Abstract
Objectives
The aim of this prospective observational study was to quantitate voluntary consumption of a nutrient-enriched water (NW) supplement and assess any improvements in serum biochemical values, clinical hydration assessments or quality of life (QoL) parameters in clinically dehydrated feline patients.
Methods
A total of 13 stable, clinically dehydrated pet cats, for which outpatient management was deemed clinically appropriate by the attending veterinarian, completed the study between May and December 2023. Cats were offered NW based on body weight, along with ad libitum water. Fluid intake was measured, and changes in blood urea nitrogen and other biochemical measures of hydration, body weight, clinical hydration and owner-perceived QoL) survey scores were assessed at baseline and at 48 h.
Results
Voluntary NW intake (median 38.9 ml/kg/day) was significantly higher than ad libitum drinking water consumption (13.7 ml/kg/day; P = 0.005). Owners perceived the NW to be well accepted by their cats. Cats had a median total fluid intake of 47.2 ml/kg/day and every participating cat exceeded a target total fluid intake of 30 ml/kg/day. There were no changes in biochemical markers or body weight over 48 h of NW intake. Clinical assessments of hydration significantly improved (P <0.004), as did owner-perceived QoL (P = 0.001).
Conclusions and relevance
Clinically dehydrated pet cats voluntarily accepted the NW and consumed more NW than water. Median NW intake exceeded commonly prescribed feline subcutaneous fluid doses of 20 ml/kg/day, and both clinical hydration and owner-perceived QoL improved over the study duration. NW provides a well-tolerated, low-stress and voluntarily accepted method to promote fluid intake in feline patients.
Introduction
Dehydration is a common clinical abnormality in feline patients with hyporexia, vomiting, diarrhea, decreased water intake or increased urinary water losses. Conventional methods of rehydration include the administration of intravenous (IV) or subcutaneous (SC) fluids. However, complications can arise from both IV and SC fluid administration, including phlebitis, dislodgement of the catheter, infection, cellulitis, local edema and pain.1,2 In addition, IV fluids require hospitalization, which can be stressful to cats and may not be necessary for cases of mild dehydration.1,3 SC fluids, with a standard dose of 20 ml/kg, require repeated outpatient visits or at-home administration by the owner. 4 This can add stress to a sick cat and contribute to the financial and logistical burdens associated with repeated veterinary visits. 3 With the cost of veterinary care increasing faster than inflation, finding lower cost and accessible alternatives to conventional treatment methods is essential. 5
Oral rehydration fluids have been demonstrated to effectively improve hydration in both human and veterinary patients.6 –12 When tolerated, oral rehydration is preferred because it is less likely to cause fluid overload.13,14 In clinically ill dogs, flavored oral fluid therapy expedites the return of voluntary appetite and improvescaloric intake compared with water. 15 Amino acid-based rehydration solutions have proven effective in restoring gut health and improving gastrointestinal signs in mice and humans.16 –18 In human pediatrics, oral or nasogastric rehydration is at least as effective as IV rehydration, with significantly fewer side effects. 13 Palatable oral hydration solutions, such as diluted apple juice, result in improved treatment outcomes, and the use of oral electrolyte hydration solutions results in reduced staff workload, shorter hospital stays and higher parent satisfaction levels compared with IV fluid therapy.19,20
Although oral rehydration is effective in other species, its viability in cats remains unclear, as their weak thirst drive can prevent them from increasing water intake sufficiently to correct dehydration.21 –23 In fact, reported normal voluntary water consumption in cats varies widely, with a range of 0.17–30 ml/kg, depending on diet.10,24,25 Nutrient-enriched water (NW) supplements are associated with significantly increased water consumption in healthy cats.10,26 However, it is not known whether NW would promote fluid intake in clinically ill cats.
The aim of this prospective observational study was to quantitate voluntary consumption of an NW supplement and assess any changes in body weight, biochemical and clinical hydration assessments, and quality of life (QoL) parameters in clinically dehydrated feline patients over a 48 h period.
Materials and methods
Cat population
Mildly dehydrated cats, deemed clinically appropriate for outpatient therapy based on history and physical examination, were recruited at the University of Wisconsin-Madison Veterinary Care (UWVC) and its affiliated spectrum of care clinic, WisCARES, between May and December 2023. The inclusion criteria consisted of cats that were 5–8% dehydrated based on clinical signs such as reduced skin turgor, tacky mucous membranes or sunken eyes.27,28 The exclusion criteria included any anorexia or adipsia reported within the previous 2 weeks, history, physical examination or laboratory findings consistent with systemic infection, new onset heart failure or diabetes mellitus, or a serum creatinine over 2.8 mg/dl. Additional exclusion criteria included SC fluid administration within the past 14 days, the need to be fed an elimination diet exclusively or the administration of new medications other than ondansetron indicated during the study interval. Owners of multi-pet households had to be able to separate the enrolled cat from other pets for the duration of the study with minimal anticipated stress. Protocols were approved by the Institutional Animal Care and Use Committee (IACUC) at the University of Wisconsin-Madison. All owners provided written informed consent.
