Abstract
Case series summary
The efficacy of medical treatment associated with single myringotomy in cats with suppurative otitis media (OM) and intact tympanum was retrospectively evaluated. Patients’ records were retrieved from cats with suppurative OM. Cats were included in this retrospective study based on fluid content within the tympanic cavity on imaging, intact tympanum on otoendoscopic examination and neutrophils on ear bulla exudate cytological examination. A total of 26 cats with middle ear suppurative otitis were included. Just over half of the cats (54%) presented bilateral involvement. Clinical signs included head tilt (13/26), otalgia (9/26), Horner’s syndrome (7/26), external ear discharge (5/26), and nystagmus and facial paralysis (1/26). Cocci were identified on cytological examination in 18/40 samples and rods in 2/40. Bacterial culture results were positive in 15/40 samples, with Pseudomonas species (4/15), Pasteurella multocida (3/15), Staphylococcus felis (3/15), Staphylococcus schleiferi (2/15), Staphylococcus canis (2/15), Escherichia coli (2/15), Staphylococcus pseudintermedius (1/15) and Serratia marcesens (1/15) isolation. After myringotomy and gentle flushing of middle ear bullae (0.5–2 ml saline per ear), all cats were treated with oral corticosteroids and a 1-month course of systemic antibiotics according to sensitivity testing. In total, 19 (73%) cats were clinically healed 60–240 days after treatment. One cat was euthanased because of failure to eradicate a Pseudomonas species infection. Another cat died a few hours after the procedure. The five remaining cats were cured after ventral bulla osteotomy.
Relevance and novel information
OM without an aural polyp is a frequent diagnosis in cats, but data on medical treatment are scarce. This simple therapeutic technique, single myringotomy procedure and mild ear flushing, offers a practical, safe and efficient first-line treatment of suppurative OM with an intact tympanum in cats with or without neurological signs.
Introduction
Otitis media (OM) is frequently diagnosed in cats; however, data on medical treatment are scarce except for polyp-associated OM.1–6 The reported literature recommends numerous abundant ear flushing with antiseptic solution, antibiotics with or without corticosteroids infused once a week or more, flushing after bulla septum fenestration and ventral bulla osteotomy.7–12 One retrospective study suggested that non-polyp-related OM can be successfully managed medically using either topical products, systemic antimicrobials or a combination. 1 In accordance with this therapeutic approach, 26 cases of suppurative OM without polyps and with a closed tympanic membrane, confirmed through cross-sectional imaging (CT or MRI) and cytological examination, were retrospectively studied to assess the efficacy and safety of the procedure.
Case series description
Animals
Data were retrospectively collected from two different private referral centres in France using the same management procedures for OM (anaesthesia, imaging, myringotomy, treatment). The research was carried out between 2018 and 2020.
Retrospective information collected from the medical records included signalment, history, treatment before and after referral, neurological and dermatological examination reports, MRI or CT findings, and cytology and bacteriology results from the affected ears and outcomes.
The inclusion criteria were as follows: fluid accumulation within tympanic cavity on CT or MRI (Figures 1 and 2), intact tympanic membrane on both ears at otoendoscopic examination (Figures 3 and 4) and neutrophils on cytological examination of bulla exudate.
