Abstract
Objectives
The aim of this study was to describe the clinical and diagnostic findings and outcome of cats with bicavitary effusion presenting to a referral centre.
Methods
Medical records of cats presenting with bicavitary effusion were identified and their history, physical examination findings, clinicopathological data, diagnostic imaging findings, aetiology of bicavitary effusions (cardiac disease, neoplasia, infectious disease, sterile inflammatory disease, severe hypoalbuminaemia, trauma, coagulopathy or ‘open’ if no definitive diagnosis was reached) and outcome were recorded. Cox regression analysis was performed to identify independent predictors of death in cats with bicavitary effusion. Kaplan–Meier curves were generated for survival analysis.
Results
In total, 103 cats with bicavitary effusion were included. Neoplasia and cardiac disease were the most common aetiologies of bicavitary effusion, in 21 (20.4%) and 20 (19.4%) cats, respectively, followed by infectious disease (n = 11, 10.7%), trauma (n = 13, 12.6%), hypoalbuminaemia (n = 6, 5.8%), sterile inflammatory disease (n = 4, 3.9%) and coagulopathy (n = 1, 1.0%). The median survival time for all cats with bicavitary effusion was 3 days. Cats with a neoplastic aetiology had a 2.03 times greater risk of death compared with cats in which no diagnosis was achieved. Neoplasia (P = 0.030) and pedigree breed status (P = 0.016) were independent predictors of death in the multivariable Cox regression model.
Conclusions and relevance
This study highlights that bicavitary effusions in cats generally carry a guarded to poor prognosis, particularly if neoplasia is the underlying aetiology or if the cat is a pedigree breed. Cardiac disease appeared to be associated with a better prognosis, suggesting that assessment for congestive heart failure should be considered early when evaluating cats with bicavitary effusion. The prognosis for cats with feline infectious peritonitis is likely to be markedly improved by the advent of novel antiviral drugs, compared with the historical cohort of cats presented here.
Introduction
Bicavitary effusion is the abnormal accumulation of free fluid within two body cavities concurrently. Effusions may develop due to various pathophysiological processes, including decreased plasma oncotic pressure, increased capillary hydrostatic pressure, increased vascular permeability, lymphatic obstruction, exfoliation of neoplastic cells, coagulopathy or traumatic injury. 1 Causes of bicavitary effusion in cats include congestive heart failure, pericardial effusion with tamponade, severe hypoalbuminaemia, neoplasia, infectious diseases, sterile inflammatory diseases, idiopathic chylothorax, coagulopathy and trauma. In some cases, a cause for bicavitary effusion may not be apparent despite diagnostic investigations, with one study reporting a definitive diagnosis in only 77% of feline cases. 2
The current literature regarding bicavitary effusion in cats is sparse, with only one study specifically evaluating bicavitary effusions in dogs and cats. In this study, patients with bicavitary effusions had a 3.3 times greater risk of death compared with those without. When patients were stratified based upon the presence of mild, moderate or severe effusions, their median survival times were 63 days, 13 days and 4 days, respectively. Neoplastic and cardiac disease were the most common aetiologies of bicavitary effusion. 3 A further study, while not specifically describing bicavitary effusions, reported almost half of cats presenting with peritoneal effusions to have concurrent pleural effusions. This study did not evaluate whether bicavitary effusion carried a worse prognosis, but again reported neoplastic and cardiac disease to be the most common aetiologies. 4
While the above literature would suggest that the prognosis for cats with bicavitary effusions is poor and a diagnosis can be difficult to obtain, studies reporting potential prognostic indicators are lacking. This is essential information to aid clinicians in providing owners with accurate prognostic information that may inform their choices for ongoing care. The aims of the present study were to report the clinical characteristics and aetiologies of bicavitary effusions in a large referral population of cats and, secondarily, to report outcome data and evaluate survival.
Materials and methods
Study design
This was a retrospective cross-sectional study.
