Abstract
Objectives
Oral squamous cell carcinoma (SCC) is the most common oral tumor in cats but treatment options that provide long-term tumor control are limited. Radiation therapy is a reported treatment option, but local tumor control is still difficult to obtain and additional treatment options are needed. Toceranib phosphate recently emerged as having biologic activity against feline oral SCC. This study is a preliminary evaluation of radiation therapy and toceranib phosphate in cats with oral SCC.
Methods
This non-blinded, retrospective, single-institutional study included all patients between 2011 and 2023 that underwent the same treatment with accelerated radiation therapy and concurrent carboplatin as previously described, with the exception of one additional fraction of radiation. Once the early side effects abated, toceranib phosphate was offered as follow-up maintenance therapy. The median survival time (MST) and progression-free interval (PFI) were assessed for the cats that received toceranib after radiation therapy and carboplatin, and were compared with the cats that received the same radiation and carboplatin protocol within the same time period but did not receive follow-up toceranib.
Results
Overall, 47 cats met the criteria for evaluation; of them, 15 received follow-up toceranib. The MST of all cats was 164 days; there was no significant difference in MST or PFI between the cats that did and did not receive adjuvant toceranib (MST 208 days vs 162 days, respectively; P = 0.35). When comparing cats with lingual tumors, the PFI was significantly longer in the cats that received toceranib than those that did not (142 days vs 104 days, respectively; P = 0.045); however, there was no difference in MST (197 days vs 147 days; P = 0.15).
Conclusions and relevance
This study suggests that most cats with oral SCC do not benefit from toceranib after radiation therapy. There may be clinical benefit to administering adjuvant toceranib after radiation in cats with lingual SCCs, but the mechanism remains unclear and additional studies are warranted.
Introduction
Oral squamous cell carcinoma (SCC) is a tumor arising from the squamous epithelium lining the oral cavity and is the most common oral tumor in cats. 1 It is considered a model tumor for human head and neck SCCs.2,3 The etiology is not entirely known but has been associated with cats living in rural areas, wearing flea collars, living in households where tobacco is smoked and eating canned food, especially canned tuna.4,5
The treatment with the longest reported survival times for oral SCC is radical surgery followed by radiation therapy, but up to 90% of cats have significant morbidity with this treatment and local recurrence is still common.1,6,7 Owing to the undesired impact of such aggressive treatment on the patient’s ability to eat and a likelihood of local recurrence, alternative treatment strategies have been investigated. Without surgery, traditional radiation strategies of 3–4 Gy per dose for a maximum dose between 40–50 Gy have been ineffective in controlling tumors.1,8 Additionally, stereotactic radiation has been used with a rapid improvement in clinical signs but disappointing responses. 9 One promising treatment option that has emerged on the human medical front is accelerated radiation protocols that shorten the interval between radiation doses to reduce cell repopulation, and the addition of chemotherapeutics to sensitize cancer cells to radiation. 10 A similar radiation therapy protocol using carboplatin was developed for cats with oral SCC with good results and acceptable toxicity, but most cats still died or were euthanized due to complications of their tumor, making clear that additional treatments are needed for these cats. 8
Tyrosine kinase inhibitors (TKIs) have revolutionized the landscape of human oncology and the ability to target specific proteins depending on the protein expression patterns of each tumor. 11 Since it received approval from the US Food and Drug Administration, toceranib phosphate (Palladia; Zoetis), a TKI, has been used for a variety of tumors, including oral SCC, in both dogs and cats.12–14 Toceranib phosphate targets the proteins KIT, PDGFRβ, VEGFR2, CSF-1 and FLT-3. 14 While studies have evaluated tumor expression of tyrosine kinases in other feline tumors, none have specifically looked at protein expression of the targets of toceranib phosphate in feline oral SCC.15,16 One study focused investigation on gene expression (mRNA) and mutations in feline oral SCCs; the tyrosine kinases targeted by toceranib were not among the most commonly mutated or dysregulated genes. 3 Feline oral SCCs have been shown to respond to the drug for short periods of time, ranging from 2 to 4 months with a response rate of 56.5%.12,13
The radiation protocol used in the study by Fidel et al 17 was modified in stages to attempt to improve patient outcomes. Initially, the accelerated protocol treated with 49 Gy in 14 fractions of radiation with treatments administered twice a day over a 9–10 day period. The MST was 86 days. 17 Adding carboplatin as a radiosensitizer to this protocol increased the MST to 163 days. 8 The current study was undertaken to determine if there was a survival benefit in oral SCC bearing cats that received accelerated radiation therapy and follow-up toceranib phosphate after radiation side effects resolved.
