Abstract
Objectives
The atrial septal pouch was first recognised in humans in 2006 and, since 2010, has been described in more detail. The first reports on the left-sided atrial septal pouch linked its occurrence with a higher risk of thromboembolic stroke resulting from left atrial thrombus formation. However, subsequent studies have challenged this theory; therefore, the consequences of septal pouch occurrence remain disputed. Currently, in veterinary species, septal pouch occurrence and morphology have been described in sheep and pigs, showing species-related differences. No studies have been conducted on cats. As cats show a species-related risk of aortic thromboembolic disease resulting from left atrial thrombus formation, we aimed to describe the interatrial septum anatomical variations as the first step in the investigation of the relationship between the septal pouch presence and left atrial thrombus formation.
Methods
The study was conducted on hearts collected post mortem from 80 cats. After formalin fixation and dissection, a detailed anatomical description of the interatrial septum for all cases was followed by a histopathological analysis of the heart in 25 cases. After dissection, the specimens were embedded in paraffin blocks and stained with haematoxylin and eosin, Masson–Goldner trichrome, Picrosirius Red and Movat pentachrome in a routine manner and evaluated via light microscopy.
Results
Our study showed a very high prevalence (95%) of the left-sided septal pouch in cats with a lower incidence (29%) of the right-sided septal pouch. We noted a high morphological variety of the left-sided septal pouch and described anatomical variants not previously reported in the literature.
Conclusions and relevance
The septal pouch is a common anatomical variation of the interatrial septum in cats. Although, based on the current study, we cannot draw further conclusions on the relationship between the left-sided septal pouch and thromboembolic events in cats, the high prevalence of this anatomical entity points to a need of further studies on the topic.
Introduction
The atrial septal pouch (SP) is a kangaroo-like pouch structure localised in the interatrial septum (IAS) that develops during septal fusion after birth. It was first recognised in humans by Breithardt et al 1 and described in more detail by Krishnan and Salazar 2 and Hołda et al. 3 It is mainly found in adults but can also be present in children. 4 An SP is formed as a result of incomplete fusion between the septum primum and the septum secundum.2,3
The consequences of an SP formation remain disputed. There are studies associating the presence of a left-sided SP (LSP) with an increased risk of rhythm disturbances (including atrial fibrillation) and/or intra-atrial thrombus formation, which can lead to a thromboembolic stroke in humans.5–29 Moreover, an LSP is considered a risk factor for a cryptogenic stroke, especially in younger patients.15,23,29 At the same time, other research teams report no association between IAS malformation and ischaemic or cryptogenic stroke.30–34 The combined role of the SP presence and other risk factors (eg, coronary disease or hyperlipidaemia) cannot be ruled out in the pathogenesis of thromboembolic ischaemic events.10,11,32,33
In veterinary medicine, the SP has been previously described in sheep and pigs,35,36 showing species differences. In sheep, both an LSP and the right-sided SP (RSP) were noted, 35 while in pigs only a RSP was reported. 36 In addition, approximately 20% of sheep or pigs exhibit either a smooth septum (without an SP on either side) or a patent foramen ovale (PFO).35,36
Although ischaemic strokes are rarely reported in feline patients, 37 aortic thromboembolism (ATE) is the second most common clinical sign in cats with cardiomyopathies,38–40 with a survival rate of only 27–45%.40,41 The thrombus formation risk factors include, among others, moderate to severe left atrial enlargement and rhythm disturbances.38–39 Nonetheless, no research has evaluated the presence, prevalence and morphology of the SP in that species. Thus, the possible contribution of the SP to ATE episodes in cats is unknown.
The aim of the study was to evaluate the prevalence and morphology of the SP in cats.
Materials and methods
The study was conducted on 80 hearts obtained from cats that underwent didactical necropsies. The sample size for the finite population (feline hearts) was calculated using an online sample size calculator, considering the assumed level of statistical significance, the size of the cat population in Wroclaw, the population proportion and the acceptable error rate. The animals were patients of various veterinary clinics in the city, donated by their owners for teaching purposes after death or euthanasia. The history of treatment of the animals was not collected, although a complete post-mortem examination was performed by students. Animals with the suspicion of congenital anomalies (including congenital cardiac defects) were excluded from the study. No other exclusion criteria were set. No animals were euthanased deliberately for this study and all samples were originally intended for use for didactic purposes.
During the necropsy, the hearts were obtained intact, sectioned transversally at the level of upper third ventricular height, washed free from blood clots with saline solution, weighed and fixed in 7% buffered formalin.