Case selection and data collection
The Small Animal Medicine service case database at Langford Vets Small Animal Referral Hospital was searched and cats with bicavitary effusion diagnosed between June 2009 and December 2021 were identified. The search terms ‘bicavitary effusion’, ‘double effusion’, ‘peritoneal effusion’, ‘abdominal effusion’, ‘peritoneal fluid’, ‘ascites’, ‘pleural effusion’ and ‘pleural fluid’ were used. For inclusion, cats were required to have free fluid identified in two or more body cavities at presentation by one or more of echocardiography, plain radiography, ultrasound or CT. Simultaneous pleural and peritoneal fluid were required for inclusion. Cats were excluded if they had undergone a laparotomy, thoracotomy or pericardiocentesis in the 2 months before referral, developed volume overload secondary to intravenous fluid therapy or if follow-up until at least 30 days after discharge was not available.
Patient signalment, body weight, body condition score (BCS), clinical signs and physical examination findings were retrieved from medical records.
Haematological and serum biochemical data, pertinent clinicopathological data and results of infectious disease testing were retrieved where available. All results, apart from patient-side feline immunodeficiency virus (FIV) antibody and feline leukaemia virus (FeLV) antigen ELISA, were obtained in reference laboratories using validated assays. Further information is provided in supplementary material 1.
Pleural and abdominal fluid analyses were performed by board-certified clinical pathologists, clinical pathology residents under supervision or qualified laboratory technicians. Fluid total protein, total nucleated cell count, infectious disease polymerase chain reaction (PCR) results, fluid cholesterol and triglyceride concentrations, packed cell volume, and bacterial culture and sensitivity testing results were recorded where available. Effusions were classified according to previously published criteria as detailed in Table 1.5 –9
Criteria for the categorisation of pleural, pericardial or peritoneal effusions

PCV = packed cell volume; TNCC = total nucleated cell count; TP = total protein
Diagnostic imaging was performed by a board-certified radiologist, cardiologist or supervised resident. Modalities via which effusions were identified were recorded. Radiographs or CT images obtained by the referring veterinarian were acceptable after review by a radiologist as above. Details regarding the diagnostic imaging systems utilised are provided in supplementary material 2.
Aetiology of bicavitary effusion and final diagnosis
Final diagnoses were recorded, and the cats were stratified into disease categories as follows:
Cardiac disease: based upon echocardiographic and/or thoracic imaging findings consistent with congestive heart failure
Neoplasia: either cytologically/histologically confirmed or strongly suspected based upon the presence of a mass lesion with or without evidence of metastasis
Infectious disease: either confirmed based upon microbiological, molecular, serological or cytological results or strongly suspected based upon compatible clinicopathological findings
Sterile inflammatory disease
Hypoalbuminaemia (serum albumin <15 g/l) 10
Trauma
Coagulopathy
Open: no definitive diagnosis or cause for bicavitary effusion identified.
Outcome
Survival was defined as the number of days from diagnosis until death.
Statistical analysis
Statistical analyses were performed using GraphPad Prism version 9 (GraphPad Software). Descriptive statistics were performed for patient characteristics, clinical signs, examination findings, clinicopathological data, diagnostic imaging findings, aetiology of bicavitary effusion and survival data. Selected clinical variables that could be obtained from a cat at the time of diagnosis were evaluated for associations with survival by performing Cox regression analysis to identify independent predictors of death. Cats with missing continuous data were excluded from analysis. Data were available for all categorical variables except for pleural and abdominal fluid classification. A missing data category was created for these variables. Data were censored if the cat was lost to follow-up. Univariable Cox regression analyses were performed initially and variables with P <0.2 were entered into a manual forward selection, stepwise multivariable Cox regression model. Coagulopathy as a disease category was excluded as it contained only one cat. The proportional hazards assumption was tested by examining log-minus-log plots. The assumption that each predictor was independent was tested by evaluating two-way interactions and the correlation matrix. The linearity assumption was tested by examining residuals. Hazard ratios with 95% confidence intervals (CIs) were determined. Kaplan–Meier survival curves were constructed for variables that remained independent predictors of death in the Cox regression model. Statistical testing of Kaplan–Meier curves was performed using the log rank test. Significance was set at P <0.05.
