Abstract
Objectives
This study aimed to describe the prominent landmarks for an in-plane ultrasound-guided lateral approach to an axillary RUMM block in cats and document the anatomical distribution of injected solution in cadavers.
Methods
Eleven mixed breed, frozen-thawed cat cadavers were used. The ultrasound probe was placed on the lateral aspect of the forelimb. A spinal needle was used to inject either methylene blue 5 mg/ml or permanent tissue dye 1:10 within the focal clustering point of the radial (R), ulna (U), median (M) and musculocutaneous (Mc) nerves. The block was approached from either a cranial or caudal direction. Specimens were immediately dissected. A successful block was defined as ⩾20 mm of continuous stain on all nerves (R, U, M and Mc), a partial block as one or more nerves stained ⩾20 mm continuously and a negative block failed to stain any nerve ⩾20 mm.
Results
The cranial approach stained the R 8/9, U 6/9, M 6/9 and Mc 8/9 nerves successfully, while the caudal approach stained the R 7/9, U 8/9, M 7/9 and Mc 5/9 nerves successfully. There were no statistical differences on staining rates between approaches or limbs used.
Conclusions and relevance
The lateral approach to an axillary RUMM block is feasible in cats and may be a useful alternative to current approaches used for brachial and antebrachial desensitisation. Both cranial and caudal approaches produced similar results; however, further research in live animals will determine the clinical applicability.
Introduction
Locoregional anaesthesia is used to produce sensory and/or motor blockage to an anatomical area of interest. The local deposition of an agent to reduce or eliminate nerve pulse transmission for surgical desensitisation, chronic pain management or diagnostic investigation is widely practised. Additional benefits include reducing the minimum alveolar concentration of volatile agents and systemic administration of opioids. 1
In the cat the brachial plexus is formed by cervical-sixth through thoracic-first spinal nerves and provides innervation to the thoracic limb. Ultrasound-guided locoregional anaesthesia of the thoracic limb either targets the brachial plexus system at the paravertebral location, 2 within the axillary space,3,4 or individual radial (R), ulnar (U), median (M) and musculocutaneous (Mc) nerves traversing the brachium distally, as a RUMM block. 5 The intended regions for desensitisation, operator experience and equipment availability largely dictate the approach. For example, the axillary brachial plexus and RUMM blocks produce reliable anaesthesia to the antebrachium, 6 whereas the paravertebral brachial plexus block may provide anaesthesia to the proximal brachium.
Traditional RUMM approaches target the R nerve laterally, and the M, U and Mc nerves medially as a single bundle at the level of the mid brachium.5,7 Leung et al 4 recently described an ultrasound-guided RUMM block in cat cadavers, using a single proximal humeral injection site from the lateral side. Targeting the R nerve first, followed by needle redirection towards the U, M and Mc nerve bundle; the authors concluded anatomical staining of all RUMM nerves was achievable, and the lateral approach may improve operator ease and potentially reduce myogenic and neurovascular trauma compared with traditional approaches.5,7–9 To our knowledge, a lateral approach to an axillary RUMM block using a single injection site, without needle redirection, has not been described in cats.
The aims of this study were to describe the prominent landmarks for an in-plane ultrasound-guided lateral approach to an axillary RUMM block in cats and to document the anatomical distribution of injected solution in cadavers. The technique was to be approached from both cranial (Cr) and caudal (Ca) directions. This novel technique was hypothesised to be both feasible and effective when described anatomically. No differences in staining success rates were expected between approaches.
Materials and methods
Eleven mixed-breed cats (seven females, two males, two male neutered) weighing a mean ± SD of 3.1 ± 1.9 kg were used. All specimens were whole, without trauma and had been thawed for 24 h prior to use. The animals died or were euthanased for reasons other than inclusion in this study and their bodies were donated by their owners to the university for research purposes. The local animal ethics authority was informed of the project, however, specific approval was not required.
