Abstract
Objectives
The study aimed to determine if an ultrasonic Doppler-guided technique (UDGT) leads to improved placement efficacy (time, success) of feline dorsal pedal arterial catheters vs the traditional palpation-guided technique (TPT).
Methods
A total of 26 adult, client-owned cats requiring sedation or general anesthesia for any reason, aged >12 months and weighing >3.0 kg, and with Doppler blood pressure measurements of at least 80 mmHg were enrolled. Each hindlimb was randomly assigned for dorsal pedal arterial catheterization using either the UDGT or TPT. With the UDGT, the location of the artery was identified by an audible sound using the Doppler. Successful catheter placement was confirmed by visualization of an arterial pressure waveform using a transducer and monitor system attached to the catheter. The Kaplan–Meier method and log-rank test were used to compare the two techniques.
Results
The overall proportion of successful arterial catheterization was 17% (9/52): 19% (5/26) via UDGT and 15% (4/26) via TPT. Among successful arterial catheterizations (n = 9), the mean time to catheterization was 339 ± 198 s: 328 ± 237 s (n = 5) with UDGT and 353 ± 171 s (n = 4) with TPT. The log-rank test showed the two techniques were not significantly different in likelihood of successful arterial catheter placement or time to successful catheterization (P = 0.698). An arterial flash occurred in 62% (32/52) of the limbs, 58% (15/26) with the UDGT and 65% (17/26) with the TPT. Complications (self-limiting bruising, hematoma formation) were observed equally between UDGT (3/26 limbs) and TPT (3/26 limbs) in six cats.
Conclusions and relevance
The UDGT did not improve the efficacy of catheter placement compared with the TPT. Few complications were associated with arterial catheterization.
Introduction
Blood pressure is one of the most important numeric vital signs, especially when treating a critical or anesthetized patient. An external cuff and either an oscillometric machine or a manual sphygmomanometer and Doppler crystal are most commonly used for non-invasive blood pressure measurement. However, these are both less accurate than direct blood pressure measurement (arterial catheter placed directly into an artery) and only provide readings intermittently.1 –7 The placement of an arterial catheter allows for gold standard direct continuous blood pressure assessment and facilitates repeated arterial blood gas sampling for analysis.
The dorsal pedal artery is relatively easy to detect on palpation and is commonly used for arterial catheterization, especially in dogs, using a blind technique guided by palpation of the pulsing blood in the vessel. 8 Dorsal pedal arterial catheterization is performed less often in cats compared with dogs9,10 for various reasons, including the difficulty associated with catheterizing these small vessels. Thus, a validated technique improving the ease of dorsal pedal catheter placement in cats is needed to facilitate both arterial blood sampling and invasive blood pressure monitoring.
In order to improve the efficacy of arterial catheterization, surgical cutdown and ultrasound-guided techniques have been described.8,11 Surgical cutdown requires skill and is an invasive procedure that can result in complications such as bleeding and local infection. 8 In contrast, ultrasound-guided techniques necessitate the use of an ultrasound machine and specialized ultrasound probe for vascular access, along with training and operator skill, making them less practical and available to practitioners. The only publication demonstrating ultrasound-guided arterial catheterization in small animals is femoral artery catheterization in dogs. 11 Rather than developing ultrasound-guided techniques for cats, use of the ultrasonic Doppler flow detector (ie, the crystal used to measure Doppler blood pressure) is attractive in veterinary medicine as it is relatively inexpensive and its use is ubiquitous in practice.
A previous study described the use of an ultrasonic Doppler flow detector for guidance of radial arterial catheterization in human patients with poor peripheral pulses; the use of the Doppler increased the likelihood of percutaneous radial artery cannulation and minimized the number of attempts at catheter placement. 12 However, in other human studies, radial arterial catheterization guided by Doppler vs palpation did not reveal a statistically significant difference in the success rate of catheterization.13,14 A recent study in dogs and cats evaluated the use of an ultrasonic Doppler flow detector to facilitate placement of dorsal pedal arterial catheters. 15 While that study did not find a statistical difference between the techniques in dogs and cats, the authors suggested that the use of Doppler might increase the opportunity for successful arterial catheter placement in critically ill patients; 15 however, the study only included five cats in its analysis, precluding a definitive conclusion in this species. 15 More data are needed to definitively determine if the Doppler flow detector can improve arterial catheterization in cats.
