Abstract
Case series summary
This case series describes seven cats that were treated with high-flow nasal cannula oxygen therapy (HFNOT). Seven cats were prospectively (n = 5) or retrospectively (n = 2) included from three veterinary university referral centers between March 2020 and September 2023. Data on signalment, medical history, clinical and diagnostic findings, treatment administered, response to HFNOT and outcomes were recorded. All cats included in this case series failed to respond to oxygen cage or flow-by oxygen therapy and were subsequently transitioned to HFNOT. After this transition, these cats demonstrated a marked improvement in respiratory parameters, including respiratory rate, effort and oxygen saturation measured by pulse oximetry, within 1 h of initiating HFNOT. All cats tolerated HFNOT well without any complications.
Relevance and novel information
There is limited literature reporting the use of HFNOT in feline patients. This is the first case series in the literature of HFNOT utilized as an advanced oxygen delivery method for feline respiratory failure. This case series indicates that HFNOT improves oxygenation in feline patients that fail to respond to conventional oxygen therapy.
Plain language summary
Seven cats with respiratory failure received high-flow nasal cannula oxygen therapy, which improved their oxygenation. Three were successfully weaned off the therapy, and one survived and was discharged.
Keywords
Introduction
High-flow nasal oxygen therapy (HFNOT) delivers humidified and heated air with a flow of up to 60 l/min and a fraction of inspired oxygen (FiO2) between 0.21 and 1.0.1,2 The advantages of HFNOT include reduction of dead space, provision of positive end-expiratory pressure and enhanced mucociliary clearance, all of which collectively improve patient comfort compared with conventional oxygen therapy (COT), such as oxygen cage, flow-by oxygen, oxygen hood or nasal cannula.1 –3 HFNOT is widely used in human medicine as an intermediary treatment for acute hypoxemic respiratory failure, providing a middle ground between COT and mechanical ventilation (MV).1,2 In recent human literature, the respiratory-rate oxygenation index (ROX), defined as the ratio of oxygen saturation measured by pulse oximetry (SpO2) to FiO2 (the SF ratio) divided by the respiratory rate (RR), has been validated as a predictive index of HFNOT success or failure. 2 A lower ROX value is associated with higher likelihood of failure to respond to HFNOT. Specific ROX cutoff values have been suggested to guide clinical decisions regarding the escalation from HFNOT to MV.2,4
In veterinary medicine, the application of HFNOT was predominantly documented in dogs with respiratory failure.3,5 –8 This technique was validated as a well-tolerated, safe and effective method of oxygen delivery in dogs with hypoxemic respiratory failure.5 –8 Despite the substantial amount of literature on HFNOT in dogs, the utility of HFNOT in feline patients is not as well-documented. The current literature primarily consists of two case reports, with Baudin et al 9 being the first to report its application in a cat. The initial placement of the nasal cannula in the report required sedation; however, subsequent heavy sedation was not required thereafter. A recent case report has highlighted the significant improvement in a cat’s respiratory parameters within 15 mins of the initiation of HFNOT. 10 In scenarios where cats do not adequately respond to COT, such as oxygen cages, HFNOT may serve as a bridging therapy to MV. Therefore, further study is warranted to establish the practicality of utilizing HFNOT in cats. The aim of the present study was to describe the application of HFNOT in cats with respiratory failures that failed to respond to COT and to provide preliminary data for future studies in a larger number of cats.
Case series description
A total of seven cats were enrolled from three veterinary university referral centers between March 2020 and September 2023. In two centers (VetAgro Sup [SIAMU] and Centre Hospitalier Universitaire Vétérinaire Animaux de Compagnie [CHUVA-AC]), five cats were prospectively enrolled. The study protocol was approved by the VetAgro Sup Ethics Committee (Number 1730). Cats were included if they were non-responsive to COT, which was defined as having persisting respiratory distress (RR above 40 rpm and increased respiratory effort) or SpO2 <95% despite initial stabilization, including medical treatment and oxygen via flow-by or oxygen cage. Primary clinicians monitored for signs of HFNOT intolerance (several attempts to remove the cannula) and any complications associated with HFNOT with routine thoracic auscultation and daily point-of-care ultrasound. In one center (Ohio State University [OSU]), electronic medical records were searched between March 2020 and September 2023 to identify cats treated with HFNOT, with keywords including ‘high-flow oxygen’ and ‘high-flow nasal cannula’. The medical records were evaluated by one author (JH), who also served as the primary clinician of the included cats, to ensure consistent application of the inclusion criteria for this study. The records were then reviewed for any complications associated with HFNOT.
