Abstract
Objectives
The aim of this study was to test the hypothesis that intranasally administered maropitant citrate would reduce the severity of clinical signs of feline upper respiratory disease (FURD) in shelter cats with naturally acquired disease.
Methods
Shelter cats with clinical signs of FURD were randomly assigned to receive either intranasal maropitant diluted in saline (maropitant citrate 10 mg/ml q12h, diluted 1:10 with sterile 0.9% saline) or intranasal 0.9% saline q12h for 7 days. Clinical disease severity was measured at entry into the study and again after completion of 7 days of treatment using a visual analogue scale to assess four separate clinical signs: conjunctivitis, blepharospasm, ocular discharge and nasal discharge. Total disease severity was also calculated. Cats received other medications for FURD as per standard shelter protocols, and all investigators were masked to group assignments. A Mann–Whitney U-test was performed to compare the clinical improvement score (CIS) between the treatment and control groups.
Results
There were 34 cats in the maropitant treatment group; 27 cats served as placebo controls. Groups did not differ in age, sex distribution, nature of disease, administration of other medications for FURD or baseline clinical disease severity. There was no significant difference in CIS between the maropitant treatment and control groups for conjunctivitis, blepharospasm, ocular discharge, nasal discharge or total disease severity after 7 days.
Conclusions and relevance
This study found no significant difference in outcomes for cats with FURD when treated with intranasal maropitant compared with treatment with intranasal saline. Further investigations would be required before intranasal maropitant could be recommended as the standard of care for FURD.
Plain language summary
Feline upper respiratory disease remains a significant challenge, impacting on shelter and owned cat populations with potential repercussions for adoption rates and housing costs. Given the demanding nature of caring for these cats, any additional intervention should offer a clear and substantial rationale. Our study on the clinical efficacy of intranasal maropitant 0.1% solution in the control of clinical signs in shelter cats with naturally acquired upper respiratory tract disease did not demonstrate any directly observable clinical benefits. Although we acknowledge the study’s limitations, including sample size and non-standardisation of additional medical therapy, we found no evidence to support the routine use of intranasal maropitant in managing feline upper respiratory disease. Further research with both timed and extended observation periods is warranted to elucidate any potential benefits of maropitant in this context.
Introduction
Feline upper respiratory disease (FURD) is common in cats, and morbidity is particularly high in kittens housed in stressful or overcrowded conditions. 1 Clinical signs associated with infection vary in severity but can include sneezing, nasal and ocular discharge, blepharospasm, conjunctivitis, reduced appetite, lethargy and fever. Pathogens involved in FURD include feline herpesvirus (FHV-1), feline calicivirus (FCV), Bordetella bronchiseptica, Chlamydia felis and Mycoplasma species,2 –7 although diagnosis is commonly based on clinical signs.
The management of upper respiratory tract infections in cats and kittens has traditionally focused on supportive care and treatment of secondary bacterial infections. 8 However, anecdotal and web-based reports suggest that intranasal or oral maropitant can be commonly employed to control clinical signs,9,10 despite a lack of evidence for its effectiveness.
Maropitant is a selective neurokinin-1 (NK1) receptor antagonist drug that acts in the central nervous system by inhibiting substance P (SP). Maropitant was developed and marketed to veterinarians as a pharmaceutical means of preventing and treating nausea and vomiting of multiple causes,11,12 for which its efficacy and safety are well established. 13 As well as its antiemetic properties, maropitant has also been found to reduce the requirement for anaesthetic gas during ovariectomy in dogs and cats.14,15 Claims have been made that it has an analgesic effect in cases of visceral pain.14 –16
The pharmacology of maropitant suggests that it could play a role in the reduction of airway inflammation associated with NK1 receptor activation. Several studies have shown positive results in the treatment of rhinitis and allergic upper airway disease in guinea pigs,17 –21 rats 22 and ferrets.23,24 The hypothesis that maropitant could be a useful adjunct therapy for cats with FURD presumably stems from the known distribution of NK1 receptor sites coupled with data from non-feline experimental trial data investigating various NK1 receptor antagonists, including maropitant. The aim of this study was to assess the clinical efficacy of intranasal maropitant as an adjunct therapy for the control of clinical signs in cats with naturally acquired upper respiratory tract disease.
