Abstract
Case series summary
This case series describes five cats with cutaneous adverse events after subcutaneous administration of frunevetmab, a felinised anti-nerve growth factor monoclonal antibody, including histopathological findings in one case. All cats displayed moderate to severe pruritus resulting in self-trauma to the neck and/or head, causing lesions ranging from superficial dermatitis to alopecia and ulcerations. There were no reactions at the injection sites. In one cat, clinical signs developed after the second frunevetmab dose the cat received, with no reaction noted after the first dose. For the remaining cats, clinical signs were observed after their first dose of frunevetmab. The onset of the first episode of pruritus and self-trauma was 3–18 days after the most recent frunevetmab injection. Three cats had one or more additional frunevetmab injections after the original adverse event and all had subsequent reactions. Subsequent reactions were either similar in time frame or occurred more rapidly, with similar or more severe pruritus compared with the original reactions. Treatments and outcomes varied between cases.
Relevance and novel information
Frunevetmab is a novel, monthly injectable monoclonal antibody for the management of pain associated with osteoarthritis in cats. This is the first published report detailing the nature of cutaneous adverse events associated with this treatment, and the first report of the histopathological findings.
Introduction
Degenerative joint disease (DJD) is highly prevalent in senior cats, with one study reporting radiographic evidence of DJD in 90% of cats aged over 12 years. 1 While chronic pain can be difficult to assess in cats, prospective studies have demonstrated improvements in mobility with analgesia.2–4 Most pharmacological analgesics require daily or multiple-times-per-day oral administration, which can be challenging for both cats and their owners, and has the potential for injury to the owner. 5 Non-steroidal anti-inflammatory drugs (NSAIDs) have traditionally been considered the mainstay for the management of chronic pain owing to osteoarthritis (OA) in dogs and cats. 6 However, as OA and chronic kidney disease (CKD) can occur concurrently in up to 70% of feline cases, there is often a reluctance to prescribe long-term NSAIDs in older cats.7,8 While a recent review has indicated that this reluctance may not be fully justified, alternative medications may be sought due to the concern about renal toxicity. 9
A recent advance in the management of chronic pain in cats has been the development of a felinised anti-nerve growth factor (NGF) monoclonal antibody (mAb), frunevetmab (Solensia; Zoetis). 10 NGF is a cytokine expressed at sites of inflammation, such as joints affected by DJD, and has been recognised as a major contributor to the generation, propagation and sensation of pain. 11 NGF is also involved in nociceptor sensitisation, potentially resulting in hyperalgesia and/or allodynia.11,12 Frunevetmab blocks the activity of feline NGF and has been shown to be effective in reducing pain in cats diagnosed with DJD.10,13,14 Frunevetmab is the only anti-NGF mAb approved for use in cats in the USA and has recently been recommended as a first-tier medication for the treatment of OA pain in cats.8,15
There is limited literature regarding the adverse events of frunevetmab administration. In two studies assessing efficacy, cutaneous adverse events were reported with significantly greater frequency in cats receiving frunevetmab vs placebo, and these findings are presented in the manufacturer’s prescribing information.13–15 In one study, 32/182 (18%) cats receiving frunevetmab developed skin-related adverse events compared with 8/93 (9%) cats receiving placebo. 13 Of the cats receiving frunevetmab, 11/182 (6%) had dermatitis and eczema, 10/182 (5.5%) had alopecia and 7/182 (3.8%) had pruritus, although it is unclear whether the cats had these conditions simultaneously and lesion distribution was not described. 13 In another study, 16/85 (19%) cats receiving frunevetmab developed dermatitis/eczema, 4/85 (5%) had alopecia (distribution not stated) and pruritus was not reported. 14 Again, it is unclear whether pruritus, alopecia and dermatitis occurred simultaneously. This latter study concluded that the adverse events were likely related to wearing an activity-monitoring collar. However, only 1/41 (2%) cats receiving placebo (also wearing collars) developed dermatitis/eczema and none had alopecia. 14
The current case series describes cutaneous adverse events after the administration of frunevetmab in one cat, including histopathological findings. It also describes four additional cats with cutaneous adverse events after the administration of frunevetmab, including the temporal course, distribution, severity, treatments and outcome.