Data were collected, including signalment, history, primary diagnosis of respiratory disease, oxygen supplementation method before HFNOT, duration of HFNOT and outcome. Treatment was defined as HFNOT success if a patient transitioned from HFNOT to COT or room air and sustained normal respiratory parameters for over 12 h. Conversely, cats that were transitioned to MV, euthanized or that died due to a progressively declining respiratory pattern and SpO2 while receiving HFNOT were categorized as HFNOT failure. HFNOT was delivered using a DRE Volumax VOS Veterinary Oxygen System (Avante; OSU) or Airvo 2 System (Fisher & Paykel Healthcare; SIAMU and CHUVA-AC). HFNOT prescription and monitoring parameters for the first hour were collected every 15 mins, including flow rate (l/kg/min), FiO2, RR, SpO2 and heart rate. SpO2 readings were verified by matching patients’ heart rates and plethysmographic waveforms by the attending veterinarian or trained veterinary technicians who used pulse oximetry daily.
All data were entered into an Excel spreadsheet (Microsoft) and analyzed using statistical SAS software (SAS Institute). The normality assumption was evaluated via inspection of QQ- and PP-plots, histograms and skewness. Normally distributed variables were summarized descriptively with the mean and standard deviation. Non-normally distributed variables were summarized descriptively with the median and range. Linear mixed models were used to compare SpO2, RR, SF ratio and ROX 2 during HFNOT (T15, T30, T45, T60) to at initiation (T0). The model had a fixed factor for time and a random intercept for each cat. Model assumptions (normality and homoscedasticity of model residuals) were evaluated by inspecting conditional QQ plots, histograms and residual plots. To meet the assumption of homoscedasticity, the SF ratio and ROX values were log-transformed before analysis. Multiple comparisons were adjusted for using Dunnett’s test. Satterthwaite’s degrees of freedom method and restricted maximum likelihood estimation were used. A value of P <0.05 was considered statistically significant.
Signalment
A total of seven cats were included in this case series. The mean age was 4.9 ± 4.6 years and the mean weight was 3.9 ± 0.6 kg. Before HFNOT, cats were determined not to respond to COT, as evidenced by SpO2 (n = 7, mean 87% ± 9%) and RR (n = 7, mean 66 ± 33 rpm) at the initiation of HFNOT (T0). The demographic information, primary respiratory diagnosis, oxygen supplementation method before HFNOT, duration of HFNOT, HFNOT success or failure, and survival are listed in Table 1.