Materials and methods
Ethical considerations
Approval for this study was obtained from the Monash Animal Research Platform Animal Ethics Committee (MARP 3, #2023-37585-91415). In addition, approval for the use of shelter cats in this study was obtained from the Animal Welfare Policy Committee of the Royal Society for the Prevention of Cruelty to Animals (RSPCA) in Victoria, Australia.
Animals
Cats and kittens weighing more than 0.6 kg with clinical signs of FURD (conjunctivitis, blepharospasm, ocular discharge and nasal discharge) were prospectively enrolled into this study between 1 June 2023 and 31 August 2023 from the population of stray and surrendered cats and kittens within a large suburban animal shelter in Melbourne, Australia. Kittens or cats with signs of disease on admission to the shelter were assigned the status of active FURD, while those that developed signs during the initial admission period (3–5 days) were labelled emergent for purposes of comparison. Age, sex, and neutering status were obtained from medical records compiled by shelter personnel at admission. All cats had a physical examination and weight measurement performed by shelter staff, although temperature, pulse and respiration were not specifically recorded in cats that appeared bright and alert apart from upper respiratory signs. Concurrent health conditions (eg, endo- or ectoparasitism, feline leukaemia virus [FeLV] and feline immunodeficiency virus [FIV] status or presence of diarrhoea) did not exclude cats from being enrolled into the study except in cases where hospitalisation was required. Cats were rejected from the study if they were insufficiently socialised to safely allow twice-daily intranasal therapy. In line with standard shelter protocols and guidelines from the International Society for Companion Animal Infectious Diseases, 8 cats with clinical signs of FURD were treated with medications prescribed at the discretion of the examining veterinarian, including doxycycline (5 mg/kg PO q12h for 7–10 days), 1% chloramphenicol eye ointment applied topically q8–12h and famciclovir (90 mg/kg PO q12h). These medications were started on the day of scoring concurrently with intranasal therapy.
Study design
At study entry (day 0), all cats and kittens were examined by 1/8 shelter veterinarians who had been familiarised with the study design and the scoring system for FURD signs. Animals were randomly assigned to either the control or maropitant treatment groups. Randomisation was achieved using Microsoft Excel for the generation of a list of random 1s and 0s, placed alongside a list of positive integers from 1 to 100. Cats were then assigned in order of enrolment to the group indicated alongside their arrival number.
A veterinarian not associated with the trial drew up 2.0 ml doses of either sterile 0.9% saline or injectable maropitant citrate (Cerenia, 10 mg/ml; Zoetis) diluted 1:10 in 0.9% sterile saline (maropitant 0.1% solution). These 2.0 ml syringes were labelled either Maropitant Trial ONE (ie, the placebo group), which was saline only, or Maropitant Trial ZERO (ie, the treatment group), which was maropitant in saline. Each cat was allocated a single syringe throughout the study.
Cats in the control group received routine FURD treatment plus one drop of 0.9% saline in each nostril twice daily for 7 days; cats in the maropitant treatment group received routine FURD treatment plus one drop of maropitant (0.1% solution) in each nostril twice daily for 7 days. Using the assumption that one drop is equivalent to 0.05 ml, this equates to a total daily dose of 0.2 mg maropitant per cat for cats in the treatment group. For the duration of the trial, investigators were blinded as to which syringes contained treatment (maropitant 0.1%) and which were control (saline) syringes.
On day 0, each cat/kitten was examined by a staff veterinarian and given a score for four FURD parameters, using a visual analogue scale. The parameters assessed were conjunctivitis, ocular discharge, nasal discharge and blepharospasm, scored 0, 1, 2, 3 or 4. The scoring system is summarised in Table 1.6,25,26 The minimum total clinical disease score possible was 1 and the maximum was 13.
