Abstract

Dear Editors,
In 2017, Dr Dobromylskyj and colleagues published an article in JFMS concerning a novel nail bed lesion named giant cell reparative granuloma (GCRG) in cats. 1 This study provided valuable insights into a previously under-recognised condition affecting the feline nail bed, contributing significantly to the understanding and diagnosis of these lesions. It has been 7 years since the publication of this paper and, while it provides excellent insights into pathological diagnosis, it appears that no further reports have been published since. Although some time has passed, we are submitting this Letter to the Editor to report our findings.
We would like to highlight a case we recently encountered that may offer additional perspectives relevant to the findings discussed by Dobromylskyj et al. 1 Our case involved a 17-year-old spayed crossbred female cat that presented with swelling of the second digit of the left forelimb. The mass, approximately 1.5 cm in diameter, was firm, ulcerated and had been present for several weeks with occasional bleeding and discharge. The middle phalanx of the second digit of the left forelimb was resected and histopathological examination was performed (Figure 1a).
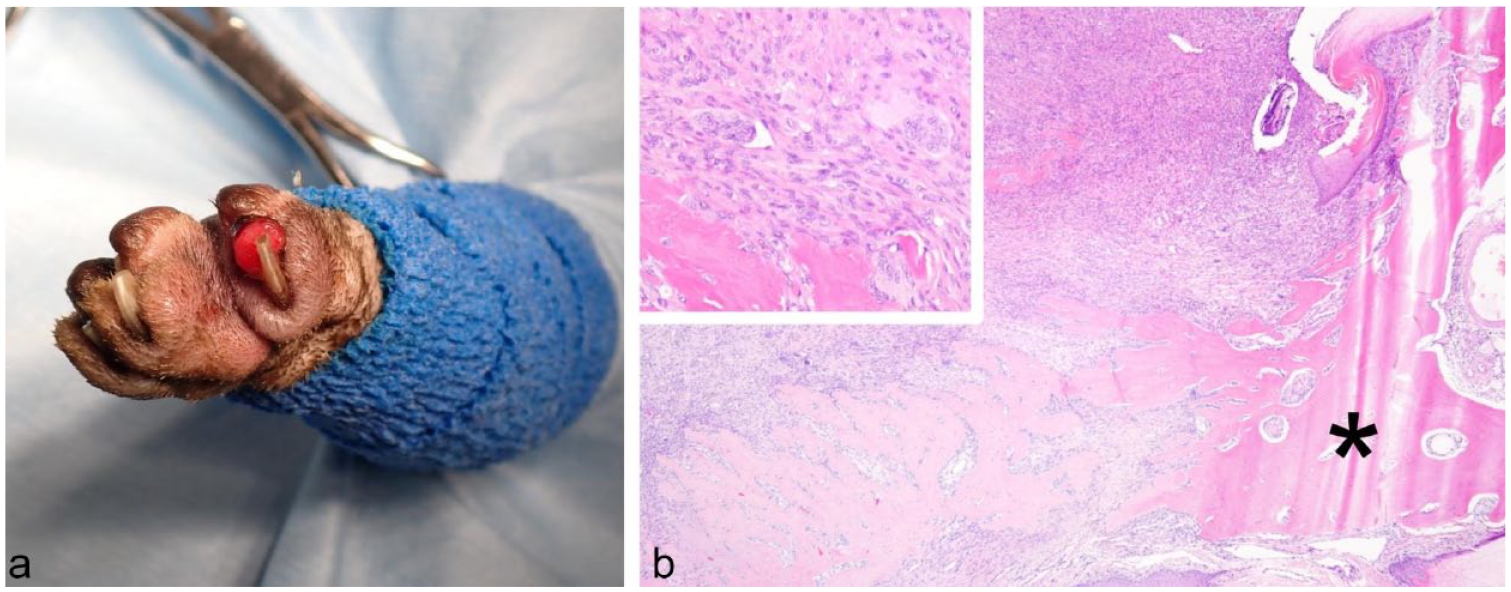
(a) Preoperative photograph showing the dorsal aspect of the second digit of the left forelimb where the mass is located. The mass is ulcerated and measures approximately 1.5 cm in diameter. (b) Low-power view (magnification: × 40) of the lesion in the second digit of the left forelimb. The distal phalanx (indicated by an asterisk) is largely preserved, although it is partially dissolved because of the proliferation of spindle-shaped cells. Inset: High-power view (magnification: × 200) of the lesion in the second digit of the left forelimb. The image shows proliferation of spindle-shaped cells with mild atypia, osteoid production and the presence of multinucleated giant cells. Hematoxylin and eosin staining
Histopathological examination revealed that the lesion was a GCRG, a rare benign entity that shares some overlapping features with lesions described by Dobromylskyj et al. 1 Although GCRG is typically benign and non-aggressive, the lesion in our case exhibited unusual bone involvement, raising differential diagnostic considerations, particularly for osteosarcoma.
Figure 1b shows a low-power view of the hematoxylin and eosin staining. The distal phalanx, indicated by an asterisk, is largely preserved, despite being partially dissolved by the proliferation of spindle-shaped cells. The inset shows a high-power view, where the proliferation of spindle-shaped cells with mild atypia, osteoid production and giant cells can be observed.