Case series description
The principal case in this report (case 1), a 6.0 kg, 14-year-old, female spayed domestic shorthair, received a frunevetmab injection (7 mg) subcutaneously between the shoulder blades in September 2022 in Queensland, Australia. It was an indoor-only cat without access to other cats. Four weeks previously, the cat had received its first dose of frunevetmab, with no adverse reaction observed. The cat had no history of pruritic skin disease, although it had experienced a mild, transient, localised reaction to transdermal methimazole over 1 year before receiving frunevetmab. Nine days after the second frunevetmab injection, an area of ulceration was noted on the right ventrolateral neck. There was no reaction at the injection site. Concurrently, the cat was noted to be moderately pruritic, using its hindlimbs to scratch its neck and the right side of its face. Twelve days after the second frunevetmab injection (40 days after the cat’s first dose), the cat presented for investigation of the neck lesion. At this time, the neck lesion appeared ulcerated and moist with suspected secondary bacterial infection. No ectoparasites were macroscopically identified. Cytology from the right ear revealed cocci and occasional Malassezia organisms. Over the next 11 weeks after presentation, the cat was treated with multiple different medications, including multiple doses of long-acting parenteral antibiotics, oral and parenteral corticosteroids, topical antiseptic application and Elizabethan collars (Table 1). The pruritus and the severity of the lesion (area affected and extent of ulceration) appeared to wax and wane, with some temporary, moderate improvement in pruritus noted after the administration of corticosteroids. However, the lesion did not resolve, and the cat continued to periodically traumatise its