A linear 15-6 MHz probe and ultrasound machine (M Turbo; Sonosite Fujifilm) was used. The specimens were placed in lateral recumbency with the forelimb in a semi-extended, neutral position (Figure 1). The glenohumeral joint, greater tubercle of the humerus and lateral epicondyle of the humerus were identified on palpation. The probe was placed caudal to and in line with the greater tubercle of the humerus, transverse to the sagittal limb. Landmarks identified on the ultrasound image included the first rib in the far field, the greater tubercle of the humerus in the cranial near field and the R, U, M and Mc nerves within their fascial plane in the mid-field, caudal to the humerus and surrounded by the musculature and fat of the brachium (Figure 2). The probe was fanned 10 until median ulnar nerve diversion was appreciable and an optimal image for the four-nerve bundle was achieved.A 22 G, 2.5 inch spinal needle (BD; Becton Dickinson) was inserted in-plane with the transducer. The stylet was removed once the dermis was penetrated. The cranial approach ran the needle over the greater tubercle of the humerus, while the caudal approach penetrated the triceps mass. When the tip of the needle was located within the visual centre of the four-nerve cluster, 0.2 ml/kg body weight solution was injected. If at initial injection the solution dispersed outside the intended fascial plane, the needle was redirected and the injection continued. No additional solution was used. Methylene blue 5 mg/ml solution (Proveblue; Provepharma) was used in specimens 3–6 and a 1:10 dilution of permanent tissue dye (Bradley Products) in sterile water for injection (Troy-Ilium) was used in specimens 7–11. Specimen 1 was used for gross anatomical dissection to identify major R, U, M and Mc nerves and associated structures of the axilla and proximal brachium. Specimen 2 was used to assess nerve staining characteristics using the permanent tissue dye, as the authors had no previous experience with the product.
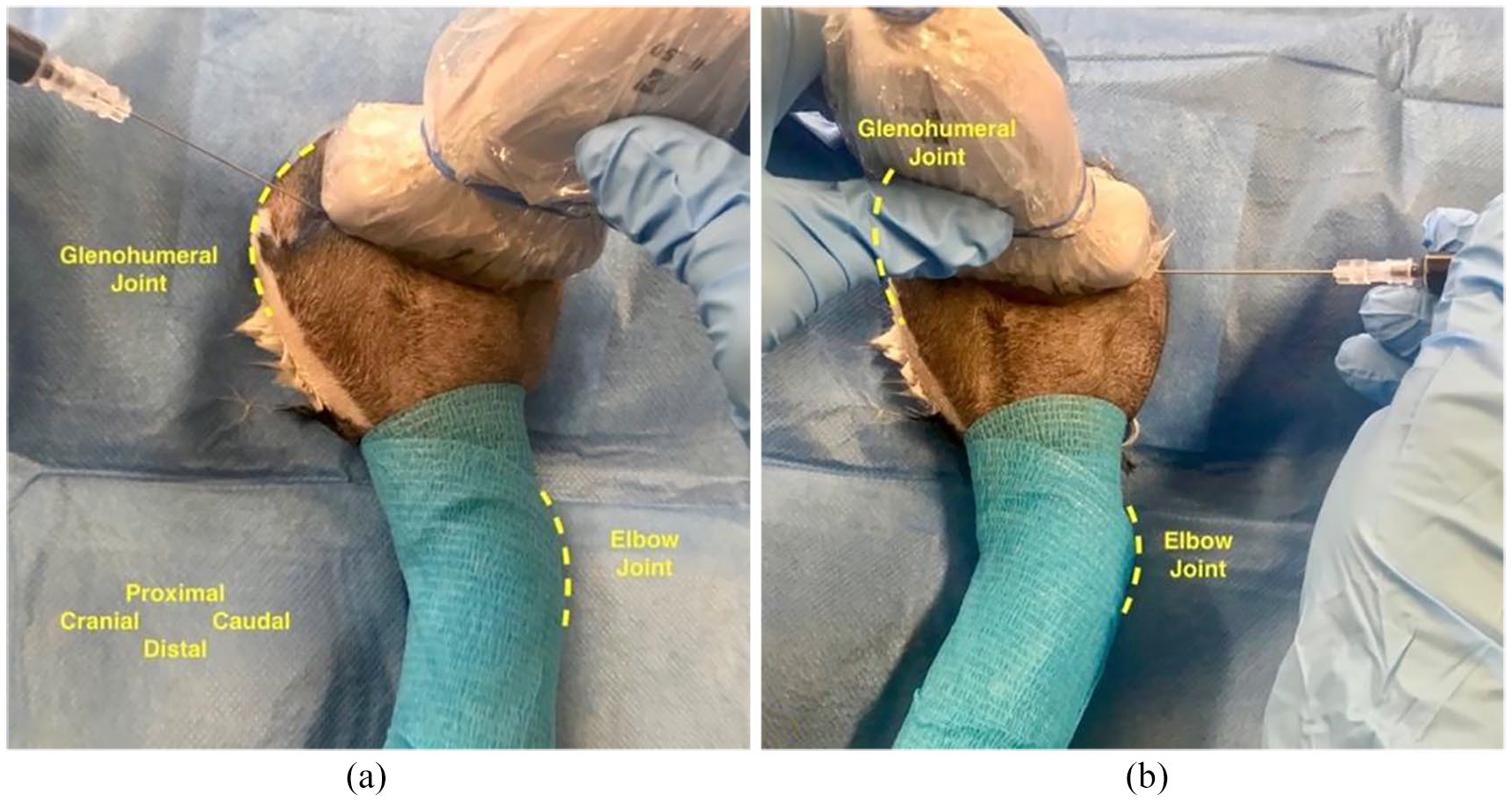
Positioning and execution of (a) cranial and (b) caudal approaches to a lateral ultrasound-guided axillary RUMM block in cat specimens. Anatomical orientation is provided in (a). RUMM = radial, ulnar, median and musculocutaneous nerves

