Abstract
Objectives
This randomised, prospective, masked clinical trial evaluated the postoperative analgesic efficacy of an ultrasound-guided transversus abdominis plane block (TAPB) with bupivacaine in cats undergoing ovariohysterectomy.
Methods
Thirty-two healthy adult female cats undergoing elective ovariohysterectomy were randomised to undergo TAPB with bupivacaine (treatment group [TG], n = 16) vs placebo (control group [CG], n = 16) in addition to preoperative analgesia with buprenorphine (0.02 mg/kg IM). All patients received a general anaesthetic and, before surgical incision, a bilateral two-point (subcostal and lateral–longitudinal) TAPB was performed using 1 ml/kg bupivacaine 0.25% (0.25 ml/kg/point) or saline. Each cat was assessed by a blinded investigator before premedication (0 h) and at 1, 2, 3, 4, 8, 10 and 24 h postoperatively using the UNESP-Botucatu Feline Pain Scale – short form. Buprenorphine (0.02 mg/kg IV) and meloxicam (0.2 mg/kg SC) were administered when pain scores were ⩾4/12. Ten hours postoperatively, meloxicam was administered to cats that did not receive rescue analgesia. Statistical analysis included Student’s t-tests, Wilcoxon tests and χ2 tests, and a linear mixed model with Bonferroni corrections (P <0.05).
Results
Of the 32 cats enrolled, three in the CG were excluded from the analysis. The prevalence of rescue analgesia was significantly higher in the CG (n = 13/13) than in the TG (n = 3/16; P <0.001). Only one cat in the CG required rescue analgesia twice. Pain scores were significantly higher in the CG compared with the TG at 2, 4 and 8 h postoperatively. Mean ± SD pain scores were significantly higher in the CG, but not in the TG, at 2 (2.1 ± 1.9), 3 (1.9 ± 1.6), 4 (3.0 ± 1.4) and 8 h postoperatively (4.7 ± 0.6) than at 0 h (0.1 ± 0.3).
Conclusions and relevance
A bilateral ultrasound-guided two-point TAPB with bupivacaine in combination with systemic buprenorphine provided superior postoperative analgesia than buprenorphine alone in cats undergoing ovariohysterectomy.
Introduction
Locoregional anaesthetic techniques have been advocated in veterinary medicine to decrease anaesthetic and analgesic requirements, as well as their related adverse effects.1,2 In a multimodal analgesic plan, the administration of a local anaesthetic in proximity to nervous structures is the only technique that completely blocks nociceptive transmission.
Ultrasound (US)-guided fascial plane blocks, such as transversus abdominis plane block (TAPB),3–6 rectus sheath block7,8 and quadratus lumborum plane block,9–12 have recently been described for abdominal surgery in small animals. The TAPB is probably the most studied and employed technique of the past decade. It relies on the diffusion of local anaesthetic in the fascial plane superficial to the transversus abdominis muscle, and on the desensitisation of the abdominal wall afferent nerve fibres. 3 It provides analgesia to the ventral and lateral abdominal wall, including skin, mammary glands, abdominal muscles and parietal peritoneum.13–17 In small animals, the TAPB has been used as part of multimodal analgesia in patients with pancreatitis, 18 and those undergoing coeliotomy,18,19 mastectomy,13,15 abdominal wall reconstruction, 20 ovariectomy14,17 or ovariohysterectomy. 16 However, there is a need for further clinical studies to evaluate specific TAPB techniques using, for example, a subcostal and lateral–longitudinal (two-point) approach with specific volumes and concentrations of bupivacaine, as well as to evaluate its efficacy for postoperative pain relief in cats undergoing surgery.
The aim of this study was to evaluate the analgesic efficacy of an US-guided bilateral two-point TAPB using bupivacaine or saline in combination with intramuscular (IM) buprenorphine in adult cats undergoing ovariohysterectomy. We hypothesised that the prevalence of rescue analgesia and postoperative pain scores would be lower after TAPB using bupivacaine than saline.