Demographics, respiratory diagnosis and outcome in cats treated with high-flow nasal cannula oxygen therapy (HFNOT)
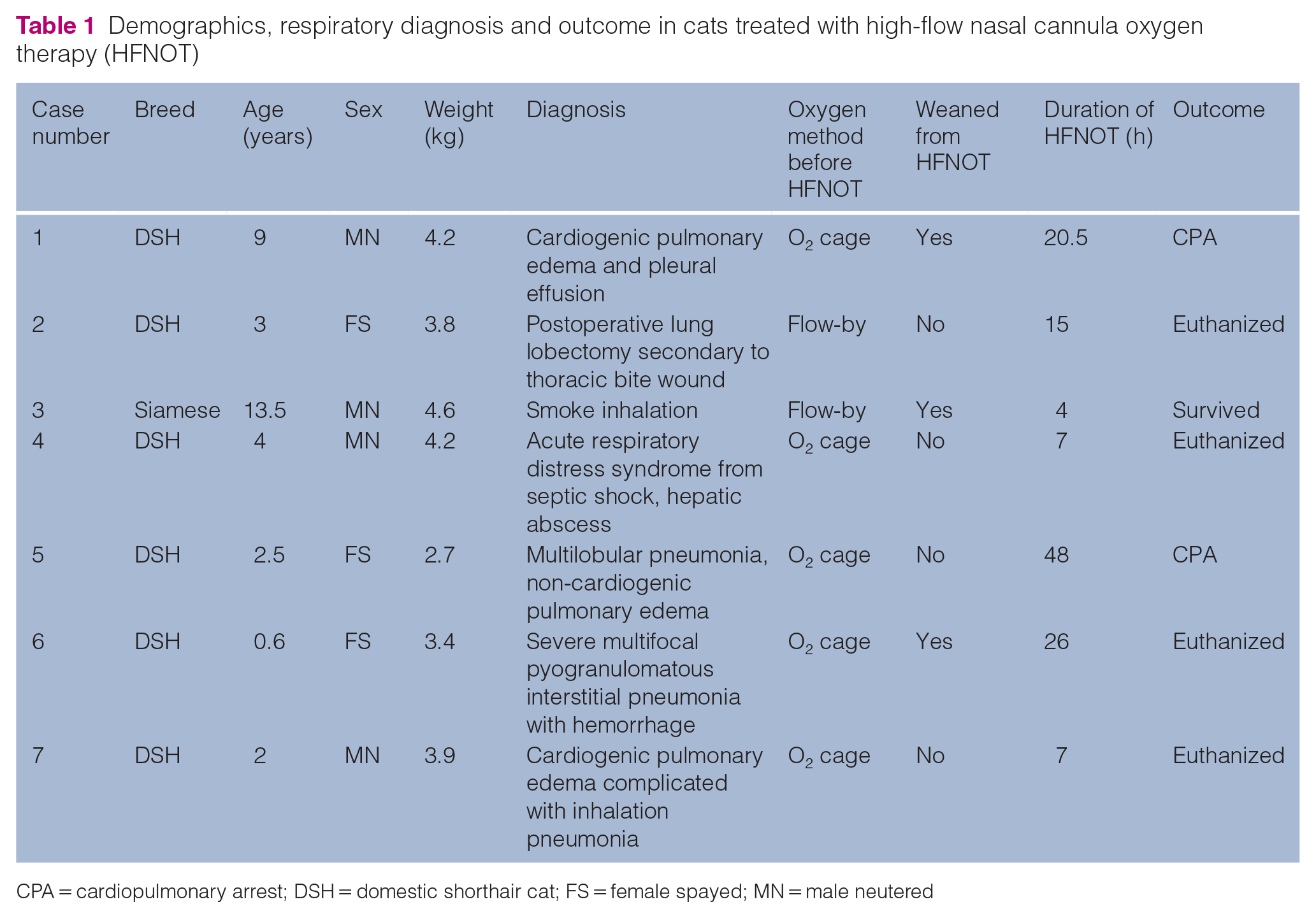
CPA = cardiopulmonary arrest; DSH = domestic shorthair cat; FS = female spayed; MN = male neutered
Cannula placement
Five cats were initially treated in an oxygen cage at a FiO2 of 0.3–0.6 for 0.5–3 h before transitioning to HFNOT, and two cats were immediately transitioned from flow-by oxygen to HFNOT upon presentation. HFNOT-specific cannulas, Vapotherm nasal cannula (Infant cannula, flow range 1–8 l/min; Vapotherm) and Optiflow+ (OPT312 Junior Premature, flow range 1–8 l/min; Fisher & Haykel Healthcare), provided by the manufacturer of DRE Volumax Veterinary Oxygen System (Avante) or Airvo 2 System (Fisher & Paykel Healthcare) were used. Although the general recommendation by the manufacturers is to choose the size of a nasal cannula occluding no more than 50% of the nares, the smallest cannula chosen for cats resulted in occlusion of approximately 50–70% (Figure 1). One cat was placed on a single-prong nasal cannula specific for the manufacturer (Single prong infant cannula; Vapotherm). The sedation protocol was chosen by the attending clinician, based on the patient’s clinical status. For the initial application of the HFNOT cannula, butorphanol (0.1–0.3 mg/kg IV) was used in three cats, dexmedetomidine (1 µg/kg IV) in one cat and acepromazine (5 µg/kg IV) in one cat. Two cats received no sedation for cannula placement. An e-collar was placed on four cats to prevent cannula removal, and two cats were placed in oxygen cages while receiving HFNOT in case of inadvertent removal of the cannulas (Figure 1). The nasal cannula was gently inserted in the nares and tightened behind the neck, and eventually secured with tape and sutured in place next to each nare (Figure 1).