Results
Patient characteristics
A total of 132 cats with bicavitary effusion were identified during initial data collection. Of them, 29 were subsequently excluded for failure to meet inclusion criteria as outlined in Figure 1. This resulted in 103 cats being included in the final population.

Flow chart demonstrating how cats were excluded from initial case selection to form the final study population
Of the 103 cats in the study, 68 were male (65 neutered, three intact) and 35 were female (33 neutered, two intact). Domestic shorthair was the most common breed (n = 67) followed by domestic longhair (n = 15), British Shorthair (n = 6), Maine Coon (n = 4), British Blue (n = 2), Bengal (n = 2) and one each of Abyssinian, Birman, Burmilla, domestic mediumhair, Selkirk Rex, Siamese and Singapura breeds. Median age was 7 years (range 1–16), median body weight was 4.2 kg (range 1.9–10.6) and median BCS was 3 (range 2–9).
Lethargy was the most common clinical sign recorded (n = 78, 75.7%) followed by inappetence (n = 64, 62.1%), tachypnoea or dyspnoea (n = 43, 41.7%), weight loss (n = 32, 31.1%), abdominal distension (n = 17, 16.5%), vomiting (n = 16, 15.5%), peripheral oedema (n = 10, 9.7%), polyuria/polydipsia (n = 9, 8.7%), abdominal pain (n = 8, 7.8%), weakness or ataxia (n = 6, 5.8%), cough (n = 5, 4.9%), diarrhoea (n = 3, 2.9%), tenesmus (n = 3, 2.9%), syncope (n = 2, 1.9%), skin wounds or abrasions (n = 2, 1.9%) and hypersalivation (n = 1, 1%).
Diagnostic imaging findings
Pleural effusion was identified in 30 (29.1%) cats using thoracic radiography, 30 (29.1%) using CT, 10 (2.9%) using echocardiography and 20 (19.4%) using thoracic ultrasound. Peritoneal effusion was identified in 81 (78.6%) cats using abdominal ultrasound and 22 (21.4%) using CT. Pericardial effusion was identified in 13 (12.6%) cats, all by echocardiography.
Clinicopathological findings
Haematological and serum biochemical findings are presented in Tables 2 and 3. Lymphopenia was the most common haematological finding, present in 66/82 (80.5%) cats, followed by neutrophilia in 29/85 (34.1%) cats and neutrophil left shift in 22/85 (25.9%) cats. Hypoproteinaemia was the most common serum biochemical abnormality, present in 74/82 (90.2%) cats for which total protein concentration was available, followed by hypoalbuminaemia in 49/83 (59.0%) cats and increased alanine aminotransferase activity in 48/82 (58.2%) cats.
Haematological findings in 103 cats with bicavitary effusion

Serum biochemical data in 103 cats with bicavitary effusion

ALP = alkaline phosphatase; ALT = alanine aminotransferase; GGT = gamma-glutamyltransferase
Infectious disease testing results are presented in Table 4. One cat tested positive for each of FIV and FeLV. Five cats had feline coronavirus detected by PCR from effusion. Two cats had a positive bacterial culture from peritoneal effusion, one with Escherichia coli and one with Yersinia pseudotuberculosis. Antibody ELISA for Borrelia burgdorferi, Anaplasma phagocytophilium, Ehrlichia canis and antigen ELISA for Dirofilaria immitis were performed in one cat and were negative.
