Abstract
Case series summary
Feline maxillary sarcomas are aggressive spindle cell neoplasms that occur within the maxilla, palate and upper lip of cats. This diagnosis includes fibrosarcoma and sarcomas with indeterminate histomorphology, excluding melanocytic tumors and sarcomas that can be differentiated by histomorphology. In this study of feline maxillary sarcomas in 25 cats, the cats’ ages ranged from 4 to 16 years (median 12.5). These sarcomas presented as smooth thickenings or mass lesions of the gingiva and palate, often involving both the right and left quadrants of the maxilla. Radiographic bone loss was typically absent to mild at the time of diagnosis. Histologically, feline maxillary sarcomas were composed of spindle cells with varying amounts of fibrous stroma and mild inflammation. Metastasis was not documented for any cat in the study, although clinical staging was limited. Cats were often euthanized because of local recurrence following incomplete tumor excision and local tumor progression. Median survival time from the date of histologic diagnosis was 70 days (n = 12).
Relevance and novel information
Feline maxillary sarcomas are aggressive neoplasms that may be difficult to differentiate from a benign, reactive process or other types of spindle cell neoplasms. Our findings indicate that feline maxillary sarcoma has distinctive clinical and histopathologic features, and the information provided in this paper will facilitate early and specific diagnosis of this tumor.
Introduction
Oral neoplasia in cats is common, accounting for 10% of feline neoplasia, and nearly 90% of feline oral neoplasms are malignant.1,2 Squamous cell carcinoma is most common and accounts for 61.2–72.5% of oral neoplasms in cats.1,2 From the 1960s to 2019, fibrosarcoma was reported as the second most common oral neoplasm in cats, accounting for 9.8–17.0% of oral tumors.1,3,4 These neoplasms have been described as locally invasive, but metastasis has not been reported.5–7 Large surveys of oral neoplasia in cats have included limited information on the typical signalment, anatomic location and clinical appearance of oral fibrosarcoma.1–4 Evaluation of clinical outcomes and calculation of survival times has occurred only in select case series following mandibulectomy or chemotherapy.5–7
Historically, the diagnosis of fibrosarcoma has been used for invasive mesenchymal neoplasms characterized by spindle cells with fibrous stroma. The histogenic origin of fibrosarcoma is the fibroblast. 8 However, it is now commonly recognized that infiltrative tumors with spindle cell morphologies may represent variable cell or tissue origin including fibrosarcoma (fibroblasts), nerve sheath tumors such as Schwannoma (Schwann cells), melanoma (melanocytes) and others.8,9 While some of these tumors can be differentiated based on histomorphologic features – such as junctional activity or pigmentation in melanomas – ancillary testing (namely immunohistochemistry) is often useful for diagnosis. Even with extensive immunohistochemical panels, the histogenic origin of some feline oral spindle cell neoplasms remains unclassified. 9 Considering this more contemporary view of mesenchymal neoplasia, it is possible that historical studies, which were based largely on histomorphology alone, may have attributed the diagnosis of fibrosarcoma to a wider range of neoplasms than those arising purely from fibroblasts. Nevertheless, the potential that oral sarcomas in cats may have a consistent clinical presentation and biologic behavior warrants further consideration.
This study defines the clinical, radiographic and histopathologic features of spindle cell sarcomas from the maxillary region of 25 cats. The diagnosis of ‘spindle cell sarcoma’ is used to refer to an invasive spindle cell tumor that is otherwise unclassified based on histomorphology and available ancillary testing. This term is inclusive of fibrosarcoma but does not exclude other sarcomas that lack a specific histomorphology. This information will define key features of feline maxillary sarcomas to improve diagnostic accuracy and prognostication for feline patients.
