Abstract
Practical relevance:
Feline inflammatory airway diseases, including (but not limited to) asthma, chronic bronchitis and bronchiectasis, are common and incurable disorders. These diseases require lifelong therapy and may result in substantial morbidity and, in some cases, mortality. Goals of therapy include reduction or resolution of clinical signs and the underlying pathologic processes driving those clinical signs. Inhalational therapy has the advantage of topical delivery of drugs to target tissues at higher doses with fewer systemic effects than oral medications. There are multiple options for delivery devices, and proper selection and training on the use of these devices - including acclimation of the cat to the device - can maximize therapeutic efficacy.
Aim:
As inhalational therapy is uncommonly used by many veterinarians and owners, this review article provides a foundation on the selection and use of devices and inhalant medications for specific feline inflammatory airway diseases. Cats present a unique challenge with respect to the use of inhalers, and easy-to-follow steps on acclimating them to the devices are provided. The review also discusses the mechanics of inhalational therapy and helps clarify why certain medications, such as albuterol (salbutamol), fluticasone or budesonide, are chosen for certain diseases. The ultimate aim is that the practitioner should feel more comfortable managing common airway diseases in cats.
Evidence base:
In compiling their review, the authors searched the veterinary literature for articles in English that discuss inhalational therapy in cats, and which focus primarily on inhaled glucocorticoids and bronchodilators. While most literature on inhalational therapy in cats is based on experimental feline asthma models, there are some studies demonstrating successful treatment in cats with naturally occurring inflammatory airway disease.
Introduction
Inflammatory airway disease, a cause of cough, wheeze and/or episodic respiratory distress, is a common reason cats present for veterinary care. Representing a wide spectrum of disorders, inflammatory airway disease can be subdivided anatomically into disorders affecting large airways (segmental and subsegmental bronchi and larger bronchioles) or small airways (bronchioles <2 mm in diameter lacking cartilage in their walls). 1 This review will focus on inhalant medications for three chronic large airway disorders in cats: allergic asthma, chronic bronchitis and bronchiectasis. Infectious bronchitis is excluded as it is not commonly treated with inhalational therapy. Modification of current inhalational therapy approaches will be required to target small airways (bronchiolar disorders) and awaits future study.
While systemic therapy remains the most common route of treatment for inflammatory airway disease in cats, there is a strong rationale for delivery of topical medications to the respiratory tract. Specifically, inhalational therapy allows for higher local doses of drug, opportunities to optimize pharmacokinetics and pharmacodynamics, and decreased systemic immune and endocrine effects.
Numerous devices exist for inhalational therapy, each having different characteristics. Major challenges arise due to patient resistance to these devices, and practitioners need to know how to mitigate these challenges in order to optimize therapy. Importantly, suggestions on how to train cats to tolerate such devices are critical to maximize acceptance and efficacy of treatment.
Respiratory disease can be a challenging prospect, but improved outcomes with inhalational therapy may bring a breath of fresh air to our feline patients.
Feline inflammatory airway diseases and general pharmacologic management
Allergic asthma
Allergic asthma in cats is orchestrated by a T helper 2 cell immune response to aeroallergens, with cytokines driving eosinophilic inflammation, airway hyper-responsiveness (defined as the tendency of airways to excessively narrow in response to a variety of stimuli that in healthy individuals would produce little or no effect) and airway remodeling. 2 The primary treatments are glucocorticoids and bronchodilators, which target inflammation and airway hyper-responsiveness/bronchospasm, respectively. Asthmatic cats develop airway eosinophilia and require lifelong treatment with glucocorticoids, but only a subset develop severe bronchospasm (‘status asthmaticus’) for which bronchodilators represent the most important life-saving therapy. 3 Glucocorticoids and bronchodilators can be given by injection, oral administration or inhalation.