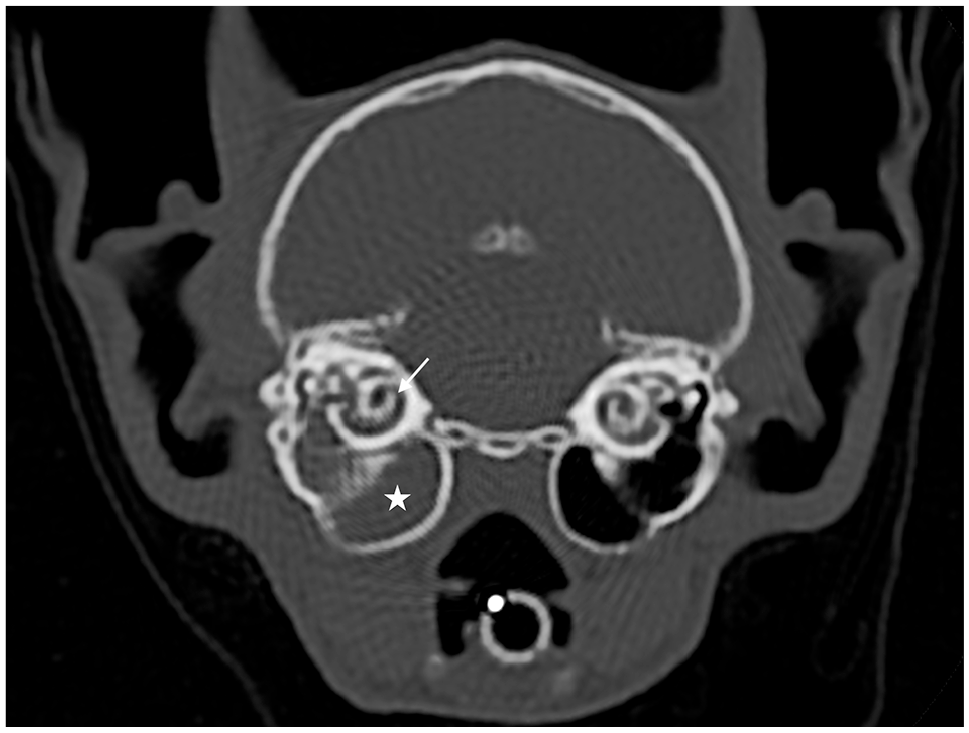
Transverse CT scanner of the head of a cat at the level of the tympanic bullae and cochleae obtained with a bone algorithm. The right tympanic bulla is fluid-filled (white star), with an intact wall. The bony structures of the cochlea are normal (white arrow)
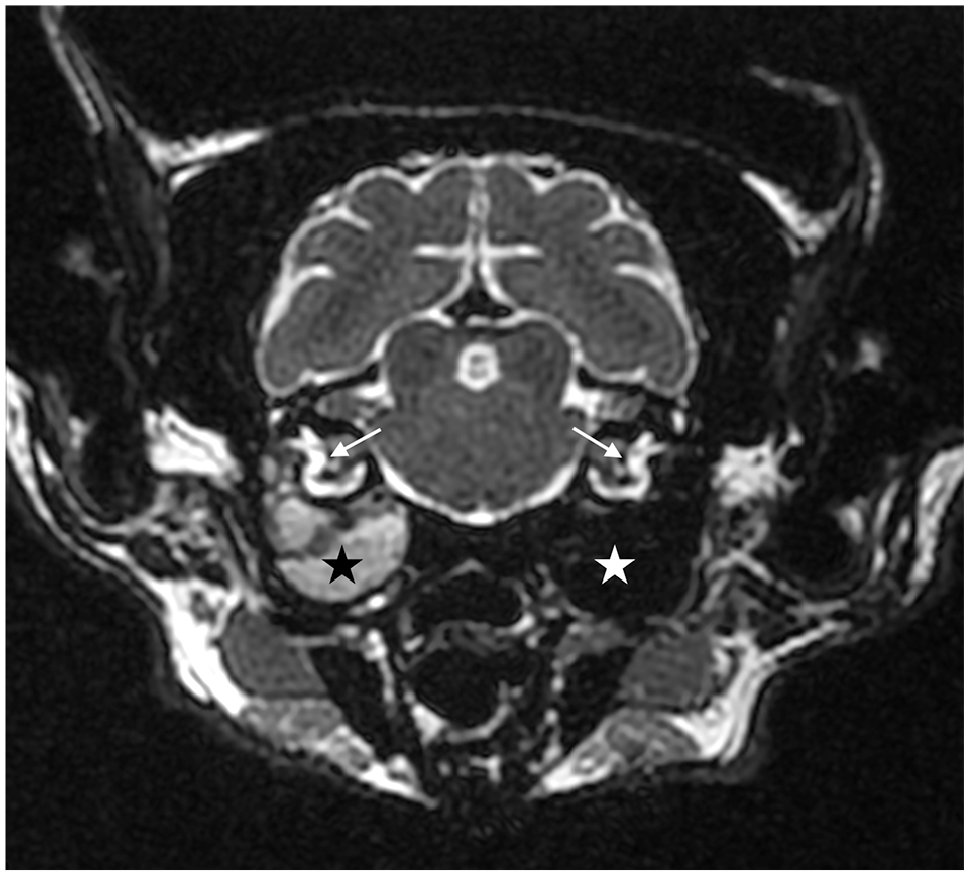
T2-weighted transverse MRI of the skull at the level of the tympanic bullae and cochleae. The right tympanic bulla is fluid-filled marked by a hyperintense signal on T2 (black star). The left tympanic bulla remains air-filled with a lack of signal (white star). The endolymph of the internal ears appears with a normal bright signal on T2 (white arrows)
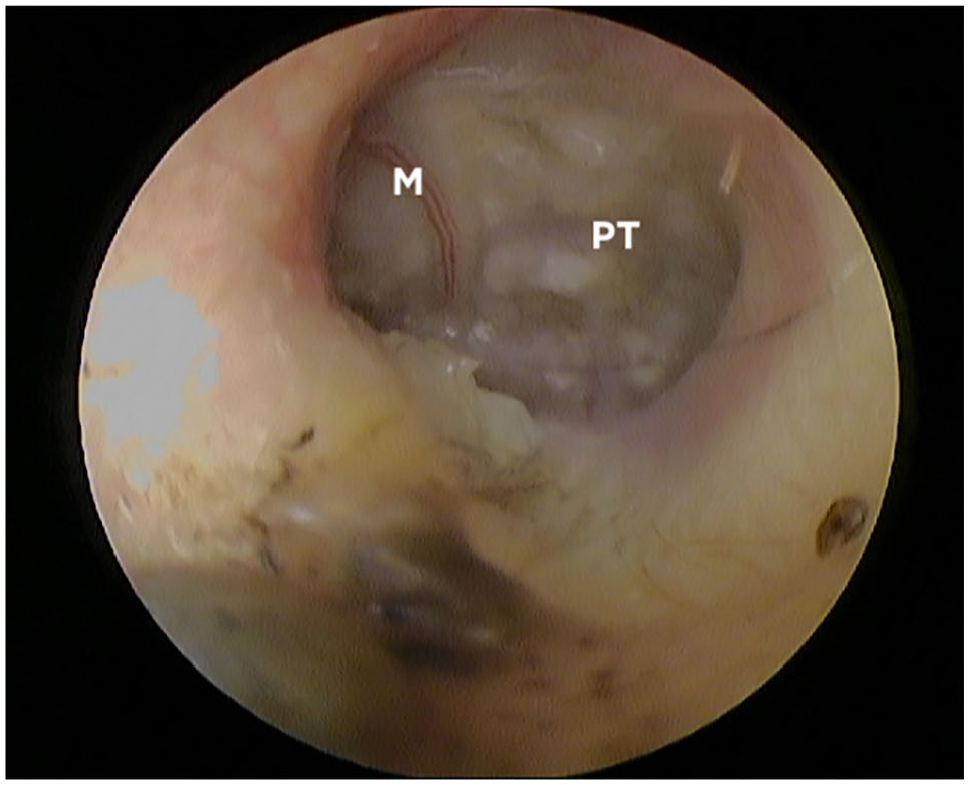
Otoendoscopic aspect of left tympanic membrane of a cat with suppurative OM. Heterogeneous pus is visible through the pars tensa. All cats with suspected or identified neoformations (ie, polyp, tumour) in the ear meatus or middle ear, tympanic perforation or non-inflammatory cytology were excluded. M = handle of malleus; PT = pars tensa