Case series description
Materials and methods
The records from the Center for Comparative Oral and Maxillofacial Pathology (CCOMP) at UW-Madison during a 20-month period were searched for cases with a diagnosis of fibrosarcoma or other unspecified spindle cell neoplasm. Inclusion criteria were: (1) a spindle cell morphology arranged in interwoven fascicles within a collagenous matrix; (2) involvement of the tooth-bearing regions of the jaw, including the gingiva, maxilla or mandible; and (3) evidence of malignancy, including histologic criteria of malignancy (nuclear pleomorphism, increased mitotic activity), histologic or radiographic evidence of invasion into surrounding tissue, and/or progression of clinical disease resulting in euthanasia or death. Exclusion criteria were: (1) neoplasms that did not involve the gingiva and tooth-bearing region of the jaw (eg, tumors confined to the lip, vestibular mucosa or tongue); and (2) spindle cell tumors with histomorphologic features and/or immunohistochemical labeling that supported a specific spindle cell neoplasm (eg, melanoma, carcinoma and nerve sheath tumor) other than presumptive fibrosarcoma.
Clinical information including patient signalment, clinical signs, history, photographs of the lesion, dental radiographs (or other diagnostic imaging), treatments administered and information regarding recurrence, progression of disease and survival (date of death) were collected. Data were obtained through review of the biopsy submission forms, a questionnaire sent to the submitting veterinarians, follow-up emails and telephone calls with the submitting and/or primary veterinarians and review of medical records when available. All radiographs were reviewed by a board-certified veterinary dentist (JWS).
Formalin-fixed specimens were received and processed to hematoxylin and eosin-stained sections of paraffin-embedded tissue. Histologic sections were evaluated for cellularity, pattern or organization, quality and abundance of matrix, type and severity of inflammation, and cellular/nuclear morphology. Six cases (cases 1, 16, 17, 18, 19 and 24) were selected for further staining with Masson’s trichrome, Alcian blue periodic acid–Schiff (PAS), reticulin and immunohistochemistry (IHC). For IHC, formalin-fixed, paraffin-embedded 5 µm-thick sections were deparaffinized and rehydrated, and antigen retrieval was performed. The following antibodies were used: anti-vimentin, anti-S100 protein, anti-smooth muscle actin (SMA) and anti-melan A. Negative and positive controls were included with each round of labeling. All histopathologic interpretation was performed by a board-certified veterinary pathologist (CMB).
Patient information
Thirty tumors from 30 cats were initially considered based on the diagnosis of a fibrosarcoma or unspecified spindle cell neoplasm. Four cases were excluded due to histomorphologic features suggestive of other neoplasms, including two osteosarcomas, one malignant nerve sheath tumor and one hemangiosarcoma. One neoplasm was excluded owing to extensive involvement of the lip and the suspicion of a primary cutaneous neoplasm as opposed to a primary oral neoplasm arising from the tooth-bearing region of the jaws. Tumors in 25 cats fitted the selection criteria, which represented approximately 18% of all oral neoplasias in cats from cases submitted to CCOMP during the designated timeframe. The mean ± SD age of the cats was 11.72 ± 2.72 years (median 12.5, range 4–16). There were 18 (72%) spayed females and seven (28%) castrated males. Sixteen were domestic shorthair (64%), three were domestic longhair (12%), two were domestic mediumhair (8%), two were Maine Coon (8%), one was a Siamese cross breed (4%) and one was Ragamuffin (4%). Data for individual cases are provided in Table 1 (see Figures 1–3 for clinical photographs, dental radiographs and histopathology of cases 16–18).