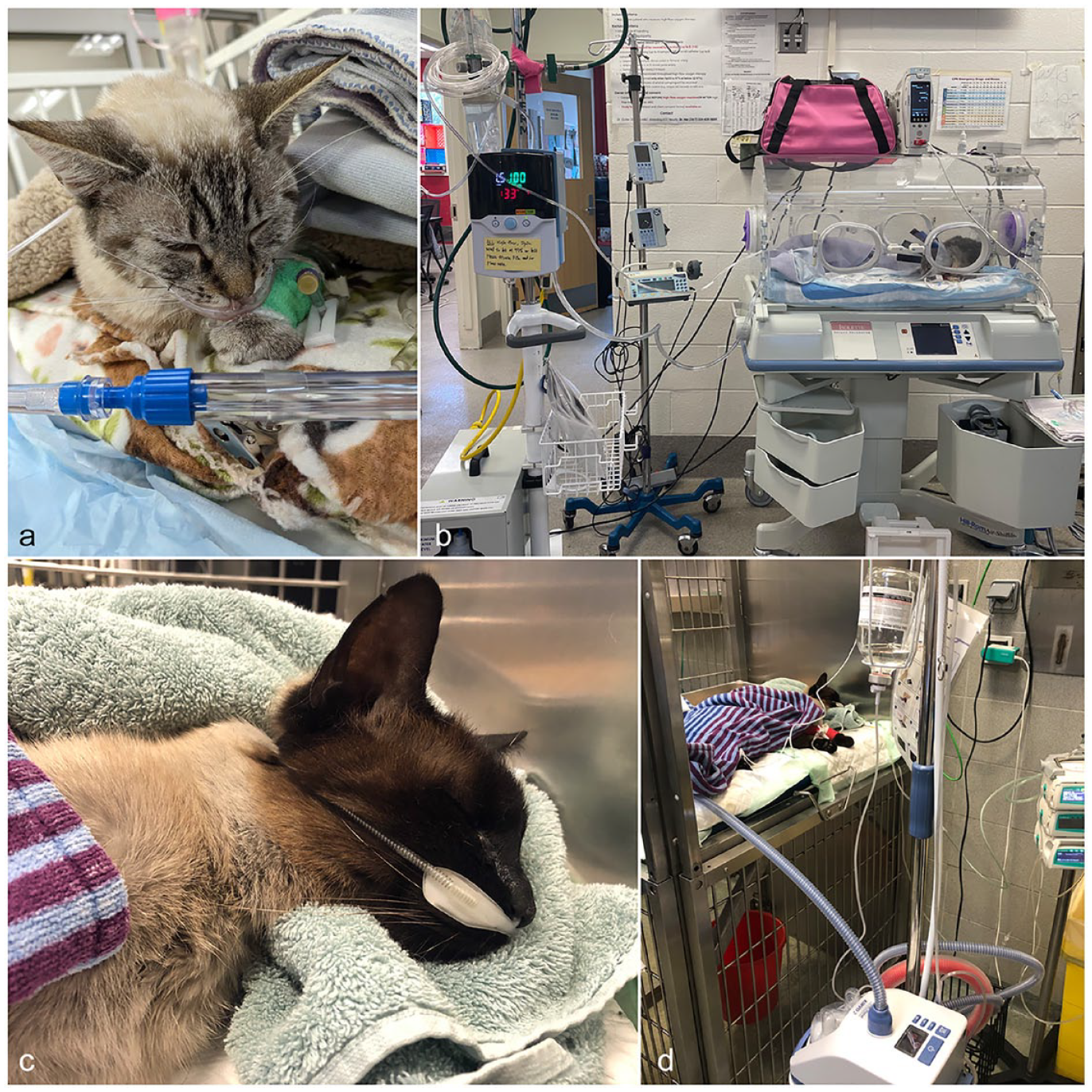
The use of high-flow nasal cannula oxygen therapy in cats with severe respiratory failure. (a) A cat placed on a Vapotherm cannula (Vapotherm); (b) the cat was placed in an oxygen cage in case of inadvertent removal of the cannulas; and (c,d) a cat with an Airvo cannula (Airvo 2 System; Fisher & Paykel Healthcare)
HFNOT period
The duration of HFNOT was in the range of 4–48 h (mean 18 ± 15 h. In total, 3/7 cats not require sedation during the HFNOT period. Among the four cats that received sedation, the most common sedative used was butorphanol. Three cats received butorphanol (0.2 mg/kg IV) 2–3 times over a HFNOT period of 15–48 h to achieve respiratory optimization and to improve cooperation with HFNOT. One cat received butorphanol as a constant rate infusion (0.2 mg/kg/h) throughout the HFNOT period. HFNOT prescription and monitoring variables are summarized in Table 2. HFNOT was initiated with a median flow rate of 1.5 l/kg/min (range 1.5–2.3) and a median FiO2 of 0.95 (range 0.4–1.0). The range of initial flow rate was selected based on the previous canine literature. 6 This approach provided flexibility for attending clinicians to adjust the flow rate based on each patient’s level of discomfort while aiming for respiratory optimization. Optimization was assessed via the patient’s respiratory rate, effort and SpO2 measurements. SpO2 measurements were available in 6/7 cats (Figure 2). HFNOT significantly improved the SpO2 levels at T15 (mean 95% ± 5%, P = 0.033), T45 (mean 95% ± 5%, P = 0.033) and T60 (mean 98% ± 3%, P = 0.004) compared with T0 (mean 87% ± 9%). The median SF ratio of cats at the initiation of HFNOT (T0) was 93 (n = 7, range 87–230). HFNOT significantly reduced the RR at T60 (mean 49 ± 21 rpm) compared with T0 (mean 66 ± 32 rpm, P = 0.041). Individual RRs are presented in Figure 3. Individual data of SpO2, SF ratio and ROX 2 are presented in Table 2. HFNOT was delivered consistently without interruption or complication in all cats. The cats did not dislodge the cannulas, and there were no instances of oxygen desaturation or pneumothorax.