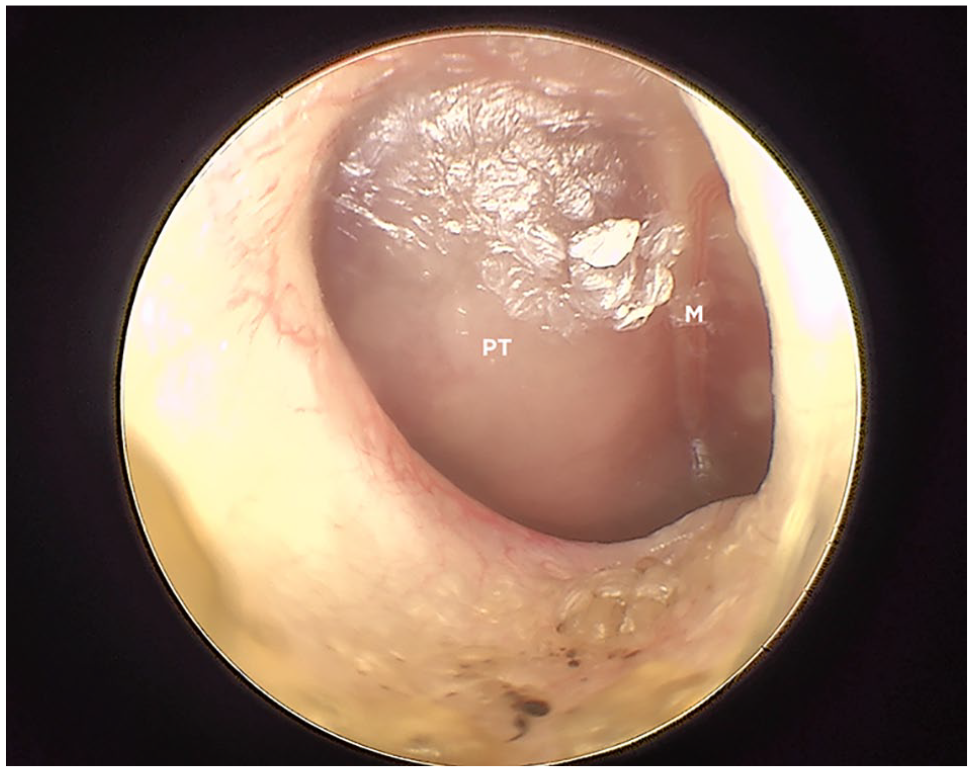
Bulging tympanic membrane of the right ear of a cat with suppurative otitis media. Exudate is visible through the membrane. M = handle of malleus; PT = pars tensa
Anaesthesia
During this period, anaesthesia for cats undergoing CT and MRI was performed using the following protocol: cats were sedated using an intramuscular (vastus lateralis) injection of midazolam (0.2–0.3 mg/kg, Midazolam 5 mg/ml; Viatris Santé), butorphanol (0.2–0.3 mg/kg, Torbugesic Vet 10 mg/ml; Zoetis) and dexmedetomidine (0.005–0.01 mg/kg 1 , Dexdomitor 0.5 mg/ml 1 ; Zoetis). Anaesthesia was induced by intravenous administration of propofol, titrated to effect (1–2 mg/kg 1 , Propovet 10 mg/ml; Zoetis). Maintenance was performed by an adjustable percentage of isoflurane in 95% oxygen diluted (Isoflurane-inspired concentration approximately 2%, Vetflurane; VIRBAC). Myringotomy practice was systematically followed by postoperative analgesia through a subcutaneous injection of buprenorphine (10–15 µg/kg SC, Bupaq; VIRBAC).