Materials and methods
The study is reported in accordance with the CONSORT guidelines. 21 The protocol of this randomised, prospective, masked clinical trial was approved by the Institutional Animal Care Committee of the Faculty of Veterinary Medicine (Comité d’éthique de l’utilisation des animaux), Université de Montréal (21-Rech-2153). The study was performed at the Centre Hospitalier Universitaire Vétérinaire of the Université de Montréal between September and October 2022.
Animals
Thirty-two female cats from three local shelter facilities were enrolled after obtaining the caregiver/shelter’s written consent for participation in the study.
Eligibility criteria included any healthy female cat of any breed, older than 6 months. Cats were considered healthy based on medical history, physical examination, up-to-date vaccination status, and haematocrit and total protein values. Exclusion criteria included body weight <2.0 kg, body condition score <3 or >6 (scale 1–9), anaemia (haematocrit <25%), hypoproteinaemia (total protein <59 g/l), clinical signs of systemic disease, cardiac dysrhythmias on auscultation, shy or fearful cats not allowing a preoperative pain assessment, or any sign/evidence of previous ovariectomy/ovariohysterectomy. Recruitment was performed by one of the investigators (HLMR). Cats were admitted approximately 17 h before general anaesthesia and housed individually in adjacent cages located in a cat ward. Each cage was equipped with water and food bowls, a cardboard box, a blanket, a litter box and a toy. Synthetic feline hormones (Feliway Classic; Ceva Animal Health) were diffused in the room from 8 h before cat admission until discharge. 22 Cats were manipulated using feline-friendly techniques.
Randomisation and masking
Each eligible cat was assigned a number (1–32) based on admission order. According to this number, the patient was allocated 1:1 to one of the two groups (treatment group [TG] or control group [CG], with 16 cats in each). Cats in the TG and CG received 1 ml/kg bupivacaine 0.25% (2.5 mg/kg; Sensorcaine 0.25%; AstraZeneca) or 1 ml/kg saline (0.9% sodium chloride injection USP; Pfizer Canada), respectively, by US-guided TAPB under general anaesthesia and before surgery. The dose and volume of local anaesthetic were based on previous studies.6,23 The one-block randomisation list was generated using a randomisation plan generator (www.randomization.com; accessed 3 August 2022). Immediately before general anaesthesia, TAPB injectate solutions (bupivacaine 0.25% or saline) were prepared using syringes labelled with the patient number. Randomisation and treatment preparation were performed by one veterinarian who was not involved in the execution of the locoregional technique or the postoperative pain assessment (RW). Individuals involved in the administration of TAPB (MG) and perioperative pain assessment (HLMR) were blinded to the injectate solution and treatment group.
Surgery and anaesthesia
Food, but not water, was withheld for 8–10 h before general anaesthesia. Each cat was premedicated with IM acepromazine (0.03 mg/kg; Atravet; Boehringer Ingelheim) and buprenorphine (0.02 mg/kg; Vetergesic; Champion Alstoe Animal Health) administered into the epaxial lumbar muscles. To facilitate venous catheterisation, a cream containing lidocaine 2.5% and prilocaine 2.5% (EMLA Cream; Aspen Pharmacare Canada) was applied over the right cephalic vein after skin preparation. Approximately 15 mins later, a 22 G catheter was inserted aseptically in the cephalic vein and the blood from the catheter was used to evaluate haematocrit and total protein. Then, the cat was transferred to the operating room and anaesthesia was induced with intravenous (IV) propofol (PropoFlo 28; Zoetis Canada) administered to effect. The patient was intubated with a supraglottic airway device (V-gel ADVANCED; DocsInnovent) connected to a capnograph and a modified Mapleson D system (Bain circuit). Anaesthesia was maintained with isoflurane (Isoflurane USP; Fresenius Kabi) delivered in oxygen.
The cat was then positioned in dorsal recumbency on a circulating warm water blanket and the abdominal area was clipped and prepared for surgery using aseptic technique. The cat’s eyes were lubricated with ocular gel before and after surgery. A multiparametric monitor (LifeWindow 6,000V; Digicare Animal Health) was used to monitor arterial oxyhaemoglobin saturation, carbon dioxide end-tidal concentrations, respiratory and heart rates, electrocardiogram, and systolic, mean and diastolic arterial pressure. Parameters were recorded every 5 mins. Rectal temperature was measured before and immediately after the end of surgery. Lactated Ringer’s solution (Lactated Ringer’s injection USP; Baxter) was administered IV at 10 ml/kg/h throughout the surgical procedure. Anaesthesia was always performed by the same veterinarian (RW).