Individual case data
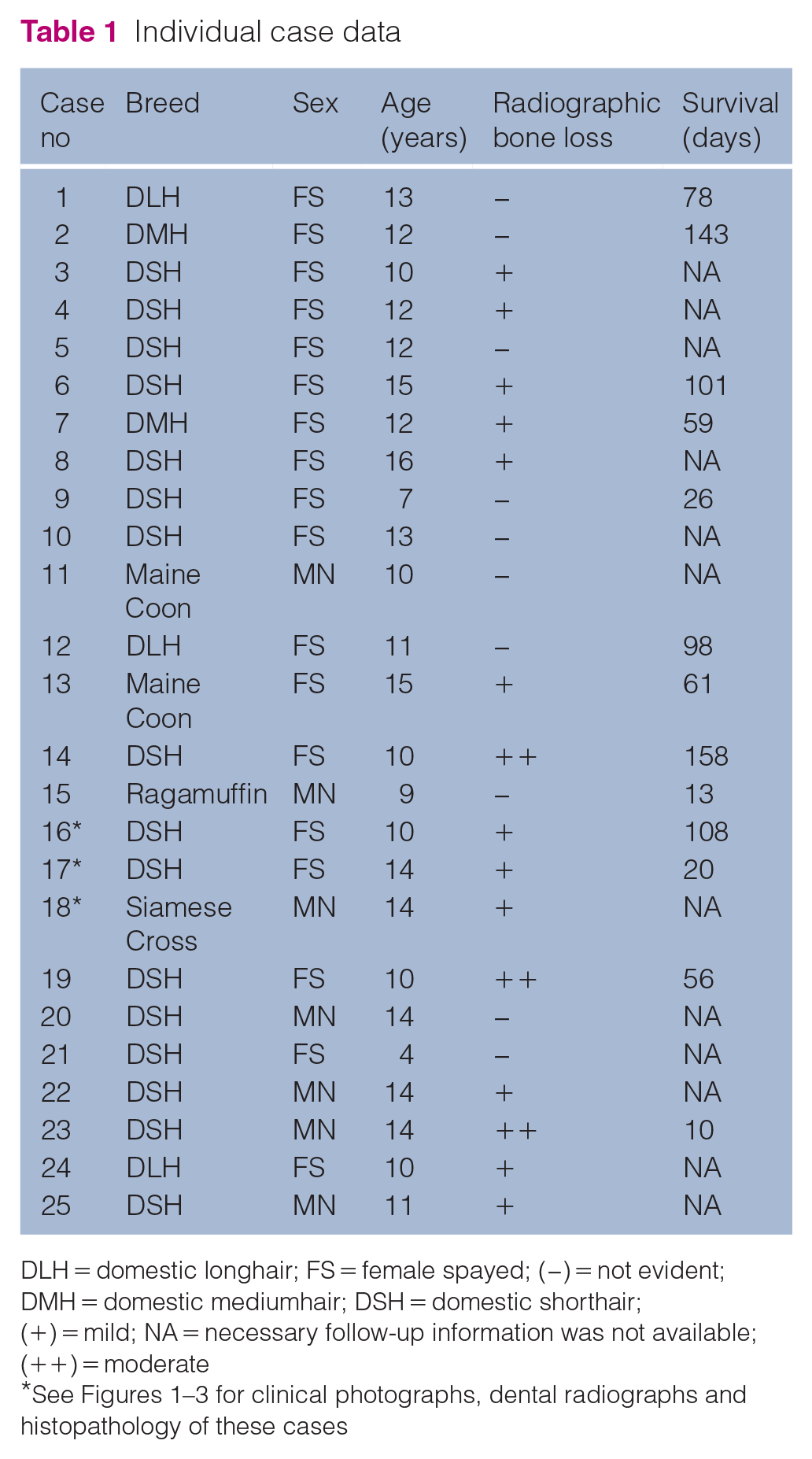
DLH = domestic longhair; FS = female spayed; (−) = not evident; DMH = domestic mediumhair; DSH = domestic shorthair; (+) = mild; NA = necessary follow-up information was not available; (++) = moderate

Feline maxillary sarcoma, case 16. (a) A poorly defined area of thickened and erythematous gingiva is centered on the right maxillary canine tooth and extends dorsal to involve the alveolar mucosa (arrows) and caudal to the mesial edge of the fourth premolar (arrowhead). (b) A dental radiograph has mild bone loss. There is a soft tissue mass (arrowheads) and fine permeative pattern of bone loss between the canine tooth and the third premolar (arrows). The second premolar is absent. (c) Neoplastic spindle cells have moderate nuclear atypia and are diffusely intermingled with small amounts of fibrillar extracellular collagen matrix (hematoxylin and eosin). (d) Neoplastic cells have strong cytoplasmic immunolabeling for vimentin. (e) Neoplastic cells have weak cytoplasmic and occasional nuclear immunolabeling for S100

Feline maxillary sarcoma, case 17. (a) A pink fleshy mass is bulging from the labial aspect of the left maxillary canine tooth, covering the left maxillary incisors and extending onto the adjacent palate (arrows). (b) A dental radiograph has mild bone loss. There is mild horizontal bone loss between the left maxillary third incisor and canine tooth (short arrows). Vertical bone loss extends along the mesial root surface of the canine (long arrows) and moderate bone loss affects the periapical region (double arrow). (c) Neoplastic spindle cells have wispy, lightly eosinophilic cytoplasm and oval nuclei with moderate anisokaryosis (hematoxylin and eosin). (d) Neoplastic cells have strong cytoplasmic immunolabeling for vimentin. (e) Neoplastic cells have moderately strong cytoplasmic and nuclear immunolabeling for S100
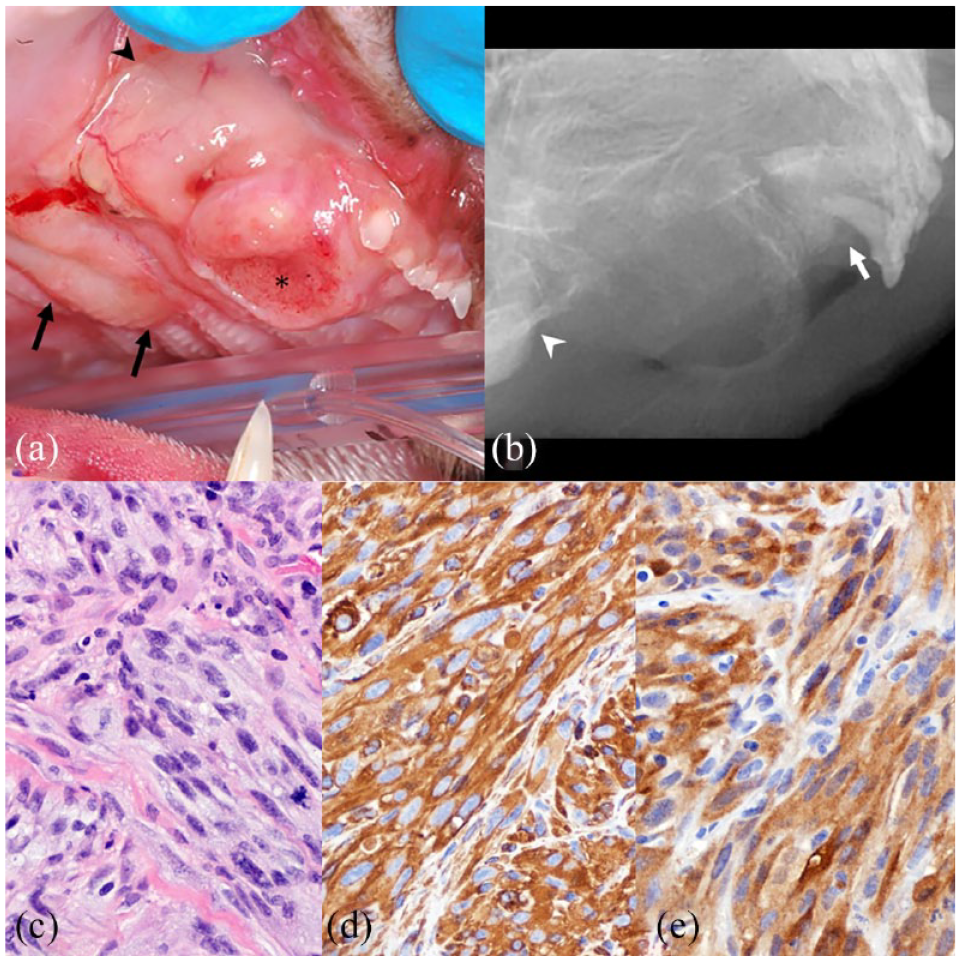