Materials and methods
Clinical study
This was a non-blinded, retrospective study involving cats that received accelerated radiation therapy and carboplatin for oral SCC. The Washington State University radiation therapy database was searched for all cats with oral SCC treated between 2011 and 2023. Beginning in 2011, all cats that received radiation therapy for oral SCC were eligible to begin toceranib phosphate 4 weeks after radiation therapy concluded and early radiation side effects had resolved. All cats were required to have a histologic or cytologic diagnosis of SCC primarily affecting the oral cavity. Cats were excluded if they received any surgery, other than excisional biopsies for diagnostic purposes, prior to radiation therapy. Data were extracted from medical records by manual review and communication with referring veterinarians or owners as needed.
All cats treated with radiation therapy between 2011 and 2023 had CT-guided plans. At the time of CT, an individualized plastic bite block and a vacuum locking bed that conformed to the patient’s body were made and used throughout radiation therapy. All radiation treatments were planned by the same radiation oncologist (JF) and were three-dimensional conformal plans. The gross tumor with roughly 1 cm margins and any known or suspected metastatic lymph nodes was included in the plan. The clinical target volume was created by adding a 3 mm margin to the gross tumor margin.
Radiation therapy was delivered with a single linear accelerator (Elekta SLi 15, Phillips Medical Systems NA). The prescribed radiation dose of 52.5 Gy was administered as 15 fractions of 3.5 Gy given once on the first day, then twice daily for 9.5 total days; the interval between treatments when given twice daily was at least 6 h. Carboplatin was dosed at 180 mg/m2 divided into two doses of 90 mg/m2 given on day 1 and day 6 of the treatment protocol. The patients’ glomerular filtration rate was not directly measured but all patients had recent bloodwork, including a blood chemistry panel with blood urea nitrogen and creatinine. If they were azotemic (creatinine >1.6 mg/dl), a urine specific gravity was obtained to better inform carboplatin dosing. 18 If a patient had International Renal Interest Society (IRIS) stage 2 or 3 chronic kidney disease (CKD), the dose of carboplatin was reduced from 90 mg/m2 to 75 mg/m2. Esophagostomy tubes were placed as needed to manage difficulties eating from tumor-associated morbidity and radiation toxicities at the attending clinician’s discretion and with the permission of the owner.
A recheck CT scan was recommended 28 days after radation therapy finished and toceranib was offered at that time if the patient was clinically well enough for oral chemotherapy and the owners elected further therapy. Patients were included in the analysis if they started toceranib within 6 months of beginning radiation therapy and there was no evidence of progressive disease prompting the initiation of other treatments. Concurrent non-steroidal anti-inflammatory drug (NSAID) therapy was allowed. The dosage of toceranib was not standardized but was intended to be between 2.5 and 2.75 mg/kg given Mondays, Wednesdays and Fridays.