High-flow nasal cannula oxygen therapy (HFNOT) prescription variables, respiratory rate (RR), oxygen saturation measured by pulse oximetry (SpO2) to FiO2 (the SF ratio), respiratory-rate oxygenation index (ROX), and sedation during the first 60 mins of HFNOT
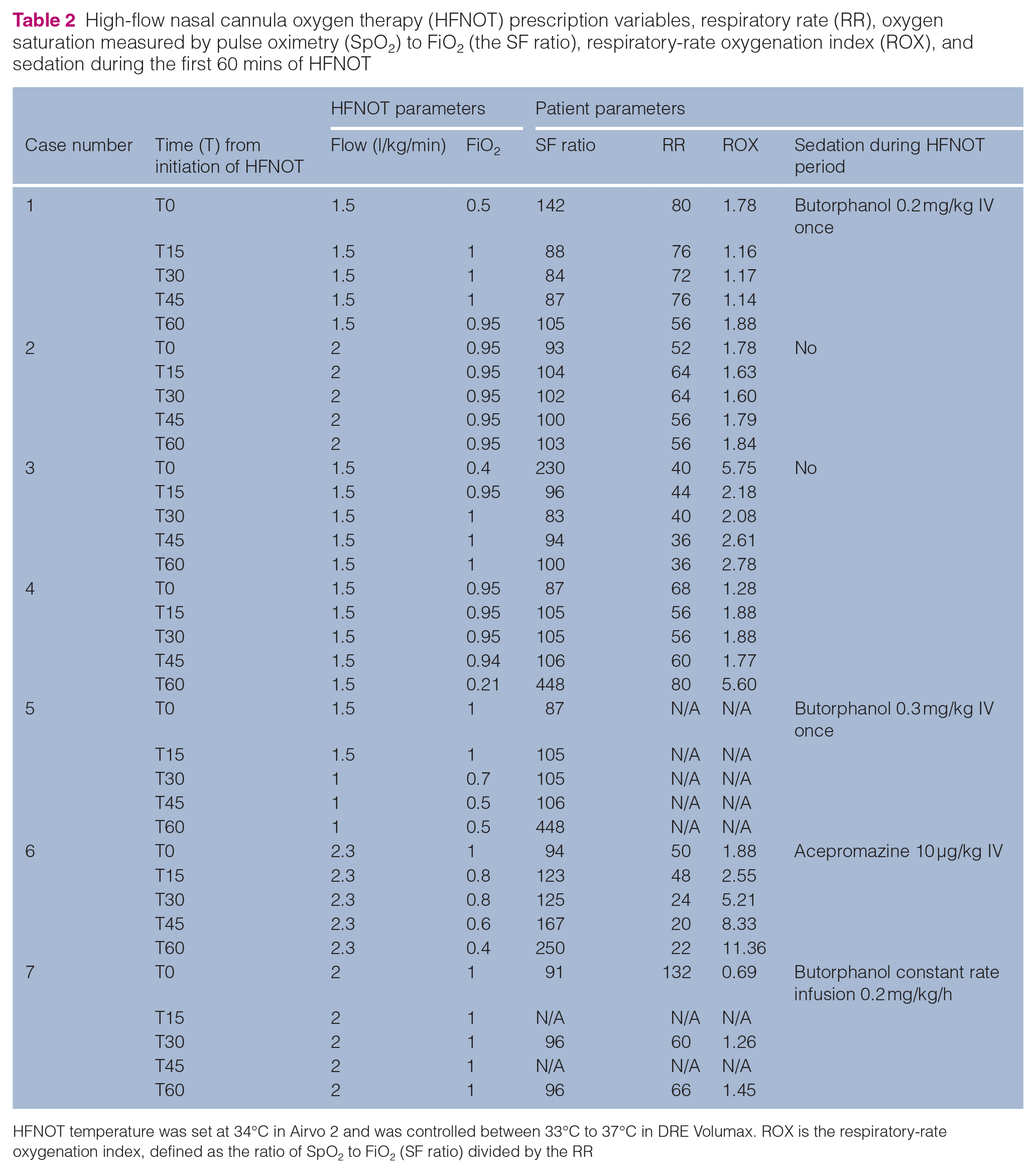
HFNOT temperature was set at 34°C in Airvo 2 and was controlled between 33°C to 37°C in DRE Volumax. ROX is the respiratory-rate oxygenation index, defined as the ratio of SpO2 to FiO2 (SF ratio) divided by the RR

Change in oxygen saturation measured every 15 mins via pulse oximetry (SpO2) for individual cats, during the first hour of high-flow nasal cannula oxygen therapy. Data from one cat are missing. T0 = values measured at the initiation of high-flow nasal cannula oxygen therapy

Changes in respiratory rates measured every 15 mins for individual cats, during the first hour of high-flow nasal cannula oxygen therapy. T0 = values measured at the initiation of high-flow nasal cannula oxygen therapy
Outcome
Overall, the success rate for HFNOT was 43% (3/7). The decision to wean HFNOT was determined by attending clinicians based on patients’ RR, effort and SpO2 during HFNOT. In general, de-escalation was considered after patients’ RR remained in the range of 20–40 rpm and their SpO2 was in the range of 94–97%, while simultaneously decreasing the FiO2 from 1.0 to 0.2–0.4 and reducing the flow rate from 1–2 l/kg/min to 0.2–0.5 l/kg/min, before transitioning to room air or an oxygen cage with an FiO2 of 0.3–0.5. Two cats (cats 1 and 6) were successfully weaned from HFNOT to an oxygen cage