Abstract
Case series summary
This study aims to describe a novel minimally invasive technique for the removal of small middle ear polyps from the auditory tube openings in cats. Five cats with clinical signs of otitis externa and/or otitis media, and/or upper respiratory tract inflammation were included. All cats underwent pharyngolaryngoscopy under anaesthesia, CT scan of the head, neck and thoracic cavity, video-otoscopic examination, retrograde nasopharyngoscopy and normograde rhinoscopy. The five cats described in this study were all found to have significant respiratory tract inflammation (rhinitis, sinusitis, nasopharyngitis, otitis media) with small polypous protrusions from the auditory tube openings. A normograde rhinoscopy-assisted traction-avulsion (RATA) removal of these small polyps was performed in all cases without complications. The rostral nasopharynx was visualised by a unilaterally normograde advanced rigid endoscope passing the choana, and polyps were removed using a grasping forceps introduced in the contralateral nostril. Telephone follow-up revealed clear improvement in all cases. One of the cases was re-evaluated with a CT scan and endoscopy 4 weeks after treatment. The CT scan showed a significant improvement with no abnormalities in both external ear canals and air opacity in both tympanic bullae. Video-endoscopic examination revealed intact tympanic membranes with mild chronic abnormalities and patent auditory tube openings upon normograde rhinoscopy.
Relevance and novel information
Rigid normograde RATA is a novel, minimally invasive and effective technique for the removal of small middle ear polyps from auditory tube openings in cats with otitis media.
Introduction
Middle ear polyps in cats are relatively common benign masses that arise from the mucosal lining of the middle ear, the auditory tube or the nasopharynx.1 –3 They usually occur in young cats but can be identified at any age.4 –6 The exact aetiopathogenesis of middle ear polyps is still unclear, but the hypothesis is that they arise from chronic inflammation of the middle ear mucosa from a viral-mediated otitis media, chronic stimulation and/or an ascending bacterial infection.4,7,8
Most cats will not demonstrate specific clinical signs when the polypous mucosal tissue remains confined to the middle ear cavity itself, 1 unless septic otitis media and interna develop. The extension of polypous tissue through the eardrum into the ear canal or down the auditory tube into the nasopharynx can lead to clinical signs of otitis externa and/or nasopharyngitis. 7
Protruding polyps themselves can be visualised using (video)otoscopy and/or nasopharyngoscopy.4,6 However, diagnostic imaging in the form of CT or MRI is advisable to not only evaluate the tympanic bullae and its chronic changes, but also the underlying and concurrent inflammation of the respiratory tract.1,4,9
For the treatment of protruding polyps in the ear canal, several techniques have been described, including per-endoscopic trans-tympanic traction-avulsion, traction-avulsion after lateral approach to the ear canal and ventral bulla osteotomy. 1 Large nasopharyngeal polyps can generally be removed using traction-avulsion after rostral retraction of the soft palate.7,8,10 Reportedly, smaller polyps can be removed using grasping forceps through a flexible endoscope or surgically via a nasopharyngotomy incision. As the auditory tube valve opening faces rostrally, visual confirmation of complete removal of the polyp and normal patency of the tube are difficult. In addition, due to the limit regarding the size of forceps that can be introduced through the working channel of a flexible endoscope, smaller non-fibrous polyps may be difficult to completely remove non-surgically. To the authors’ knowledge, there has been no report so far on normograde rigid rhinoscopy-assisted traction-avulsion (RATA) removal of middle ear polyps from auditory tube openings as part of the treatment of otitis media in cats.
The aim of the present study was therefore to describe this unreported and minimally invasive technique of removal of middle ear polyps protruding from the auditory tube in cats.
Case series description
Study design
The medical records of five cats that were presented to a private referral hospital between January 2022 and July 2022 for the evaluation of clinical signs of otitis externa, otitis media and/or nasopharyngitis were evaluated retrospectively. The inclusion criteria for this study were cats that had undergone CT imaging of the head, neck and thoracic cavity, demonstrating otitis media with ipsilateral small protrusions of polypous tissue from the opening of the auditory tube into the nasopharynx that were treated by normograde RATA removal.
All cats underwent pharyngolaryngoscopic evaluation under general anaesthesia, with subsequent intubation and CT imaging in sternal recumbency. Different anaesthetic protocols consisting of a combination of medetomidine, butorphanol, propofol and isoflurane were used, depending on the American Society of Anesthesiologists anaesthesia classification of the individual animal. This protocol provided adequate perioperative analgesia. Postoperative pain medication was not prescribed routinely in any of the reported cases, as the procedure was deemed minimally invasive and short, and anti-inflammatory medication in the form of steroids was prescribed afterwards. All CT images were obtained with the same multi-slice helical scanner (Aquilion CX 128; Toshiba Medical System). The CT examinations were performed in helical acquisition mode with a slice thickness of 0.5 mm, pitch of 0.641–0.656, tube rotation time of 0.5 s, 120 kVp, variable mA (75–250 mAs) and a matrix of 512 × 512, and both bone and soft tissue algorithms were available. All CT images were reviewed in transverse planes and axial and sagittal multiplanar reconstruction images with a pre-set bone (width of 1500 Hounsfield units [HU] and level of 300 HU) and soft tissue window (width of 400 HU and level of 40 HU) using commercially available software (Zillion PACS/RIS/XDS solutions; Rogan-Delft).
A retroflexed nasopharyngeal endoscopic examination using a 5-mm flexible endoscope (Karl Storz, 11001BB), 11 a video-otoscopic examination using a 2.7-mm 30° rigid scope (Karl Storz, 64029 BA) and a normograde rhinoscopic examination using the same rigid scope were performed in all cases (Figure 1). 11 Both auditory tube valve openings could be rhinoscopically visualised in all cases (Figure 2).

