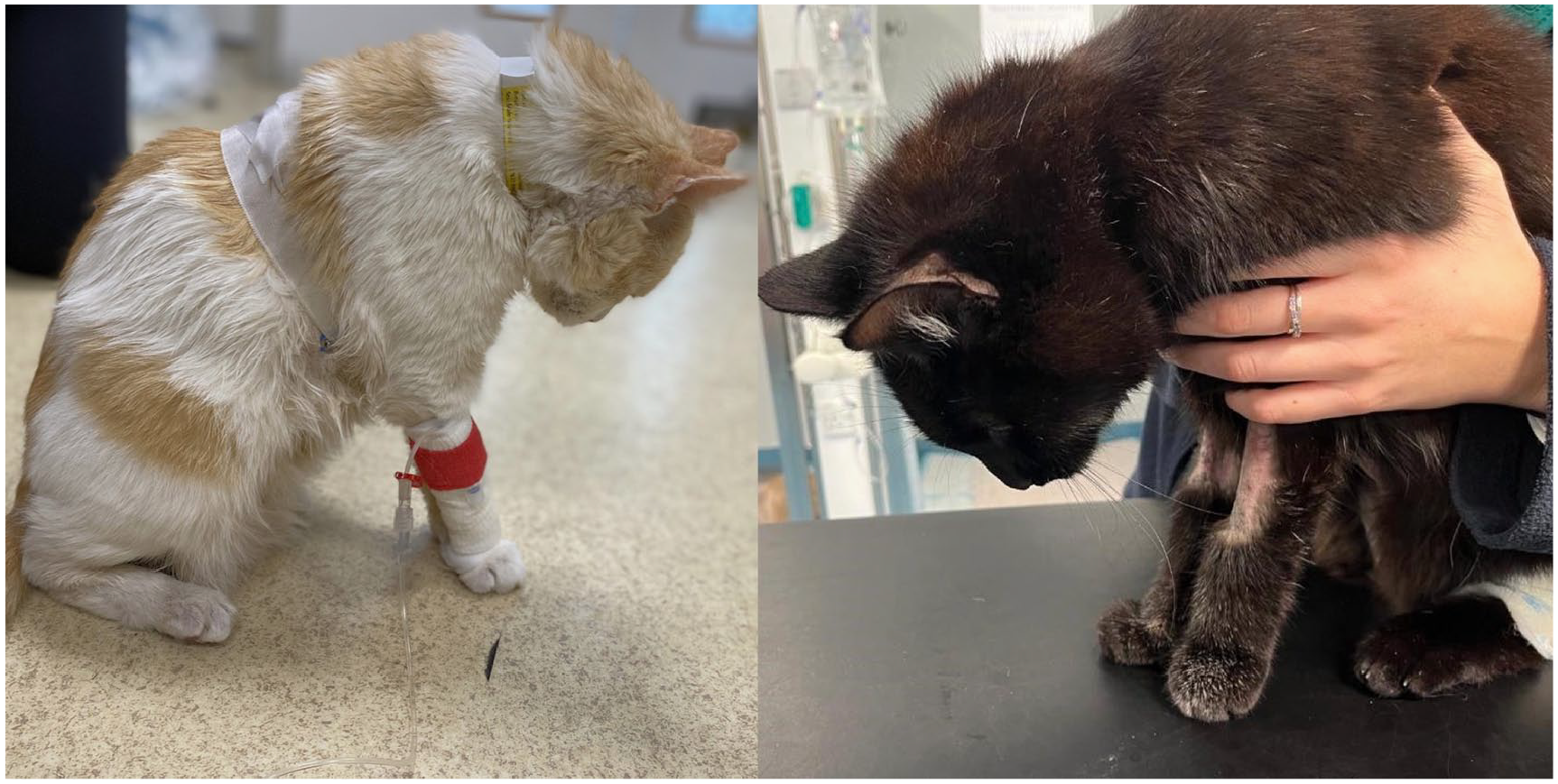
Cats with cervical ventroflexion
In humans, dropped head syndrome (DHS) is a rare condition characterised by passive flexion of the cervical vertebral column.2,10 It is most commonly associated with neuromuscular disorders or neuronopathies, including amyotrophic lateral sclerosis, myasthenia gravis, polymyositis and motor neuron disease. 2 Hypokalaemia and hypothyroidism are rare causes of DHS in humans. 10 When cervical muscle weakness is the only sign, it is referred to as isolated neck extensor myopathy.2,10
In cats, cervical ventroflexion is typically seen with neuromuscular disease. 1 Other findings include protrusion of the dorsal scapulae when weight bearing, reluctance to move, short-strided gait and wide-based pelvic limb stance.1,5,11,12 Diseases associated with cervical ventroflexion in cats include cervical ischaemic myelopathy (IM), immune-mediated and infectious polymyositis, immune-mediated polyneuropathy (IMPN), acquired myasthenia gravis, organophosphate intoxication, ammonium chloride intoxication, hereditary myopathies, feline hypokalaemic myopathy (FHM), hypernatraemic polymyopathy, hypocalcaemia, thiamine deficiency, hepatic encephalopathy due to portosystemic shunt and hyperthyroidism.1,4,5,12 –18
To date, no retrospective studies have been conducted to investigate cervical ventroflexion in cats. The objective of this study was to characterise this clinical sign and any diseases associated with its occurrence, and to evaluate treatment outcomes.