with an FiO2 of 0.3–0.5, and the other cat (cat 3) was weaned from HFNOT to room air. Out of the three cats that were successfully treated with HFNOT, only one (cat 3) survived to discharge, resulting in an overall survival rate of 14% (1/7). The cat presented with respiratory distress due to smoke inhalation and was treated with HFNOT for 15 h before transitioning to room air. The cat remained stable and eupneic in room air for 4 days and was discharged from the hospital. Another cat (cat 1) developed sudden cardiopulmonary arrest due to aortic thromboembolism 68 h after the completion of HFNOT. The other cat (cat 6) was euthanized due to a recurrence of respiratory signs warranting reinitiating HFNOT, 20 h after the completion of HFNOT. Among the four cats (cats 2, 4, 5 and 7) that could not be weaned off HFNOT, two cats (cats 4 and 7) were euthanized due to a poor prognosis from continued decline in respiratory status. Another cat (cat 5) developed cardiopulmonary arrest and died during HFNOT. The other cat (cat 2) was transitioned to MV for severe hypoventilation (PvCO2 of 120 mmHg) during the postoperative period and then was euthanized shortly after due to a poor prognosis.
Discussion
Based on the literature search performed, this is the first case series of cats treated with HFNOT. HFNOT is widely used to treat respiratory diseases in people, and its utility has already been proven in healthy and hypoxemic dogs.1,5 –8,11 In the present case series, seven cats with severe respiratory failure that failed to respond to COT were escalated to HFNOT. All of the cats tolerated HFNOT well without clinically relevant complications. As described in dogs,5,8 HFNOT improved SpO2 and decreased RR within 1 h in most cats (Figures 2 and 3). Although trained personnel verified the SpO2 measurements with matching heart rate and plethysmographic waveforms, arterial blood gas analysis is considered the gold standard for assessing oxygenation impairment. This is due to inherent inaccuracies of SpO2 in various clinical scenarios, such as differences in pigmentation and peripheral vasoconstriction. 