Feline maxillary sarcoma, case 18. (a) A poorly defined area of thickened and focally ulcerated (asterisk) tissue is centered on the area of the absent right maxillary canine tooth and extends to the rostral midline, well beyond the mucogingival junction (arrowhead) and caudal to involve the palate (arrows). (b) A dental radiograph of the right maxilla has bone loss that extends from the third incisor tooth (arrow) to the level of the rostral edge of the zygoma (arrowhead). The canine tooth and all premolars are absent. (c) Neoplastic spindle cells have pale-staining cytoplasm, oval nuclei and occasional stromal collagen fibers (hematoxylin and eosin). (d) Neoplastic cells have strong cytoplasmic immunolabeling for vimentin. (e) Neoplastic cells have moderate-to-strong cytoplasmic and rare nuclear immunolabeling for S100
Clinical and radiographic findings
Each of the 25 neoplasms was located within the maxilla and none involved the mandibles. The specific location and at least one clinical photograph of the lesion was available in 24 cases. Fourteen tumors (58%) occurred as a solitary mass, while 10 (42%) presented as multiple maxillary gingival swellings. In 21 cases (88%), both the right and left maxillae were involved, and 15 lesions (60%) also involved the palate (Figure 4). Affected sites on the gingiva and/or palate appeared as regionally extensive areas of irregularly thickened, pink, smooth and often ulcerated tissue or, less often, as a single discrete mass (Figures 1a, 2a, 3a and 5).

Anatomic distribution of feline maxillary sarcomas. Shading represents the location and approximate size of each tumor in this study. Areas of darker shading represent overlap of two or more tumors in different cats; therefore, density of shading correlates to the relative frequency of tumor location among the 25 cats (ie, dark gray areas were the most often affected)

Feline maxillary sarcomas. Representative photographs of tumors from 20/25 cats in the study