Materials and methods
This retrospective two-centre study was conducted at two referral hospitals in the UK spanning the period 2003 to 2024. Search terms included: ‘ventroflexion’ and ‘neck’ or ‘cervical’ and ‘cat’ or ‘feline’. The inclusion criteria were as follows: (1) complete medical records; (2) presence of cervical ventroflexion upon admission; (3) minimum database of complete blood count (CBC) and serum biochemistry, or packed cell volume (PCV), total solids (TS) and venous blood gas analysis, including electrolytes, glucose, creatinine and urea; and (4) diagnosis.
Variables collected included signalment, presenting complaints, onset of clinical signs, progression of clinical signs, neurological examination findings, lateralisation of neurological signs, neuroanatomical localisation, electrolyte concentration (potassium, sodium, calcium), primary and concurrent diagnoses, and comorbidities. Cervical ventroflexion was considered a neurological rather than clinical sign. MRI, cerebrospinal fluid (CSF) analysis, histopathological findings and clinicopathological results including total thyroxine (TT4) were recorded when available. Onset was categorised as hyperacute (<24 h), acute (1–7 days) and chronic (>7 days). Clinical signs were classified as progressive if deteriorating or non-progressive if static. MRI devices included high-field magnets (1.5 Tesla Hallmarq Small Animal MRI system, Hallmarq Veterinary Imaging; 1.5 Tesla Intera, Philips Healthcare). Sequences included, at a minimum, transverse and sagittal plane T2-weighted (T2W) images, fluid attenuation inversion recovery (FLAIR) (for brain studies), short tau inversion recovery (STIR) (for spinal cord studies) and pre- and post-contrast T1-weighted (T1W) images (gadopentetate dimeglumine, 0.1 mmol/kg IV). All clinical and neurological examinations were performed by a board-certified specialist or a resident in emergency and critical care or neurology.
Primary diagnosis was defined as a disease with a well-established or suspected pathophysiological mechanism leading to cervical ventroflexion.4,5 Cats with hyperthyroidism or thiamine deficiency with mild hypokalaemia were considered the primary diagnosis group.12,19 Hyperthyroidism was diagnosed based on elevated serum TT4 concentration (reference interval [RI] 19–62 nmol/l). 20 Thiamine deficiency was diagnosed presumptively based on history (eg, diet), clinical signs, MRI findings (when available) and response to treatment.21 –23 Absolute confirmation was not achieved because of the technical challenges, limited laboratory availability and inherent limitations of the available diagnostic tests. Erythrocyte transketolase activity can be influenced by other systemic diseases, while thiamine pyrophosphate measurement via high-performance liquid chromatography, although more sensitive and specific, does not quantify all forms of thiamine in the body. 21 FHM was diagnosed based on serum potassium levels below 3.5 mmol/l (RI 3.6–4.6) and response to potassium supplementation. 24 Clinically significant hyponatraemia and hypernatraemia were determined by serum sodium levels below 125 mml/l and over 170 mmol/l (RI 140–153), respectively. 24 Acquired myasthenia gravis was diagnosed through a positive neostigmine or edrophonium chloride response test and positive serology for nicotinic acetylcholine receptor antibodies (RI <0.3 nmol/l).16,25 Cervical IM, ischemic cerebrovascular accident (CVA) and congenital hydrocephalus were diagnosed by MRI.5,26 –30 A presumptive diagnosis of IMPN was made based on clinical presentation, response to treatment and electrodiagnostic findings, and muscle and nerve biopsies when available. 18 Dysautonomia was definitively diagnosed by post-mortem histopathology.31 –33 Immune-mediated polymyositis was diagnosed after unfixed muscle cryosections histopathology. Feline infectious peritonitis (FIP) meningoencephalomyelitis was diagnosed based on history, typical neurological signs, clinicopathological findings, albumin:globulin ratio, exclusion of other differential diagnoses, MRI findings (when available) and response to antiviral treatment.34,35 Extraneural comorbidities included conditions either indirectly related or unrelated to cervical ventroflexion. Treatment, when administered, was documented. Outcome at discharge was categorised as improvement, static or deterioration, depending on whether cervical ventroflexion had resolved, remained unchanged or worsened, respectively. Follow-up was obtained through clinical re-examination or phone records, when available.