In an attempt to provide more species-specific information, this study focuses on arterial catheterization in sedated/anesthetized cats. Our objective was to determine if using an ultrasonic Doppler-guided technique (UDGT) would improve the efficacy of dorsal pedal arterial catheter placement vs the traditional palpation-guided technique (TPT) in sedated/anesthetized cats. Efficacy was measured by both likelihood of successful arterial catheter placement and time to successful catheterization. We hypothesized that dorsal pedal arterial catheterization using an ultrasonic Doppler would be more efficacious vs catheterization using a traditional palpation technique.
Material and methods
This was a randomized controlled trial performed at the Purdue University Veterinary Hospital.
Animal and study design
Adult, client-owned cats aged at least 12 months, weighing more than 3.0 kg and requiring sedation or general anesthesia for any reason were eligible for enrollment. To be included, cats needed normal healthy hindlimbs (ie, no wounds or past/current disease affecting the hindlimbs) with bilaterally strong femoral pulses. Recruited cats received a full physical examination by the primary investigator (SH), were assigned an American Society of Anesthesiologists Physical Status Classification System (ASA) score 16 and received a non-invasive blood pressure measurement on a hindlimb while sedated or under general anesthesia before the procedure. Cats enrolled in the study were deemed cardiovascularly stable based on examination, systolic blood pressure readings of at least 80 mmHg 17 and assignment of an ASA score between I and III. 16 The exclusion criteria included an ASA score of IV–VI, those with systolic blood pressure below 80 mmHg or cats with any abnormality of the hindlimbs that would preclude placement of an arterial catheter in the dorsal pedal artery.16,17
All clients signed a written informed consent form before enrollment. The protocol and informed consent forms were approved by the Purdue Animal Care and Use Committee (PACUC; approval number 0223002354) and the Purdue Veterinary Clinical Study Committee.
Placement of dorsal pedal arterial catheter
After enrollment, the techniques (UDGT vs TPT) and limbs (right vs left) for the initial dorsal pedal arterial catheterization were chosen randomly per cat by drawing a slip of paper on which ‘traditional placement’ or ‘ultrasonic Doppler-guided placement’ and ‘left limb’ or ‘right limb’ were written. The technique and limb were in equal numbers on the paper slips. The second dorsal pedal arterial catheterization on each cat was always the other limb using the other technique. Each cat received either sedation or general anesthesia for the purpose of their diagnostic and/or therapeutic procedure(s), as well as immobilization when performing arterial catheterization. The drug protocol selected was at the primary clinician’s discretion for that cat’s primary presenting complaint. The study began after the cat’s primary procedure(s) were completed. Each cat was placed in dorsal recumbency and the fur on the dorsal metatarsal area was clipped and aseptically prepared on both limbs before catheterization. Dorsal recumbency was chosen for the purpose of consistency throughout the study as all the urethral obstruction cats enrolled were already in that position.
The procedure time was measured per limb by the veterinary technician who was monitoring the sedated/anesthetized cat. The time started when the primary investigator (SH) started to localize the arterial pulsation before the first arterial catheterization attempt and ended with either successful arterial catheterization by the primary investigator, 10 mins or immediate complications that prevented catheter placement occurred, whichever came first. Immediate complications were defined as severe hematoma formation or excessive bleeding that prevented placement of the arterial catheter and/or detection of arterial pulsation. Each catheterization attempt was defined as the moment the catheter was inserted into the skin until the catheter was completely removed from the skin. A redirection of the catheter under the skin did not delineate the start of a new catheterization attempt. If catheterization attempts were unsuccessful after a 10 min period per leg or immediate complications were seen, all further catheterization attempts on that limb were aborted. Successful arterial catheterization was confirmed by visualizing pulsatile arterial blood flow from the catheter and an arterial blood pressure waveform using a transducer (TruWave 3 ml/60 in [150 cm]; Edwards Lifesciences) attached to an invasive blood pressure monitor (iMEC8 Vet; Mindray DS USA). 15 Time to arterial flash was defined as the time from starting to localize the pulse to when there was noticeable blood in the hub of the catheter stylet. Regardless of successful arterial catheterization in the first limb, the alternate placement protocol was applied to the second rear limb by the same primary investigator immediately after the first limb.