A previous biopsy of the lesion had been performed for six cats (24%). In four cats (cases 4, 12, 16 and 21), the previous histologic interpretation was a hyperplastic inflammatory lesion. One cat (case 19) had been diagnosed with squamous cell carcinoma at the affected site, which was not confirmed in the specimen evaluated and included in this study. One cat (case 1) had a previous histopathologic diagnosis of oral fibrosarcoma.
Dental radiographs were available for evaluation in 17 cases (68%). There was no evidence of alveolar bone loss in five cases (29%), 12 cases (71%) showed mild alveolar bone loss and three cases (18%) showed moderate alveolar bone loss (Figures 1b, 2b and 3b). Individual case data are provided in Table 1.
Microscopic features
Tissue for histologic evaluation was available in all cases. Consistent histologic findings included moderate-to-high cellularity of a solid neoplasm that was composed of spindle cells with fasciculated pattern of organization and fibrillar collagenous matrix that ranged from scant to moderately abundant (Figures 1c, 2c and 3c). The distribution of scant PAS-positive material and reticular fibers correlated with basement membranes of stromal vessels, and the collagenous nature of the stroma was further confirmed by staining blue with Masson’s trichrome (not shown). Mitotic count ranged from 0 to 75 mitotic figures per 10 high-power fields (HPFs; × 400 magnification, 2.37 mm2), with a mean of 21 ± 20 mitotic figures/10 HPFs (median 14).
Neutrophilic inflammation was common (n = 19 [76%]), while approximately half of tumors had perivascular lymphoplasmacytic infiltrates (n = 13 [52%]) in addition to, or instead of, neutrophils. Invasion into maxillary bone was observed in four (16%) tumors; however, many biopsy samples were superficial and did not adequately represent the relationship of the neoplasm to the underlying bone.
Of the six tumors for which IHC was performed, each had strong cytoplasmic labeling for vimentin in >75% of neoplastic cells (Figures 1d, 2d and 3d) and none had positive labeling for SMA or melan A. S100 labeling of cytoplasm and nuclei was moderate to strong in 5/6 tumors tested (Figures 1e, 2e and 3e).