Myringotomy
If the proximal portion was obstructed by a ceruminous plug, the meatus was irrigated with peroxidised cerumenolytic solution (Otoprof; ICF). The plug was then extracted using sterile saline solution forced irrigation and the meatus was abundantly rinsed and then dried using suction.
A sterile, rigid polypropylene catheter with a diameter of 2 mm was cut at an angle of 60° with an incision blade providing a sharp point. This catheter tip was advanced under good visualisation using an otoendoscope and the caudal pars tensa was punctured (Figure 5).
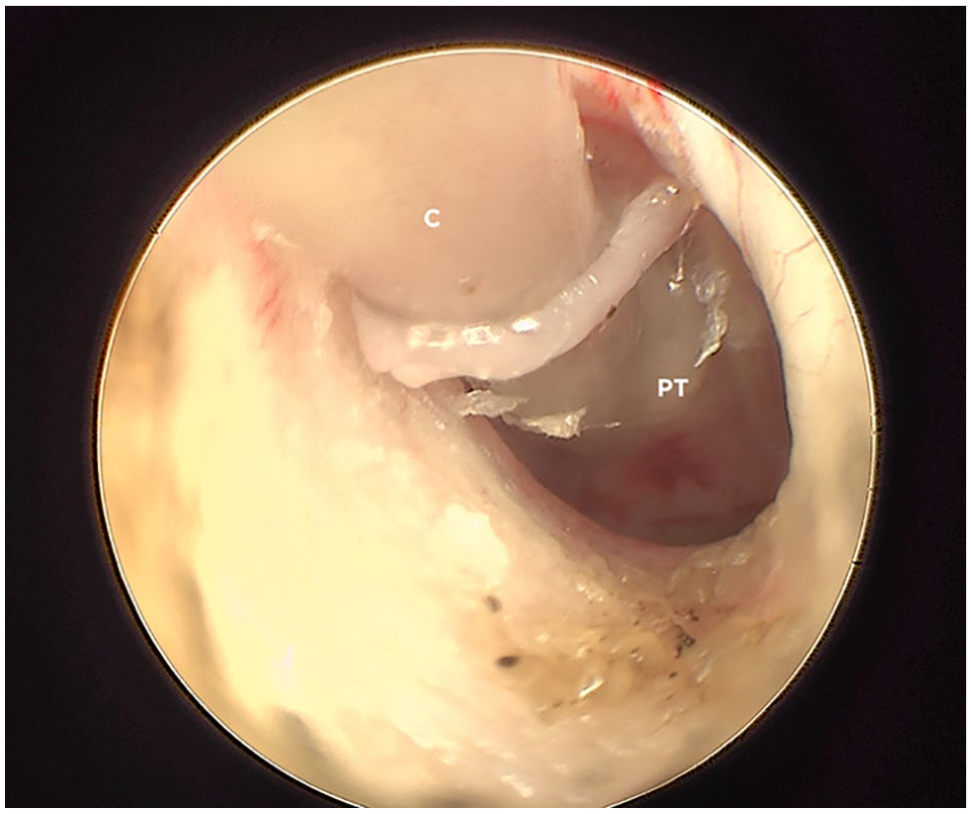
Myringotomy of the right ear with a rigid polypropylene catheter with a diameter of 2 mm. Aspiration of middle ear material. C = catheter; PT = pars tensa
Tympanic bulla contents were immediately gently aspirated. The catheter was removed and the contents were submitted for cytology, bacterial culture and antibiotic sensitivity.