Study interventions
Eligible cats in the outpatient clinics were screened with a complete blood count, serum chemistry panel, complete urinalysis using urine collected by cystocentesis and body weight measured using the same scale during both the initial and follow-up visits. We also calculated serum osmolality (mOsm/kg) as (2[sodium + potassium] + [glucose/18] + [BUN/2.8]), with electrolytes expressed in mEq/l and glucose and blood urea nitrogen (BUN) in mg/dl.29,30 Hydration was assessed independently for each cat by two members of the study team (AN and MP) and was scored in the range of 0–10% (Table 1). 27 Hydration scores were then shared between the two assessors and any differences were resolved via consensus.
Clinical assessment of dehydration (modified from Matthews) 27

Owners of eligible cats completed selected questions from a validated feline QoL survey at baseline (see Appendix S1 in the supplementary material). 31 Owners were provided pouches of the NW supplement (Hydra Care; Nestlé Purina PetCare). Each packet of NW contains 85 g and is 97% moisture and 2% protein. The primary three ingredients are water, glycine and whey protein. Label instructions were to offer one pouch per day for small cats (up to 5 lbs), two pouches per day for medium sized cats (5.1–10 lbs) and three pouches per day for larger cats (>10 lbs).
Owners were asked to record the cat’s intake of the NW each day by using a provided calibrated gram scale to weigh the empty bowl, the bowl plus NW and the same bowl plus NW after 24 h. The investigators confirmed a 1:1 relationship between NW weight and liquid volume, by weighing a 50 ml syringe, drawing up 50 ml of NW and reweighing the syringe plus NW. In total, 50 ml of NW weighed 50 g; therefore, ml units were used for analyses.
The owners replenished the bowl with the label dose of NW each study day and repeated the weighing procedure. Owners were also asked to rank on a 5-point Likert-type scale how much they felt their cat enjoyed the NW each day. Options were ‘not at all’, ‘a little’, ‘somewhat’, ‘very’ and ‘extremely’.
Owners were instructed to use the same method to provide a measured amount of water, weighing the bowl at the start and end of 48 h. If the water bowl was emptied in the interim, owners refilled and reweighed accordingly. Cats were continued on their current diet(s) and owners recorded the types of diets offered (dry, canned or mixed). Owners were also asked to note any water spillage or if their cat drank from an additional source (eg, faucet) during the study period. A phone call 24 h after the initial examination was conducted to ensure cats were not showing signs of worsening diarrhea, or persistent or worsening hyporexia, vomiting or lethargy.
The time for each in-person recheck was planned for approximately 48 h after the start of the NW intervention for each cat. Cats were reassessed in the clinic 2 days after starting the NW with a physical examination, including a body weight and hydration estimate, packed cell volume (PCV)/total protein (TP), abbreviated serum chemistry panel (sodium, potassium, glucose, BUN, creatinine, TP, albumin), calculated serum osmolality and urine specific gravity (USG). Owners also completed the same selected questions from the QoL survey after 48 h of treatment. 31
Statistical analyses and sample size
Estimated dehydration was encoded as an ordinal variable: 0, <5% dehydrated; 1, 5–6% dehydrated; 2, 7–8% dehydrated; and 3, >8% dehydrated. For each cat, Wilcoxon signed-rank tests were used to compare baseline and 48 h biochemical markers of hydration (BUN, albumin, total protein, PCV, USG, calculated serum osmolality); body weight, clinical hydration assessment and calculated fluid deficit; and QoL scores, as well as differences between measured NW and water intake for each cat. P <0.05 was considered significant.
Although this was an observational study, we performed an a priori sample size calculation to establish power to detect improvements in BUN during NW treatment. We estimated that a typical clinically ill cat would present with mild prerenal azotemia, for example, a BUN of approximately 36 mg/dl, and hypothesized that NW would lead to a 20% improvement in BUN, to approximately 29 mg/dl, over 48 h. A recent study that evaluated NW in healthy cats found a small standard deviation in baseline BUN (1 mg/dl). 26 We assumed a five-fold greater variability (SD 5 mg/dl) in presenting BUN in clinically ill cats for the present study. Based on these assumptions, we estimated the need to enroll nine cats to show a 20% decrease in BUN as significant, with a power of 80% and P <0.05 (www.stat.ubc.ca/~rollin/stats/ssize/).