12 Due to technical challenges in obtaining arterial blood samples from critically ill or distressed cats, arterial blood gas samples were not collected in the present study. It would have been interesting to document the progression of partial arterial pressure of oxygen to verify the improvement of SpO2 observed in this study and to investigate the changes in partial pressure of carbon dioxide (PaCO2) in cats undergoing HFNOT. Canine studies have demonstrated a mixed effect of HFNOT on PaCO2.5,6,8
The ROX has shown its value as a prognostic indicator for predicting HFNOT success or failure in humans and dogs.2,4,13 Roca et al 4 reported that a ROX value of 4.88 or higher at the 2, 6 or 12 h mark indicated successful weaning from HFNOT in human patients with acute hypoxemic respiratory failure. Since then, the index has proven its value in predicting HFNOT success or failure in patients with conditions such as pneumonia, COVID-19, thoracic injuries and those with compromised immune systems. 14 Similar findings were observed in a recent retrospective study of dogs, suggesting that the ROX is a useful predictor of HFNOT outcome in dogs with hypoxemic respiratory failure. 13 The present study reports the ROX in cats treated with HFNOT (Table 2). Due to the limited sample size, our study could not assess the predictive utility of the ROX in cats. This index was easy to measure at the bedside and may have great clinical utility in guiding clinicians in managing cats treated with HFNOT. Future studies are warranted to investigate the utility of ROX in a larger population of cats undergoing HFNOT.
