Abstract
Objectives
Adrenal gland size and its association with body weight have been rarely evaluated in cats. This study was undertaken to assess the association between feline body weight and adrenal gland thickness, and to propose reference intervals (RIs) for adrenal gland thickness in healthy cats.
Methods
This was a cross-sectional study in which 39 healthy cats were included. The cats were divided into two weight categories, classified as ⩽4.0 kg and >4–8 kg of ideal body weight (with 13 and 26 cats in each group, respectively), which took into consideration the body condition score of the cats. All cats underwent an ultrasound examination that was taken from a subcostal position. Maximum dorsoventral thicknesses of the left (MTL) and right (MTR) adrenal glands were measured in a sagittal plane. RIs were obtained for the maximum thickness (MT), which included the MTLs and MTRs of each cat. RIs with the 90% confidence intervals were calculated according to American Society for Veterinary Clinical Pathology guidelines on RIs.
Results
No statistical differences for adrenal gland thickness were observed between the left and right (P = 0.543) adrenal glands or between male and female cats (P = 0.943). Mean MT was significantly greater in the group of cats weighing >4–8 kg compared with the group of cats weighing ⩽4 kg (3.7 ± 0.6 vs 3.2 ± 0.4 mm; P <0.005). The lower limit of the RI for MT was 2.4 mm (range 2.2–2.6 mm) in the group weighing ⩽4 kg and 2.6 mm (range 2.4–2.8 mm) in the group weighing >4–8 kg. The upper limit of the RI for MT was 3.9 mm (range 3.7–4.1 mm) in the group of cats weighing ⩽4 kg and 4.8 mm (range 4.6–5.1 mm) in the group of cats weighing >4–8 kg.
Conclusions and relevance
The use of RIs based on two group sizes allows for a more accurate ultrasonographic evaluation of adrenal gland thickness in cats. The maximum normal adrenal gland thickness is lower in smaller cats (3.9 mm for those weighing ⩽4 kg and 4.8 mm for those weighing >4–8 kg).
Introduction
Ultrasonography is a useful diagnostic technique for the assessment of adrenal gland disorders in cats. An important use is the detection of adrenal asymmetry as the finding of a unilateral adrenal mass is suspicious for adrenal neoplasia.1–3 Primary hyperaldosteronism (Conn syndrome) is the most common adrenocortical disorder in cats; less common feline adrenal neoplasias include adrenal-dependent hyperadrenocorticism, sex hormone-secreting neoplasia, phaeochromocytoma, non-secreting neoplasia and metastasis.2,4
Ultrasound evaluation may also be of help to identify adrenomegaly consistent with bilateral hyperplasia; however, this finding might be less reliable as it can occur not only in cats with pituitary-dependent hyperadrenocorticism and hyperaldosteronism, but also in cats with chronic stress or non-adrenal disorders (acromegaly and hyperthyroidism).3,5–7
Adrenal hyperplasia can cause diffuse adrenal enlargement; however, hyperplastic nodular lesions might also occur in endocrine diseases such as hyperaldosteronism. Therefore, adrenal size should be considered and the evaluation of other ultrasonographic changes of adrenal structure might be helpful in the detection of adrenal lesions.
The ultrasonographic appearance of normal feline adrenal glands can be bean-shaped, oval or fusiform, and they are usually hypoechoic compared with surrounding tissues. Normal adrenal gland size has only been evaluated in four studies, and the maximum normal thickness reported varied from 4.5 to 5.3 mm.8–11 As no differences have been found for adrenal thickness between sick and healthy cats, some of these studies combined both groups for the assessment of adrenal gland size.8,9 However, it has been reported that chronic stress resulting from long-term illness may cause increased adrenal gland function and size, as has been observed in cats with hyperthyroidism. 5
Additionally, the relationship between adrenal gland thickness and body weight has not been extensively studied in cats. One study retrospectively assessed CT images of adrenal gland size in healthy cats, and no association was found between body weight and adrenal gland size in 30 cats. 12 Similarly, another study reported no correlation between adrenal thickness and body weight, and between adrenal thickness and body surface area in diseased cats. 10
In one study, most of the adult cats (82%) weighed >4 kg, whereas only 18% weighed <4 kg. 13 Therefore, cats with a body weight of ⩽4 kg could represent a less frequently observed population characterised by a smaller size. We hypothesised that these smaller cats could have a smaller adrenal thickness and therefore using reference intervals (RIs) established for the entire population of cats might not be appropriate for accurate detection of adrenomegaly in smaller cats. The aims of this study were to elucidate whether or not body weight may affect adrenal gland thickness in cats and to propose RIs for adrenal gland thickness in two weight categories (⩽4 kg and >4–8 kg) of healthy cats. These two weight categories could avoid large variations of weight within each group.