A bilateral two-point TAPB 6 was performed by a veterinarian with advanced training in anaesthesia and analgesia, and with experience in locoregional anaesthesia (MG). All locoregional techniques were performed using a 22 G, 50 mm Quincke spinal needle (BD spinal needle; Becton Dickinson) connected by a T-port (Med-RX extension set with T-connector; CHS) to a prefilled syringe (Terumo syringe 5 ml; Terumo). The needle was introduced under US visualisation using a portable US machine (Sonosite Edge II Ultrasound System; Sonosite) and a 13–6 MHz linear array probe (HLF-38; Sonosite). The scanning depth was set at 1.9 cm (the minimum allowed by the US machine) and the gain was manually adjusted to optimise image quality. The four TAPB injections were performed in the following order: subcostal and lateral approaches on the right and the left hemiabdomen, respectively. The probe was initially positioned perpendicular to the ventral midline at ∼1 cm caudal to the xiphoid process. It was then slid laterally, parallel and caudal to the costal arch, to perform the TAPB subcostal approach. The needle was introduced in-plane in a ventromedial to dorsocaudal direction until its tip was visualised between the rectus abdominis and transversus abdominis muscles. For the lateral approach, the probe was slid further laterally, following the transversus abdominis muscle, and rotated parallel to the abdominal midline at ~1–2 cm lateral to the mammary line and ~2 cm caudal to the last rib. The needle was introduced in-plane in a cranial to caudal orientation until its tip was visualised between the transversus abdominis and obliquus internus abdominis muscles. After negative aspiration, 0.1 ml of the solution was slowly injected under US visualisation to assess the correct position. If hydro-dissection of the target plane was not visualised, the needle was redirected and the same volume injected until correct positioning was achieved. A volume of 0.25 ml/kg/point (volume of test dose included) of either bupivacaine 0.25% or saline was administered according to the treatment group (Figure 1).

Ultrasound-guided transversus abdominis plane block (TAPB) in a cat. (a) Schematic representation of the probe position (grey rectangles) and needle orientation (black arrows) to perform a bilateral TAPB combining subcostal and lateral approaches. (b,c) Ultrasound images of the TAPB subcostal approach (b) before and (c) after administration of 0.25 ml/kg of injectate solution (IS) between the rectus abdominis (RA) and transversus abdominis (TA) muscles. (d,e) Ultrasound images of the TAPB lateral approach (d) before and (e) after the administration of 0.25 ml/kg of IS between the obliquus internus abdominis muscle (OIA) and the TA. Cd = caudal; Cr = cranial; D = dorsal; L = lateral; M = medial; OEA = obliquus externus abdominis muscle; V = ventral
Approximately 5 mins after TAPB, an experienced veterinarian (BPM) performed ovariohysterectomy using the pedicle tie technique via a ventral midline incision. 24 The abdominal wall was closed using a simple continuous pattern with poliglecaprone 25 (Monocryl 3-0 or 4-0; Ethicon). The skin and SC tissues were sutured using a continuous intradermal pattern using the same suture. The surgical incision length was measured using a plastic ruler. Then, a 1–2 cm green tattoo was performed parallel to the surgical incision for identification of a neutered cat.
At the end of surgery, the administration of isoflurane was stopped. The cat was disconnected from the anaesthesia machine, moved to another table for recovery and positioned in sternal recumbency on a circulating warm water blanket. Monitoring was performed using a portable pulse oximeter (Rad-5v; Masimo). Extubation was done by an individual dedicated to the anaesthetic recovery (AM) when strong palpebral reflexes were present.
The following parameters were recorded for each cat: duration of anaesthesia (time from beginning to cessation of isoflurane administration); time to perform the TAPB (time from probe positioning to injection of the last aliquot of injectate); duration of surgery (time from first incision to placement of the last suture); and length of the incisional site. Any complications in performing the TAPB, such as vascular puncture, poor hydro-dissection or misidentification of the target fascial plane, intraperitoneal injection or abdominal organ puncture, were recorded.