After the 24–48 h fixation, the hearts underwent pathological analysis. The thickness of ventricular walls, interventricular septum and ventricular internal diameters were measured at the third ventricular height. Subsequently, both atria were dissected to visualise the IAS. The presence of an LSP, RSP, double SP (DSP), PFO or smooth septum was recorded. If present, the SP was measured: the pouch depth and orifice width measurements were taken three times and the mean value was calculated. All measurements were taken using a manual calliper with an accuracy of 0.1 mm. Photographs of each case were taken using a camera (D700; Nikon).
Further, the SP area was dissected in 25 cats (Figure 1) and the sections were embedded in paraffin blocks in a routine manner, cut into sections of 4 µm and stained with haematoxylin and eosin, Masson–Goldner trichrome, Picrosirius Red and Movat pentachrome. The specimens were evaluated via light microscopy and polarised light microscopy using a microscope (CX41; Olympus).

(a) The dissection of a septal pouch (SP) with (b) a schematic presentation. The left-sided SP is marked with a black arrow. The right-sided SP is marked with a blue arrow. Bar = 1 cm. LA = left atrium; RA = right atrium
The statistical analysis was performed using Statistica 13.3 software (Tibco). Data normality was tested using the Shapiro–Wilk test. The variables were compared using the Mann–Whitney U-test and the Kruskal–Wallis analysis. Correlation was tested using Spearman’s correlation analysis. The level of significance was set at P ⩽0.05.
Results
The examined group comprised 80 adult cats (53% male; age range 1–14 years; median 8 years). Domestic shorthair was the most common breed (n = 66, 82.5%), followed by Maine Coon (n = 4, 5%), domestic longhair (n = 3, 3.75%), British Shorthair (n = 3, 3.75%), Siamese (n = 2, 2.5%) and one each of Ragdoll and Neva Masquerade (n = 1, 1.25%). The mean weight of the examined cats was 3.85 ± 1.84 kg (range 1.5–10) and the mean heart weight was 18.3 ± 6.6 g (range 9–33).
Prevalence and morphology of the septal pouch
The presence of an RSP was noted in 23 (29%) animals and an LSP in 76 (95%) animals. Of the above-mentioned animals, five (6%) showed a distinct DSP and a smooth septum was noted in four (5%) animals.
When observed on the right side of the IAS, the SP presented either as a thick muscular ridge protruding into the right atrial lumen (n = 6) (Figure 2a,b) or, more frequently, as a delicate ridge on the surface of the septum (n = 17). The SP on the left side of the septum presented more often as a distinct structure (n = 68) (Figure 2c,d) than only as a ridge (n = 8). The orifice diameter and depth of the RSPs are presented in Table 1.

The morphology of the septal pouch (SP) in cats: (a,b) a right-sided SP; (c,d) a left-sided SP; (b,d) with transillumination. The marker indicates the localisation of the SP. Bar = 1 cm
Dimensions of a right-sided septal pouch (RSP) and left-sided septal pouch (LSP) in 80 cats (in mm)

Orifice diameters were measured in both distinct pouches and septal ridges, while depths were measured only in distinct septal pouches
P = 0.01
The distinct LSP demonstrated a variable morphology including a simple kangaroo-like pouch structure (n = 25) (Figure 2c,d), a pouch connected to the endocardium with an endocardial band (n = 7) (Figure 3a–d), a structure with a distinct endocardial crest present at the bottom of the pouch (n = 6) (Figure 3e–h), a thin membranous pouch closely connected to the IAS (n = 3) (Figure 3i,j), a very shallow pouch (n = 3), a deep and narrow SP that in part presented as a septal ridge (n = 2), a pouch connected to the endocardium with a net-like structure (n = 1) and a very large pouch, partially forming a ridge with a very long endocardial band connecting it to the atrial endocardium (n = 1). The orifice diameter and depth of the LSPs are presented in Table 1.