Infectious disease testing performed out of 103 cats with bicavitary effusion
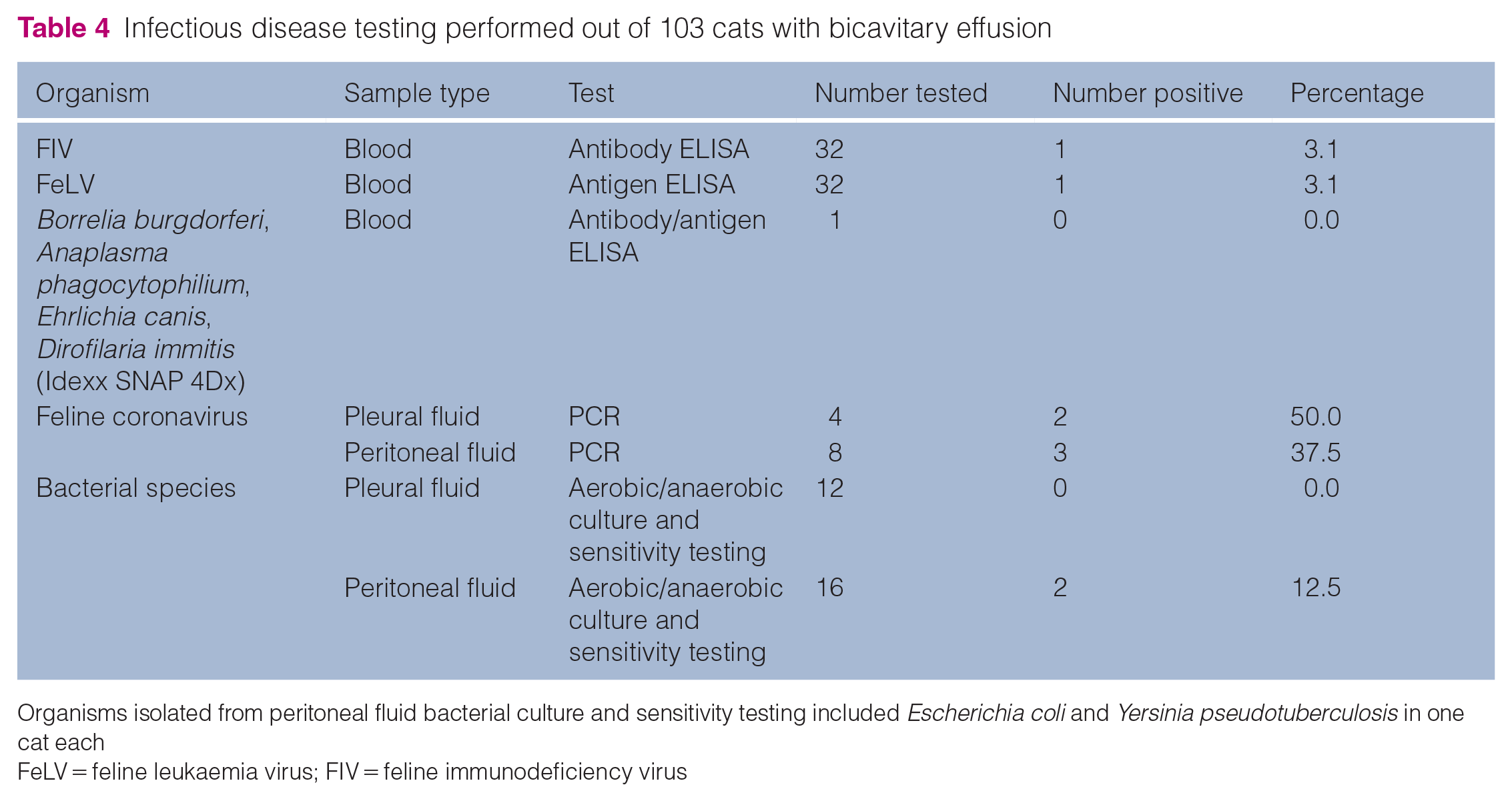
Organisms isolated from peritoneal fluid bacterial culture and sensitivity testing included Escherichia coli and Yersinia pseudotuberculosis in one cat each
FeLV = feline leukaemia virus; FIV = feline immunodeficiency virus
The results of body cavity fluid analyses are presented in Table 5. Protein-rich transudate was the most common fluid type identified for both pleural and peritoneal effusions. In total, 15 cats had concurrent pleural and peritoneal fluid analysis performed, in which 11 (73%) had the same fluid type identified in both the pleural and peritoneal cavities and four (27%) had different fluid types as presented in Table 6.