Treatment and follow-up
Each case was submitted as an incisional biopsy and/or surgical debulking specimen without marginal tissue. Excisional biopsy with curative intent was not attempted for any of the 25 cats. In 17 cases, the submitting veterinarian completed the study questionnaire. Of these cats, 16 (94%) received supportive care only and one (6%) received palliative radiation therapy (800 cGy × four doses). Four cats were later lost to follow-up, leaving a total of 13/25 cats (52%) with adequate follow-up information. As disease progressed, thickening of adjacent lips was reported for four cats (31%) and ocular clinical signs were reported for three cats (23%), including one that had decreased retropulsion of the globe. Eleven (85%) of the cats were euthanized owing to recurrence of the mass or complications due to local progression, primarily an impaired ability to eat due to local mass effect and/or occlusal pain. One (7.7%) cat died at home 10 days after surgical biopsy, with no cause confirmed. One (7.7%) cat was alive at the time of last follow-up (78 days following diagnosis). In the cat that was treated with palliative RT, a decrease in measured tumor size was achieved and maintained at 2 months following treatment. However, recurrence (increase in tumor size) was noted 3 months after treatment. Ultimately, the cat was lost to follow-up following recurrence at 3 months and survival time was unavailable. One other case in which local recurrence was noted at 2 weeks was also lost to follow-up.
Metastatic disease was not documented. Regional lymph node enlargement was not reported in the clinical history for any cat; however, most cats did not have full clinical staging. Two cats (cases 9 and 19) had thoracic radiographs and one cat (case 8) had a cranial, cervical and thoracic CT scan to check for metastatic disease. One cat (case 15) had cytologic evaluation of aspirates from the mandibular lymph nodes. None of the cats in the study were submitted for post-mortem evaluation (ie, necropsy).
Survival time was calculated from the time of diagnosis to the time of death. Death that occurred 10 days following biopsy was censored, owing to the possibility of perioperative complications. Median survival time was 70 days (range 13–158) and mean survival time was 77 ± 47 days (95% confidence interval 51–103 days) (Figure 6).

Kaplan–Meier survival plot (n = 12). Survival data were available for 13/25 cats. One cat was censored due to proximity of death to the time of biopsy and suspected surgical complications. Median survival time for the remaining 12/25 cases was 70 days (range 13–158). Mean ± SD survival time was 77 ± 47 days (95% confidence interval 50–103)
Discussion
Feline maxillary sarcoma is invasive and predominantly a tumor of the maxillary gingiva that progresses to invade the bone, palate and/or upper lips. Although spindle cell neoplasia occurs in the mandible of cats, this data set suggests that mandibular tumors are likely to be identified as a specific type (eg, melanoma, osteosarcoma and nerve sheath tumor), and that unspecified mandibular spindle cell sarcomas are rare in cats. Feline maxillary sarcoma occurs in middle aged to older cats. The average age of 12 years is similar to the reported mean age of 12.5 years for oral squamous cell carcinoma in cats. 2 In contrast to previous studies that reported a more equal sex distribution for oral fibrosarcoma in cats, female spayed cats were over-represented in this study population; the significance of this is unclear.1,2 Breed distribution was interpreted as reflecting the overall cat population in the USA, with a majority of domestic shorthair cats.
Feline maxillary sarcoma can be difficult to recognize as a neoplasm, which is a significant barrier to prompt and accurate diagnosis. Clinically, there is often a poorly circumscribed maxillary and/or palatal swelling rather than a distinct mass lesion. The differential list for a maxillary mass or swelling in a cat includes malignant neoplasia, odontogenic tumors, odontogenic cysts and benign entities such as feline epulides, granulation tissue and other forms of reactive gingival hyperplasia.3,10,11 The presence of multifocal swellings or masses within one or both maxillary quadrants is presumed to be the result of multifocal mass lesions arising from a single regionally extensive neoplasm rather than true multicentric neoplasia.