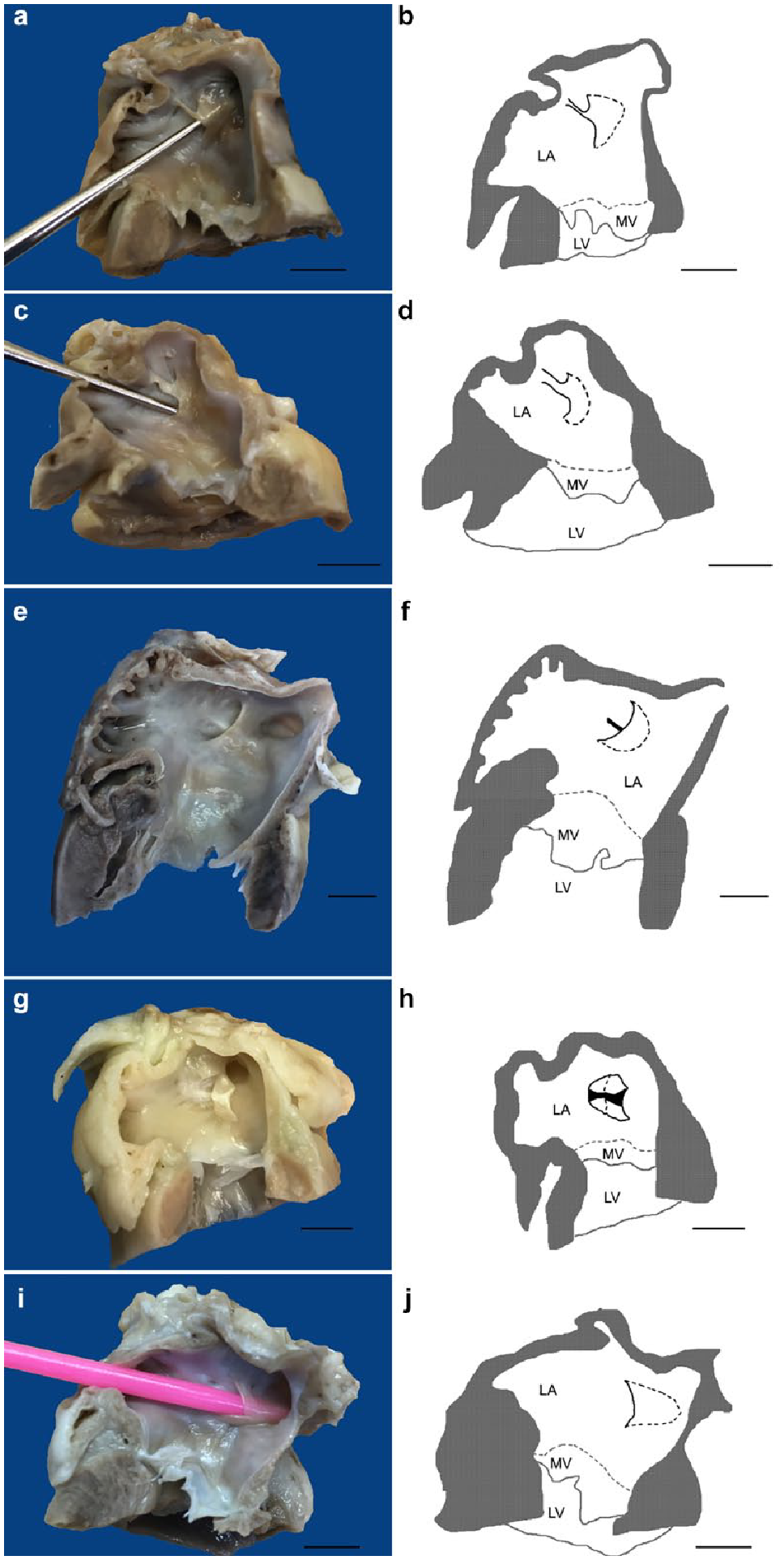
Various morphologies of a left-sided septal pouch (LSP) in cats with schematic presentation: (a–d) an LSP connected to the endocardium with (a,b) a narrow or (c,d) wide endocardial band; (e–h) an LSP with a distinct endocardial crest at the bottom: (e,f) before and (g,h) after dissection of the septal pouch; (i,j) a thin membranous pouch. Cardiac contour is grey, septal pouch is. Bar = 1 cm. LA = left atrium; LV = left ventricle; MV = mitral valve
A detailed description of various combinations of SPs on both sides is presented below and in Figure 4. An isolated RSP was not observed in any of the cats studied. Only one animal showed a distinct RSP with a left septal ridge (Figure 4a). An isolated distinct LSP (Figure 4b) was observed in 47 animals. A distinct LSP combined with a right septal ridge (Figure 4c) was observed in 16 animals and a left septal ridge with a smooth septum on the right side (Figure 4d) was found in six animals. As mentioned before, concomitant distinct RSPs and LSPs (Figure 4e) were observed in five animals, leading to the formation of a DSP. The presence of septal ridges on both sides (Figure 4f) of the IAS was observed in one animal and a smooth septum (Figure 4g) was observed in four animals.