A variety of other treatments have been investigated in experimental feline asthma models including, among others, ciclosporin, cyproheptadine, cetirizine, zafirlukast, maropitant, lidocaine, N-acetylcysteine, allergen-specific immunotherapy, adipose-derived mesenchymal stem cells, tyrosine kinase inhibitors and immunomodulators.4-16 While cyproheptadine, cetirizine, zafirlukast and maropitant failed to reduce airway inflammation, and N-acetylcysteine had unacceptable side effects, the other treatments hold promise and await clinical trials in pet cats.5,6,13-15 Most of these treatments are administered systemically, except for lidocaine and N-acetylcysteine, which have been evaluated as topical therapies.10,14
Chronic bronchitis
A variety of infectious, inflammatory and toxic insults can induce damage to ciliated airway epithelial cells, leading to self-perpetuating mucociliary dysfunction, inflammation characterized by non-degenerate neutrophils, mucus hypersecretion and airway remodeling.3,17-21 The initial insult is generally not identified at the time of diagnosis. Glucocorticoids represent the primary treatment modality. While bronchodilators have anecdotally been used, cats with chronic bronchitis (in contrast to feline asthma) do not develop life-threatening bronchospasm. 3
Because chronic bronchitis and asthma have different airway cytology (neutrophilic vs eosinophilic, respectively), the main way to differentiate them is via airway sampling. A study investigating the potential for non-invasive clinical differentiation showed that signalment, a single clinical sign, hematologic parameters or changes on thoracic radiography were not discriminatory, underscoring the key role of airway cytology in diagnosis. 18 Blood eosinophilia was more common in patients with asthma compared with those with bronchitis, with numerous other factors, including radiographic findings, being unhelpful for differentiation.
Bronchiectasis
Bronchiectasis, defined as a pathologic dilation of bronchi due to destruction of the elastic and muscular components of the airway walls, is uncommon in cats. 22 Impairment of mucociliary function leads to a cycle of inflammation and predisposition to bacterial infection. After identifying and addressing the underlying cause, where possible, treatment focuses on glucocorticoid and, if secondary bacterial infection is documented, antimicrobial therapy.
Parasitic bronchitis
Inflammation of the airways can result from infection by Aelurostrongylus abstrusus, Dirofilaria immitus (ie, heartworm-associated respiratory disease) and Toxocara cati, among others.21,23 Identification of the specific type of parasite will guide anthelmintic therapy. Additionally, patients with this group of disorders benefit from antiparasitic prophylaxis.21,23,24 In some cases, anti-inflammatory doses of glucocorticoids may help control bystander damage to lung tissue during acute parasite die-off. Glucocorticoids are administered systemically, as the time to onset of action of inhaled glucocorticoids is delayed (see later). There is no clear role for broncho dilators, with conflicting results in the published literature.23-26 Unlike other types of large airway disease, the pharmacologic goal in cases of parasitic bronchitis is cure.
Bronchiolar disorders (small airway disease)
A group of disorders targeting the small airways (<2 mm diameter) interposed between the large airways and pulmonary acini has been recently recognized and characterized. 1 In cats, clinical signs of bronchiolar disease overlap with those of many other respiratory diseases, thoracic radiography is of low utility diagnostically and there are no pathognomonic cytologic findings, making thoracic CT and histopathology critical, 1 although often impractical. There have been no studies investigating treatment (which in large part will depend on the underlying cause), but there is reason to believe that inflammatory small airway disorders would be amenable to modified therapy with inhalants (ie, extrafine particle formulations). 27
Devices for inhalational therapy
Drugs must be delivered to their site of action within the lower respiratory tract, requiring passage without excessive loss through the upper aerodigestive tract. Feline patients present a challenge as it is not possible to coordinate the timing between the release of medication (actuation) and inhalation. Delivery devices help optimize medication delivery while minimizing deposition in the throat (ie, the amount swallowed vs inhaled); they also slow the velocity of inhaled drugs to improve delivery deep in the lung. This can be particularly helpful in cats with respiratory disease associated with altered breathing patterns leading to an abnormally low volume of air moving in and out of the lungs during a typical breath.