Materials for the normograde rigid rhinoscopy-assisted traction-avulsion technique (RATA). Top: a 2.7-mm 30° rigid endoscope (Karl Storz, 11001BB). Bottom left: small plane cupped biopsy forceps. Bottom right: small alligator foreign body removal forceps

Normograde rigid rhinoscopic examination of the nasopharynx. On the right of the image, the normal left auditory tube valve opening can be seen
Normograde rigid RATA
All cats were rhinoscopically examined in sternal recumbency. The rigid video endoscope entered the nasopharynx through the contralateral nasal cavity (opposite the side from which the polyp was protruding from the auditory tube) (Figure 3 and Supplementary Video 1). Polypous tissue protruding from the auditory tube was identified in all cats (Figure 4). Biopsy forceps were subsequently blindly introduced through the ipsilateral nostril into the nasal cavity and ventrally advanced towards the nasopharynx. The biopsy forceps were visualised coming from the choana and advanced towards the auditory tube opening (Figure 5). Under endoscopic visualisation, the protruding tissue was grabbed and removed with traction from the auditory tube opening and removed via the nose (Figure 6).

The set-up for normograde rigid rhinoscopy-assisted traction-avulsion (RATA) removal of middle ear polyps from the auditory tube opening

Rhinoscopic view of polypous tissue protruding from the right auditory tube opening in case 4

Rhinoscopic view of rhinoscopy-assisted traction-avulsion (RATA) removal of a middle ear polyp from the right auditory tube opening in case 4. On the left, a small cupped biopsy forceps is shown grasping the polypous tissue

Polypous tissue that was removed with the use of a rhinoscopy-assisted traction-avulsion (RATA) from the left auditory tube opening in case 3
All cats were discharged with a 10-day course of doxycycline (Doxybactin; AST Farma) and a tapering course of prednisolone (Prednoral; AST Farma).
Cases
This study included five client-owned cats. Age, sex, breed, principal presenting signs, clinical findings, CT findings, endoscopy findings, histology and follow-up were recorded and are summarised in Table 1.
Case descriptions