Postoperative pain assessment
Pain assessment was performed using the UNESP-Botucatu Feline Pain Scale – short form (UFEPS-SF) 25 at least 1 h before premedication (0 h, baseline) and at 1, 2, 3, 4, 8, 10 and 24 h after the end of surgery by the same veterinarian (HLMR), who was blinded to the treatment groups. This observer has 15 years of experience in clinical practice and 5 years in pain research in cats, and participated in the multilingual validation of the UFEPS-SF. 26 Rescue analgesia was provided with buprenorphine (0.02 mg/kg IV) and meloxicam (0.2 mg/kg SC; Metacam; Boehringer Ingelheim) when pain scores ⩾4/12. Pain was reassessed 30 mins after the administration of rescue analgesia to ensure that the patient was comfortable and did not require further pain relief. Data collected after the administration of rescue analgesia were not included in the statistical analysis, but pain assessment continued up to patient discharge. Meloxicam was administered after the 10 h time point to cats that did not require rescue analgesia during the study. A second dose of meloxicam (0.05 mg/kg; Metacam oral suspension; Boehringer Ingelheim) was administered orally to all cats 24 h after the first dose and before discharge.
Sample size calculation and statistical analysis
The prevalence of postoperative rescue analgesia was considered the primary outcome of the study as it is an important indicator of analgesic efficacy in the clinical setting. Therefore, sample size estimation was based on the prevalence of rescue analgesia in cats undergoing ovariohysterectomy and on an opioid-free protocol (57%) 27 or buprenorphine in the context of multimodal analgesia (6%). 28 A power analysis with the Fisher’s exact test indicated that a sample size of 12 cats per group was necessary 80% of the time to detect a difference with the alpha level at 5%. The population size was increased of ~25% to 32 cats (n = 16/group) to account for possible dropouts.
Data were analysed by a data scientist using R software within the integrated RStudio environment (version 4.1.0). Data normality was assessed using the Cramer–Von Mises test. Normally distributed variables were compared between groups using the two-tailed Student’s t-test and reported as mean ± SD. Variables that were not normally distributed were analysed using a two-tailed Wilcoxon test and reported as median (interquartile range). The difference in the frequency of rescue analgesia between groups was analysed using the χ2 test. Pain scores were analysed using multilevel generalised linear models adjusted according to Poisson distribution in which the best model was identified according to the Bayesian information criterion. ‘Time’, ‘group’ and their interaction were considered fixed effects; ‘cats’ were considered as random effect, and ‘age’ was added as a covariate to the model. The Bonferroni adjustment procedure was used after multiple comparisons to the post-hoc test. For all tests, a P value <0.05 was considered to be statistically significant.
Results
Demographic data
Three cats were excluded from the CG (one cat was already spayed, one presented melaena postoperatively and one did not allow a postoperative pain assessment). Therefore, 29 cats completed the study (Figure 2).