Devices available to deliver inhaled medications include pressurized metereddose inhalers (pMDis), breath-actuated (BA)-pMDis, dry powder inhalers (DPis) and nebulizers. The following discussion focuses on pMDis and nebulizers. BA-pMDis and DPis require a conscious inhalation effort to draw in the medication as the lips are pursed around the mouthpiece; as such, they are not applicable for clinical use in cats.
pMDIs
A pMDI, colloquially known as an ‘inhaler’, delivers medication via a compressed propellant that aerosolizes the drug. in cats, pMDis work with spacers (hollow tubes regulating flow of aerosolized drugs) called valved holding chambers (VHCs), which are fitted to a face mask (Figure 1).

Use of a pressurized metered-dose inhaler (pMDI) with a spacer (valved holding chamber) attached to a tight-fitting face mask in a cat
Ideally, pMDIs require good actuation-inhalation coordination, an inspiratory flow of around 30 l/min, and a postinhalational breath-hold. 28 Feline patients are unable to coordinate inhalation with actuation, and cannot be instructed to take slow, deep breaths or use a postinhalational breath-hold. Use of a VHC with face mask allows owners to actuate drug into the holding chamber, and the one-way valve permits the cat to breathe in the drug during tidal breathing. An unpublished study, discussed within an article by one of the present authors (CRR) and colleagues, 14 documented the amount of aerosol reaching the lungs using nuclear scintigraphy, and found it to be only 1% in cats where a spacer with a tight-fitting face mask was used. There are, however, fewer systemic side effects when drugs are delivered by pMDI compared with other routes.29,30 Local reactions, specifically cutaneous demodicosis of the muzzle, have been described with inhaled glucocorticoids (Figure 2). 31

(a) Cutaneous demodicosis of the muzzle of a cat secondary to long-term fluticasone inhalational therapy. (b) A skin scrape of the muzzle of the cat, confirming the diagnosis
To ensure that a cat can tolerate a pMDI with a tight-fitting face mask, acclimation to the device is recommended (see box on ‘Training cats to tolerate inhalational therapy’).


(a,b) Training cats to acclimate to an inhaler device using treats placed in the face mask. Images courtesy of Dr Sarah Ellis
Nebulizers
Nebulizers are devices that turn liquids into an aerosolized mist (Figure 4) that can be delivered to cats using a face mask. Handheld nebulizers powered with batteries are recommended for at-home use. ‘Flow-by’ inhalation of nebulized drugs without the use of a face mask, while appealing in cats for ease of administration, has not been studied, but would be expected to result in substantial drug loss into the environment.
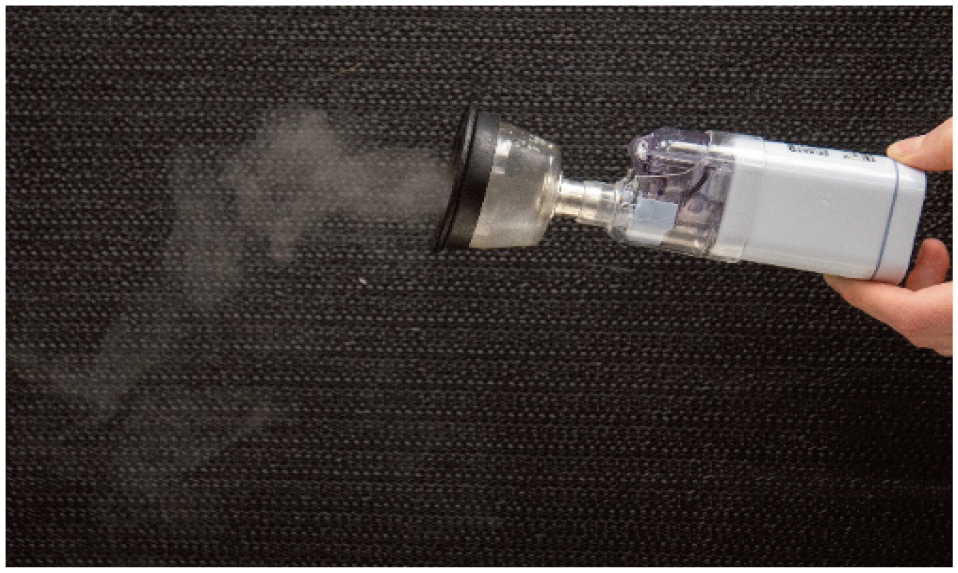
A plume of nebulized aerosol being generated by a handheld, battery-powered nebulizer. The device would be attached to a face mask for drug delivery to a cat
Compared with other inhalant devices, nebulizers are considered the most user-friendly. 32 They can be divided into three broad types: jet nebulizers, ultrasonic nebulizers and vibrating mesh nebulizers. 32 Jet nebulizers use compressed air to aerosolize medications; while relatively inexpensive, treatment times are longer, and the technology is comparatively inefficient and noisy. Ultrasonic nebulizers utilize a high-frequency vibrational piezoelectric crystal to generate waves in the liquid medication, resulting in fine droplet formation. They are more expensive than jet nebulizers, but are more efficient (with shorter treatment times) and are quiet. The third type of nebulizer generates aerosols by using a piezoelectric element to vibrate a mesh in contact with the medication. Vibrating mesh nebulizers are similarly expensive, but are efficient (with the shortest treatment times) and quiet, and importantly are portable for at-home use. To date, there is no scientific literature comparing the efficacy of different nebulizer types in cats, but two studies have been performed using one brand of vibrating mesh nebulizer,10,33 which is what the authors also use in clinical practice.