FURD scoring chart used in a study on the effects of intranasal maropitant in shelter cats with naturally acquired FURD

FURD = feline upper respiratory disease; N/A = not applicable
For the duration of the 7 days of the trial, each cat was treated with either intranasal maropitant or intranasal saline at the rate of one drop into each nostril every 12 h, in addition to other FURD treatment prescribed by the attending veterinarian. The intention was to test the efficacy of intranasal maropitant as an adjunct therapy for FURD, which is how it is typically given, rather than as a sole treatment. On day 7, cats were again examined by a shelter veterinarian and scores were assigned for each of the four measured parameters.
Statistical analysis
Categorical variables are presented as numbers and percentages. Parametric data are presented as mean ± SD, whereas non-parametric variables are presented as median (range). For each clinical parameter, a clinical improvement score (CIS) was calculated by subtracting the day 7 score from the baseline day 0 score. A score difference of 0 indicated no improvement, a positive value (>0) signified improvement and a negative value (<0) indicated deterioration. Total disease severity on day 0 was calculated for each cat by summing scores assigned for each clinical parameter (conjunctivitis, blepharospasm, ocular discharge, nasal discharge). Age, baseline CIS, total disease severity score and change in CIS were compared between groups using the Student’s t-test for parametric data and Mann–Whitney U-test for non-parametric data. The total number of clinical signs per cat was calculated and group comparisons were made (Prism version 10.2.0; GraphPad).
The number of cats receiving additional medication was compared between the maropitant treatment and control groups using Fisher’s exact test. The number of cats considered actively vs emergently affected was compared in a similar fashion.
Based on statistical analyses from previous studies,25,26 the number of cats needed to assess a treatment that could improve clinical signs would be between 55 and 78 to achieve a power of 0.80 with a 0.05 probability of a type I error.
Results
Animals
Between 1 June 2023 and 31 August 2023, 529 cats and kittens were admitted to the shelter. Shelter veterinarians were asked to enrol animals into the trial based on the presence of one or more of four clinical signs of FURD (conjunctivitis, blepharospasm, ocular discharge and nasal discharge). Of the 61 cats that completed 7 days of intranasal therapy, 34 (56%) were randomly assigned to the maropitant treatment group and 27 (44%) to the control group. Most cats were classified as domestic shorthairs (43/61, 70.5%) or domestic mediumhairs (8/61, 13%). Of the remaining cats, there were six (10%) Ragdolls, two Birmans, one Burmese and one Persian cat. There were 33 females (27 spayed) and 28 males (four castrated). Age was not available for one cat in the maropitant treatment group. Of the remaining 33 cats, 10 (29%) were aged less than 1 year, compared with 12/27 (44%) in the control group. There were no significant differences in age or sex distribution between the two study groups (Table 2). In the placebo group, 11/27 cats were considered to have emergent disease, while 17/34 maropitant-treated cats were emergent.
Age and sex distribution of cats treatment and control groups in a study on the effects of intranasal maropitant in shelter cats with naturally acquired feline upper respiratory disease

Data are n (%) or median (range)
None of the cats was positive for FeLV and one cat in each group was positive for FIV. Standard therapy for FURD included doxycycline in 29/34 maropitant-treated cats and 21/27 placebo cats (P = 1.0), ocular chloramphenicol ointment in 12/34 maropitant-treated cats and 6/27 placebo cats (P = 0.40), and oral famciclovir in 17/34 maropitant-treated cats and 11/27 placebo cats (P = 0.12).
Clinical parameters
The number of clinical signs per cat at study entry was similar between the groups (Table 3). For each of the cats enrolled into the study, the severity of disease on day 0 varied from mild to marked. Total disease severity score at study entry did not differ between maropitant-treated cats (median 4, range 1–8) and placebo controls (median 3, range 1–7; P = 0.20).