right ventrolateral neck, causing the lesion to enlarge and worsen in appearance (Figure 1). Thirteen weeks after the second frunevetmab injection, a surface swab for bacterial culture and an incisional biopsy for histopathology were performed. Bacterial culture yielded a moderate growth of Staphylococcus felis (susceptible to all antimicrobials tested) and a light growth of the environmental organism Stenotrophomonas maltophilia (antimicrobial susceptibility testing was not performed).
Treatments trialled in case 1: a pet cat that developed ulcerative skin lesions 9 days after a second monthly injection of frunevetmab, and before excisional biopsy at 18.5 weeks after the injection; an incisional biopsy was performed at 13 weeks after the injection

Topical or oral interventions/medications were not tolerated well by the patient and were either discontinued or not repeated
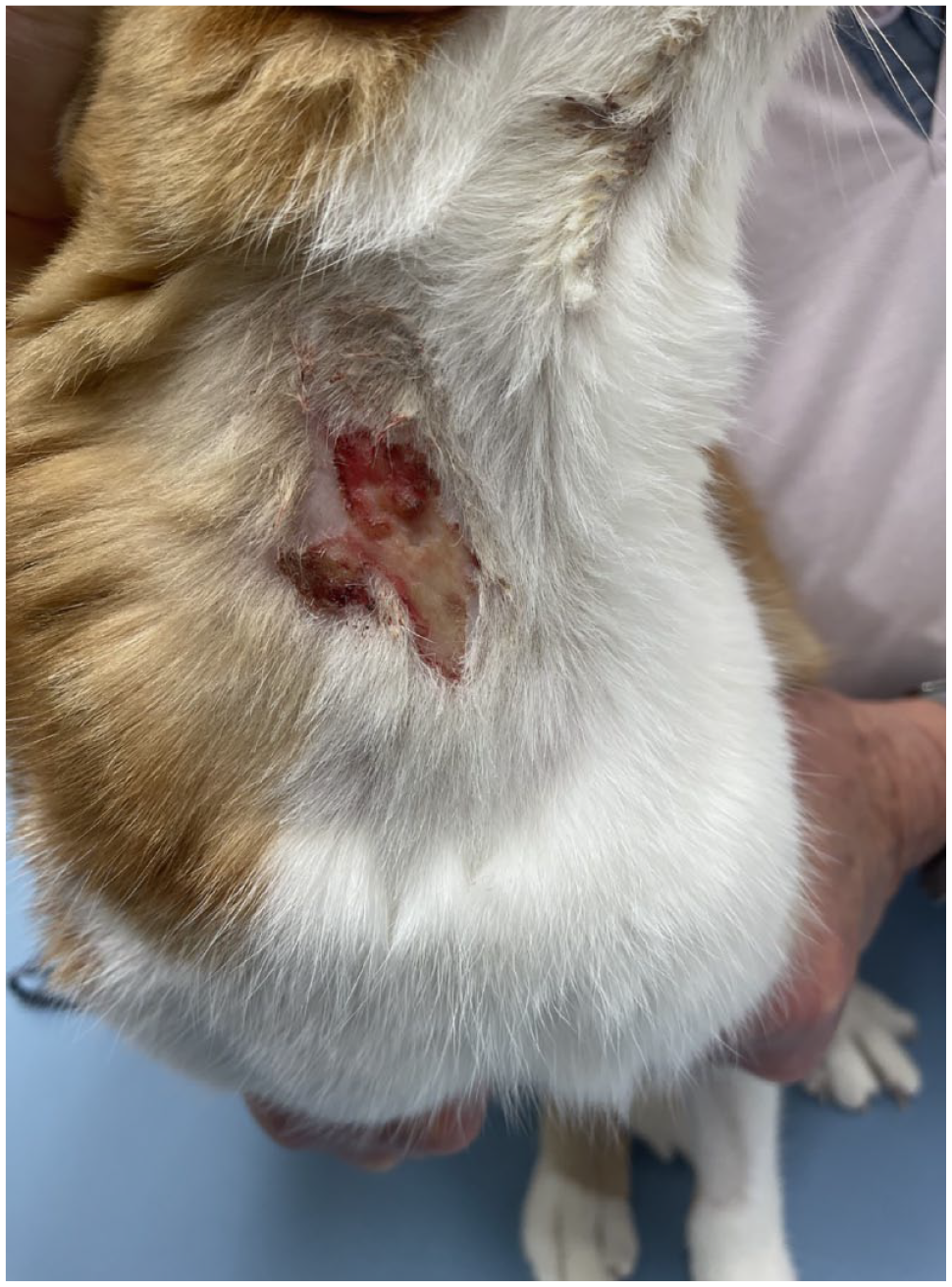
Case 1: 16 weeks after the second frunevetmab injection (3 weeks after the incisional biopsy). There is a demarcated area of ulceration on the right ventrolateral neck
Histopathology revealed epidermal ulceration overlain by a serocellular crust (Figure 2). Necrosis extended to the superficial dermis. Beneath the ulceration, the superficial dermis contained a relatively mild perivascular inflammatory infiltrate consisting of mainly neutrophils with low numbers of mononuclear cells and mast cells. There was minimal extension of inflammation into hair follicles at the junction of ulcerated and intact epidermis, and in one follicle, luminal keratin contained coccoid bacteria. Additional changes included minor superficial dermal fibroplasia. Deeper levels in the tissue block revealed a subcorneal pustule at the junction of ulcerated and intact epidermis. A periodic acid-schiff (PAS) stain did not reveal fungi.