Statistical analysis
The statistical analysis was performed using standard statistical software (SPSS Statistics 26; IBM Corporation). Data were assessed for normal distribution using the Shapiro–Wilk test for normality. Non-normally distributed numerical variables were represented as median, interquartile range (IQR) and range. Categorical variables were summarised as counts and percentages. The groups were compared using the Mann–Whitney U-test (two groups) or Kruskal–Wallis H test (more than two groups) in the case of numerical variables, and by the maximum likelihood G test or Fisher’s exact test (if the expected count in any cell of the contingency table was less than five) for categorical variables. The significance level (alpha) was set at 0.05 and all statistical tests were two-tailed. For the univariable analysis, the cats were divided into two groups based on their primary diagnosis: ‘FHM’ and ‘other diagnoses’. Multiple variables (sex, neuter status, age at presentation, onset of clinical signs, presence of additional neurological signs, neurological findings, outcome) were compared between groups. Variables that were significant in the univariable analysis were further analysed by multivariable analysis using logistic regression with the χ2 method.
Results
Signalment
The study included 86 cats: 47 (54.7%) females and 39 (45.3%) males. Of them, 70 (81.4%) were neutered. In total, 71 (82.6%) cats were non-purebred. The 15 purebred cats included British Shorthair (n = 4), Abyssinian (n = 2), Siamese (n = 2) and one each of Bengal, Birman, Burmese, Maine Coon, Norwegian Forest Cat, Ragdoll and Scottish Fold. The median age at presentation was 8 years (range 1.9 months to 22 years, IQR 11 years).
Presentation
Common presenting complaints included unspecified weakness (53/86, 61.6%), lethargy (41/86, 47.7%), anorexia (32/86, 37.2%), weight loss (18/86, 20.9%), gastrointestinal signs (16/86, 18.6%), polyuria/polydipsia (10/86, 11.6%) and ataxia (7/86, 8.1%). Onset of cervical ventroflexion was hyperacute in 22/86 (25.6%) cases, acute in 36/86 (41.9%) cases and chronic in 28/86 (32.6%) cases. The clinical signs were progressive in 66/86 (76.7%) cases and non-progressive in 20/86 (23.3%) cases. Three cats were fed solely chicken and/or fish and three had a history of having gone missing for 6–12 weeks, all of which were later diagnosed with thiamine deficiency.
Neurological examination findings
All 86 (100%) cats had cervical ventroflexion. Additional neurological signs were present in 63/86 (73.3%) cats and included abnormal gait (49/86, 57%), abnormal mentation (34/86, 39.5%), postural reaction deficits (25/86, 29.1%), cranial nerve deficits (18/86, 20.9%), hyporeflexia (12/86, 14%) and spinal hyperaesthesia (7/86, 8.1%). Neurological signs were lateralised in 8/86 (9.3%) cases. The most common neuroanatomical localisations were neuromuscular (45/86, 52.3%), followed by multifocal (30/86, 34.9%), central vestibular system (6/86, 7%) and C1–C5 spinal cord segments (3/86, 3.5%). A summary of the signalment, presentation, additional neurological deficits and neuroanatomical localisation can be found in Table 1.