Proliferative spindle cells showed mild atypia and few mitotic figures. Immunostaining revealed that the Ki-67 positivity rate of proliferative cells was very low (2.1%). Despite bone involvement, the low proliferation index, mild cellular atypia and preserved structure of the distal phalanx supported the diagnosis of GCRG rather than a more aggressive neoplasm. This underscores the importance of careful histopathological evaluation and consideration of the differential diagnoses in similar cases. One month postoperatively, no recurrence or metastasis was observed.
We believe that including such cases with atypical presentations in future studies will further enhance our understanding of feline nail bed lesions and their potential variations. We commend the authors for their thorough and insightful work and look forward to continuing research in this area.
1Laboratory of Clinical Diagnostics, School of Veterinary Medicine, Azabu University, Sagamihara-shi, Kanagawa, Japan
2Veterinary Teaching Hospital, Azabu University, Sagamihara-shi, Kanagawa, Japan
3Laboratory of Veterinary Pathology, School of Veterinary Medicine, Azabu University, Sagamihara-shi, Kanagawa, Japan
4Laboratory of Small Animal Surgery, School of Veterinary Medicine, Azabu University, Sagamihara-shi, Kanagawa, Japan
The corresponding author responds:
Many thanks to the authors of this letter for sharing their interesting case, and to the Editors of JFMS for the opportunity to reply. Since publication of the original study in 2017, anecdotally I have received communications from pathologist colleagues worldwide expressing gratitude for undertaking the study into these apparently not uncommon but poorly described lesions affecting cats’ toes. This suggests to me that pathologists are regularly diagnosing these lesions and probably have been for some time. But anecdotal data such as these are notoriously difficult to document.
One issue we have with many veterinary studies is there are often few or no subsequent publications unless something significantly novel is discovered. Hence our initial findings are rarely further validated, and we consequently base our diagnostic and prognostic ‘rules of thumb’ on what are critically small studies (as well as any personal and anecdotal evidence we accumulate as our careers progress!). Therefore, any case reports such as this one, and future publications, are always to be warmly welcomed and we must support the reporting of such cases in our veterinary literature.
There is still so much that remains unknown about these lesions; for example, the aetiology and any contributory factors. I have several points that I feel we should consider carefully here. First, when researching these lesions, I found the classification of similar lesions in humans to be rather complex, and it was difficult to find an exact equivalent to our cat toe lesions. We decided on the name ‘giant cell reparative granuloma’ as the closest match, although we omitted to add terms such as ‘central’ or ‘peripheral’, which are often used in human lesions of this nature, because the cat toe lesions did not fit well into that part of the human classification system. Hence, it is difficult and potentially dangerous to extrapolate between species (and even between different anatomical sites in the same species) for these lesions in terms of their behaviour.
We should also remember that in biology, most lesions form a spectrum rather than discrete entities. Despite our (very human) attempts to ‘pigeon-hole’ every mass or lesion we see precisely into one category or another, unfortunately biology is not as neat as we would like it to be. Therefore, it is still possible that these masses form such a spectrum, and they may not all behave in the same fashion. Every case should be approached with an open mind. And we have not even considered whether there is potential for malignant transformation, something that cats appear to have a propensity for, particularly in the face of a chronic inflammatory process.
When diagnosing such masses, both the clinician and the pathologist must bear in mind the potential differential diagnoses. Very importantly, given the site, an osteosarcoma (multinucleate cell type), a nerve sheath tumour with osseous metaplasia and an undifferentiated pleomorphic sarcoma must be thoroughly excluded – from personal experience, I have diagnosed all of these entities at this site in cats.
In our previously published study, one of the key features we found included an absence of any underlying bone involvement both on radiography and histologically. Hence, the findings of the current case, which describes some bone involvement, need to be very carefully considered. Distinct bone lysis and destruction should be clearly differentiated from any periosteal reaction and remodelling of the pre-existing distal phalanx. I very much agree with the authors’ statement ‘this underscores the importance of careful histopathological evaluation and consideration of the differential diagnoses in similar cases’. The authors of the letter describe low mitotic activity and very low Ki-67 positivity as well as mild atypia in their case. The masses we reviewed in the 2017 study had a very variable mitotic count, sometimes surprisingly high for a non-neoplastic process, but no atypia, so clearly there is a range of possible features we might expect to see histologically. 1 An exophytic growth pattern, the presence of ulceration and of neutrophilic inflammation were also key and in some ways were more consistent features in the cases we reviewed in the 2017 study. 1 Thus, it is important to always consider all of these features together.
If clinicians are investigating such a lesion, detailed radiographic assessment is strongly advisable. If any bone involvement is seen, I would personally consider digital amputation and submission of the sample for histological assessment.
Finn Pathologists, Diss, Norfolk, UK
Footnotes
Acknowledgements
We thank Dr Yumiko Kagawa of North Lab for her valuable suggestions. The cat described in this work belonged to the corresponding author, Dr Kamiie. The owner is fully aware of and agrees with this case and is grateful to the cat.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