The method for TPT is as previously described: 8 (1) palpate a dorsal pedal pulse over the metatarsal bones; (2) insert a 24 G intravenous (IV) catheter (Terumo SurFlash; Terumo Medical) into the dorsal pedal artery at an angle of 15°–20°; (3) subsequent to a visible flash of blood in the catheter hub, advance the stylet and catheter a short distance and then gradually advance the catheter off the stylet into the artery; and (4) attach a Luer lock T-port extension set (MicroCLAVE Small Bore T-Set; MILA International) (pre-flushed with heparinized saline [heparin sodium injection 10,000 U/ml; Sagent Pharmaceuticals]) to the catheter hub and connect it to a pressure transducer (TruWave) attached to a monitor (iMEC8 Vet) to generate a visible waveform for confirmation of placement.
The method for placement using UDGT is as previously described: 15 (1) apply the Doppler probe (Ultrasonic Doppler Flow Detector, model 811-B, infant flat probe; Parks Medical Electronics) with sterile ultrasound gel (Ultrasound Gel; Medline Industries) to the skin parallel to the dorsal pedal artery and locate an audible Doppler sound; and (2) adjust the location of the probe to find the most intense audible sound and insert a 24 G IV catheter into the dorsal pedal artery as described in steps (2)–(4) above.
Each cat’s signalment, primary complaint, physical examination findings, ASA score, choice of sedation or anesthesia protocol, time to complete procedure, time to obtain arterial flash through the catheter stylet, number of arterial catheterization attempts and confirmation of arterial catheterization were documented.
After completion, any successfully placed indwelling arterial catheters were removed and a pressure bandage was placed for 10 mins. Cessation of bleeding was confirmed visually and recorded. If catheter placement was unsuccessful, a light wrap was placed over the catheter site for 10 mins. For all cats, any complications noted during catheterization attempts, catheter removal or bandage removal, while hospitalized or reported after hospital discharge by owners, were recorded.
Statistical analysis
D’Agostino–Pearson tests were used to examine whether continuous variables were normally distributed. If a variable was normally distributed, the mean ± SD was reported, otherwise the median (range) was reported. The Kaplan–Meier method was used to estimate the cumulative proportion of success over time while accounting for censoring (ie, unsuccessful catheterizations aborted before the 10 min allotted time). The log-rank test was used to compare the Kaplan–Meier curves between groups. McNemar’s test was used to compare the proportion of successful catheterization and arterial catheter flash between groups. The t-test (or non-parametric test, if the data were not normally distributed) was used to compare the time to catheterization between groups in successful limbs. Random-effect logistic regression was conducted to examine whether blood pressure affected the odds of successful arterial catheter placement. A P value <0.05 was considered significant.
A sample size calculation suggested that a total of 44 arterial catheterizations (22 of each technique) would be sufficient to identify a difference in arterial catheterization time of at least 120 s between the traditional and Doppler methods at a 95% level of confidence and 90% power, assuming the population SD is 120 s.
Results
A total of 28 cats were enrolled between May and September 2023 at the Purdue University Veterinary Hospital. Two cats were excluded due to low blood pressure (<80 mmHg), leaving a total of 26 cats (52 hindlimbs) for arterial catheterization and further statistical analysis. The mean body weight of enrolled cats was 5.4 ± 1.4 kg with a median age of 66 months (range 12–193). Neither body weight nor age were statistically significantly different in each group. The median blood pressure while under sedation or general anesthesia before the procedure was 96 mmHg (range 80–205). Cats were given a variety of medications, including methadone (18/26 cats), propofol (19/26 cats), midazolam (13/26 cats), isoflurane (13/26 cats), ketamine (8/26 cats), dexmedetomidine (6/26 cats), alfaxalone (5/26 cats), hydromorphone (3/26 cats) and acepromazine (2/26 cats). Five cats received a single agent and 21 cats received multiple agents. One cat’s dorsal pedal artery was located lateral, rather than medial, to the visible dorsal pedal vein, which was confirmed with the UDGT technique.