Results
A total of 14 cats presenting to the primary care and WisCARES outpatient clinics were considered eligible based on history and physical examination and were further screened for the study with blood work. One cat was excluded for enrollment because of a new and significant azotemia (creatinine >2.8 mg/dl) on screening blood work. Of the 13 cats enrolled, all owners completed the QoL surveys and rated their perception of their cat’s enjoyment of the NW, 12 owners recorded daily NW and fluid intake, and 11 cats had both before and after blood and urine samples collected. Two cats did not have post-laboratory samples drawn: one because no NW intake data were completed, and the other because the cat was not amenable to repeated sampling.
The 13 enrolled cats had a median age of 7 years (range 6 months to 17 years) and consisted of four spayed females and nine castrated males. Of the cats, 12 were domestic shorthair cats and one was an Oriental Shorthair. Of the 13 cats, four (31%) were fed an exclusively dry diet, one (8%) was fed an exclusively canned diet and eight (62%) were fed a mixture of canned and dry food. A total of 10 cats presented with gastrointestinal signs (including hyporexia, weight loss, diarrhea, vomiting, tenesmus), one with increased drinking and urination, and two with upper respiratory signs. Of the 13 cats, four had a USG below 1.030 at presentation (Table 2): two with historical, stable International Renal Interest Society stage 2 chronic kidney disease (one of these cats also had controlled hyperthyroidism and hypertension, with no changes in medications during the study period), one with newly documented mild azotemia (serum creatinine 2.1 mg/dl) and one with a serum creatinine of 0.8 mg/dl.
Biochemical markers and body weight of dehydrated cats given a nutrient-enriched water supplement on an outpatient basis

Reference intervals are as follows: sodium (148–157 mmol/l), potassium (3.5–5.1 mmol/l), BUN (15–35 mg/dl), creatinine (0.9–2.3 mg/dl), albumin (2.7–3.9 g/dl), total protein (5.5–7.1 g/dl), PCV (31–51%), osmolality (290–330 mOsm/kg). P values compare pre- and post-NW administration
BUN = blood urea nitrogen; PCV = packed cell volume; USG = urine specific gravity
No cats were removed from the study because of worsening clinical signs. There were no owner-reported adverse effects of the NW. Food consumption remained consistent from baseline for each individual cat. One cat was perceived to drink out of the toilet briefly before the owner intervened. There was no other report of unrecorded water consumption or spillage events. Owners’ perception of NW acceptance over 48 h was high, with a median score of 4 (range 1.5–4.5) on a 5-point Likert-type enjoyment scale (Figure 1).

Owner-perceived enjoyment of the nutrient-enriched water supplement in clinically ill pet cats, using a 5-point Likert-type scale averaged over 48 h
Biochemical measures of hydration (n = 11) and body weight (n = 12) compared before and after NW administration showed no significant differences over the 48 h period (Table 2). Of the 12 cats, eight gained weight over the study period and 7/11 cats that had before-and-after blood work had an observed decrease in calculated serum osmolality over the study period.
Clinical assessment of hydration improved over the study period (median 2, range 1–2 at baseline and median 1, range 0–2 at 48 h; P = 0.004). No patient appeared less clinically hydrated after 48 h (Figure 2).