Ulcerated and inflamed spindle cell sarcomas can mimic granulation tissue or benign gingival hyperplasia. Of the six cats with previous biopsies in this case series, four were diagnosed with benign, hyperplastic inflammatory lesions. An ideal incisional biopsy would sample multiple affected areas and extend deep into submucosa or alveolar bone. In cases where overt invasion or histologic features of malignancy are not evident, it is important to carefully consider all patient data. Important factors to consider are the age of the cat, concurrent dental and orofacial disease, occlusion, duration and growth rate of the lesion, and findings on dental and maxillofacial imaging.
Radiographic evidence of bone loss is generally mild; therefore, feline maxillary sarcoma may not have the appearance of an aggressive lesion, somewhat similar to the histologically low grade yet biologically high grade fibrosarcoma that often affects the maxilla of dogs. 12
Microscopically, feline maxillary sarcoma is composed of spindle cells without specific histomorphologic features. Most of the tumors had only small amounts of collagen and it cannot be known if it was produced by neoplastic cells. Histologic differentials include other spindle cell neoplasms, including fibrosarcoma, nerve sheath tumors, including Schwannoma, vascular wall tumors, smooth muscle tumors, feline restrictive orbital myofibroblastic sarcoma, osteosarcoma, melanoma and spindle cell squamous cell carcinoma.8,13 These neoplasms have significant histologic overlap and ancillary immunohistochemistry is indicated when a specific tumor type cannot be determined by histomorphology alone. While immunohistochemistry can be helpful in some circumstances, the sensitivity and specificity of the available immunohistochemical markers in veterinary medicine are limited. Consideration of other factors – such as the gross appearance, radiographic changes and overall clinical picture – are important when ruling in or out these differentials. Restriction of PAS-positive and reticulin-positive staining to the basement membranes of the stromal vessels suggests that the tested sarcomas were neither smooth muscle nor nerve sheath tumors. Immunohistochemistry was limited in this study, but none of the tested tumors had labeling characteristic of a smooth muscle tumor or melanoma. Variable positive labeling with S100, which is a marker of neural crest cells, is not surprising for a mesenchymal tumor of the maxillofacial region, which is derived largely from neural crest cells. 14
Even with immunohistochemistry, it is not always possible to classify malignant spindle cell neoplasms within the feline oral cavity. In a study by Saverino et al, 9 the majority of neoplasms suspected to be melanoma based on histomorphology were recategorized as unclassified malignant neoplasms following immunohistochemical evaluation, which showed lack of expression of diagnostic IHC markers. As in this case series, many of the neoplasms had a predominantly spindle cell morphology and occurred in the maxilla. While Saverino et al 9 provide information regarding the IHC labeling patterns of feline oral spindles cell sarcomas, their IHC panel was relatively limited (melan A, S100, PNL2, laminin, CD34, cytokeratin) and largely aimed at ruling in or ruling out an oral malignant melanoma. A more complete evaluation of the immunohistochemical phenotype of feline maxillary sarcomas is warranted, and may provide additional insight into the histogenesis, aid in establishing a diagnostic panel and/or identify prognostic markers for these neoplasms. At present, the gross, radiographic, and histologic features of these neoplasms remain the primary means of establishing a diagnosis that will inform clinical treatment and prognostication.
In humans, sarcomas of the maxillofacial regions are uncommon and approximately 80% arise within soft tissues.15,16 Metastasis is rare and the rate of local recurrence is high due to infiltrative growth of the neoplasm, as well as surgical limitations imposed by vital anatomic structures.15,16 The term spindle cell sarcoma is used in humans for locally invasive spindle cells neoplasms of the oral cavity that are diagnosed by exclusion following extensive immunohistochemical and molecular testing.15,16 While histopathology remains central to the diagnosis of sarcomas, molecular genetic and cytogenetic testing has become routine and is often considered more prognostically important than histomorphology.16,17 Genetic analysis includes conventional karyotype analysis, RT-PCR, fluorescence in situ hybridization and targeted sequencing.16,17 Investigation into specific genomic alterations in cats with maxillary sarcomas would likely provide the most sensitive and specific characterization of these neoplasms; however, these technologies are largely unavailable in veterinary medicine.