The catheter was replaced in the tympanic cavity and 0.5–2.0 ml of sterile saline solution were injected and immediately aspirated.
Cytology
The sample was placed onto a slide glass and smeared with an additional slide, dried, fixed and stained using a modified Romanowsky-type rapid staining solution kit (RAL555, RAL).
Bacterial and fungal culture and sensitivity
Sterile swabs with liquid Amies culture medium were used and sent to the same laboratory under refrigerated conditions (Cerba Vet) for aerobic and anaerobic bacterial culture and fungal culture. Antibiotic susceptibility testing was performed by disk diffusion on Mueller-Hinton culture medium and Mueller-Hinton supplemented with 5% blood depending on the bacteria species.
Medical treatment
As a postoperative procedure, patients received dexamethasone disodium phosphate injection subcutaneously after the procedure (0.1 mg/kg, Dexadreson; MSD). Prednisolone was prescribed the next day (1 mg/kg PO q24h) for 7 days. Antibiotics were prescribed initially according to cytological bacteria morphology: amoxicillin/clavulanic acid 12.5 mg/kg PO q12h (Kesium; CEVA) for cocci or lack of bacteria and marbofloxacin 2 mg/kg PO q24h (Marbocyl; Vetoquinol) for rods. The prescription was then adapted according to bacterial culture and antibiotic sensitivity. Antibacterial therapy was maintained for 30 days.
Follow-up
The cats’ follow-up was carried out through physical examination for the first 3 months and later through telephone calls. Clinical examination included neurological examination and otoendoscopy (Figure 6). For economic reasons, CT or MRI were rarely performed at check-up visits.
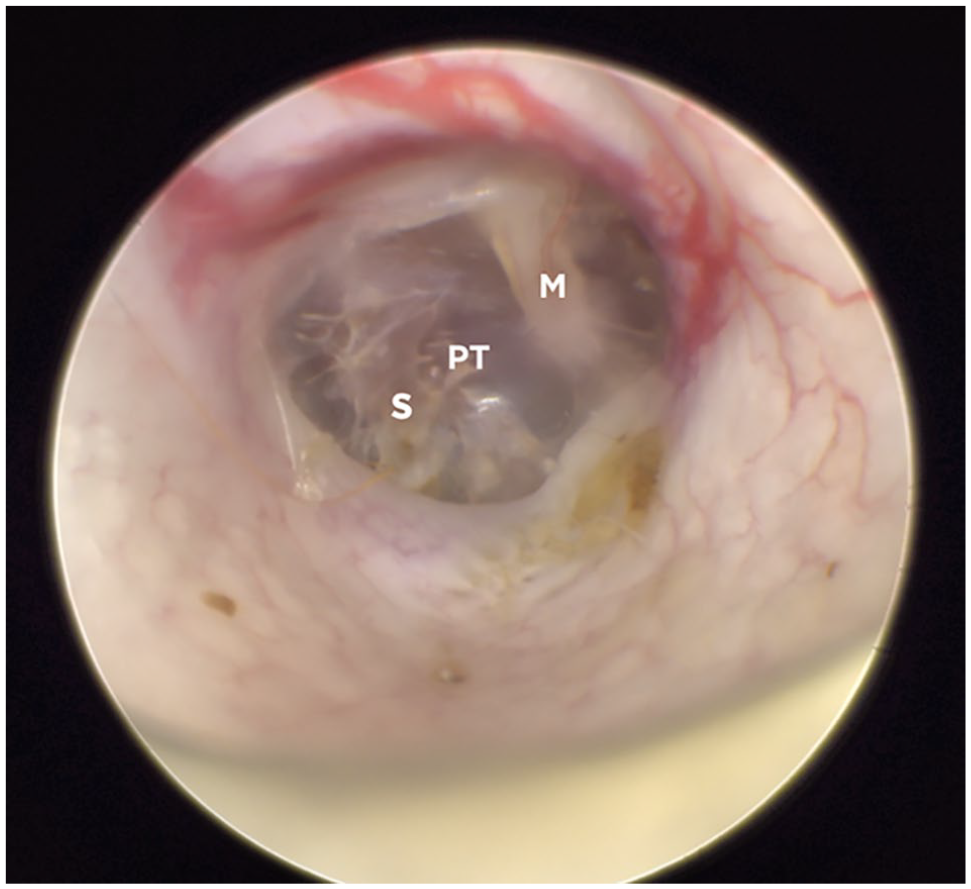
Otoendoscopic aspect of the tympanic membrane of the right ear of a cat 4 weeks after myringotomy. Large fibrotic scar on the caudal part of the pars tensa. M = handle of malleus; PT = pars tensa; S = scarce
Animals
A total of 26 cats met the inclusion criteria between 2017 and 2021 (54% had bilateral OM) (Table 1).