Statistical analysis
All medical records from the patients were reviewed and incomplete records were completed as much as possible by contacting referring veterinarians or owners. Data recorded included prior treatments, treatments after radiation therapy, mitotic index (if histopathology was performed), cause of death, radiation toxicities according to the Veterinary Radiation Therapy Oncology Group (VRTOG) 2.0 criteria, time to local disease progression; local and distant metastasis were documented if they occurred. 19 Toceranib toxicity was assessed when available using Veterinary Cooperative Oncology Group-Common Terminology Criteria for Adverse Events (VCOG-CTCAE) v2 criteria. 20
Progression free interval (PFI) was defined as the time from the start of radiation therapy to local or metastatic disease progression. Survival time was defined as the time from the start of radiation therapy to the date of death. Cats were censored from PFI analysis if they were lost to follow up with no evidence of disease recurrence at the last known date of contact or if they died of other known causes. If the cause of death was uncertain and cats had evidence of SCC at the time of death, death was attributed to SCC and they were included in all analyses. Cats were also removed from comparison between the toceranib and no toceranib groups if they died before 37 days (9 days of therapy and 28 days of recovery) as they could not have received toceranib according to the protocol, but they were included in the overall survival analysis. Additional therapies were allowed after disease progression. Statistical analysis was performed using commercial statistical software (GraphPad Prism v10.3.1). Survival and PFI data were plotted using Kaplan–Meier curves and comparisons between group variables were analyzed with the log-rank test with significance set at P <0.05.
Results
Medical records from 72 cats with oral SCC that received the prescribed radiation protocol of 15 fractions of 3.5 Gy in 9.5 days were identified in the radiation database. Twenty-five cats were excluded from the study for various reasons, leaving records from 47 cats for analysis all of which received the radiation and carboplatin protocol. Of the 47 cats, there were 25 castrated males and 22 spayed females. There were 37 domestic breeds, including three Maine Coons, two Siamese, two Sphynxes, one Norwegian Forest Cat, one Himalayan and one Persian. The locations of tumors were mandible (16), maxilla (12), lingual (14), tonsil (four) and cheek (one). The median age at the start of radiation was 13 years (range 6–19).
Five cats had suspected or confirmed metastasis (one pulmonary, four nodal) at the time of radiation and three developed metastasis (one pulmonary on necropsy and two nodal) after treatment for the 8/47 cats with metastasis. Of the cats with pulmonary metastasis, one cat had radiographic evidence of pulmonary nodules suggestive of metastatic disease as well as lymph node involvement (without confirmation) at the time of radiation therapy, and one had pulmonary metastatic lesions that were not radiographically apparent 3 days prior to death but were evident on necropsy after radiation. For the cats with lymph node metastasis, two cats had confirmed lymph node metastasis (one with histopathology and one with cytology) at the time of radiation and two more had suspected lymph node involvement by CT characteristics. Two more developed lymph node metastasis after treatment (one confirmed with histopathology and the other suspected based on CT findings).
Radiation side effects were not uniformly assessed, but a recheck physical examination was recommended 4 weeks after therapy and the noted side effects were graded via the VRTOG v2.0 criteria; the toxicities are summarized in Table 1. 19 Late side effects were graded by VRTOG v2.0 through follow-up appointments and records and are summarized in Table 2. Of 47 cats, 13 had pre-emptive esophagostomy tubes placed and these were classified as grade 4 oral toxicities though not every cat may have needed them because of radiation alone. Oral toxicities ranged from patchy mucositis to severe mucositis with ulceration necessitating an esophagostomy tube in most patients with one patient developing tongue fibrosis as a late toxicity. Early toxicities in the skin were generally erythema and alopecia and late toxicities were largely leukotrichia and fibrosis; a notable toxicity in one cat was a grade 3 late skin toxicity of fibrosis and lymphedema, which is often reported after oral radiation in humans. 21 Early ocular toxicities included conjunctival hyperemia and corneal ulceration. Late ocular toxicities included mild mucoid discharge and persistent conjunctival hyperemia without ulcer formation. Central nervous system toxicities included mild lethargy, a seizure (possibly drug-induced) and dull mentation responsive to glucocorticoids. Peripheral nerve toxicities included Horner’s syndrome (likely from tumor infiltration seen surgically) and a decreased pupillary light reflex. The only bone toxicity noted was pathologic fracture evidenced by sudden severe oral pain months after radiation with or without bony deviation of the mandible (eight) or maxilla (one) that caused euthanasia in some (see Table 2).