Summary of pleural and peritoneal fluid analyses performed in cats with bicavitary effusion

Data are n (%) unless otherwise indicated. Neoplastic cells identified in pleural fluid were round cells in two (50.0%) cats and a neoplastic epithelial/mesothelial population in two (50.0%) cats. In peritoneal fluid, four (80.0%) cats had a neoplastic round cell population identified and one (20.0%) cat had a neoplastic epithelial/mesothelial population
Summary of four cats with incongruent pleural and peritoneal fluid analyses

Aetiology of bicavitary effusion and final diagnosis
The aetiology of bicavitary effusion and the final diagnoses are presented in Table 7. In 23 (22.3%) cats, a definitive diagnosis was not reached. In cats with a final diagnosis, neoplasia was the most common aetiology, identified in 21 (20.4%) cats, followed by cardiac disease in 20 (19.4%), infectious disease in 15 (14.6%), trauma in 13 (12.6%), severe hypoalbuminaemia in six (5.8%), sterile inflammatory disease in four (3.9%) and coagulopathy in one (1.0%).
Aetiologies and final diagnoses in 103 cats with bicavitary effusion

Data are n (%)
Outcome
Survival data were available for 84 cats. A total of 17 cats were lost to follow-up and two cats were still alive at the time of data collection. The overall median survival time for cats with bicavitary effusion was 3 days (range 1–311). The following variables were significant in the univariable Cox regression analysis as predictors of death in cats with bicavitary effusion: pedigree breed (P = 0.043); female neutered status (P = 0.003); neoplastic aetiology (P = 0.049); and presence of abdominal septic exudate (P = 0.024). The results of the univariable Cox regression analysis, including hazard ratios and 95% CIs, are presented in Table 8. In the final multivariable Cox regression model, neoplasia (P = 0.030) and pedigree breed (P = 0.016) remained