Signalment and clinical characteristics of all 26 cats
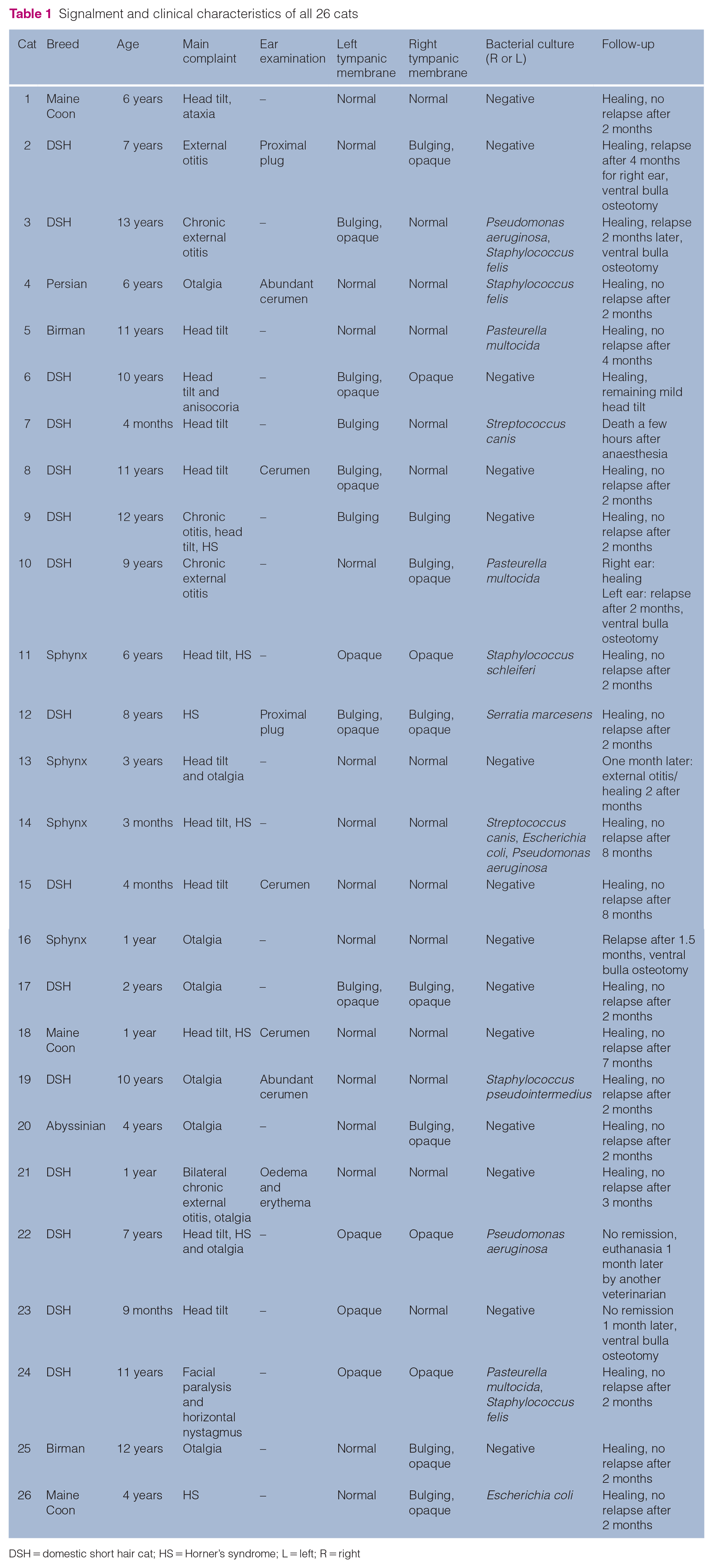
DSH = domestic short hair cat; HS = Horner’s syndrome; L = left; R = right
The median age of cats was 6 years (range 3 months to 13 years). There were 14 females (three intact, 11 spayed) and 12 males (three intact, nine castrated; male:female ratio = 0.86).