Graded early radiation toxicities by Veterinary Radiation Therapy Oncology Group 2.0 of all patients

Graded late radiation toxicities according to Veterinary Radiation Therapy Oncology Group 2.0 of all patients

The five total toxicities were two confirmed late bone toxicities and three unlikely grade 4 toxicities
The four total toxicities were two unlikely and two possible grade 5 toxicities
After radiation, many cats received other treatments aside from toceranib at the discretion of the referring oncologist, including NSAIDs (23), carboplatin (16), bisphosphonates (five), bleomycin (four), carboplatin polymer injections (one) and mitoxantrone (one). Six cats received only NSAIDs after discharge and six cats received no further antineoplastic therapies. Eight cats began toceranib after disease progression but for data analysis were not included in the toceranib group. Of 47 patients, five had incomplete records – their cause and date of death were available, but PFI could not be assessed so these were not included in PFI analysis. In the 31 cats with available data on cause of progression, there was local progression in 27 cats and nodal progression in three cats; one cat had both local and nodal progression.
Out of 47 cats, there were three cats lost to follow-up, two immediately after discharge, one after relapse of disease at 142 days and their dates and causes of death are unknown, leaving 44 cats with known outcomes for outcome analysis. Forty-two cats died and two cats were still alive (284 and 648 days from starting radiation) at the time of writing with no evidence of disease. The cause of death in 31 cats was attributed to the local tumor and in one cat due to both local disease and suspected lymph node metastasis. Three cats died of unknown causes while the owners were not present (two in remission at the time), one cat was euthanized for an oronasal fistula at the site of the original tumor with no evidence of tumor regrowth, one cat died of suspected cardiac disease, one cat died suddenly of a suspected internal bleed or thromboembolic event, one cat died of massive adrenal necrosis evident on necropsy, one cat died of congestive heart failure, one cat died of pancreatitis and one cat died of a suspected acute kidney injury.
In the group of cats treated with toceranib that met the inclusion criteria for analysis, there were 17 that initially began toceranib; two were excluded (one owner was only able to administer two doses of medication, and one began to receive toceranib only after disease progression) leaving 15 cats in the toceranib group. There were 27 cats in the radiation group; five cats died or were lost to follow-up after radiation before toceranib could be offered. In the 15 evaluable cats in the toceranib group, the tumor location included mandible (six), maxilla (three) and tongue (six). The median toceranib dosage was 2.52 mg/kg (range 2.12–2.88) all given on Mondays, Wednesdays and Fridays. The median duration of time between starting radiation and starting toceranib was 43 days (range 22–158). During its treatment, one cat in the toceranib group developed pulmonary metastasis which was only evident on necropsy, and at the time of radiation and toceranib one other had nodal metastasis which was the source of progression without additional lymph node metastasis. While the 32 (30 plus the two cats removed from the toceranib group) cats in the radiation treatment group without toceranib included five cats with confirmed or suspected metastasis at the time of radiation, this group also included two cats that developed suspected or confirmed lymph node metastasis during treatment. Further treatments in the toceranib group after disease progression was noted included carboplatin (three), carboplatin added to toceranib simultaneously (one) carboplatin polymer injections (one), bleomycin (one) and mitoxantrone (one).
Toceranib toxicities (see Table 3) were generally mild and similar to previous reports of toceranib use in cats.12,13 One cat receiving concurrent meloxicam progressed from IRIS CKD stage 2 to 3 while receiving toceranib (a possible grade 3 toxicity). The median duration of toceranib administration was 111 days (range 27–474). In the eight cats whose tumors relapsed before they were given toceranib, and then received toceranib, their responses were unable to be assessed and the median duration of administration was 49.5 days (range 13–82).