Material and methods
Selection of animals and study design
A cross-sectional study was performed at the Veterinary Teaching Hospital (University of Las Palmas of Gran Canaria [ULPGC]) and included healthy adult cats. This study was part of a larger, more extensive study. 14 This study was approved by the Animal Welfare Ethics Committee, ULPGC, Spain (reference number 10/2018). Cats were recruited after owners voluntarily signed an informed consent form. Cats were considered healthy based on a clinical questionnaire, and a normal physical examination, biochemical profile and abdominal ultrasound.
Cats were classified as ⩽4 kg and >4–8 kg of ideal body weight. The ideal body weight of cats was calculated with the following formula: current weight × (100/[100 + 10 × (current body condition score – 5)]). 15 The body condition score was based on a nine-point scale.
Ultrasound evaluation
An ultrasound system (LOGIQ P5; GE Healthcare) with a linear transducer (3–11 MHz) and an ultrasound system (Z.one Pro; Mindray) with a curve array transducer (3–9 MHz) were used to perform the abdominal ultrasonographic examinations. All cats underwent an ultrasound examination that was taken in a subcostal position. Maximum craniocaudal lengths and maximum dorsoventral thicknesses of the left (MTL) and right (MTR) adrenal glands were measured in a sagittal plane (Figure 1). The same person (LPL) performed all the measurements. Images were reviewed and, when necessary, measurements were corrected using ImageJ 1.51s (National Institutes of Health).

(a) Left and (b) right adrenal glands of a 5-year-old neutered male Siamese cat that weighed 3.7 kg. (a) Measurement of the maximum dorsoventral thickness (3.5 mm) of the left adrenal gland; (b) measurement of the maximum dorsoventral thickness (3.6 mm) of the right adrenal gland
Statistical analyses
The normality of the parameters was assessed using Q-Q plots and the Kolmogorov–Smirnov test. Qualitative variables are described as n (%). Quantitative variables are given as mean ± SD when they were normally distributed, or as median and interquartile range (IQR; 25th–75th percentile) when they were non-normally distributed. For comparisons, the t-test or pairwise Mann–Whitney U-test were used, as appropriate. Percentages were compared using Fisher’s exact test. These statistical analyses were performed with SPSS Statistics 25 software (IBM). Two-tailed P values <0.05 were considered to be statistically significant.
RIs were obtained for the maximum thickness (MT) that included both the MTL and MTR of each cat. These RIs were calculated with a parametric method according to the American Society for Veterinary Clinical Pathology’s RI guidelines, 16 and outliers were identified using the Cook method. These analyses were performed using the R package, version 3.6.1 (R Development Core Team, 2019).
Results
A total of 39 cats were included: 15 neutered males and 24 (23 neutered) females. Median age was 8 years (IQR 6–10 years). According to actual body weight, 10 cats were included in the ⩽4.0 kg group and 29 cats in the >4–8 kg group. Three of the 29 cats initially included in the >4–8 kg were reassigned to the ⩽4.0 kg group once ideal body weight was calculated. Therefore, according to ideal body weight, 13 cats (two males, 11 females) were included in the group weighing ⩽4.0 kg and 26 cats (13 males, 13 females) in the group weighing >4–8 kg. Data regarding actual and ideal body weight, age and breed in both groups are shown in Table 1.