The overall proportion of successful arterial catheterization was 17% (9/52 limbs). Successful arterial catheterization via UDGT occurred in 5/26 (19%) limbs and in 4/26 (15%) limbs using TPT (P = 1.0). All the successful catheterizations occurred in cats that did not receive dexmedetomidine as part of their multi-modal sedation protocol. No cats received dexmedetomidine as a single agent.
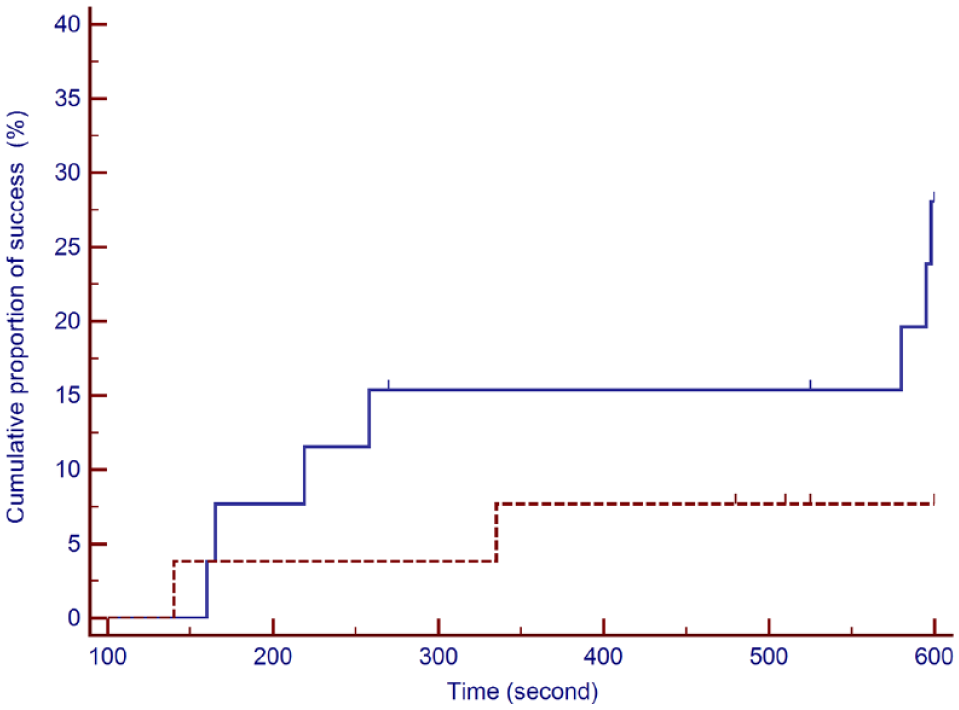
The mean procedure time among successful arterial catheterizations was 339 ± 198 s (n = 9): 328 ± 237 s (n = 5) for UDGT and 353 ± 171 s (n = 4) for TPT (P = 0.862). When the mean times to successful catheterization with each technique were compared using the Kaplan–Meier method and log-rank test (Figure 1), the time to successful catheterization between the two techniques was not statistically significantly different (P = 0.698).

Kaplan–Meier curves of the cumulative proportion of successful arterial catheterization over time via the ultrasonic Doppler guidance technique (UDGT) and traditional palpation-guided technique (TPT). The x axis represents the procedure time (in seconds), and the y axis shows the cumulative proportion of successful arterial catheterization (%), which were not statistically significantly different between the two techniques (P = 0.698). The solid line indicates the UDGT (n = 26) and the dashed line indicates the TPT (n = 26)
The proportion of successful arterial catheterization via UDGT in the left hindlimb was 31% (4/13) vs 7.7% (1/13) in the right hindlimb. Similarly, the proportion of successful left arterial catheterization using the TPT was 23% (3/13) compared with 7.7% (1/13) in the right hindlimb. Figure 2 shows the Kaplan–Meier curves of the cumulative proportion of successful arterial catheterization between the two hindlimbs; the two curves were not significantly different (P = 0.078).