All cats in our study were young to middle-aged (range 10–110 months) with varying upper respiratory tract and external and/or middle ear signs. CT findings included enlargement of the tympanic bulla, thickening of the bulla wall, soft tissue density filling of the middle ear cavity, and small masses of variable size of soft tissue density at the auditory tube opening (Figure 7). Findings in all cats were consistent with otitis media with polyp formation protruding through the auditory tube opening and, in three cases, into the ear canal. All nasopharyngeal polypous protrusions were removed using the RATA technique without complications. Case 1 developed Horner’s syndrome after removing polypous tissue from both the left external ear canal and the left auditory tube opening. Follow-up by telephone 1 month postoperatively revealed that the cat still had mild signs of left-sided Horner’s syndrome. Case 3 (Figure 8a) was re-examined 1 month after treatment and was free of clinical signs. A CT scan showed a significant improvement with no abnormalities in either external ear canal and air opacity in both tympanic bullae (Figure 8b). Video-otoscopy revealed no further signs of otitis externa and media.
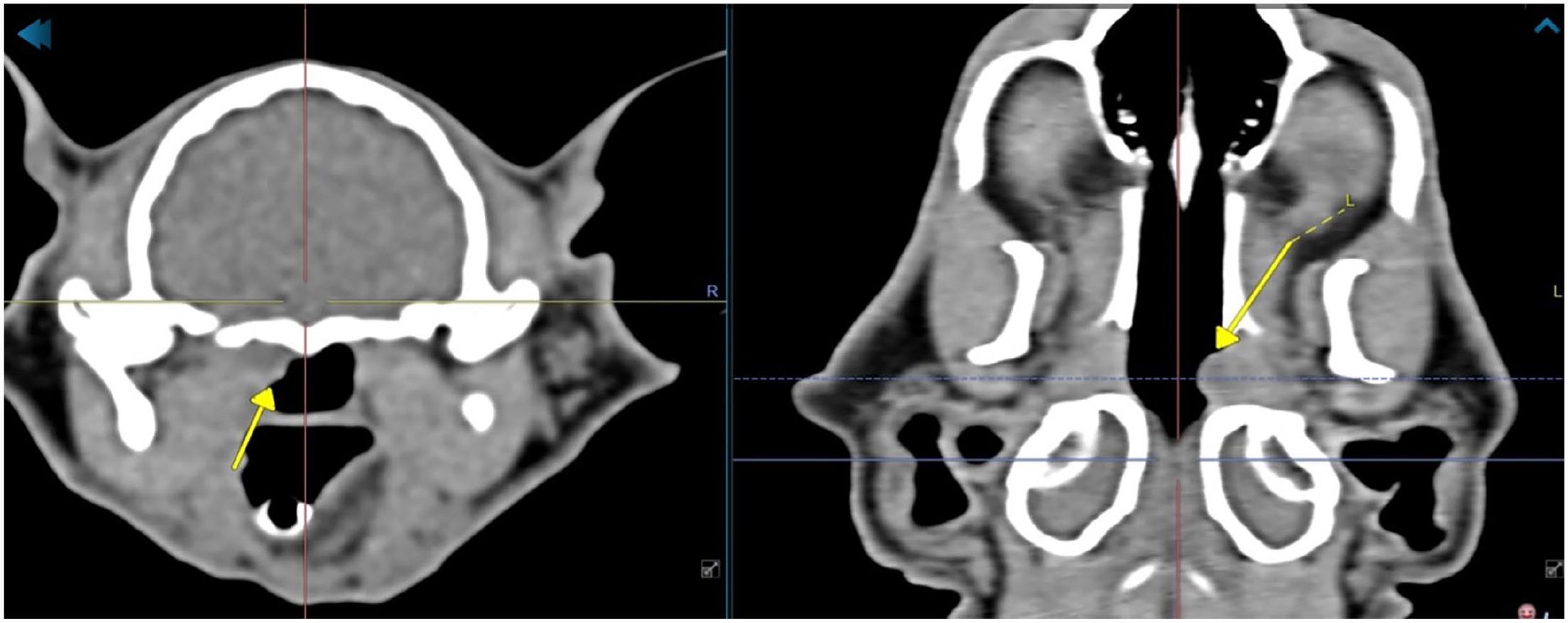
CT images of case 3. Transverse view (left) and dorsal view (right) in the soft tissue window, showing a small soft tissue mass protruding from the left auditory tube opening (arrow) into the nasopharynx