Summary of the signalment, presentation, presence of additional neurological deficits and neuroanatomical localisation based on the primary diagnosis

Data are n (%) or median (range) [IQR]
A = acute; As = asymmetric; C = chronic; CVA = cerebrovascular accident; E = entire; F = female; FIP = feline infectious peritonitis; H = hyperacute; M = male; MB = mixed breed; N = no; NE = neutered; NP = non-progressive; P = purebred; PR = progressive; S = symmetric; Y = yes
Diagnostic test findings
All cats had CBC and serum biochemistry, or PCV, TS and venous blood gas analysis. MRI was performed in all cats diagnosed with congenital hydrocephalus (2/2), cervical IM (3/3) and ischaemic CVA (1/1), 4/9 cats with thiamine deficiency and 1/3 cats with FIP meningoencephalomyelitis. The cat with ischaemic CVA showed a left forebrain lesion on MRI involving the caudate nucleus, internal capsule and cerebral cortex, which was T2W and FLAIR hyperintense, T1W hypointense, hyperintense in diffusion-weighted imaging and hypointense in the apparent diffusion coefficient map. CSF analysis was performed in one cat each with thiamine deficiency, IM and congenital hydrocephalus, and the cat with ischaemic CVA. Immune-mediated polymyositis was confirmed via muscle histopathology, which showed endomysial and perimysial mixed mononuclear cell infiltrations, regional fibre loss and fibrosis.
Post-mortem examination findings
Diagnosis was confirmed via post-mortem examination in two cats. Histopathological examination of the cervical spinal cord in one cat with IM revealed a large area of necrosis and cavitation involving the central and ventral grey matter at the level of the C2 vertebra. Hyaline arteriopathy was also observed, consistent with an ischaemic infarct. 36 Histopathological examination of the small intestine and ileum in the cat with dysautonomia showed multifocal neuronal loss and apoptosis.
Primary diagnoses
The most common primary diagnoses among the 86 cats were FHM (n = 42, 48.8%), hyperthyroidism (n = 10, 11.6%), thiamine deficiency (n = 9, 10.5%), IMPN (n = 6, 7%), cervical IM (n = 5, 5.8%), acquired myasthenia gravis (n = 3, 3.5%) and FIP meningoencephalomyelitis (n = 3, 3.5%). Of the remaining cats, two were diagnosed with congenital hydrocephalus, two with hypernatraemia and one cat each with dysautonomia, hyponatraemia, ischaemic CVA and immune-mediated polymyositis. Concurrent hypokalaemia was observed in 6/10 (60%) hyperthyroid cats and 1/9 (11.1%) thiamine-deficient cat.
Extraneural comorbidities
Comorbidities were more frequent in cats with FHM and included hyperaldosteronism (11/42, 31%), chronic kidney disease (7/42, 16.7%), congestive heart failure (3/42, 7.1%), diabetes (2/42, 4.8%) and renal tubular acidosis (2/42, 4.8%). The only Burmese cat was diagnosed with periodic hypokalaemic myopathy.
Treatment and outcome
In total, 67/86 (77.9%) cats received treatment. Specifically, 30/42 (71.4%) cats with FHM received intravenous or oral potassium. All treated hyperthyroid cats (6/10, 60%) received thiamazole, with two also receiving potassium. All thiamine deficient cats (9/9, 100%) received oral thiamine, with one also supplemented with potassium. All cats with cervical IM (5/5, 100%) and IMPN (6/6, 100%) received physiotherapy, with two of the latter also receiving oral prednisolone. All cats with acquired myasthenia gravis (3/3, 100%) were treated, two (66.7%) with oral pyridostigmine and one (33.3%) with dietary supplements. Cats with FIP meningoencephalomyelitis (3/3, 100%) received antiviral treatment, with two (66.7%) also receiving oral prednisolone. Both cats with congenital hydrocephalus (2/2, 100%) underwent ventriculoperitoneal shunting. One cat with hypernatraemia and one cat with hyponatraemia received intravenous fluid therapy. Of the 86 cats, nine (10%) also received treatment for the identified comorbidities.
All but two cats (65/86, 75.6%) that received treatment for the primary diagnosis showed an improvement of the cervical ventroflexion upon discharge. The two cats that failed to improve, one with thiamine deficiency and one with cervical IM, were euthanased before discharge, along with the 21/86 (24.4%) untreated cats.