Kaplan–Meier curves depicting the cumulative proportion of successful arterial catheterization regardless of technique in each limb over time (P = 0.078). The x axis represents the time to catheterization (in seconds), and the y axis shows the cumulative proportion of success (%). The solid line represents the left hindlimb and the dashed line represents the right hindlimb
An arterial flash occurred in 62% (32/52) of the limbs: 58% (15/26) using the UDGT and 65% (17/26) using the TPT (P = 0.754). With UDGT, the mean time to flash was 281 ± 173 s, whereas the mean time to flash was 289 ± 184 s with the TPT (P = 0.859). The results of arterial catheterization are summarized in Table 1.
Results of arterial catheterization in 26 cats using the ultrasonic Doppler-guided technique (UDGT) and traditional palpation-guided technique (TPT)*

Data are mean ± SD or mean (range)
Cats first received catheterization by a randomly assigned technique in a randomly assigned limb with the other technique used on the opposite limb
Allotted time for unsuccessful catheterization was set at 10 mins. Two cases in the UDGT and two in the TPT were aborted due to a complication a (hematoma) before the 10 min period
Three cats were successfully catheterized on the first attempt via UDGT
Side effects were observed equally between UDGT (3/26 limbs) and TPT (3/26 limbs) in six cats in total (12% of limbs) and included self-limiting bruising or hematoma formation.
Discussion
This study investigated whether the use of the UDGT for dorsal pedal arterial catheterization would improve the efficacy of catheterization (success rate or time to catheterization) over the TPT in sedated/anesthetized cats. Contrary to our hypothesis, the UDGT did not significantly improve the efficacy of arterial catheter placement over the TPT. This is similar to previous human studies that did not find a difference in the efficacy of arterial catheterization between the UDGT and TPT.13,14 A previous veterinary study evaluating arterial catheterization techniques in dogs and cats also reported no significant differences in success rates between these two techniques. 15 That veterinary study also reported an overall arterial catheterization success rate in cats identical to that of our study (17%). 15
Although our study did not find a statistical difference in the efficacy of catheterization between the techniques, the Kaplan–Meier curves consistently showed a trend suggesting that the UDGT allowed for faster catheter placement compared with TPT (Figure 1) and was still a worthwhile technique to use. In addition, although this did not always translate to successful catheterization, the UDGT was particularly useful in cases of unique anatomy, such as the study subject whose dorsal pedal artery was located lateral, rather than medial, to the visible dorsal pedal vein or when pulses were hard to palpate. Thus, both techniques are complementary, and clinicians can try one technique if the other does not work.
It is worth noting that the majority of attempts in this study (62%) involved an arterial flash in the catheter even if the catheter could not be placed. The overall success rate of obtaining an arterial flash was similar to the study previously reported in dogs and cats (67%). 15 We suspect that an arterial flash would still allow for collection of arterial blood samples. We partially attribute the low proportion of successful catheterizations to difficulty feeding the catheter into the small superficial feline vessel after the flash was obtained. A study of the Egyptian domestic cat (Felis catus) reported that the diameter of the dorsal metatarsal artery was 1.3 mm compared with the 0.7 mm diameter of a 24 G catheter. 18 In addition, many of the drugs used in these cats, such as dexmedetomidine, will vasoconstrict the arteries peripherally, making the vessel even smaller and hence catheterization more difficult. 19
Interestingly, the likelihood of successful arterial catheterization in the left limb (31%) was more than four-fold higher than in the right limb (7.7%) with both the UDGT and TPT. As the anatomy of the limbs should be a mirror image of one another, we suspect this finding is attributable to a more ergonomic angle of placement for the right-handed primary investigator (SH) when using the left limb when the cats were positioned in dorsal recumbency. A differing successful catheterization rate of right vs left limbs was not documented in a previous veterinary paper, 15 but the cats were positioned differently in our paper (dorsal recumbency) vs the previous paper (lateral recumbency). Thus, right-handed operators placing catheters in cats in dorsal recumbency may want to use the left limb preferentially; alternatively, cats could be placed in lateral recumbency to improve the success rate in the right limb.