There are no set guidelines for HFNOT settings in cats. Individualizing HFNOT settings is recommended in human medicine, to improve effectiveness and tolerance. 15 The flow rate adjustments, FiO2 and gas temperature should be based on SpO2, respiratory characteristics, patient tolerance and arterial blood gas analysis, if available. 15 At the authors’ institutions, we typically start at flow rates of 1–2 l/kg/min with a FiO2 of 1.0 and temperature between 34°C and 37°C and adjust HFNOT settings based on the abovementioned parameters. The initial flow rate of 1–2 l/kg/min was determined based on the previous canine literature, which suggests that the flow rate provides continuous positive airway pressure in dogs, 3 despite the absence of similar studies in cats. This highlights the need for future research on cats to evaluate the impact of flow rate on airway pressure and patient comfort in both experimental and clinical settings.
In this study, starting HFNOT at a median flow rate of 1.5 l/kg/min (range 1.5–2.3) and a median FiO2 of 0.95 (range 0.4–1.0) improved oxygenation and reduced RR in cats; these effects lasted during the first hour of HFNOT. Although this study focused on reporting the oxygenation parameters every 15 mins for the first hour, the cats were monitored throughout the entire HFNOT period. Further research is needed to evaluate the trend of oxygenation over the entire duration of HFNOT in cats and to determine the optimal initial settings, stabilization procedures and weaning strategies for HFNOT.
There is limited research on the use of HFNOT in cats, making our understanding of their acceptance of the HFNOT interface limited. Baudin et al 9 were the first to suggest its application in cats as an alternative to continuous positive airway pressure delivered by a helmet. The authors stated that while the initial placement of the nasal cannula required sedation, further heavy sedation was not necessary once the cat accepted the HFNOT interface. In addition, a recent case report involving a cat with cardiogenic pulmonary edema treated with HFNOT highlighted the usefulness of sedation for initiating HFNOT. 10 Although the cat had accepted the placement of the HFNOT cannula without sedation, starting the HFNOT flow rate (1.8 l/kg/min) necessitated a short period of heavy sedation with propofol. In line with the previous reports, five cats in this study received sedation for both HFNOT cannula placement and flow rate initiation, highlighting the need for sedation for the smooth initiation of HFNOT in cats. Cats 2, 3 and 4 did not necessitate any sedation; however, they were all critically ill and depressed because of their primary condition. Sedation was given to four cats throughout the HFNOT period; however, infrequent sedation was sufficient to keep the cats on HFNOT, except for one cat that received a constant rate infusion of butorphanol mainly due to markedly increased respiratory effort. Of note, the cats in this study may have appeared to be more compliant with HFNOT due to their systemic illness in addition to the sedation administered for HFNOT cannula placement. Future investigation is warranted to evaluate the feasibility and tolerability of HFNOT in conjunction with the level of sedation, utilizing both the tolerance and the sedation score in cats.
Complications of HFNOT appear to be minimal in canine literature.3,6 –8 Most of the reported complications in dogs are minor, such as aerophagia, intolerance of the nasal cannula and persistence of a pre-existing pneumothorax. These complications were resolved with HFNOT discontinuation. Similar to the canine studies, this study noted no clinically relevant complications in cats. The primary concern with HFNOT in these cats was the potential for them to remove the cannulas. Therefore, standard procedures included securing the HFNOT interface with sutures or staples and fitting an e-collar. In addition, 2/7 cats were housed in an oxygen cage to decrease the risk of desaturation resulting from accidental removal. This approach might be suitable for cats less accepting of the HFNOT interface than those described in this study. Future studies are warranted to assess the complications associated with HFNOT in a larger number of cats.
Conclusions
To the authors’ knowledge, the present study enrolled the largest number of cats treated with HFNOT published thus far. In this case series, HFNOT improved respiratory status in cats with respiratory failure not responding to COT. No clinically significant complications associated with HFNOT were observed. While the advantages of HFNOT have been well-documented in humans and dogs, its application in cats warrants further investigation in a larger number of cats.
Footnotes
Acknowledgements
The authors are grateful to Dr Deborah Keys for assistance with the statistical analysis.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
CPN and PV received financial support from L’institut Agronomique, Vétérinaire et Forestier de France (AGREENIUM) for the research and publication of this article. The authors thank Agreenium, ENVA, ENVT, Oniris and VetAgro Sup for the financial support of this study (National Program of Clinical Research 2020).
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