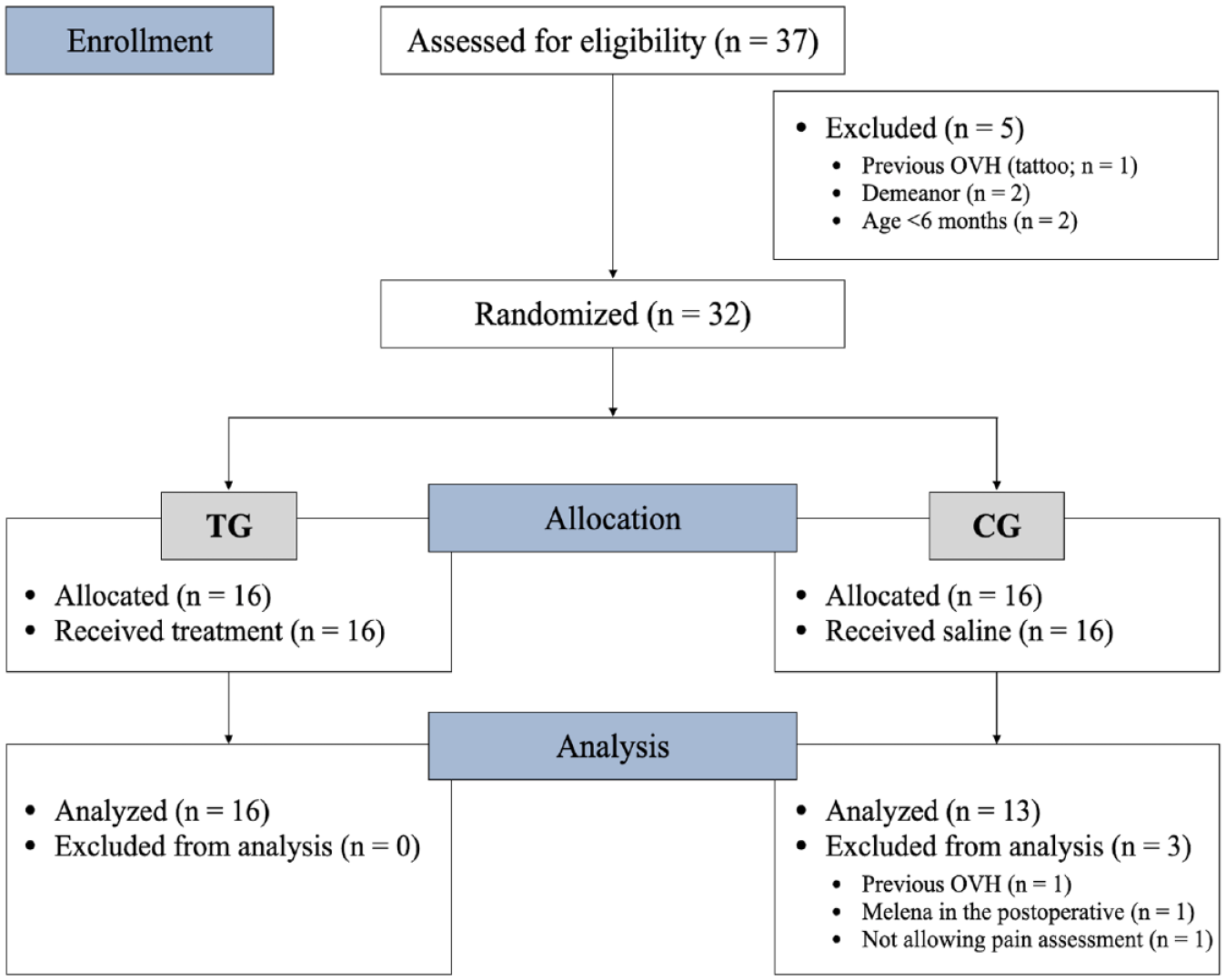
CONSORT flow diagram showing study randomisation, group allocation and data analysis. Cats received ultrasound-guided bilateral two-point transversus abdominis plane block with 0.25 ml/kg/point of either bupivacaine 0.25% (treatment group; TG) or saline (control group; CG) and underwent ovariohysterectomy (OVH)
Patient demographics, haematocrit, total protein, propofol dose, duration of anaesthesia and surgery, and length of incisional site did not differ between the groups (Table 1). The time taken to perform the TAPB was significantly shorter in the CG compared with the TG (P = 0.04; Table 1). The TAPB was performed in all cats; in two cats in the TG and in three in the CG, one injection resulted in limited hydro-dissection of the target plane/partial IM injection (Figure 3). No other complications related to the TAPB technique were observed. Two cats, one from each group, were in mid-stages of pregnancy. Two cats in the TG and one in the CG were lactating. All cats were discharged from the hospital after the 24 h time point evaluation and returned to the shelters for adoption.