independent predictors of death as presented in Table 9. The Kaplan–Meier survival curves for the variables found to be significant in the final multivariable Cox regression model are presented in Figure 2. A significant difference was identified between non-pedigree and pedigree cats when Kaplan–Meier curves were analysed using the log rank test (P = 0.028). A significant difference was also identified between survival curves for different disease categories when Kaplan–Meier curves were analysed using the log rank test (P = 0.008) as presented in Figure 3. The median (range) survival for cats with bicavitary effusion of different aetiologies is presented in Table 10.
Results of the univariable Cox regression analysis of predictors of death in cats with bicavitary effusion
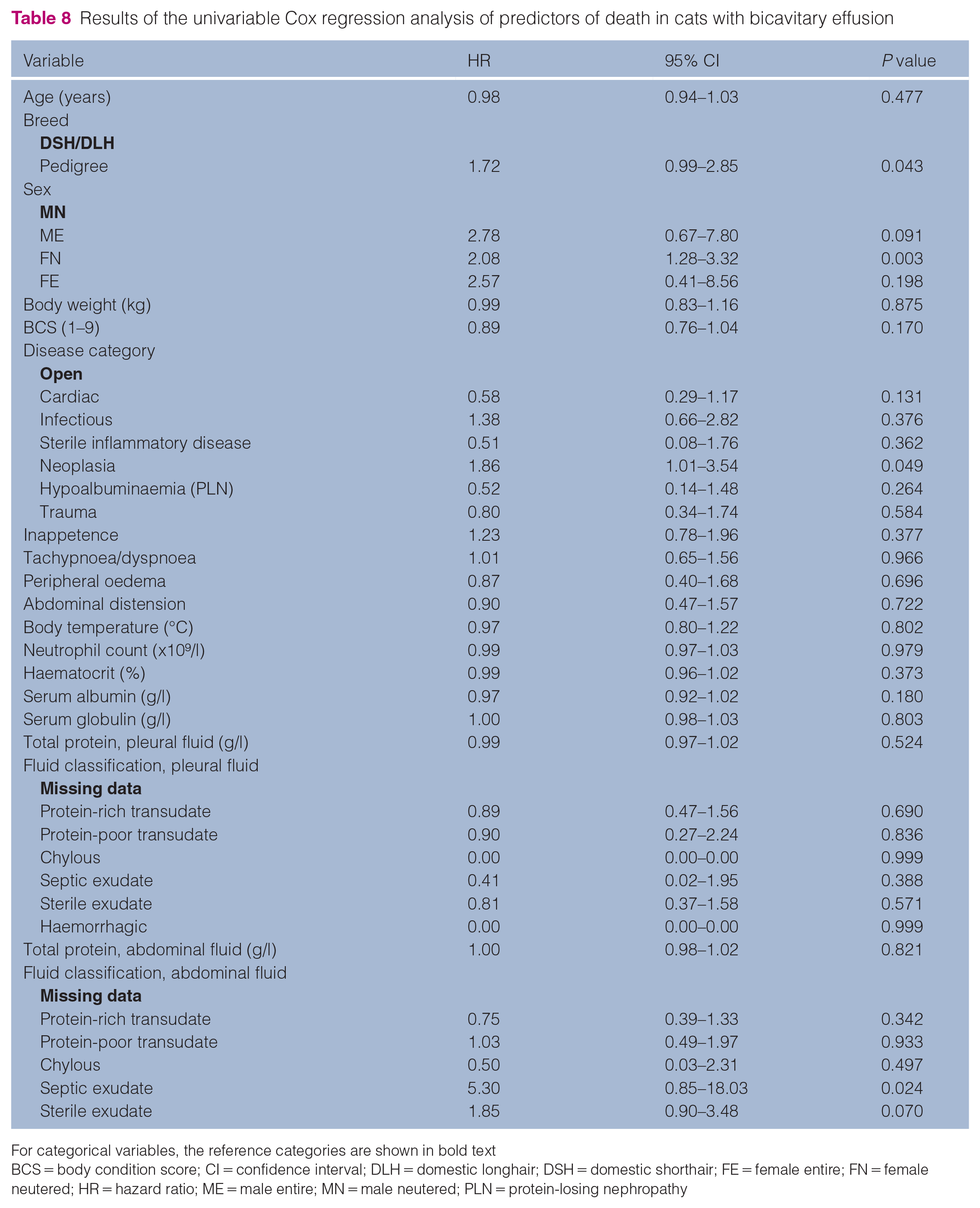
For categorical variables, the reference categories are shown in bold text
BCS = body condition score; CI = confidence interval; DLH = domestic longhair; DSH = domestic shorthair; FE = female entire; FN = female neutered; HR = hazard ratio; ME = male entire; MN = male neutered; PLN = protein-losing nephropathy
Results for final multivariable Cox regression model for predictors of death in cats with bicavitary effusion

CI = confidence interval; DLH = domestic longhair; DSH = domestic shorthair; HR = hazard ratio; PLN = protein-losing nephropathy; SE = standard error

Kaplan–Meier survival curves for variables that remained independent predictors of survival in the multivariable Cox regression model. DLH = domestic longhair; DSH = domestic shorthair

Kaplan–Meier survival curves for different aetiologies of disease causing bicavitary effusion
Median survival times for different aetiologies of bicavitary effusion

PLN = protein-losing nephropathy
Discussion
The study reported here describes in detail a large referral population of cats with bicavitary effusion.