Actual and ideal body weight, age and breed of cats in two weight categories (⩽4.0 kg and >4–8 kg)

Data are provided as median and interquartile range (25th–75th percentile). Qualitative variables are given as n (%)
BCS = body condition score
Of the 39 cats, MTL and MTR were not significantly different (3.5 ± 0.7 vs 3.6 ± 0.6 mm; P = 0.543). The mean MT of the 78 adrenal glands from the 39 cats was 3.5 ± 0.6 mm. MT was not significantly different between 15 male cats and 24 female cats (3.7 ± 0.6 mm vs 3.5 ± 0.6 mm; P = 0.943). MT was significantly greater in cats weighing >4–8 kg compared with cats weighing ⩽4 kg (3.7 ± 0.6 mm vs 3.2 ± 0.4 mm; P <0.005). Other ultrasonographic measurements are given in Table 2. RIs for adrenal gland thickness in the cats are shown in Table 3.
Ultrasonographic adrenal gland measurements of cats in two weight categories (⩽4.0 kg and >4–8 kg)
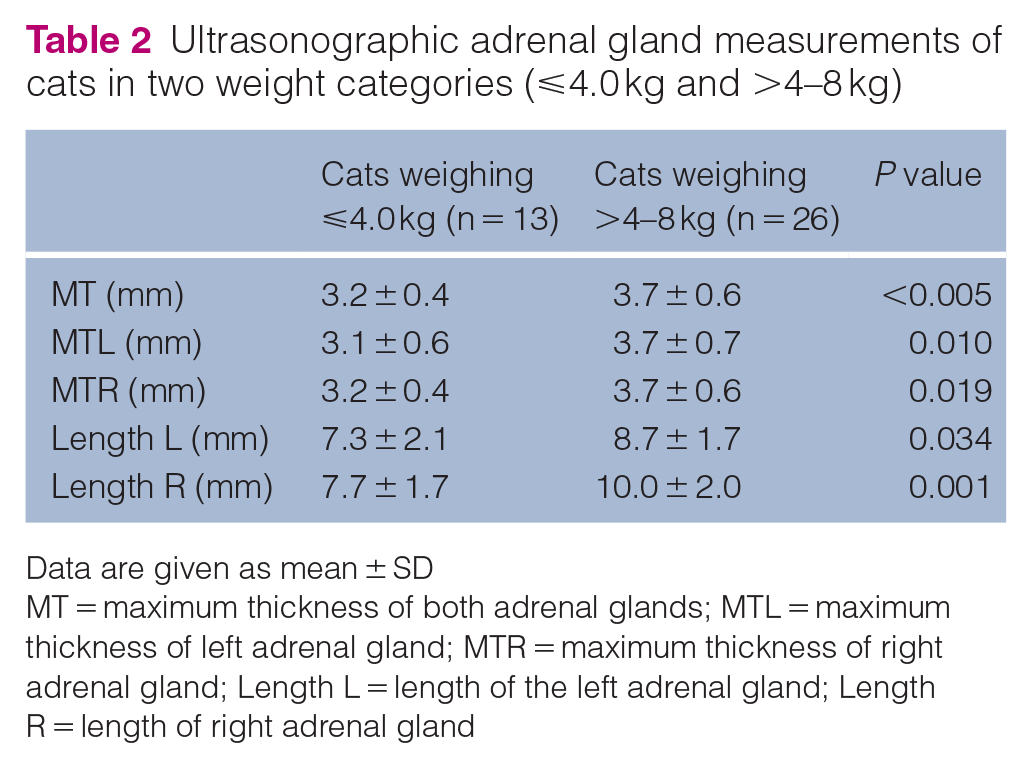
Data are given as mean ± SD
MT = maximum thickness of both adrenal glands; MTL = maximum thickness of left adrenal gland; MTR = maximum thickness of right adrenal gland; Length L = length of the left adrenal gland; Length R = length of right adrenal gland
Reference intervals (upper limit and lower limit) and their 90% confidence intervals following the parametric method (American Society for Veterinary Clinical Pathology reference interval guidelines) for maximum gland thickness in 39 healthy cats based on two weight categories

Number of adrenal glands included to calculate the reference interval
Discussion
This study shows that the RIs for feline adrenal gland thickness should be based on size groups. Smaller size cats (ideal body weight ⩽4.5 kg) had a lower MT than larger cats (ideal body weight >4–8 kg), (3.9 vs 4.8 mm). Similarly, in previous studies, the maximum thickness of feline adrenal glands ranged from 4.0 to 5.3 mm.8–11 However, feline adrenal gland thickness has not been previously evaluated according to body weight categories and the results of this study showed an association between cats size and the adrenal thickness. As previously reported, 9 no statistical differences were detected between the left and right adrenal glands of healthy cats. In contrast to dogs, the left and right adrenal glands of cats do not show an apparent difference in morphology on ultrasound examination. In dogs, the left adrenal gland has been described as being ‘peanut’ shaped, whereas the shape of the right adrenal gland is more variable (oval or more complex).17,18 In cats, the appearance of the adrenal glands can vary slightly from mildly bean-shaped to oval or fusiform. 9 However, differences from one gland to another can be slight and subjective, and, to date, marked differences between left and right adrenal glands have not been described in this species. Therefore, it is considered that the size of both glands can be interpreted under the same RI. In contrast, the interpretation of adrenal gland thickness according to the size of the cat could be important. Ultrasonographic evaluation of the adrenal glands is a useful tool to support the diagnosis of adrenal disorders in cats, and precise interpretation of adrenal gland thickness based on the size of the cat may be crucial to detect adrenomegaly in cats, especially at initial stages of adrenal neoplasia or in disorders that can present adrenal hyperplasia.1–4