The population characteristics of cats undergoing ovariohysterectomy after the administration of either bupivacaine 0.25% (treatment group; TG) or saline (control group; CG) by a two-point bilateral transversus abdominis plane block (TAPB) in combination with systemic buprenorphine

Data are reported as mean ± SD or median (interquartile range) as appropriate. Superscript letters indicate a significant difference between groups with a > b
Haematocrit reference interval = 28–47%; total protein reference interval = 59–81 g/l

Ultrasound images showing unsatisfactory distributions of the injectate solution (IS; either bupivacaine 0.25% or saline) administered by an ultrasound-guided two-point transversus abdominis plane block (TAPB) in cats before ovariohysterectomy. (a,b) Ultrasound images of the TAPB subcostal approach showing partial intramuscular spread of the IS into the transversus abdominis (TA) muscle. (c,d) Ultrasound images of the TAPB lateral approach showing limited spread of the IS in the transversus abdominis plane between the obliquus internus abdominis muscle (OIA) and the TA. Cd = caudal; Cr = cranial; D = dorsal; L = lateral; M = medial; OEA = obliquus externus abdominis muscle; RA = rectus abdominis muscle; V = ventral
Pain scores
The UFEPS-SF scores were higher in cats in the CG than those in the TG at 2 h (P = 0.02), 4 h (P = 0.0005) and 8 h postoperatively (P <0.0001; Figure 4). In the CG, pain scores (mean ± SD) were higher at 2 h (2.1 ± 1.9; P = 0.016), 3 h (1.9 ± 1.6; P = 0.011), 4 h (3.0 ± 1.4; P = 0.0009) and 8 h postoperatively (4.7 ± 0.6; P = 0.0001) vs baseline (0.1 ± 0.3). In TG, postoperative pain scores were not different from baseline (P = 1.0).
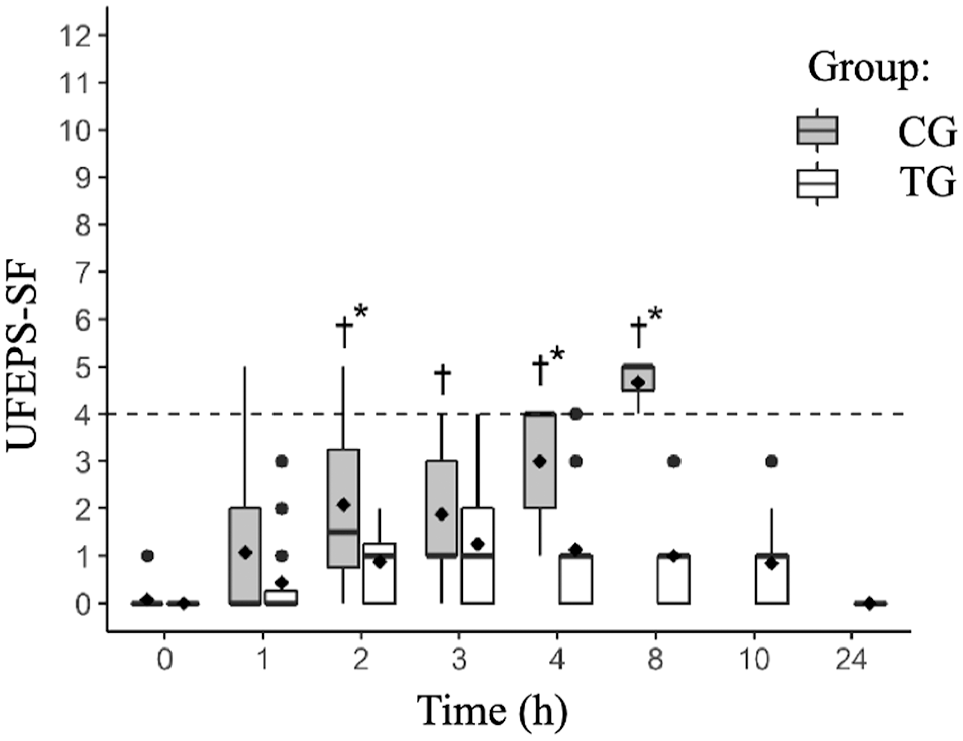
Boxplot of the UNESP-Botucatu Feline Pain Scale – short form (UFEPS-SF) scores before (0 h) and at several time points after ovariohysterectomy in cats receiving preoperative buprenorphine and a bilateral ultrasound-guided transversus abdominal plane block with bupivacaine 0.25% (treatment group; TG) or saline (control group; CG). Circles indicate outliers; diamonds indicate means; the dotted line indicates the analgesic threshold of the UFEPS-SF. *Significant difference between groups at specific time. †Significant difference in CG when compared with baseline values (0 h)
Prevalence of rescue analgesia
The prevalence of rescue analgesia was significantly higher in cats in the CG (n = 13/13) compared with those in the TG (n = 3/16; P <0.001). Rescue analgesia was administered in the CG at 1 h (n = 1), 2 h (n = 3), 3 h (n = 2), 4 h (n = 4) and 8 h (n = 3), and in the TG at 3 h (n = 2) and 4 h (n = 1). One cat in the CG required the administration of buprenorphine twice (at the 1 h and 10 h time points).