Case 1: incisional biopsy of skin 13 weeks after the second frunevetmab injection. There is epidermal ulceration that is overlain by a serocellular crust. Necrosis extends to the superficial dermis. Beneath the ulceration, the superficial dermis contains a relatively mild perivascular inflammatory infiltrate, consisting of mainly neutrophils. There is an abrupt transition to intact, hyperplastic epidermis. Haematoxylin and eosin, ×4
After the incisional biopsy, further medications were trialled, including oral antibiotics and multiple doses of parenteral corticosteroids (Table 1). Again, pruritus improved only temporarily after corticosteroid doses and the skin lesion did not resolve. A total of 18.5 weeks after the second frunevetmab injection, owing to a lack of response to medical treatment, the neck lesion was surgically resected. Histopathology of the excised specimen revealed epidermal ulceration, superficial bacterial infection (cocci consistent with Staphylococcus species) and superficial dermal fibrosis and fibrovascular hyperplasia accompanied by relatively mild inflammation (Figures 3 and 4). Additional changes included patchy subcorneal pustules, and atrophy and loss of hair follicles. After excision of the lesion, the pruritus improved dramatically and the skin healed completely. The cat continued to demonstrate occasional, very mild pruritus, which was managed with subcutaneous dexamethasone injections for the first 2 months postoperatively. While the pruritus occurred spontaneously, the owner also described pruritus/muscle twitching being triggered by touch around the cat’s face/neck. At the time of submission of this manuscript (6 months after the excisional biopsy), no further skin lesions have developed. Furthermore, pruritus has now completely resolved and the cat has not required any medications for the past 4 months.

×4 Case 1: excisional biopsy of skin 18.5 weeks after the second frunevetmab injection. There is a demarcated epidermal ulcer, the edge of which is overlain by a serocellular crust. Necrosis extends to the superficial dermis. Beneath the ulceration, the superficial dermis exhibits fibrosis and fibrovascular hyperplasia accompanied by relatively mild inflammation. Haematoxylin and eosin, ×4

×20 Case 1: excisional biopsy of skin 18.5 weeks after the second frunevetmab injection. The ulcerated area is overlain by a serocellular crust. Necrosis extends to the superficial dermis. Within the crust and necrotic debris, there are numerous clusters of coccoid bacteria. Haematoxylin and eosin, ×20
Additional cases of cutaneous adverse reactions to frunevetmab were identified by searching for reports on Veterinary Information Network message boards and via online veterinary dermatology forums. Three veterinarians contributed four additional cases (cases 2–5), with a total of nine adverse events documented between August 2022 and January 2023. The contributing veterinarians provided information via a questionnaire regarding signalment, clinical signs, cutaneous lesions, previous dermatological disease, treatments and outcome (Table 2). Two cats had three adverse reactions each, one cat had two reactions and one cat had one reaction. None of these additional cases had received a dose of frunevetmab without experiencing a cutaneous reaction. A previous history of skin disease was reported in case 3 (transient facial pruritus after administration of a behavioural supplement containing l-theanine, magnolia/phellodendron and whey protein concentrate) and case 5 (seasonal pruritus).