Execution of (a) cranial and (b) caudal approaches to a lateral ultrasound-guided axillary RUMM block in cat specimens. Anatomical orientation is provided in image (a). RUMM = radial (R), ulnar (U), median (M) and musculocutaneous (Mc) nerves
The injections were performed by the same novice operator (SBP) on two separate occasions 7 days apart (specimens 1–6 on day 1 and specimens 7–11 on day 8). The order of approach (Cr or Ca) and limbs used for each approach (right or left) were individually randomised by random draw. A second, non-blinded investigator (FM-T) dissected the specimens immediately after injections. The dissection method followed previous descriptions by Anson et al. 11 A successful block was defined as ⩾20 mm of continuous stain on all nerves, a partial block was defined as one or more nerves stained ⩾20 mm continuously and a negative block failed to stain any nerve ⩾20 mm.
Data management and statistical analysis
Nerve-staining success rates remained as population proportions. Thirty-two individual contingency tables were created in Prism 8 (Graphpad Software). Fisher’s exact tests were used to investigate statistical differences (P <0.05) in staining success rates between approaches (Cr and Ca) and limbs used (right and left).
Results
Ultrasonographic visualisation of the R, U, M and Mc nerves using the described technique was acceptable in all frozen-thawed cadaver specimens and all injections were successfully executed. At specimen dissection, the injected coloured dye distributed extensively within the central axillary space bound by the subscapularis muscle deep, the pectoralis muscles superficial, the common insertion of the latissimus dorsi and teres major tendons caudally and the greater tubercle of the humerus and coracobrachialis muscle cranially. Extension of dye towards the distal brachium was bounded by the biceps brachii muscle cranially and the medial head of the triceps muscle caudally (Figure 3). In specimens 5 (Ca, methylene blue) and 7 (Cr, permanent tissue dye), however, injected solution was deposited deeper, between the triceps muscle bellies. No intrathoracic or intravascular injections were noted. In specimen 2, the 1:10 dye dilution was found to acceptably stain nerve bundles and was not susceptible to physical removal at dissection.