Discussion
This clinical trial demonstrated that postoperative analgesia is superior with an US-guided bilateral two-point TAPB with bupivacaine combined with systemic buprenorphine than the opioid used alone in cats undergoing ovariohysterectomy. Both pain scores and the prevalence of rescue analgesia were lower after the TAPB using bupivacaine when compared with saline, and according to the original hypothesis of the study.
The two-point TAPB technique employed in this study (subcostal and lateral–longitudinal approaches) was feasible and resulted in reliable nerve staining from the tenth thoracic to the second lumbar in a previous anatomical cadaveric study in cats, 6 wider than a one-point (lateral–longitudinal) TAPB technique 6 and similar to a three-point (one subcostal and two lateral–transversal approaches) TAPB technique. 4 In a clinical setting, the decision of whether to use a one-, two-, or even three-point TAPB approach depends on the location and extent of the surgical incision, as each technique provides different dermatomal coverage.4,6 Furthermore, increasing the number of injections increases the likelihood of blockade failure, as one of the injections could produce partial or no blockade, compromising the efficacy of the technique. The anaesthetic diffusion in a fascial plane can be unpredictable, despite the use of US guidance to enhance landmark identification and visualisation. 29 In the present study, three cats in the TG required rescue analgesia. In two of these cats, one of the four TAPB injections resulted in limited spread within the target plane/partial IM injection, likely leading to an incomplete analgesic block. The prevalence of incomplete block (n = 2/13) and rescue analgesia (n = 3/16) in the TG is higher than previously reported in a similar investigation using a bilateral one-point TAPB in combination with a systemic opioid and a non-steroidal anti-inflammatory drug (NSAID) in cats undergoing ovariectomy. 14 However, these data are not comparable as a NSAID was administered as part of the multimodal approach in the latter study. It could be expected that the addition of a NSAID to our analgesic plan would perhaps eliminate the need for rescue analgesia, as was the case when buprenorphine, meloxicam and intraperitoneal bupivacaine–dexmedetomidine, but not buprenorphine–meloxicam or buprenorphine–intraperitoneal bupivacaine, were used for analgesia in cats undergoing ovariohysterectomy.28,30,31
The prevalence of rescue analgesia observed in our study was lower than in dogs undergoing ovariohysterectomy after a bilateral one-point TAPB with bupivacaine and dexmedetomidine (44.4%) and with liposomal bupivacaine (33.3%). 16 Heterogeneity of the type of surgery, species, TAPB approaches, experience in pain assessment, anaesthetic drugs, and dosage regimens may impact the interpretation of these findings.