Number of clinical signs per cat and total disease severity score at study entry in a study on the effects of intranasal maropitant in shelter cats with naturally acquired feline upper respiratory disease

Data are median (range)
Clinical improvement score
Boxplots displaying CIS data for conjunctivitis, blepharospasm, ocular discharge and nasal discharge are presented in Figures 1–4. In each figure, the box represents the interquartile range between the first quartile (Q1) at the bottom of the box and the third quartile (Q3) at the top, with the median (Q2) in between. The ‘whiskers’ extending up and down from each box indicate the minimum and maximum data points. There were no statistically significant differences in CIS between the control and treatment groups for conjunctivitis (P = 0.92), blepharospasm (P = 0.39), ocular discharge (P = 0.55) and nasal discharge (P = 0.98). Similarly, total CIS for disease severity did not differ between control (median 3, range −1 to +7) and treatment (median 2, range −1 to +6) groups (P = 0.32).
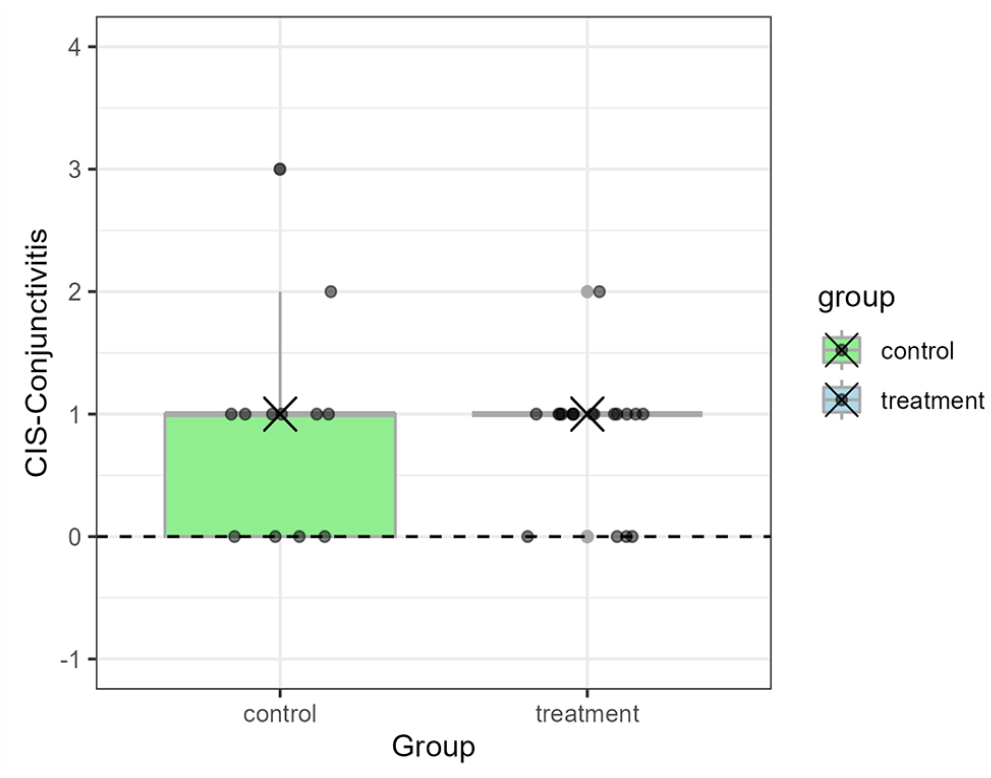
Boxplot displaying the CIS of cats with conjunctivitis after 7 days of treatment between the control and treatment groups in a study on the effects of intranasal maropitant in shelter cats with naturally acquired upper respiratory disease. The small circles denote the CIS of individual cats whereas the cross denotes the median CIS for each group. Conjunctivitis was scored as no (0), mild (1), moderate (2) or severe (3) conjunctival redness. CIS was calculated as day 7 score – day 0 score. CIS = clinical improvement score; FURD = feline upper respiratory disease

Boxplot displaying the CIS of cats with blepharospasm after 7 days of treatment between the control and treatment groups in a study on the effects of intranasal maropitant in shelter cats with naturally acquired upper respiratory disease. The small circles denote the CIS of individual cats whereas the cross denotes the median CIS for each group. Blepharospasm was scored as follows: 0 = eyes open; 1 = eyes <25% closed; 2 = eyes 25–50% closed; 3 = eyes 50–75% closed; 4 = eyes completely closed. CIS = clinical improvement score; FURD = feline upper respiratory disease

Boxplot displaying the CIS of cats with ocular discharge after 7 days of treatment between the control and treatment groups in a study on the effects of intranasal maropitant in shelter cats with naturally acquired upper respiratory disease. The small circles denote the CIS of individual cats whereas the cross denotes the median CIS for each group. Ocular discharge was scored as follows: 0 = no discharge; 1 = minor watery discharge; 2 = moderate mucous discharge; 3 = marked green-yellow discharge. CIS = clinical improvement score; FURD = feline upper respiratory disease