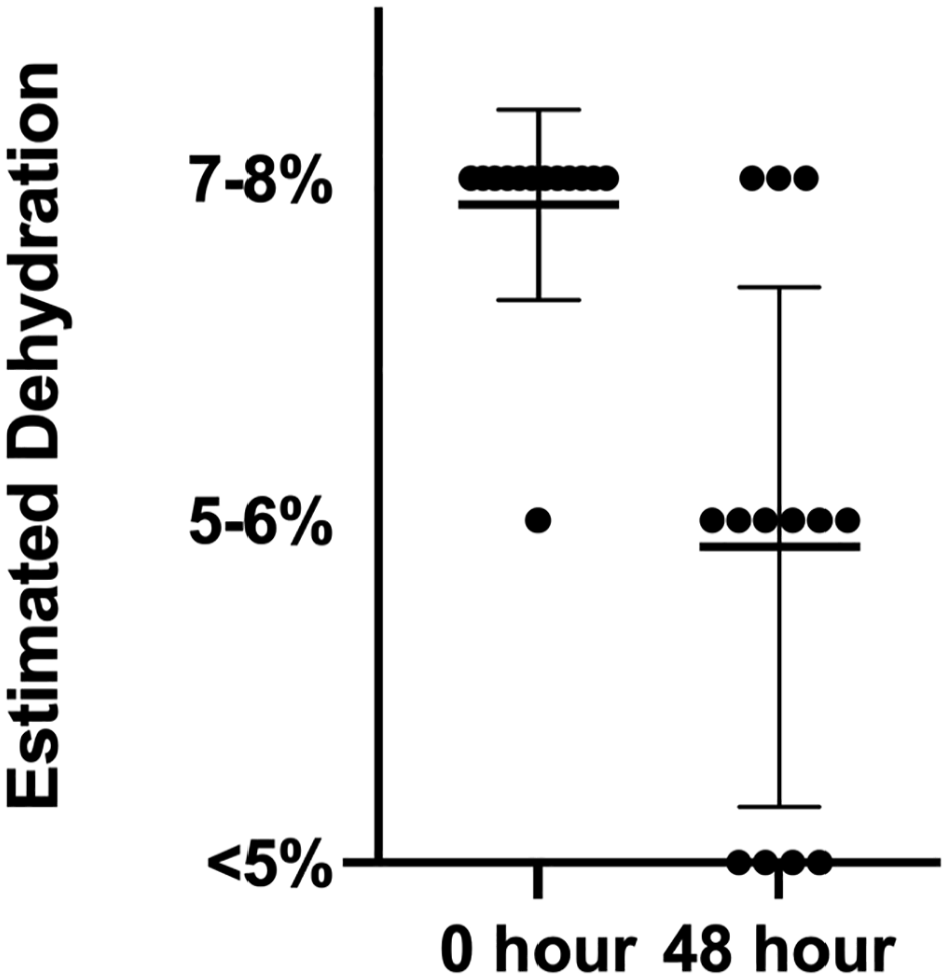
Estimated level of clinical dehydration in cats before administration of nutrient-enriched water and at the 48 h recheck
The median calculated fluid deficit at baseline was 293 ml (75 ml/kg) (Table 2). The median calculated fluid deficit at 48 h was 199 ml (51 ml/kg), which was lower than at baseline (P = 0.006). The median total fluid consumed over 48 h of the NW plus water was 179 ml (range 109–304). The cats had a median total fluid intake of 47.2 ml/kg/day (range 32.6–122.4), which was greater than our target fluid intake of 30 ml/kg/day.
Five cats consumed the entire volume of NW offered. Cats consumed significantly more NW (median 38.9 ml/kg/day, range 8.4–66.4) than water (median 13.7 ml/kg/day, range 2.37–56; P = 0.005) (Figure 3).

Scatterplot depicting the total fluid intake (nutrient-enriched water [NW] and water) measured in ml/kg body weight, the amount of NW intake and the amount of water intake in 12 clinically ill pet cats over 48 h
Owner-perceived QoL scores significantly increased from a median of 27 (range 21–35) to a median of 32 (range 25–35; P = 0.001) over the 48 h period (Figure 4). No owners reported a worsening in QoL score over the 48 h period.

Line graph showing change in owner-perceived quality-of-life (QoL) scores (out of 35 total points) in 13 clinically ill cats, at baseline and at 48 h after receiving nutrient-enriched water. Each line shows an individual response; two pairs of cats have the same scores at both time points and are indicated by a single line
Discussion
Our study found that an NW supplement was a well-tolerated, voluntarily accepted oral hydration solution for dehydrated pet cats fed a variety of diets. This aligns with previous studies showing that this supplement was palatable in laboratory-housed healthy cats fed a uniform diet.10,26 The tested NW supplement was accepted even in cats presenting for gastrointestinal signs, and no cats had to withdraw from the study for worsening gastrointestinal or other clinical signs.
Cats in this study had a median fluid intake of 47 ml/kg/day over 48 h. This exceeds previous estimates of normal fluid consumption in cats, which had a wide range 0.17–30 ml/kg/day, depending on diet.24,26 In fact, every study cat consumed over the target fluid intake of 30 ml/kg/day, as would be expected in cats presenting with mild dehydration. In addition, the NW made up over 80% of the overall fluid intake in our study cats. This aligns with pediatric studies demonstrating the importance of palatability on compliance and effective rehydration treatment. 19
We did not find significant changes in biochemical measures of hydration with NW administration at the label dosages over 48 h. We had predicted a 20% decrease in BUN with NW treatment, but only one cat (cat 3) met this standard. Decreases in BUN can be seen as early as 8 h after oral rehydration in dehydrated dogs; 8 however, in that study, the volume of oral rehydration was individualized based on patient-estimated fluid deficit. It is possible that if we had individualized the amount of NW offered based on fluid deficits, or offered it ad libitum, we would have seen changes in biochemical measures in more cats.