Follow-up was available for 33/86 (38.4%) cats, with a median time of 30 days (range 7–2190, IQR 34). Of these, 30 (90.9%) remained improved, while three deteriorated and were euthanased, including one each of FHM, acquired myasthenia gravis and FIP meningoencephalomyelitis. A summary of all clinical findings, diagnoses and treatments are provided (see Table 1 in the supplementary material).
Statistical analysis
Univariable analysis was performed to compare the group of cats with FHM vs a group with all other diagnoses combined. The absence of additional neurological deficits was significantly associated with a diagnosis of FHM (P = 0.02). Presence of abnormal gait (P <0.001), postural reaction deficits (P <0.001), segmental spinal reflex deficits (P = 0.016) and cranial nerve deficits (P <0.001) was negatively associated with a diagnosis of FHM. Lateralisation of the neurological signs was again negatively associated with a diagnosis of hypokalaemic myopathy (P = 0.004) (Table 2). Neuroanatomical localisation differed significantly between the two groups (P = 0.009). In FHM, the most common localisation was neuromuscular, whereas localisations such as cervical spinal cord segments or central vestibular system were observed exclusively in the other diagnoses group. Multivariable analysis did not reveal any statistical significance among the variables.
Univariable analysis comparing variables in cats with feline hypokalaemic myopathy vs all other diagnoses
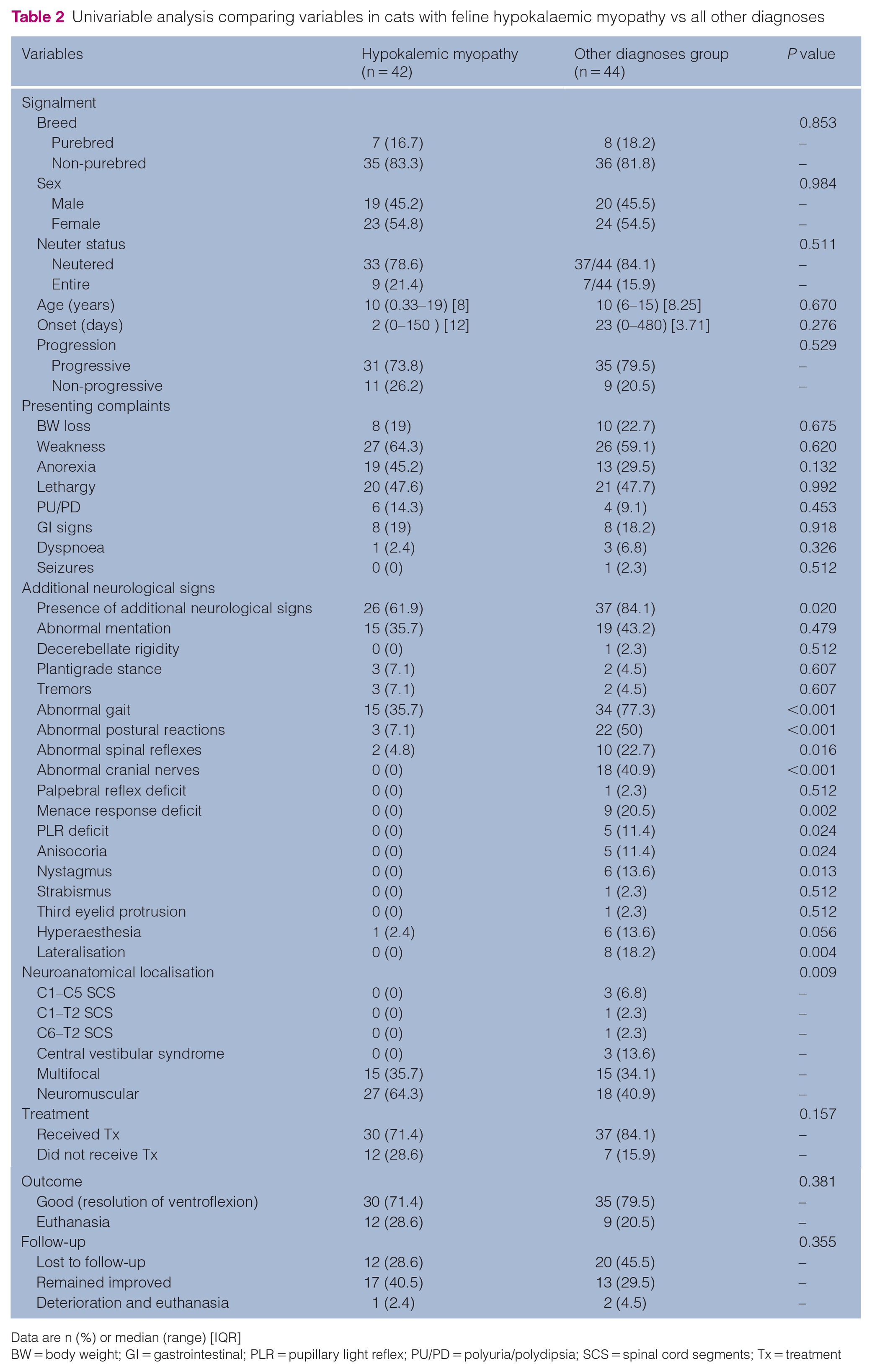
Data are n (%) or median (range) [IQR]
BW = body weight; GI = gastrointestinal; PLR = pupillary light reflex; PU/PD = polyuria/polydipsia; SCS = spinal cord segments; Tx = treatment
Discussion
Our study provided a prioritised list of differential diagnoses that can aid clinicians when encountering cats with cervical ventroflexion. FHM, the most common primary diagnosis in our study, was significantly associated with the absence of additional neurological deficits or lateralisation of signs when compared with the other diagnoses – two easily identifiable features that can strongly suggest this diagnosis.
As previously reported, FHM was the most common cause of flaccid cervical ventroflexion. 4 Onset of cervical ventroflexion was predominantly acute progressive, with the semiology limited to cervical ventroflexion, occasionally accompanied by obtundation. Hyperaldosteronism was the most prevalent comorbidity in this group, followed by chronic kidney disease, consistent with previous findings.1,12 Cervical ventroflexion was completely reversible in treated FHM cats, as previously described.1,12,14
Hyperthyroid cats mainly presented with hyperacute onset of cervical ventroflexion. Hypokalaemia was identified in 60% of them, higher than the 20% reported in a previous study. 37 This electrolytic abnormality is a rare consequence of hyperthyroidism, likely secondary to potassium wasting through polyuria, or potassium intracellular shifting. 38 Although hyperthyroidism has a direct myopathic effect, concurrent hypokalaemia and thiamine deficiency can exacerbate the resultant weakness.19,38,39