Possible combinations of the septal pouch presence in cats: (a) a dominating right-sided septal pouch (RSP): a distinct RSP with a left septal ridge; (b–d) a dominating left-sided septal pouch (LSP); (b) an isolated distinct LSP; (c) a distinct LSP with a right septal ridge; (d) a left septal ridge with a smooth septum on the right side; (e–g) a balanced bilateral morphology; (e) a concomitant distinct RSP and LSP; (f) a concomitant presence of a septal ridge on both sides of the interatrial septum; and (g) a smooth septum. Red arrows are LSPs; red arrowheads are left septal ridges; blue arrows are RSPs; blue arrowheads are right septal ridges
Statistical analysis
There were no differences in the orifice diameter and depth (in both RSPs and LSPs) with regard to the animals’ sex and breed (P >0.05; Mann–Whitney U-test and Kruskal–Wallis test, respectively). The LSP had a significantly wider orifice than the RSP (P = 0.01; Mann–Whitney U-test) (Table 1).
The LSP orifice diameter positively correlated with heart weight (P = 0.004, r = 0.36), left ventricular wall thickness (P = 0.03, r = 0.27), interventricular septum thickness (P = 0.03, r = 0.28) and right ventricular wall thickness (P = 0.0001, r = 0.44). The LSP orifice diameter negatively correlated with right ventricular internal diameter (P = 0.03, r = −0.28). The LSP depth positively correlated with heart weight (P = 0.02, r = 0.28) and with right ventricular wall thickness (P = 0.03, r = 0.27). Moreover, a positive correlation was found between the LSP orifice diameter and the LSP depth (P = 0.0001, r = 0.52). No correlations were found between the RSPs and heart diameters.
When hearts were divided into two groups based on the heart weight, a significant difference in the mean LSP orifice diameter was noted between normal (weight ⩽20 g; n = 56) and hypertrophied (weight >20 g; n = 24) hearts (4.69 ± 1.5 mm vs 5.72 ± 1.5 mm; P = 0.02; Mann–Whitney U-test).
Histological analysis
Histologically, the LSP presented as a thin transverse muscular layer covered with fibrous tissue and on both sides with endocardium (Figure 5). When present, the RSP presented as a blunt thick muscular protrusion also covered with endocardium. A various amount of connective tissue was observed within the area of origin of the LSP (Figure 6).

The histological image of a routinely stained left-sided septal pouch. A transverse muscular layer (m) covered with a fibrous tissue (f) and on both sides with the endocardium (e). Haematoxylin and eosin stain; magnification ×100. al = atrial lumen; spl = septal pouch lumen

The origin of the left-sided septal pouch (LSP) and its relation to the interatrial septum: (a,b) direct with a mild amount of connective tissue; (c,d) separated by a moderate amount of connective tissue; (e,f) wide, separated by a thick layer of connective tissue. The connective tissue is marked with arrowheads and the LSP is marked with asterisks. (a,c,e) haematoxylin and eosin stain; (b,d,f) Picrosirius Red stain. LA = left atrium; RA = right atrium
Discussion
Our study showed that an LSP is a very common finding in feline hearts, and presents with variable morphologies.
In humans, an LSP is more common than an RSP and is encountered in 17–42% of the examined cases (vs 4.3–12.3% of the RSPs).2,3,9,11,19,26,29,31,42 In the current study, an LSP was more common than an RSP, but the prevalence of both LSP and RSP was higher than in humans. In sheep, LSP occurs as frequently as RSP, 35 while in pigs, LSP has not been reported, whereas RSP has an occurrence of over 22%. 36 Moreover, approximately 5% of humans exhibit a DSP, 3 but no such cases have been reported in pigs and sheep.35,36 In our study, a DSP was present in 6% of cases.
Pigs and sheep exhibit a left-sided septal ridge (15% of sheep and 28% of pigs) or an RSP with a left-sided septal ridge (10% of sheep).35,36 We also observed this type of pouch morphology in cats, although at a lower prevalence (7.5% for a left-sided septal ridge and 1.25% for an RSP with a left-sided septal ridge). In addition, 10% of the examined sheep exhibit a left-sided septal bridge. 35 This type of SP morphology was not noted in the current study; however, in 10% of cats there was an LSP associated with a tissue band connecting the LSP with the left atrial endocardium. Furthermore, in the current study, we observed a right septal ridge, which has not been reported previously in any species, including humans.
In humans, sheep and pigs, a PFO was encountered in over 22% of cases,2,3,19,35,36 while in our study, none of the examined animals exhibited this type of septal morphology.