Graded toceranib toxicities for all patients by Veterinary Cooperative Oncology Group criteria

One toxicity was confirmed and one was only present after concurrent carboplatin was added
Considered a possible toxicity
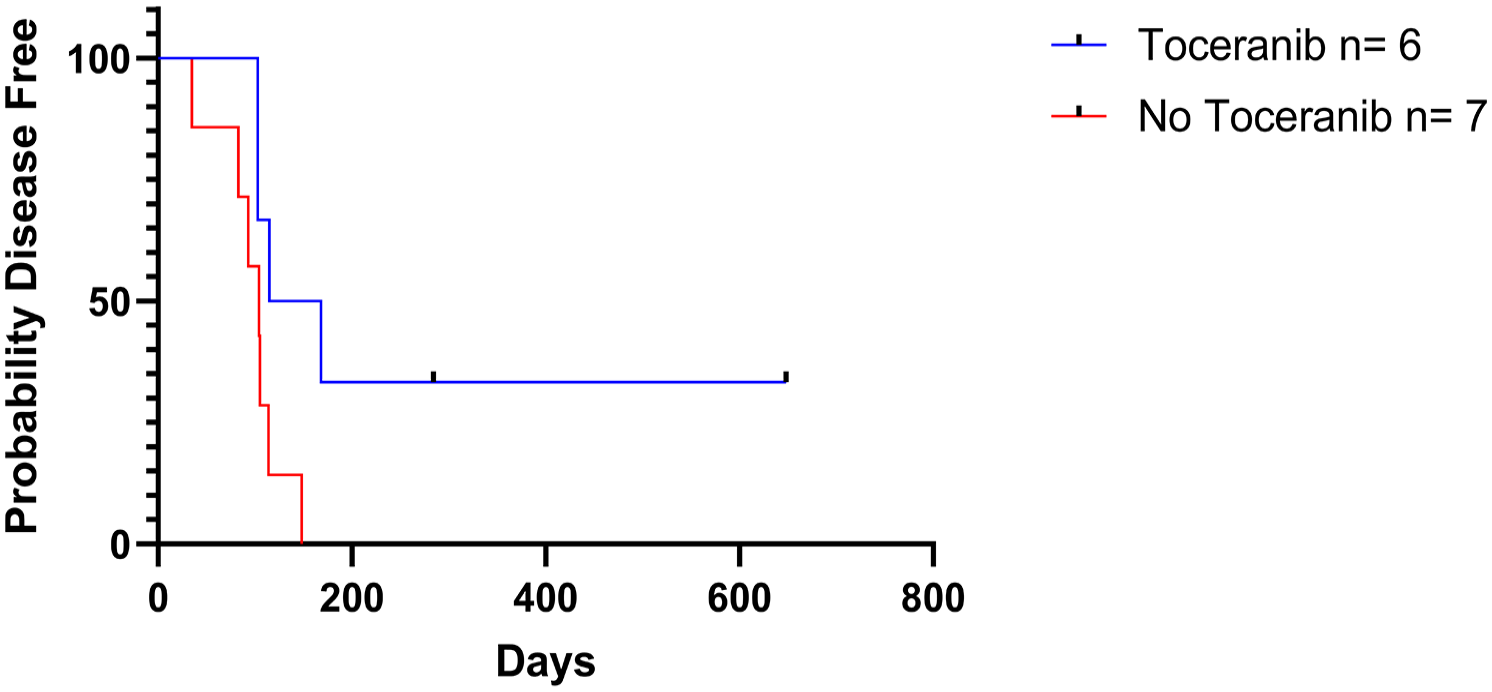
The MST of all cats in the study was 164 days and the median PFI of all cats was 121 days, as shown in Figures 1 and 2. There was no significant difference in MST between cats that received toceranib after radiation (n = 15) and those that did not as shown in Figure 3 (n = 27) (208 days vs 162 days, respectively, P = 0.35). There was no significant difference in PFI between cats that received follow up toceranib (n = 15) and those that did not (n = 23) (121 days vs 144 days, respectively P = 0.45). When comparing the 13 cats with lingual tumors from the two treatment groups evaluable (one cat died before 37 days so was not included making six in the toceranib group and seven in the radiation therapy group), there was a significantly longer PFI in cats that received adjuvant toceranib compared with those that did not as shown in Figure 4 (142 days vs 104 days, respectively P = 0.045) but there was no difference in MST (197 days vs 147 days, P = 0.15).