Thiamine deficiency was associated with a chronic, progressive presentation. Abnormal mentation, abnormal gait, nystagmus, mydriasis and menace response deficits were frequently reported, similar to previous reports.6 –8,23 Thiamine-deficient cats present spastic cervical ventroflexion alongside muscle hypertonicity, torticollis and head tilt. 4 Experimental studies suggest that handling or suspending affected cats can induce seizures, exacerbating neck flexion.6 –8,40,41 Pathological findings support involvement of the caudal colliculi, vestibular, oculomotor, red and lateral geniculate nuclei, although the basal nuclei may also be affected.6 –8,41,42 Spastic cervical ventroflexion could therefore represent structural focal dystonia involving the cervical musculature (anterocollis). 9
Cats with IMPN typically presented acute, progressive and symmetrical neurological signs. Median age at onset was 12 months; in a previous study, 91% of cats were aged under 3 years. 17 Neck weakness and ventroflexion were documented in 25.7% of cats with IMPN in a recent study. 18 Cervical ventroflexion was reversible in all cats of this group after treatment, reflecting the favourable prognosis previously described.17,18
Cats diagnosed with cervical IM mainly presented with hyperacute, lateralised neurological signs. Median age at onset was 11 years, consistent with previous studies.15,26 All cats received supportive treatment, with all but one improving. As previously described, cervical ventroflexion in these cases likely results from dysfunction of the lower motor neurons due to cervical spinal cord grey matter involvement, leading to cervical dorsal epaxial muscle flaccid paresis. 15
Three cats each were diagnosed with acquired myasthenia gravis and FIP meningoencephalomyelitis. Cervical ventroflexion in cats as a sequela of acquired myasthenia gravis results from skeletal muscle weakness of the epaxial cervical musculature due to impaired neuromuscular transmission.16,25,39,43,44 Consistent with previous literature, cats diagnosed with FIP meningoencephalomyelitis in this study were young and presented mainly with vestibulocerebellar signs.34,35,45,46 Although cervical ventroflexion has not been previously associated with FIP, several mechanisms can explain it. As perivascular neutrophilic and lymphoplasmacytic infiltrates can be present within the cervical spinal cord parenchyma, ventral grey horn and ventral white matter damage may result in denervation of the paraspinal muscles.34,41 In our study, MRI in one FIP cat revealed ill-defined intramedullary contrast-enhancing lesions affecting both grey and white matter, supporting this theory. Cervical ventroflexion has also been reported in cats with central cord syndrome. 47 Cervical syringomyelia, reported in 17/24 cats with FIP in a previous study, may also contribute to the sign. 34 Notably, both syringomyelia and cervical spinal cord lesions have been associated with neck weakness and abnormal postures in dogs.48,49 Although MRI of the cats with congenital hydrocephalus in this study did not include the cervical vertebral column, given the high incidence of associated syringomyelia, a similar mechanism could explain the presence of cervical ventroflexion in this group.29,50 –52
Except for the two cats in our study, cervical ventroflexion associated with hypernatraemia has been documented in only one other case. 53 Hypernatraemia can rarely induce myopathy in cats, by increasing intracellular sodium and reducing potassium, leading to signs that resemble those of hypokalaemia. 4 Although hyponatraemia-induced myopathy is a known entity in human medicine, and has been associated with muscle weakness in the veterinary literature, cervical ventroflexion is not commonly reported in this context.11,24,54 –56