The length of the right and left adrenal glands were statistically different between groups of cats (those weighing ⩽4 kg vs those weighing >4–8 kg). However, previous studies have shown that the length of the adrenal gland is an ultrasonographic measurement with greater intra- and inter-observer variability than the thickness of adrenal gland in dogs and cats19,20 and, owing to this limitation, adrenal gland length is considered to be less useful for assessment of adrenal gland size. Therefore, further statistical analyses of the length of adrenal glands were not conducted.
This study has some limitations. First, the sample size included to calculate the RIs for cats ⩽4 kg was smaller than the sample size included for cats weighing >4–8 kg (26 vs 52 adrenal glands). Furthermore, most cats included in the ⩽4 kg group were females (two males vs 11 females) as it is rare that male cats weigh ⩽4 kg; sex was equally distributed in the >4–8 kg group. However, in our study no statistically significant differences were found for adrenal gland thickness between male and female cats. A previous study that included a larger number of cats did not find differences between males and females. Therefore, this potential limitation might be partially mitigated.
Finally, another limitation of this study is that sensitivity and specificity of the proposed RIs have not yet been evaluated in cats with adrenal diseases and cats with non-adrenal diseases (eg, cats with hyperthyroidism).
Conclusions
This study proposes new RIs for adrenal gland thickness in cats based on two groups of body weight. Maximum adrenal gland thickness should be considered as 3.9 mm in cats weighing ⩽4 kg and 4.8 mm in cats weighing >4–8 kg.
Footnotes
Author note
This study was presented as an oral communication at the European College of Veterinary Diagnostic Imaging 2020 Online Conference
Conflict of Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
A grant from Consejo Social of the University of Las Palmas de Gran Canaria helped with the measurement of some of the biochemical parameters. In addition, the study was performed during a pre-doctoral fellowship (LP-L) granted by the University of Las Palmas de Gran Canaria.
Ethical approval
This work involved the use of non-experimental animals only (including owned or unowned animals and data from prospective or retrospective studies). Established internationally recognised high standards (‘best practice’) of individual veterinary clinical patient care were followed. Ethical approval from a committee, while not necessarily required, was nonetheless obtained, as stated in the manuscript.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedure(s) undertaken (either prospective or retrospective studies). For any animals or humans individually identifiable within this publication, informed consent (either verbal or written) for their use in the publication was obtained from the people involved.