In our research, a smooth septum was noted in only 5% of cases, while in humans, it has been reported in 22–29% of the examined specimens,2,3 in sheep in approximately 18% of cases and in pigs in over 26% of cases.35,36
The reported frequencies of various septal morphologies in humans differ depending on the research method, with higher values noted in post-mortem studies compared with diagnostic imaging methods (CT and transoesophageal echocardiography).19,43 No similar comparisons are available in animal research. In addition, in our study, only a post-mortem analysis was performed. The possibilities and sensitivity of diagnostic imaging in animals should be further evaluated.
LSP presents with variable morphologies in humans,3,44–46 although the morphological types do not influence the risk of stroke. 11 Similarly, in our research, LSP presented with various morphological types, including those that have not been previously reported. Apart from a typical LSP, we have encountered an LSP with a fibrous crest and an LSP with an endocardial band.
In humans and sheep, the LSP is deeper than the RSP,3,19,26,35 with a comparable width of both LSP and RSP orifices (Table 2).3,35 Although similar results for pouch depth have been obtained in our study with the LSP deeper than the RSP, no statistical significance was noted. Moreover, we have noted a significantly wider LSP compared with the RSP (Tables 1 and 2), which is in contrast with the results obtained in humans and sheep.
The dimensions of a right-sided septal pouch (RSP) and left-sided septal pouch (LSP) in relation to heart weight in humans and various animal species

Data are mean ± SD
Although it is difficult to compare the dimensions measured in cats with other animal species and humans because of the differences in body and heart size, when related to heart mass, the LSP is relatively wider and deeper in cats than in humans, sheep and pigs (Table 2). Moreover, as mentioned before, a correlation of the LSP diameters and cardiac dimensions was noted. The lack of correlations between the RSP and heart diameters was probably attributable to a small number of RSP-positive animals. The histological appearance of the SP in cats was similar to that observed in humans. 3
As mentioned before, an LSP is considered a risk factor for ischaemic stroke in humans due to an increased risk of thrombus formation.5–29 Although cardiac-related neurovascular ischaemic events are not reported in cats, 37 another serious thrombus-related disorder is ATE, as a result of cardiomyopathies, among other causes.38,39 Small thrombi originating from within the left atrium pass within the blood stream to the aortic trifurcation leading to the occlusion of the iliac arteries. This results in an acute ischaemia of the hindlimb(s) and has a poor prognosis.38–41 The literature provides two case reports of people with either upper or lower limb thrombosis associated with the presence of an LSP,47,48 suggesting the possible role of the SP in thrombus formation underlying ATE. As mentioned before, the role of the SP in thromboembolic events is disputable. Nonetheless, it could be one of the factors predisposing to such episodes in humans and animals.10,11,25,32 This theory is also supported by the observed correlation between LSP diameters and feline heart weight together with a significant difference in these diameters between normal and hypertrophied hearts. One can hypothesise that cardiac hypertrophy may lead to the widening of an existing LSP, increasing the risk of intra-atrial thrombus formation. Nonetheless, this hypothesis requires further verification in additional clinical studies. We have not found similar comparisons or relationships in humans or other animals.
As a limitation, the hearts used in the research were obtained during didactic necropsies after a complete flushing of blood; therefore, no further conclusions could be drawn regarding thrombi formation within the SP. Another limitation resulting from the use of hearts obtained during didactic necropsies was the lack of clinical history of the cats. Therefore, no further conclusions can be drawn regarding the relationship between the presence and size of an SP and heart disease or thromboembolic events.
Conclusions
The SP is frequently present in feline hearts with a higher frequency of LSPs. The SP morphology is diverse, presenting as types not previously reported. Taking into account a relatively high frequency of LSPs and a high risk of ATE in cats, the topic requires further examination. Subsequent studies should evaluate the relationship between the presence of an LSP and thromboembolic events in cats (possibly with other risk factors, eg, left atrial enlargement or presence of arrhythmias) and the possibilities of ante-mortem imaging of atrial septum morphology. Because of a high prevalence of LSPs in cats and a high risk of thrombotic events, in the future, cats might be considered a model animal for studies on the relationship between the SP and ischaemic episodes in humans.
Footnotes
Acknowledgements
The authors would like to thank Krzysztof Kwieć for his technical support in collecting the samples and performing the study. We would also like to thank Hanna Kozłowska for her assistance in preparing the final version of the manuscript.
Author note
The results of the study were presented in part at the III Polish Association of Veterinary Cardiology Congress in 2024.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