The ischaemic CVA in the cat in this study involved the caudate nucleus, internal capsule and cerebral cortex. The quality of the ventroflexion (flaccid vs spastic) was not documented, making it challenging to determine a possible mechanism. Focal cervical dystonia has been associated with caudate nucleus lesions in humans.57,58 Reversible DHS has been reported in a human patient with an ischaemic CVA affecting the caudate nucleus and putamen. 59
One cat had a final diagnosis of dysautonomia. Interestingly, cervical ventroflexion associated with dysautonomia was previously reported in a retrospective study. 60 Although dysautonomia primarily affects the sympathetic and parasympathetic ganglia, milder histopathological changes have been described in the general somatic efferent or afferent neurons, and specifically in the dorsal root spinal ganglia, motor nuclei of several cranial nerves and ventral horn grey matter of the spinal cord.61 –63
Immune-mediated polymyositis is a recognised cause of flaccid cervical ventroflexion in cats.1,4,14 Histopathological examination in our case revealed mononuclear infiltration with myofibre necrosis, consistent with previous descriptions. 12
The limitations of our study include its retrospective nature. It is also important to note that the quality of cervical ventroflexion (flaccid vs spastic) was not consistently recorded. Primary diagnoses and extraneural comorbidities were classified based on the current veterinary literature introducing bias as this classification may ignore any unknown potential impact of these diseases to the cervical ventroflexion. Follow-up information was only available in a small portion of the population, and in some instances was based on owner reports, thus introducing a degree of subjectivity.
Conclusions
FHM, hyperthyroidism, thiamine deficiency, IMPN, cervical IM, acquired myasthenia gravis and FIP meningoencephalomyelitis were the most common primary diagnoses of cervical ventroflexion in cats. The absence of additional neurological deficits or lateralisation of neurological signs was significantly associated with a diagnosis of FHM. Thiamine deficiency and ischaemic CVA may lead to spastic cervical ventroflexion. This study sheds light on the clinical characteristics of cats with cervical ventroflexion and their associated underlying diagnoses that could further assist clinicians for decision-making, particularly when referral is not an option.
Supplemental Material
Table 1
Presenting complaints, concurrent hypokalaemia, comorbidities, neurological examination findings, clinicopathological findings, treatment and outcome based on the primary diagnosis
Footnotes
Acknowledgements
We wish to thank IVC Evidensia, via the IVC Evidensia Research Fund, for providing funding for this study.
Supplementary material
The following files are available as supplementary material:
Table 1. Presenting complaints, concurrent hypokalaemia, comorbidities, neurological examination findings, clinicopathological findings, treatment and outcome based on the primary diagnosis
Video. Cat with cervical ventroflexion
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received financial support for publication of this article by IVC Evidensia.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtaiend from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues or samples) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