In general, numerous medications can be used to treat a variety of respiratory disorders via a nebulizer, including (but not limited to) sterile saline, short- and long-acting beta-2 (β2) agonists, certain glucocorticoids, lidocaine, N-acetylcysteine and various antibiotics. Many of these have not been investigated in cats, aside from a few studies discussed below in the section ‘Inhalant medications used in cats with inflammatory airway disease’. It is not recommended to nebulize N-acetylcysteine as monotherapy in asthmatic cats as it has been shown to increase airway resistance in experimentally induced feline asthma. 14
Device care
Regardless of the device used, keeping it clean is important to maintain efficacy of the device and minimize the risk of inadvertent nebulization of microbes. Following the manufacturer’s recommendations for cleaning VHCs was reported to be sufficient for bacterial decontamination. 34 A recent prospective study culturing spacers and masks from 66 client-owned cats with inflammatory airway disease found that 58% had bacterial growth, with most contamination occurring within the mask. 35 Owner-reported cleaning frequency was not associated with contamination of the device nor the degree of contamination. 35 The study was not designed to determine whether any of the cats had bacterial growth in their airways contributing to clinical signs; however, most bacteria were considered part of the microbiota of the skin or those present in the environment. 35 Further research is needed to determine the clinical significance of cultivable bacteria from delivery devices and masks.
Optimizing inhalational therapy
Factors determining therapeutic efficacy
All types of inhalational therapy rely on several factors to determine efficacy:
✜
✜
✜
✜
Drug deposition
Most inhalational devices produce particles in the 1-5 µm range, which have the greatest potential to deposit in the lungs. 42 Compared with adult humans, cats have narrower airway diameters and higher intraluminal airflow, which likely makes the optimal particle size range for aerosols smaller. Healthy cats administered a radiopharmaceutical agent using a nebulizer, spacer and face mask were documented to have uptake in the ‘lungs’; 43 however, the study did not attempt to discriminate or calculate the percentage of the radiopharmaceutical in the large airways, small airways or pulmonary parenchyma.
Computational fluid dynamics represents a means of non-invasively modeling conditions to study aerosol transport and deposition, and has been applied to cats. 44 Nine different combinations of devices were modeled using CT images of client-owned cats, allowing for assessment of pMDI delivery of albuterol (salbutamol). The influence of spherical vs conical masks over the muzzle, shorter vs longer spacer devices and actuation through an endotracheal tube was specifically investigated. 44 Not surprisingly, independent of the model, the most drug was deposited within the device and upper airways.