Median survival time for all cats in the study was 164 days

Progression-free interval for all cats in the study was 121 days

Median survival time of cats that received follow-up toceranib (208 days) vs those that did not (162 days). There was no significant difference between the groups
Progression-free interval for cats with lingual squamous cell carcinoma that received adjuvant toceranib. There was a significant difference between the groups (142 vs 104 days, P = 0.045)
When looking within the adjuvant toceranib group, there was a significant survival difference based on tumor location (mandible = 248 days, maxilla = 117 days, lingual = 197 days, P = 0.025). Within this group, there were also significantly different PFIs based on location (mandible = 183 days, maxilla = 88 days, lingual = 142 days P = 0.032). There was no significant difference in PFI or MST in cats comparing toceranib with radiation therapy for each location except lingual, between cats that had or developed metastasis, or between cats that did or did not receive NSAIDs after radiation therapy.
Discussion
This is a single institutional retrospective study with a standardized chemoradiation protocol to evaluate the efficacy of adjuvant toceranib in cats with oral SCC. The most significant finding in this study is that the majority of cats did not benefit from adjuvant toceranib after radiation, but there was an improved PFI in cats with lingual SCC. With few exceptions, most of these patients still died of their disease, their survival was not significantly improved by toceranib treatment and the survival times are similar to those previously reported with this protocol. 8 This could be due to a number of limitations of this study. It should be noted that all but one cat in the toceranib group started toceranib within 3 months of radiation therapy, and the one that started after 5 months is likely an outlier, as it was still alive at the time of writing.
Some of the limitations reflect the retrospective nature of the study. These limitations include, but are not limited to, multiple patients immediately lost to follow-up, various and inconsistent follow-up appointments, and other treatments, including toceranib, given to patients by clinicians at the time of tumor relapse, making comparisons difficult. The multiple different post-radiation treatment protocols were given at the discretion of referring clinicians, adding more variables to this study. Additionally, multiple patients were euthanized because of bone necrosis or fracture, which was attributed to tumor recurrence but not confirmed with histopathology, making it difficult to assess if bone necrosis was a side effect of treatment (radionecrosis), if the bone became destabilized after reduction of tumor within bone or a failure of the treatment to control the tumor. Another important limitation was that the groups were of different sizes, and the clinicians and owners were not blinded to which patients were receiving toceranib.
If our data are an accurate reflection of response in a population of cats treated with radiation and toceranib, it shows most cats will not benefit from use of toceranib after radiation. Previous reports show response rates of 56.5% if used without radiation, begging the question as to why there is a potential difference. It may be that the primary response reported with toceranib alone is due to the effect it has on the suppression of tumor blood supply, a known prognostic factor in human SCCs. 22 Radiation can also compromise blood supply to a tumor and perhaps this is why there seems to be no benefit when toceranib is added. It is also possible that a compromised blood supply limits the delivery of the drug to the tumor, reducing its effectiveness. Another possibility is that there is some other change in the tumor induced by radiation or chemotherapy or other therapies that were used in these patients. Further studies are needed to determine optimal therapies and timing in these patients.
Additionally, 8/47 (17%) of patients in this study had metastatic disease at diagnosis or it developed throughout their treatment. While metastasis was the cause of death in only one patient this also serves as a reminder that it does occur and clinicians should be aware of the possibility. Accelerated radiation protocols are not generally indicated aside from tumors with the ability to rapidly repopulate, but the general low toxicity profile of toceranib in this study after radiation suggests a possible role as follow-up to other radiation protocols in tumors sensitive to toceranib.
Conclusions
The majority of cats with oral SCCs did not seem to benefit from follow-up toceranib after radiation; however, toceranib administration should be considered as a treatment option following radiation therapy for feline lingual SCCs. Standardized studies are needed to better elucidate the mechanism underlying the lack of benefits and to identify which patients might benefit from adjuvant therapies such as toceranib.
Footnotes
Acknowledgements
The authors would like to thank Kasey Burton for her phenomenal care for over 70% of the patients in the study and Caroline Hohlman for managing multiple patients in this study. The authors would also like to thank Washington State University for their support in providing the statistical software used.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