Despite no significant changes in BUN, 7/11 cats did show a reduction in serum osmolality over the 48 h, which is consistent with observed correlations between serum osmolality and hydration status in human patients. 32 It is possible that a higher osmolality of NW compared with drinking water blunted an expected decrease in serum osmolality in the remaining cats or that our treatment period was too short to detect improvements across all cats.
Overall body weight did not increase significantly over 48 h for these cats, although 8/12 cats did gain weight. The median total amount of fluid consumed over the 48 h observation period was 179 ml, while the median calculated fluid deficit at baseline was 293 ml. Although median estimated fluid deficits did decrease significantly, the lack of complete response could be due to either lack of sensitivity of clinical evaluation of hydration or that 48 h might not have been enough time to fully detect the effects of oral rehydration on body weight. 33
Although we did not find changes in biochemical markers of hydration, we did find a significant improvement in clinical hydration, albeit through subjective, non-blinded assessments. Some, but not all, human and veterinary studies support the use of clinical hydration parameters, such as skin turgor, to assess small changes in interstitial hydration.14,33,34 –37 The improvement in clinical hydration assessments is consistent with the measured fluid intake of the study cats.
The QoL survey considered owners’ perceptions of their cats’ energy and behaviors with the goal of assessing clinical signs of early or mild dehydration. Dehydration in humans negatively affects mood and cognition, with even 2% dehydration causing cognitive impairment and fatigue.38,39 Although early signs of dehydration in cats are poorly characterized, our findings of owner-perceived improvement in QoL could indicate resolution of mild dehydration not easily assessed with biochemical hydration assessments. However, owner bias is an important consideration, as clients were aware of the study’s intervention, and the absence of a masked control group limits the ability to conclude that NW directly improved QoL in these cats.
This observational study is limited by several factors, including a small sample size, lack of blinding for both clinicians and clients, and the absence of an untreated control group for ethical reasons. Differences in dietary moisture have been shown to affect voluntary fluid intake, but we did not attempt change to a uniform diet during the study period because of possible food rejection in these clinically ill cats.24 –26 Given that almost 70% of the cats in this study were fed some canned food, we were likely underestimating true fluid intake for most of our study cats. In addition, we did not account for evaporative water loss over the 48 h period. The timing of the sample collection could have underestimated the efficacy of NW, as most cats consumed the NW immediately after being offered, and blood and urine samples were collected at the end of 48 h (ie, at the potential nadir of NW effect). Finally, the amount of NW provided was based on manufacturer recommendations, and allowing owners to offer additional pouches of NW could have further improved hydration assessment.
Despite its limitations, our study found a preference for NW over water in dehydrated cats, with a voluntary NW intake of 39 ml/kg/day, which could complement or provide an alternative to parenteral fluid administration in mildly dehydrated cats.
Conclusions
We found that an NW supplement can be used in dehydrated cats to help promote fluid intake in the outpatient setting. The NW was well tolerated, voluntarily accepted and preferred over water, with cats consuming about three times more NW than water. Cats also consumed almost double the volume of NW (39 ml/kg/day) than typically administered volumes of SC fluids (20 ml/kg/day). While the volume of NW consumption did not solely correct for calculated fluid deficits over 48 h, this study demonstrates that NW supplementation offers an affordable and accessible alternative or adjunctive therapy to SC fluids for outpatient management of stable dehydrated cats, which may be less stressful for both the cat and owner. Future studies including a control group only offered water should evaluate the effects of longer administration durations in a larger population of clinically ill cats.
Supplemental Material
Appendix S1
Selected questions from a validated feline qualityof- life survey.
Footnotes
Acknowledgements
Nestlé Purina PetCare provided the nutrient-enriched water supplement Hydra Care for the study, but did not participate in the study design, data analyses or conclusions drawn.
Author note
Some of these data were presented in abstract form at the 2024 ACVIM Forum in Minneapolis.
Supplementary material
The following file is available as supplementary material:
Appendix S1: Selected questions from a validated feline quality-of-life survey.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Nestlé Purina PetCare provided funding to RM in the form of a Veterinary Student Summer Research Fellowship.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