CT images in the transverse plane of case 3: (left) in the bone window and (right) in the soft tissue window. (a) Images before treatment and (b) images from the control CT scan 1 month after treatment. Before treatment, middle ear effusion was present on both sides (arrows in the upper left image). One month later the middle ears were aerated. Only a small line of soft tissue opacity ventral in the left tympanic bulla was found (arrow in the lower left image), most likely consistent with thickening of the mucosal tissue
Discussion
Complete removal of small nasopharyngeal polyps protruding from the auditory tube opening can be challenging when using grasping forceps through a flexible endoscope retroflexed into the nasopharynx, as the auditory tube valve openings face rostrally and only limited-sized forceps can be introduced through the working channel. Alternatively, surgical removal with a nasopharyngotomy incision would provide sufficient access but is more invasive. The present study describes a minimally invasive RATA technique to remove small middle ear polyps from auditory tube openings in five cats.
Ensuring patency of the auditory tube and subsequent ventilation and drainage of the middle ear cavity is essential for successfully treating otitis media in cats. In veterinary medicine, the treatment of otitis media in cats is generally described as being medical, generally with antibiotics. 4 When obvious polyps have formed and cause clinical signs, removal is generally recommended. 3 To date, there are no diagnostic work-up or treatment recommendations for the removal of small polypous protrusions from the auditory tubes in cats with otitis media that do not cause clinical signs based on their presence alone, such as gagging/retching, snoring or reverse sneezing. However, as these protrusions will lead to blockage of the auditory tube, they prevent drainage of the mucus normally produced by the middle ear mucosa. This probably leads to a vicious cycle of the development of chronic otitis media with a further risk of recurrence of polyp formation and/or development of otitis interna. Obstruction of the auditory tube and inoculation of staphylococci, for instance, has been shown to induce otitis media in cats. 12 In addition, the high incidence of otitis media seen in children has also been shown to be related to a functional obstruction of the auditory tube and decrease of ventilation and drainage of the middle ear.13,14
Complications in the current study were only observed in case 1. This cat developed Horner’s syndrome after removing polypous tissue from both the left external ear canal and the left auditory tube opening. Horner’s syndrome is a well-described complication after traction-avulsion of polyps and is usually temporary.1,5,8
Although the use of corticosteroids is not commonly recommended for the medical treatment of otitis media in cats, it has been recommended for reducing the risk of polyp recurrence. In the study by Anderson et al, prednisolone was significantly more effective in reducing the recurrence rate of polyps after traction-avulsion compared with antibiotics alone or no medical treatment. 2 It is likely that steroids decrease recurrence rates by decreasing the inflammation of the respiratory mucosal lining of the middle ear and entire respiratory tract that may lead to polyp formation. Further studies are required to investigate the effect of and need for steroids in the treatment of otitis media in cats.
The collection of middle ear material for bacterial culture was not performed in our study, but doxycycline is legally the first-choice antibiotic in the Netherlands and is widely used as an antibiotic for agents causing ear, nose and throat infections (including otitis media) in cats. 4 Besides being a broad-spectrum antibiotic, 15 it has also demonstrated anti-inflammatory effects on its own in humans with respiratory tract infections. 16 Although side effects, such as oesophageal ulcerations, are known to possibly occur and can be decreased by using the monohydrate salt formulation, these side effects were not encountered in our study. The need for antibacterial medications after restoring auditory tube patency needs to be established.
The present study has several limitations. Due to the retrospective nature of the study, CT follow-up was not available for all the cases included and follow-up was very short. A statistical analysis was not performed due to the relatively small group size and the lack of a control group.
Conclusions
Even inflammatory middle ear polyps protruding from the auditory tube openings into the nasopharynx that have not led to obvious local clinical signs should be removed to restore patency of the auditory tubes to help successfully treat otitis media and prevent recurrence. This study describes a novel and minimally invasive technique for the removal of small polyps from auditory tube openings in cats with otitis media.
Footnotes
Supplementary material
The following file is available online: Video 1:
This video shows the procedure for visualising the auditory tube openings and introduction of the forceps up until the level of the auditory tubes as part of the normograde rigid rhinoscopy-assisted traction-avulsion technique (RATA).
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
IInformed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