An operator’s learning curve did not affect the success rate over time, as witnessed by the fact that successful catheterization events were sporadic throughout the course of the entire study and the frequency of success did not improve towards the end of the study period. This finding is in contrast to a previous veterinary study, which demonstrated a learning curve for all investigators when placing a central venous line via ultrasound-guided and palpation techniques. 20 However, in the previous veterinary study comparing the TPT and UDGT for dorsal pedal arterial catheterization, the skill set and experience of the operator also did not statistically significantly affect the likelihood of successful arterial catheterization in dogs and cats via the UDGT. 15
In the present study, the only complications observed were self-limiting bruising and hematoma formation (12% of limbs). Most complications occurred in the first third of enrolled cats and fewer complications were seen as the study progressed. This is likely due to the primary investigator (SH) learning to provide proactive compressive hemostasis in the midst of catheterization attempts. This complication rate is similar to that in a retrospective study of arterial catheterization, primarily in dogs and mainly in the dorsal pedal artery, that observed bruising in 8.6% of study subjects. 9 A retrospective study of arterial catheterization in cats, equally placed in both the dorsal pedal and coccygeal arteries, showed a similar complication rate (17%); however, the complications reported in that study were cold or swollen paws rather than bruising. 10 No indications of ischemia or necrosis were observed in the cats in our study after arterial catheterization; this was similar to the findings reported in another feline case report. 21
The present study has some limitations. First, the power analysis was conducted with the expectation of a higher successful catheterization rate compared with the actual proportion of successful arterial catheter placements documented in this study. Our lower success rate resulted in a much smaller analytical sample size than the initially expected sample size of 44 total successful procedures. This suggests that the present study would not have sufficient power to detect a clinically important difference. The results in this study cohort implied that there was a small trend toward faster time to successful catheterization via the UDGT compared with the TPT, although all differences between the two techniques were small.
The second limitation to our study was that the sedation and general anesthesia protocols used in these cats were not standardized but were at the discretion of the primary clinician. All successful arterial catheterizations were performed in cats that did not receive dexmedetomidine. This suggests that the vasoconstrictive effects of this medication may affect the ability to catheterize the artery. However, in all but one cat, and regardless of the drug protocol, one limb could be catheterized with one technique and the other limb could not be catheterized with the other technique. This suggests that failures in catheterization were usually due to the operator rather than the cat and its vessels, indicating the sedation or anesthesia protocol was less important.
Third, the fact that the left limb was successfully catheterized more often than the right limb might indicate that the results of our study would be different if we conducted arterial catheterization in only the left limb or had positioned the cats differently depending on which leg was being catheterized. Using only left limbs might have increased our overall success rate or the success rate might have increased in the right limbs with different positioning; both of these scenarios would have increased our numbers of successful placements. More successful placements might have changed our overall study conclusions but more likely would have simply increased the overall number of successful catheter placements while still not revealing a significant difference between the techniques.
Lastly, the study population was not standardized and had a variety of different disease processes. However, since arterial catheters are used in patients with diverse disease processes, we do not believe that this detracted from our study results.
Conclusions
This study revealed that the use of the UDGT did not improve the efficacy of placing arterial catheters in feline patients compared with the TPT. Our study implied that the UDGT may be clinically helpful in locating the position of an unusually placed artery and, in some cases, might locate the artery when it cannot be palpated. Few complications were associated with arterial catheterization.
Footnotes
Acknowledgements
The authors would like to thank all the staff in the emergency and critical care service for their support in enrollment of patients and assistance during procedures related to this study.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals and procedures that differed from established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient. The study therefore had prior ethical approval from an established (or ad hoc) committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