Airway obstruction and altered breathing patterns, which are common in cats with inflammatory airway disease, limit therapeutic deposition of drugs. Unlike in people where mouth (vs nasal) breathing and slow, deep respirations to enhance drug retention time are used to improve drug distribution, cats cannot perform these maneuvers on command. 45
Prognosis
A case control study involving 54 cats with and 121 cats without inflammatory airway disease (the latter group inclusive of cats with respiratory distress of other etiologies) that presented to the emergency room, showed that cats diagnosed with inflammatory airway disease had a survival rate of 94%. 46 Of note, most patients in this study did not undergo a comprehensive diagnostic evaluation to fully characterize the underlying etiology of their respiratory disease. In general, cats with inflammatory airway disease can be well managed and seem to have a fair to excellent prognosis in the long term.
Inhalant medications used in cats with inflammatory airway disease
Common medications for inhalational therapy in cats, with suggested doses, are listed in Table 1.
Inhalant medications used to treat feline asthma

LABA = long-acting 32 agonist; SABA = short-acting 32 agonist
Glucocorticoids
Lifelong therapy with glucocorticoids is key to attenuating the airway inflammation that drives clinical signs and leads to airway hyper- responsiveness and remodeling. While oral glucocorticoids (OGCs) are commonplace, inhaled glucocorticoids (IGCs) have appeal because of their enhanced topical potency and minimal systemic bioavailability. For cats that may not tolerate OGCs because of comorbid conditions, such as congestive heart failure or diabetes mellitus, IGCs could represent a treatment option. 47
In healthy pet cats, IGC treatment (flunisolide 500 µg q12h) suppressed the hypothalamic-pituitary-adrenocortical axis, but to a lesser extent than OGCs, and had minimal systemic immunologic effects. 30 In pet cats with asthma or chronic bronchitis, inhaled budesonide (400 µg q12h) led to hypothalamic-pituitary-adrenocortical axis suppression in a minority (3/15) of study participants. 17 Unfortunately, studies with long-term follow-up are not available, with most evaluating response 8-12 weeks after treatment. This precludes the ability to extrapolate long-term effects of such medications.
Most investigations of IGCs have used experimental feline models of allergic asthma. One study showed that both oral and inhaled (flunisolide 250 µg q12h) glucocorticoids significantly attenuated airway eosinophilia when compared with placebo. 13 Another study investigated an IGC with a long-acting β2 agonist (fluticasone 500 µg/salmeterol 50 µg q12h) and determined it was equipotent to OGCs in reducing airway inflammation. 48 To determine the effective dose of IGCs, experimentally asthmatic cats were administered fluticasone at dosages of 44, 110 or 220 µg q12h, and airway eosinophilia was blunted at all three strengths. 49 These results suggest that a plateau effect can occur, such that giving more IGC does not always translate into better control of airway inflammation. A final study investigated adjunct glucocorticoid therapy in experimentally asthmatic cats undergoing allergen-specific immunotherapy; the aim was to control clinical signs during the period required for immunotherapy to induce immunologic tolerance to aeroallergens. 50 Cats receiving an OGC (prednisolone 10 mg q24h) - but not cats receiving an IGC (fluticasone 250 µg q12h) - had diminished efficacy of allergen-specific immunotherapy, suggesting that IGCs were critical to administer during immunotherapy. 50
Results of available studies performed using IGCs in cats with spontaneous inflammatory airway disease can be briefly summarized as follows:
✜ Cats in a research colony that had spontaneously developed chronic bronchitis were treated with 250 µg of fluticasone q24h and showed diminished airway inflammation. 19
✜ In pet cats with asthma or chronic bronchitis, administration of 400 µg of inhaled budesonide q12h led to improvement in both clinical signs and metrics of non-invasive pulmonary function testing. 17
✜ Pet cats with spontaneous asthma treated with 110 µg of fluticasone q12h had resolution of clinical signs and similar reductions in airway eosinophilia as cats treated with prednisolone (5 mg PO q24h). 51
✜ In terms of owners’ perceptions of improvement in clinical signs, the frequency and severity of cough were significantly lower after 2 months of treatment for asthma and chronic bronchitis, with most cats receiving OCGs, IGCs or both as a component of therapy. 40
✜ The barometric whole-body plethys-mography parameter Penh (‘enhanced pause’) is used as an indirect measure of airflow limitation. In 25 cats with inflammatory airway disease treated with 12 different protocols (all but one involving glucocorticoids), significant improvements in a 12-point clinical score and Penh were noted. 52
The results of these last two studies must be interpreted with caution as owner-perceived improvement in clinical signs does not always correlate with resolution of airway inflammation in cats with asthma and chronic bronchitis. 53 It is also important to note that when IGCs are administered, they are not immediate-acting; it is recommended to overlap treatment for 7-10 days with OGCs before discontinuing the latter.