Maxillary sarcomas represent a progressive disease in cats, and a surgical cure is rarely feasible. Affected cats are frequently euthanized, owing to local tumor growth, and clinical signs are associated with oral pain, anatomic distortion and difficulty eating. Previous studies of spindle cell tumors in the jaws of cats, most of which are reported as feline oral fibrosarcoma, suggest that mandibular lesions are more likely to have a favorable outcome with treatment. Cats with oral fibrosarcoma have been treated with surgical excision, radiation therapy and/or chemotherapy. Survival times ranged from 60 days to >2 years.5–7 The median survival time of the four cases with reported survival times was 180 days.5,6 Cats with the longest survival times were treated with mandibulectomy ± radiation therapy or chemotherapy. 7 The median survival time (70 days) and mean survival time (77 ± 47 days) in this case series are notably shorter than these previous reports. It is possible that the cats lost to follow-up included those with the longest survival times. Additionally, the majority of the cats in this case series (16/17 with treatment information) were treated with palliative supportive care alone, while the cats in the previous reports were treated with chemotherapy and/or radiation therapy. This discrepancy suggests that ancillary treatments may play a role in prolonging survival. This case series may serve as a basis for comparison to future clinical studies investigating ancillary treatment options for these cats. Furthermore, all but two of the cases reported in these cited studies occurred in the mandible, while all cases in this series occurred in the maxilla. In general, mandibular tumors are more amenable to surgical excision than maxillary tumors and this fact may account for some difference in outcome. Further research investigating the significance of location relative to prognosis for feline oral sarcomas is also warranted.
Metastasis of feline maxillary sarcoma has not been documented in a cat from this study or previous studies of feline oral fibrosarcoma.5–7 A limitation of this study is the low number of cats that had thoracic imaging or evaluation of lymph node aspirates. None of the cats in the study was fully staged and none had a post-mortem necropsy performed. Had these procedures been performed for each cat, the data would have provided a much more accurate picture of the true metastatic potential. The lack of clinical staging/lymph node evaluation or necropsy in these cases precludes determination of metastasis, and local tumor progression clearly impacts prognosis more significantly than metastatic disease for cats with maxillary sarcomas. The rapid progression of clinical disease may have also contributed to a lack of reported metastatic disease as the patients may not live long enough to develop clinically relevant metastatic disease.
Owing to the retrospective nature of the investigation, there was no standardization of the information included in the submission forms and medical records. As this is a retrospective analysis of samples submitted to a referral pathology laboratory, complete medical records and information regarding survival data were obtained via a voluntary questionnaire that was sent to the submitting veterinarian. While the response rate to the questionnaires was 68%, some cases were lost to follow-up so that adequate follow-up information was available for only 52% of the total cases. Because most cats in this study were biopsied by a specialist in veterinary dentistry, the data may be biased towards cats with advanced disease. This may contribute to the relatively shorter median survival time in this study vs the survival times reported in other case series.
Conclusions
Feline maxillary sarcoma is an invasive neoplasm that occurs in middle-aged to geriatric cats and often involves the palate and/or upper lip. These neoplasms carry a poor prognosis, with most cats succumbing to the effects of local disease following tumor recurrence and/or progression. These neoplasms can be difficult to recognize clinically owing to the gross appearance of smooth or ulcerated thickenings that may be bilateral within the maxilla and mild radiographic bone loss at the time of biopsy. Histologically, these neoplasms can be easily misdiagnosed as hyperplastic or inflammatory lesions. The cell or tissue of origin is not clear, although spindle cell morphology and biologic behavior are characteristic of a sarcoma. High-quality biopsy samples and correlation of histopathology with the clinical and radiological findings are key to an early and accurate diagnosis.
Footnotes
Acknowledgements
The authors wish to thank the many clinical veterinarians who submitted these cases, granted permission for the use of the clinical photographs and provided invaluable follow-up information.
Author note
This paper was presented, in part, at the 2016 American College of Veterinary Pathologists Annual Meeting in New Orleans, LA, USA.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or humans individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