Boxplot displaying the CIS of cats with nasal discharge after 7 days of treatment between the control and treatment groups in a study on the effects of intranasal maropitant in shelter cats with naturally acquired upper respiratory disease. The small circles denote the CIS of individual cats whereas the cross denotes the median CIS for each group. Nasal discharge was scored as follows: 0 = no discharge; 1 = minor watery discharge; 2 = moderate mucous discharge; 3 = marked green-yellow discharge. CIS = clinical improvement score; FURD = feline upper respiratory disease
Discussion
To the authors’ knowledge, this is the first prospective placebo-controlled study to evaluate the utility of intranasal maropitant in cats with upper respiratory tract disease. Our study investigated whether intranasal maropitant provided any additional benefits in the treatment of FURD beyond that achieved using a standard shelter protocol of doxycycline with or without famciclovir or ocular medication. Findings indicated that, after 7 days, intranasal maropitant treatment showed no significant difference in the reduction of clinical signs compared with an intranasal saline control. Although it is possible that the maropitant treatment group experienced a more rapid reduction in FURD scores, this study was not designed to test that hypothesis.
Maropitant has several properties that make it attractive for consideration in the management of infectious or inflammatory upper respiratory tract disease. SP and the NK1 receptor are closely associated with the cell membranes of sensory neurons found within the gastrointestinal, urinary and respiratory tracts.27,28 Activation of these NK1 receptor sites triggers vasodilation and neurogenic inflammation and, given the abundant presence of NK1 receptor sites in the respiratory tract, it has been suggested that SP may contribute to clinical signs of respiratory diseases. As a result, maropitant, as an NK1 receptor antagonist, might play a role in preventing or alleviating respiratory signs. Several studies have shown positive results in the treatment of rhinitis and allergic upper airway disease in laboratory animals such as rats and guinea pigs.20,29,30 Evangelista et al 20 found a marked increase in vascular permeability of the upper airways of the guinea pig with systemic administration of SP and reported that pre-emptive intranasal administration of an NK1 receptor antagonist, FK888, completely abolished the SP response. Rahman et al 30 found that topical administration of SP to rat nostrils resulted in a dose-dependent increase in sneezing and nasal rubbing, and that intravenous administration of an NK1 receptor antagonist (L-732,138) 5 mins before SP administration significantly reduced the subsequent sneezing and nasal rubbing response. However, these studies were limited to laboratory, disease-induced models and treatment before the insult rather than clinical, naturally acquired disease in cats. Our study does not support the clinical use of intranasal maropitant in the setting of FURD despite anecdotal reports of efficacy.