Bronchodilators
Bronchodilators target the smooth muscles within the airway walls, which constrict either in response to specific sensitizing allergens (in allergic asthma) or a non-specific irritant/bronchoprovocant. They are thus critical to reverse airway hyper-responsiveness. In cats, broad classes of bronchodilators include β2 agonists, methylxanthines and, less commonly, anticholinergics.
As bronchodilators generally have weak or absent anti-inflammatory effects, they should not be administered as monotherapy in cats with inflammatory airway disease. Additionally, not all cats with inflammatory airway disease have clinically impactful bronchospasm. The authors’ recommendation is that bronchodilators be used primarily for patients with wheeze or expiratory respiratory distress.
In cats with status asthmaticus, bronchodilators represent the single most important treatment to directly address severe bronchospasm (see box, ‘Use of bronchodilators in status asthmaticus’).
β2 agonists
Administration of an albuterol pMDI in the home setting should be restricted to ‘rescue’ use in emergency situations, and not regular use. Paradoxically, β2 agonist overuse in humans has been linked to an increased risk of morbidity and mortality from bronchoconstriction. 57 This may in part be explained by the racemic mixture of albuterol in pMDIs, consisting of R- and S-enantiomers with opposite effects: R-albuterol is bronchodilatory with weak anti-inflammatory properties and is metabolized quickly from the lung, whereas S-albuterol is bronchoconstrictive and pro-inflammatory, with slower metabolism and clearance from the lung, allowing for its preferential accumulation and dominant effects.33,58 In healthy research cats, q12h use of aerosolized R,S-albuterol (but not R-albuterol) induced airway neutrophilia. 33 In experimentally asthmatic cats, q12h use of aerosolized R,S-albuterol (but not R-albuterol) exacerbated airway eosinophilia. 33 Thus, if inhaled albuterol is needed on a regular basis to control clinical signs, it is recommended that the non-racemic product R-albuterol (leval-buterol) pMDI is used.

Studies using inhaled bronchodilators in pet cats with inflammatory airway disease are lacking, and none report direct measures of improvement in airway resistance. In asthmatic pet cats, a published abstract suggested that use of an albuterol pMDI improved Penh. 59 The aforementioned study on client experiences associated with medications to treat lower airway disease documented that in 88% of cats administered bronchodilators, pMDIs were used; challenges in bronchodilator pMDI administration were reported by 31% of owners. 40 Notwithstanding that many of these cats concurrently received glucocorticoids with bronchodilators (in conjunction with environmental changes focused on air quality management), clients perceived significant improvements in treatment response after at least 2 months of therapy. 40 Other retrospective studies in naturally developing asthma and chronic bronchitis suggest bronchodilators (as a component of therapy) improve clinical signs; however, all except one have assessed oral bronchodilators.60-64 The sole retrospective study to include an inhaled bronchodilator (nebulized albuterol) reported this was administered in a single cat with ‘good effect’. 61
Methylxanthines
Methylxanthines are not recommended to be administered by inhalation in people, as they have poor bioavailability and are not well tolerated. 65 There are no studies investigating inhaled methylxanthines in experimental feline asthma or spontaneous feline inflammatory airway disease.