Male cats were over-represented in our study. This might be due to the relatively high frequency of trauma cases observed (12.6% of the study population), for which male non-pedigree cats are reported to be at greater risk. 11 Male cats are also over-represented for cardiomyopathies.12,13 Indeed, 10/13 trauma cases and 17/20 cardiac cases in our study were male. In addition, a recent study reported that cats with cardiomyopathy as the aetiology of their pleural effusion were significantly more likely to be male. 14 The aforementioned study evaluating bicavitary effusions in both dogs and cats also reported males to be more commonly affected. 3 Male cats, however, did not have an increased risk of death compared with female cats in the final multivariable model.
The median age of cats in our study was 7 years, which likely reflects the heterogeneous aetiologies of their bicavitary effusions, as cats presenting with trauma and infectious disease are likely to be younger whereas cats with neoplasia are likely to be older.4,14 The mean BCS was 3/9, which likely reflects the fact that neoplasia and cardiac disease were common, and both commonly cause weight loss or cachexia. The BCS was not reported in any of the previous studies identified regarding this topic.
Clinicopathological findings in this population were attributable to their underlying diseases. Lymphopenia, neutrophilia and neutrophil left shift were the most common haematological findings, likely reflecting systemic illness or systemic inflammation. The high prevalence of hypoproteinaemia in this population of cats can be explained by third-spacing of serum proteins into effusions. Interestingly, given that neutrophil left shift was identified in 22 cats, only eight of the study cats had hyperglobulinaemia. This is unexpected, as a polyclonal gammopathy resulting in hyperglobulinaemia might be expected where there is marked systemic inflammation resulting in neutrophil left shift.
Infectious disease testing was performed inconsistently in this population, possibly due to cost constraints or because in some cases a rapid alternative diagnosis was made that precluded infectious disease testing. As expected, due to the now widespread use of vaccination against FeLV, none of the cats diagnosed with lymphoma (or indeed neoplasia) in this study were FeLV positive. A total of 12 (11.65%) cats had PCR for feline coronavirus performed, of which five were positive. This differs from previous studies regarding body cavity effusions in cats, likely because this test has only more recently become widely available. Nonetheless, a more recent study evaluating pleural effusions in cats reported that only 4.2% of cases had a PCR for feline coronavirus performed. 14 Feline infectious peritonitis (FIP) was the most common infectious disease diagnosed in the present study population, comprising 11/15 (73%) of all infectious disease cases.
One cat each was diagnosed with pyothorax and septic peritonitis, which typically would not be expected to result in bicavitary effusions. Possible explanations for bicavitary effusion in these cases could be systemic inflammation and/or vasculitis, extension of infection by direct or hematogenous spread, or hypoalbuminaemia associated with sepsis that may have led to reduced plasma oncotic pressure. Unfortunately, neither of these cases had concurrent pleural and peritoneal fluid analyses performed so it was not possible to identify the type of bicavitary effusions.
Neoplastic disease was the most common aetiology of bicavitary effusion in our study, followed by cardiac disease. This is in line with the aforementioned studies regarding bicavitary effusions in dogs and cats, peritoneal effusions in cats (in which almost half of cases had concurrent pleural effusions) and a more recent study evaluating solely pleural effusions in cats.3,4,14 Cats with a neoplastic aetiology in our study also had the highest risk of death (hazard ratio [HR] 2.03, P = 0.030) This may be because neoplasia truly carries a worse prognosis or because owners may be more likely to euthanase their pets when presented with a diagnosis of neoplasia. Other studies regarding effusions in cats report variable prognoses associated with neoplastic aetiologies, with one study demonstrating a 100% mortality rate within 1 week of diagnosis, except for cats with mediastinal lymphoma. 2 In contrast, a further study reported cats with neoplasia as a cause of pleural effusion to have a 24% mortality rate before discharge. 14 These differing methods of presenting outcome data, however, limit direct comparisons between our study and those mentioned above. Despite advances in cross-sectional imaging and diagnostic techniques, similar to previous studies, a final diagnosis was not achieved in almost one-quarter (22.3%) of our cases. 2
Pedigree cats were at 2.10 times greater risk of death compared with non-pedigree breeds (P = 0.016) in this population. This finding has not been described previously, to the best of the authors’ knowledge, in cats with effusions but may be explained by the fact that pedigree cats are over-represented for diseases such as FIP (which did not, for the majority of the cats in this study, have an effective treatment available at the time of diagnosis). Of the 11 cats with confirmed or strongly suspected FIP in this study, eight (72.7%) were of a pedigree breed.