The most common breed of cat was domestic shorthair (15/26, 57%), followed by Sphynx (n = 4), Maine Coon (n = 3), Birman (n = 2), Persian (n = 1) and Abyssinian (n = 1).
Clinical signs
The most commonly presented signs were neurological signs (15/26), otalgia (9/26) and external ear discharge (5/26). Dermatological examinations were otherwise normal in all cats.
Neurological signs included head tilt (13/26) (Figure 7), Horner’s syndrome (HS; 7/26) and facial paralysis with horizontal nystagmus (1/26). Clinical examinations confirmed peripheral vestibular syndrome in all cases of head tilt and HS.
Right head tilt of cat with otitis media/interna
Otalgia was identified on palpation of the submandibular area in 7/26 cats. Erythematous otitis externa was observed in 4/26 cats and proximal ceruminous ear plugs without external otitis in another 4/26 cats.
Imaging
Out of 26 cats, 22 (85%) had a CT examination. Fluid attenuation within the tympanic cavity was observed in all cases. Irregular periosteal proliferation was noticed in six cases and tympanic bulla partial lysis in four cases. Four patients had an MRI examination rather than a CT scan, of which 100% showed enhanced material in the bullae. No cat had evidence of otitis interna nor meningitis.
In total, 14 cats showed bilateral OM. Both tympanic bullae compartments, on each side of the bony septum, were affected in almost all cases (92.5%).
Otoendoscopy
Out of 40 affected ears, only 23 (57%) showed evidence of post-tympanic exudate: both bulging and opaque (12/40), opaque (10/40) and bulging (1/40) tympanic membranes (Figures 3 and 4).
Cytology
Microscopic cytological evaluation identified degenerated neutrophils on diagnostic samples (n = 40) with activated macrophages in 13/40 samples. Cocci were identified in 18/40 (45%) smears and rods in 2/40 (5%) smears (Table 2).
Correlation between cytology and bacterial culture

Bacterial culture
Bacterial culture was positive in 15/40 (37%) ear samples, with Pseudomonas species (4/15), Pasteurella multocida (3/15), Staphylococcus felis (3/15), Staphylococcus schleiferi (2/15), Streptococcus canis (2/15), Escherichia coli (2/15), Staphylococcus pseudintermedius (1/15) and Serratia marcesens (1/15) isolation. Of the 15 positive samples, a single bacteria species was isolated in 87% of cases and a combination of bacteria in 13% of cases. No resistance to initially prescribed antibiotics was noticed. All strains were sensitive to common antibiotics that are usually effective on each species, including Pseudomonas species and Serratia species, both sensitive only to fluoroquinolones.
Treatment
All 26 cats were treated with antibiotics and prednisolone (7 days). Clavulanate-potentiated amoxicillin (13–25 mg/kg PO q12h) for 1 month was used in 24/26 cats. The remaining two cats were treated with marbofloxacin (2–2.5 mg/kg PO q24h) for 1 month for Pseudomonas species and Serratia species infections.
Outcomes
In total, 19 (73%) cats were clinically healthy 60–240 days after the start of treatment, with full healing of the tympanic membrane at the 4-week checkup. One of the 19 cats still had an attenuated head tilt despite apparent healing from the ear infection several months after the procedure. One cat was euthanased at the request of the owners because of difficulties in controlling the Pseudomonas species infection. A second cat died at home a few hours after anaesthesia without any clinical signs nor determined cause. The five remaining cats were successfully treated with a ventral bulla osteotomy.