Other studies have investigated the role of maropitant in lower respiratory diseases, including in an experimental model of feline asthma. Neither acute nor chronic administration of maropitant relieved clinical signs of airflow limitation or airway eosinophilia in cats with experimentally induced feline asthma,31,32 and no studies have been performed on the naturally occurring disease. Given that life-threatening bronchoconstriction can occur in naturally occurring feline asthma, ineffective therapy could be associated with substantial morbidity and even mortality. Therefore, the use of maropitant cannot be recommended for lower respiratory tract disease, and our study indicates that it is not effective in controlling signs of acute upper respiratory tract disease in cats and kittens. The pattern that emerges from the foregoing is that NK1 receptor antagonists might show promise in experimental and in vitro settings. However, this potential has not translated into clinical success in feline respiratory tract disease.
The dose of maropitant used in this study (one drop of a 10% dilution of the injectable drug administered to each nostril every 12 h) amounts to 0.05 mg per drop, and therefore 0.2 mg maropitant per cat per day. This dose was chosen as it is most commonly utilised based on anecdotal and web-based reports.9,10 This dose is much lower than the prescribed systemic dose of 1.0 mg/kg as described in the accompanying manufacturer’s literature, and no studies have been conducted to address intranasal absorption in healthy cats, let alone absorption or activity in the presence of mucoid or infected secretions. Other topical agents, such as gentamicin, have poor penetration into mucus and are ineffective in an acidic environment such as that associated with infection. 33 Therefore, further investigation into the specific properties of topical maropitant is required before intranasal use can be recommended.
Costs involved in the use of maropitant are not insignificant, not only because of the base price of the drug but also because of drug compounding. In a shelter situation, considerable resources are involved in administering intranasal therapy, and the potential for worsened spread of disease through the use of communal medication vials always has to be considered. The stress of twice-daily handling could worsen the disease in certain cats. Finally, in a home environment, the possibility of injury to household members during administration must be considered.
One limitation of our study is the absence of daily scoring between days 0 and 7, which would have allowed us to assess the rate of response to therapy. This is an important consideration in a shelter situation when financial resources for housing must be considered, along with the adoptability of the animals. Future research could address this limitation by incorporating daily CISs and employing a randomised controlled design with a predefined therapy protocol. The heterogeneous nature of our study population was partly mitigated by enrolling a large number of cats and kittens into both groups, allowing control over variables such as age, sex, breed and disease severity so that baseline characteristics did not differ between groups. The recruitment of additional cats was limited by the planned length of the study within the shelter situation. In addition, other shelter populations could have different pathogens involved in the disease process, which could allow a therapeutic response. We did not specifically test for pathogens involved owing to the difficulty in interpreting molecular tests as well as the expense involved. Another limitation of the study is the lack of a dose escalation for maropitant. The dose of maropitant used in this study was recommended on veterinary-related websites, but other doses might be found to be efficacious. Finally, the non-standardisation of drug therapy presents a limitation of this study. Our study did not have an untreated group, as we considered this unethical in the shelter situation, 26 and an additional control group that did not have intranasal therapy administered might have added further information regarding this limitation. Regardless of these limitations, the effect of intranasal maropitant for FURD in our trial appeared to be either negligible or minimal.
Conclusions
FURD remains a significant challenge, impacting on shelter and owned cat populations with potential repercussions for adoption rates and housing costs. Given the demanding nature of caring for these cats, any additional intervention should offer a clear and substantial rationale. Our study on the clinical efficacy of intranasal maropitant 0.1% solution in the control of clinical signs in shelter cats with naturally acquired upper respiratory tract disease did not demonstrate any directly observable clinical benefits. Although we acknowledge the study’s limitations, including the sample size and non-standardisation of additional medical therapy, we found no evidence to support the routine use of intranasal maropitant in managing FURD. Further research with both timed and extended observation periods is warranted to elucidate any potential benefits of maropitant in this context.
Footnotes
Acknowledgements
The authors gratefully acknowledge the assistance, advice and guidance of the veterinarians, veterinary nurses and animal attendants of the RSPCA Burwood shelter for graciously allowing us to add to their already substantial workload.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Publication costs were supported in part by the Bailey Wrigley Fund, University of California, Davis, USA.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