Summary of cutaneous adverse reactions in five cats after administration of frunevetmab*

In case 1, clinical signs developed after the second frunevetmab dose the cat received, with no reaction noted after the original dose. For cases 2–5, clinical signs were observed after the cats’ first dose of frunevetmab
Principal case
Treatments/measures believed to benefit case/result in clinical resolution
DSH = domestic shorthair; EOD = every other day; FS = female spayed; IBD = inflammatory bowel disease; MN = male neutered
For these additional cases (cases 2–5), the cats were aged 8.5–14 years. Frunevetmab (7 mg) was administered subcutaneously, either between the shoulder blades or over the hip region for the management of OA pain. The adverse reactions began 3–18 days after the cats’ first frunevetmab injections. Clinical signs in all cases consisted of mild to severe pruritus of the neck and/or head, distant from the site of injection. Signs of self-trauma, including excoriation with crusting, occurred in all cases. Of the four cats, three (cases 2, 4 and 5) also developed alopecia in the regions of pruritus. Cases 2, 3 and 4 received subsequent doses of frunevetmab, which elicited further cutaneous reactions with similar or more severe pruritus occurring in the same time frame or more rapidly compared with prior reactions. Treatments varied among cases (Table 2). In the principal case (case 1), although corticosteroids were found to improve pruritus, surgical resection was the only treatment that resolved the skin lesion. In case 2, amoxicillin/clavulanic acid, oral antihistamine and topical hydrocortisone did not appear to improve the clinical signs. After the third episode, pruritus resolved after oral prednisolone at a dose of 1 mg/kg q24h. In this case, prednisolone was continued in the long term at 1 mg/kg every second day as it improved the cat’s mobility. In case 3, severe pruritus after the second frunevetmab injection improved rapidly after the administration of oclacitinib (Apoquel; Zoetis) for 10 days. Specifically, pruritus resolved in less than 24 h, which compared favourably with the 26 days for resolution of pruritus after the cat’s first adverse reaction was treated with topical fatty acids and ceramide. After 10 days of treatment with oclacitinib, the medication was discontinued for 3 days before starting again due to the relapse of pruritus. Four days later, it was possible to discontinue the medication without any further pruritus. Case 4 did not receive any specific treatment and clinical signs resolved 4 weeks after the third adverse reaction. In case 5, prednisolone administered at 2.5 mg every third day for three doses did not improve pruritus. Nail trimming was believed to limit the damage caused by scratching, resulting in clinical resolution. In cases 2–5, resolution of clinical signs occurred within 2–6 weeks after the final dose of frunevetmab.
Discussion
This case series describes cutaneous adverse events in five cats after the administration of frunevetmab, including the histopathological findings in one cat. Pruritus/self-trauma was a consistent feature, and it is suspected that skin lesions or alopecia were a consequence of self-trauma in all cases. Histopathology in case 1 demonstrated ulceration with superficial dermal fibrosis and neutrophilic inflammation. These findings could also be caused entirely by self-trauma with secondary superficial bacterial infection. There was no eosinophilic inflammation to specifically indicate a hypersensitivity reaction. The histological lesion had similarities to feline idiopathic ulcerative dermatitis (FIUD), a poorly understood condition associated with self-trauma. FIUD is characterised by a demarcated, crusted, non-healing ulcer that occurs most commonly on the dorsal neck or between the scapulae. Lesions have also been reported on the lateral neck and temporal areas.16 –20 While some cases of FIUD have been shown to respond to glucocorticoids, topiramate, oclacitinib, surgical resection, bandaging/prevention of self-trauma or environmental enrichment, some authors regard the prognosis as guarded, as lesions may be refractory to medical treatment or may recur despite surgical resection.16–22 In the present case series, a range of responses was noted, from spontaneous resolution in case 4, to the principal case (case 1) in which the skin lesion was refractory to medical treatment and required surgical resection.
In FIUD, it has been speculated that cats self-traumatise due to hyperstimulation of nerve endings (neuropathic itch syndrome).17,20 It is believed that the face, interscapular and neck regions are predisposed to neuropathic itch, as they contain dense aggregates of sensory nerves and are also easily accessible for cats to scratch.17,19 As NGF acts on sensory afferent nerves, it is conceivable that some cats receiving frunevetmab could develop altered nerve sensation, leading them to self-traumatise. 23 This would explain why the lesion distribution and histopathology are similar to FIUD. The human literature reports cases of paraesthesia, hyperaesthesia and a burning sensation in 5–10% of patients receiving anti-NGF mAbs.24,25 In these studies, most of the sensory abnormalities resolved within 1 month, although reports of persistent symptoms lasting up to 5 months have also been documented. The relatively transient nature of these symptoms has been speculated to be due to a temporary change in the sensitivity, or ‘tone’, of afferent nerve fibre populations. 26 A temporary change in sensitivity could also explain why the clinical signs resolved without treatment in one case in the present report (case 4).