Discussion
The main clinical signs of suppurative OM with an intact tympanum were neurological: head tilt and HS. Pain was reported in one-quarter of these cats and was the second chief complaint. This observation is comparable with those reported in other forms of OM in cats (non-intact tympanic membrane, ear polyps).11,13
Compared with studies of OM in cats, otitis externa is much less frequent (10% vs 47%). 14 This is probably due to the inclusion of cats with an intact tympanum. Most cases of suppurative otitis externa in cats that are associated with suppurative OM have a tympanic rupture or ear mass and rarely result from a ‘descending’ infection. 11
To diagnose OM in cats, CT or MRI are considered the gold standard;9,15 however, it is necessary to confirm the inflammatory and infectious nature of the liquid using a myringotomy and cytological examination.
The high magnification offered using otoendoscopy can help identify the signs of OM: an opaque or bulging tympanic membrane; and fluid and air bubbles seen behind the intact tympanic membrane.7,8 However, the sensitivity of otoendoscopy in this study was mild (57%); therefore, a lack of tympanic membrane alterations cannot exclude a diagnosis of OM.
It was not possible to obtain a positive bacterial culture despite the suppurative nature of the exudate in 25/40 (62.5%) samples studied. These data can be compared with the results of a study of 282 ventral bulla osteotomies in which the percentage of negative bacterial cultures was observed (65.2%). 16 This could partly be due to previous anti-infective treatment or sample collection, which can minimise anaerobic bacterial isolation.
Isolation of bacteria on cytology was also poorly sensitive (50%). Such low sensitivity could be due to the protection of bacteria by mucus. 8 This can also explain discrepancies between cytology and bacterial culture results.
Lack of sensitivity of both techniques could also reflect the implication of other microorganisms such as mycoplasma.8,17 Molecular methods, such as next-generation sequencing (NGS), could enhance the sensitivity of bacterial identification in ear effusion.
In most studies of feline OM, the bacteria involved were quite variable (Staphylococcus species, Streptococcus species, Pasteurella species, Pseudomonas species and E coli).1,14,16
Malassezia species yeasts were not isolated on cytology. This could be due to the selection of OM without tympanic rupture limiting middle ear infections by Malassezia species from the external ear canal. This is not surprising, as these yeasts require a lipid substrate that is not present in the middle ear. 18 It is only during tympanic rupture that colonisation of the middle ear by Malassezia species can be observed.
Our study confirms previous observations on the efficacy of treatment based on systemic antibiotics.1,14
This therapeutic approach, associating myringotomy, systemic antibiotics and corticosteroids, is much easier, less invasive and more comfortable than multiple ear flushing with antibiotics or ventral bulla osteotomy.7–12,16 It limits the risk of ototoxicity from solutions being dropped into the tympanic cavity and discomfort due to liquid present in the bulla. 19 It also limits the risk of acquired drug resistance from the frequent use of antibiotics.
Myringotomy with a large catheter (2 mm diameter) was well tolerated and tympanic healing was fast (less than 4 weeks) in all cases. Because the recheck was carried out at week 4, it was impossible to assess the true speed of healing of the tympanic membrane.
The role of a myringotomy (indispensable for diagnosis) in the healing of OM is difficult to evaluate, but it at least allows a reduction in pressure within the middle ear. This can contribute to rapid improvement in neurological signs and the comfort of cats. It also provides the drainage of fluids for several days. This is why, despite the lack of direct access to the medial compartment, a myringotomy should always be recommended when neurological signs are present.
The use of systemic steroids is debatable as well as the dose and duration. Further studies are needed in cats with OM, with and without neurological signs. In human medicine, guidelines for acute OM treatment do not mention the use of steroids20–22 and their addition is not beneficial. 23 Treatment is limited to short-term (11–14 days) empirical systemic antibiotics.20,24
However, the therapeutic approach varies significantly between cats and humans. In the case of humans, the observation of tympanic lesions during otoscopic examination is the main diagnostic element. Thus, cross-sectional imaging and myringotomy are seldom employed in these cases, unlike in cats. In cats, by performing a myringotomy, we can both promptly alleviate pain and prescribe targeted antibiotic therapy.
Conclusions
A single myringotomy combined with medical treatment successfully treats the majority of cases of closed, suppurative OM in cats. Further studies are necessary to assess the need for systemic antibiotic therapy or the duration of corticosteroid treatment.
Footnotes
Acknowledgements
The authors thank Dr Mitchell Song and Dr Aurore Laprais for their reviewing the English language, Dr Yannick Ruel for the CT and MRI photos and interpretation, Dr Kristina Museux (Cerba Vet) for bacterial culture technical details, Dr Kirsten Gnirs, Dr Stella Papageorgiou and Dr Léa Arti for neurological examination, Dr Luca Zilberstein and all the veterinary and nurses of the anaesthesia team at ADVETIA for their daily help.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