In this study, a significant difference in the time taken to perform the TAPB was observed between groups. However, this difference was not clinically relevant (60 s). The longest time taken to perform the TAPB in cats in the TG was about 410 s in three cats, two of which were lactating. The presence of highly developed mammary glands made the TAPB more challenging, especially with the lateral approach when avoiding the mammary tissue. The lateral approach was performed approximately 3–4 cm dorsal to the mammary line, whereas the subcostal approach was done by introducing the needle through the mammary tissue in these two individuals. In a clinical setting, bupivacaine could be excreted in the milk and absorbed by the kittens. Although the pharmacokinetics of bupivacaine and its transfer into milk have not been reported in lactating queens, breast milk transfer of racemic and liposomal bupivacaine infiltration after TAPB is small in women having a caesarean section. In this case, the relative bupivacaine neonatal dosage is <1% the maternal dose. 32
Based on the distribution of the local anaesthetic solution, it is generally accepted that the TAPB is only effective for the treatment of somatic pain. Nevertheless, a preoperative TAPB decreased anaesthetic requirements in women undergoing total hysterectomy, 33 and allowed better intraoperative haemodynamic control in high-risk abdominal surgery than general anaesthesia alone. 34 Furthermore, it was considered effective for abdominal pain relief in humans 35 and dogs 18 with acute pancreatitis, suggesting that this locoregional technique might provide some visceral analgesia. The mechanism of action of fascial plane block is still not fully understood. 36 While findings in humans suggest that TAPB decreases the perioperative neuroendocrine stress response, 37 a recent investigation in dogs undergoing ovariectomy after a one-point TAPB failed to show any difference in serum cortisol compared with placebo. 17 In our study, the haemodynamic intraoperative parameters were monitored but not analysed as it was assumed that all cats underwent the same nociceptive stimuli, and no other medication was administered intraoperatively. However, to improve pain relief and eliminate the need for postoperative rescue analgesia, it would be interesting to investigate the clinical efficacy of TAPB combined, or not, with intraperitoneal instillation of local anaesthetic 31 in cats undergoing ovariohysterectomy. In this case, care should be taken to use appropriate doses and concentrations of local anaesthetic when combining more than one locoregional technique. Moreover, consideration should be given to adding an adjuvant analgesic (ie, dexmedetomidine) to TAPB in order to increase the duration of action and improve the magnitude of analgesia.16,38
This study has limitations. Firstly, the TAPB was performed by the same anaesthesiologist with advanced training in the speciality. Further studies are required to determine the success rate of the regional technique when performed by a less experienced operator, and how this would influence outcomes in terms of postoperative analgesia and complications. Secondly, pain recognition is challenging in cats.39,40 In this study, an experienced observer with training performed postoperative scoring. Subtle changes in behaviour could have been missed by a less experienced observer, even when using a validated pain-scoring instrument. Results could have been different. Moreover, the study population involved female cats older than 6 months undergoing ovariohysterectomy. Kittens may express pain behaviours differently from adults or show fewer behavioural signs of pain. 41 For this reason, our findings may not be extrapolated to kittens or male cats undergoing abdominal surgery. Thirdly, the cohort of this study was restricted to cats with a body weight between 2.5 and 5 kg. Therefore, it is not known if the efficacy and complications of TAPB would have been similar in cats outside this weight range. Indeed, this clinical investigation supports our previous concerns about performing the TAPB in patients weighing <3 kg. 6 In the present study, 5/128 TAPB injections resulted in partial IM spread or limited local anaesthetic distribution in the target fascial plane. Performing the TAPB in small cats is challenging, and the efficacy of the blockade could be compromised. However, carrying out the technique in obese feline patients can also be difficult due to the presence of layers of fat within the abdominal muscles.42,43 Finally, this proof-of-concept study aimed to demonstrate the superior postoperative analgesia of a TAPB with bupivacaine when combined with systemic buprenorphine compared with the opioid alone. We do not support the use of buprenorphine alone for pain management after ovariohysterectomy as it provides inadequate analgesia.27,30,44 Additionally, the administration of a feline-approved NSAID is recommended to minimise postoperative pain as part of multimodal analgesia.
Conclusions
Systemic buprenorphine in combination with an US-guided bilateral two-point, subcostal and lateral–longitudinal TAPB with 1 ml/kg bupivacaine 0.25%, but not saline, provided effective analgesia in most cats after ovariohysterectomy. Postoperative pain scores and the prevalence of rescue analgesia were lower in the group treated with bupivacaine and buprenorphine than in the group treated with the opioid alone.
Footnotes
Acknowledgements
The authors thank Pedro H Trindade for statistical analysis.
Author note
This manuscript represents a portion of a PhD thesis of the first author to the Université de Montreal.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Paulo V Steagall’s laboratory is funded by a Discovery Grant of the Natural Sciences and Engineering Research Council of Canada (RGPIN-2018-03831).
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