The pruritus exhibited by one cat (case 3) after its second reaction to frunevetmab resolved rapidly (within 12 h) after off-label treatment with oclacitinib, compared with its previous (first) reaction when only topical treatments were used. Oclacitinib is a Janus kinase inhibitor, which blocks JAK1-dependent cytokines involved in inflammation and pruritus. This medication is used in the treatment of canine atopic dermatitis and has experimentally been shown to reduce pruritus in 63% of cats if administered 1 h before an injection of IL-31, a pruritogenic cytokine. 27 Oclacitinib has also been reported to be successful in the treatment of one case of refractory FIUD. 22
In the present case series, other treatments considered by veterinarians to improve the clinical course were surgical resection (1 case), subcutaneous dexamethasone (1 case), oral prednisolone (2 cases) and trimming the nails on the hindfeet to prevent further self-trauma (1 case). In case 1, other medical treatments were not trialled as the cat was not amenable to oral medications. Surgery resolved the lesion, although ongoing, very mild episodic pruritus continued for a further 2 months. Ongoing self-trauma and/or lesion recurrence has previously been documented in cases of FIUD treated with surgical resection. 21
Topiramate, an anticonvulsant medication, was administered in a reported case of refractory FIUD, leading to improvement within 2 weeks and resulting in long-term remission. 17 While it is difficult to draw conclusions on potential therapeutic options in cats, treatments that are effective for FIUD may also prove effective for cutaneous reactions secondary to frunevetmab. Gabapentin or topiramate could be considered due to their inhibitory effects on neuronal excitability.17,28–30 Whether these medications could be administered concurrently with frunevetmab to reduce the severity of a cutaneous reaction in cats that have experienced prior episodes warrants further investigation.
As previously mentioned, an early study of the efficacy and safety of frunevetmab suggested that activity-monitoring collars were the cause of observed skin disorders, including dermatitis/eczema and alopecia. 14 This assumption is also repeated in the manufacturer’s prescribing information. 15 However, a significantly greater number of cats receiving frunevetmab were affected compared with those receiving placebo, despite all cats wearing the collars. In addition, none of the cats in the present case series had collars in place at the time of their frunevetmab administration or their subsequent cutaneous reactions. These findings suggest that the administration of frunevetmab results in an abnormal sensation around the neck, which collars may or may not exacerbate.
In the present case series, of the cats that received multiple frunevetmab injections, cutaneous adverse reactions were often evident more rapidly at subsequent injections. This could indicate a sensitisation component or a cumulative pharmacological effect. To determine whether this is a true pattern, larger, prospective studies are required.
It should be noted that all four of the veterinarians who contributed cases to this series continue to recommend frunevetmab for cats diagnosed with reduced mobility due to OA. They do, however, warn owners of possible cutaneous adverse events. For cats with a single episode of pruritus and traumatic skin lesions after frunevetmab, owners should be warned that subsequent injections may lead to repeat cutaneous reactions. For each cat, the risk of recurrent adverse events must be weighed against the clinical benefits of the drug.
Conclusions
This case series further characterises cutaneous adverse events that may occur after the administration of frunevetmab. We postulate that the mechanism is primarily hyperaesthesia/paraesthesia with neuropathic itch resulting in self-trauma, causing alopecia, ulceration and possible secondary bacterial infection. There is clinical and histological similarity to FIUD, as the lesions in these cases and FIUD appear to be fundamentally self-traumatic in nature. Larger studies including further histopathological evaluation are required to better understand the pathogenesis, guide treatment and anticipate the outcome.
Footnotes
Acknowledgements
The authors acknowledge and thank the veterinarians who supplied their medical records for this series. Participating clinics include The Cat Clinic, Mount Gravatt, Brisbane, Queensland, Australia; Bridge West Animal Hospital, Napanee, Ontario, Canada; and 518 House Call Vet PLLC, Albany, NY, USA. A special thanks to the owner of the principal case used in this series for supplying detailed information regarding her own pet.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