Anticholinergics
Two anticholinergic medications (ipratropium pMDI and tiotropium DPI) have been assessed in research cats.54,55,66,67 In healthy research cats, ipratropium alone or with a SABA as a pMDI attenuated muscarinic (carbachol)-induced bronchospasm when measured using the Penh parameter; the combination of the two drugs was synergistic. 55 Additionally, ipratropium was nebulized and, whether administered via a nebulizer or a pMDI, was considered short-acting compared with the tested LABA, and had a dose-dependent effect. 55
In an experimental feline asthma model in which allergen challenge was used as the bron-choprovocant, salbutamol, ipratropium or a salbutamol/ipratropium combination administered by a pMDI failed to reverse allergen-induced bronchospasm, as assessed using the Penh parameter. 54 However, these results contrasted with allergen-challenged cats with experimentally induced asthma that underwent bronchoscopy and bronchoalveolar lavage (procedures shown to induce airflow limitation); an improvement in Penh was documented in these cats when administered a salbutamol/ipratropium pMDI. 66
Using a feline model of acute cigarette smoke-induced airway inflammation, investigators evaluated tiotropium administered via a DPI under anesthesia (the drug was pumped in, not spontaneously inhaled). Beneficial effects on inflammation and reduced airway hyper-responsiveness in response to methacholine challenge were documented. 67
Miscellaneous medications
Nebulized lidocaine has been investigated as a novel anti-inflammatory agent, with beneficial effects on pulmonary mechanics in people with intractable cough and asthma. 68 In an experimental feline asthma model, nebulized lidocaine (2% solution administered at a dosage of 2 mg/kg q8h for 2 weeks) significantly reduced airway resistance induced by bronchoprovocation with methacholine, although there was no effect on airway eosinophilia. 10
Mucus hypersecretion is a prominent feature of inflammatory airway disease in cats. It can cause airflow limitation by narrowing the luminal diameters of the intrathoracic airways and, in cases where lobar bronchi are occluded, can lead to atelectasis. N-acetylcysteine has both mucolytic and antioxidative properties and was assessed in pet cats nebulized through a 38 l barometric whole-body plethysmograph chamber. 69 The authors of this published abstract reporting on 27 pet cats (only two of which were asthmatic) concluded that it was safe and did not adversely affect the Penh. 69 However, nebulization into a 38 l chamber likely resulted in negligible delivery to the airways, prompting a follow-up study in experimentally asthmatic cats; these received directly nebulized N-acetylcysteine through an endotracheal tube, with recording of ventilator-acquired pulmonary mechanics. 14 A mean ± SD increase in airway resistance after aerosol delivery of N-acetylcysteine of 150 ± 18% over baseline values was recorded, with every cat experiencing adverse effects. 14 Thus, inhaled N-acetylcysteine cannot be recommended for clinical use in cats.
Conclusions and future directions
Inhalational therapy should become more commonplace as practitioners, clients and pet cats become accustomed to devices and medications. Such therapy will hopefully have many benefits: improving measurable outcomes such as survival times, owner perceptions and/or the need for rescue medications; improving quality of life; and providing therapy for patients that might otherwise succumb to their disease without topical therapies. Exciting new research is expected as the use of inhalational therapy increases and the costs decrease.
Although significant data already exist in human medicine to guide clinical practice, the veterinary inhalational therapy literature, especially with respect to clinical trials in pet cats, is still in its infancy. A large proportion of the pubished feline studies involve experimentally induced asthma, which represents an important starting point but has limitations. Future studies should ideally be randomized, prospective and blinded, investigating large populations of naturally occurring inflammatory airway disease in cats. While this review should help veterinarians in their day-to-day work, additional study and expanding clinical expertise will shape future guidance.
Key Points
✜ Feline inflammatory airway disease encompasses a spectrum of large and small airway disorders which are considered chronic and manageable but require lifelong therapy.
✜ Inhaled therapies have clear advantages over oral medications for cats with chronic inflammatory airway disease - including delivery of higher concentrations of drug locally, and fewer systemic endocrine and immune effects.
✜ Training owners and cats to use inhalers effectively is a critical component of improving patient outcomes and client satisfaction. Adequate time and resources must be provided for client education to ensure the success of inhalational therapy.
✜ The most common inhaled medications for cats with inflammatory airway disease are glucocorticoids and bronchodilators. Inhaled glucocorticoids represent the mainstay of therapy for feline asthma, chronic bronchitis and bronchiectasis.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work did not involve the use of animals and therefore ethical approval was not specifically required for publication in JFMS.
Informed consent
This work did not involve the use of animals (including cadavers) and therefore informed consent was not required. No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