The median survival time for all cats with bicavitary effusion was 3 days. This suggests that bicavitary effusion carries a poor prognosis, in line with the existing literature.3,4 Based on our study, the exception may be cardiac disease (HR for death of 0.50). However, this narrowly missed reaching statistical significance in the multivariable model (P = 0.059). Further studies may be warranted to explore whether cats with bicavitary effusion caused by congestive heart failure truly do have a better prognosis than those with other aetiologies. If so, then clinicians might consider evaluating for congestive heart failure early on in their investigations to identify cats that may respond more favourably to treatment. In addition, cats with infectious disease had a median survival time of just 2 days (range 1–84), which likely reflects the fact that a definitive treatment for FIP was not available for the majority of our study period. It is likely that more favourable survival times would be identified if a similar population of cats were evaluated at the present time.
The present study has some limitations, many of which are inherent to its retrospective nature. Diagnostic investigations were performed at the discretion of the attending clinician and therefore not standardised. This, however, likely reflects the situation encountered in clinical practice in that the diagnostic evaluations would be initially guided by the history and examination findings and therefore would differ in every case. In terms of identifying effusions, it is possible that the use of thoracic radiographs rather than CT or ultrasound may have missed small pleural effusions, as radiography is generally regarded as less sensitive for the detection of free pleural fluid. 15 It is likely that many of our cats would have undergone thoracic point-of-care ultrasound (POCUS) at presentation, but these examinations would not have been carried out by a board-certified radiologist, and therefore such cases would not have been suitable for inclusion based on POCUS alone. The survival of cats recruited at different time points between 2009 and 2021 may have been affected by advances in diagnostics and treatments throughout that period, such as the availability of PCR assays for feline coronavirus and treatments for FIP. In addition, for cats that survived to discharge but subsequently died or were euthanased, it is possible that the cause of death was not related to bicavitary effusion. However, given that most of the reported aetiologies for bicavitary effusion cannot generally be cured, this would appear relatively unlikely.
Future studies to evaluate the prognostic relevance of single vs bicavitary effusions for cats with common aetiologies, such as neoplasia or cardiac disease, may be useful and help guide prognosis for owners. In addition, a study evaluating whether the volume of pleural, pericardial or peritoneal effusions may be useful in determining an underlying cause may be helpful to general practitioners who do not have access to advanced diagnostics.
Conclusions
This study highlights that bicavitary effusion in cats is commonly caused by neoplasia or cardiac disease. Our data suggest that, overall, cats with bicavitary effusion likely have a guarded to poor prognosis, particularly if there is a neoplastic aetiology. It suggests that cats with cardiac disease might have a better prognosis, which might prompt clinicians to evaluate for evidence of congestive heart failure early on in their diagnostic evaluation. Overall, the information presented here should aid clinicians in decision-making and prognostication when bicavitary effusion is identified in a feline patient.
Supplemental Material
Supplementary material 1:
Details regarding laboratory analysers for generation of haematological, serum biochemical and other clinicopathologic data.
Supplemental Material
Supplementary material 2:
Details regarding ultrasound, CT and radiography systems utilised for image acquisition.
Footnotes
Author note
A version of this abstract was presented at the British Small Animal Veterinary Association (BSAVA) Congress, 2021, Manchester, UK.
Supplementary material
The following files are available as supplementary material:
Supplementary material 1: Details regarding laboratory analysers for generation of haematological, serum biochemical and other clinicopathologic data.
Supplementary material 2: Details regarding ultrasound, CT and radiography systems utilised for image acquisition.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