Medial view of the left foreleg of a cat specimen with the pectoralis muscles reflected. Structures of interest have been highlighted. The leg was stained with methylene blue 5 mg/ml using a lateral ultrasound-guided axillary RUMM approach. RUMM = radial (R), ulnar (U), median (M) and musculocutaneous (Mc) nerves
Population statistics and nerve staining results are presented in Tables 1 and 2. A cranial approach was performed three times on the right limb (CrR) and six times on the left limb (CrL). A caudal approach was performed six times on the right limb (CaR) and three times on the left limb (CaL). The cranial approach stained the R 8/9, U 6/9, M 6/9 and Mc 8/9 nerves successfully, while the caudal approach stained the R 7/9, U 8/9, M 7/9 and Mc 5/9 nerves successfully. A successful block was achieved in 6/9 cranial and 5/9 caudal approaches. A partial block occurred on five occasions: two caudal individual Mc nerve failures; two cranial combined M and U nerve failures; and one caudal combined R, M and Mc nerve failures. A complete negative block occurred once cranially and once caudally. In both, the dye was found within the fascial planes intersecting the triceps muscle bellies. There were no statistical differences on staining rates between approaches or limbs used.
Population statistics for a lateral ultrasound-guided axillary RUMM block in 11 cat cadavers using a cranial and caudal approach

R = radial nerve; U = ulnar nerve; M = median nerve; Mc = musculocutaneous nerve; DSH = domestic shorthair; Cr = cranial; l = left; r = right; Ca = caudal; DMH = domestic mediumhair
Statistical analysis for a lateral ultrasound-guided axillary RUMM block in eleven cat cadavers using a cranial and caudal approach

Successful blocks stained nerves ⩾20 mm continuously
CrR = cranial approach right limb; CrL = cranial approach left limb; Cr = cranial; CaR = caudal approach right limb; CaL = caudal approach left limb; Ca = caudal; RUMM = radial, ulnar, median, musculocutaneous nerves
Discussion
This is a novel description of a lateral ultrasound-guided axillary RUMM block in cat cadavers. The ultrasound image was easily visualised by a novice operator and both cranial and caudal approaches generated promising R, U, M and Mc nerve staining results. Distribution of injected dye solutions was generally within the targeted proximal axillary region and major collateral staining was not described.
In this study, combined M and U nerve stain failures using the cranial approach (specimens 8 and 10) may be explained by the nerves’ close proximity to each other within the proximal brachium, caudal to the R and Mc nerves. Similarly, repeat caudal Mc nerve stain failures may have occurred owing to the nerves’ cranial location. The isolated successful staining of the U nerve using the caudal approach (specimen 11) may be explained by the caudal location of this nerve as it splits away from the median–ulnar nerve bundle and traverses the caudal brachium distally to pass over the olecranon. 11 It remains unclear whether these stain failures are suggestive of individual variability in tissue plane distribution, or whether inappropriate volumes of stain were used. Evidence for an axillary sheath surrounding the R, U, M and Mc nerves has recently been described in dogs. 8 Despite no visualisation of one in this study, its existence in the cat is plausible and may explain the variability in nerve stain characteristics between specimens, as well as the complete stain failures in specimens 5 and 7. In these specific specimens (5 and 7), injected solution between the triceps muscles indicate incorrect needle placement, possibly outside a fascial plane.
Evans and de Lahunta 12 summarised location and target innervation of the major nerves of the brachial plexus in the dog, and Anson et al 11 correlated ultrasonographic images to anatomical dissection of the feline thoracic limb. The proximal approach described in this study aims to target nerve bundles within the axilla prior to major nerve division (Figure 3). In combination with individual variations in anatomy, nerve divergence presents a major problem for partial brachial and antebrachial desensitisation with traditional RUMM approaches. For example, complete cutaneous innervation is often difficult to achieve clinically as axillary and Mc nerve divisions innervate distal brachial and antebrachial cutaneous tissues.12,13 While a full brachial plexus block (which must include the axillary, subscapular and suprascapular nerves) is unlikely, the proximal spread of dye using this technique may achieve a more complete sensory block over traditional RUMM approaches.
The electrical impulse blocking potential of local anaesthetics depends largely on drug potency and the length of nerve exposed. Using dissected frog sciatic nerves, Raymond et al 14 demonstrated a minimal 6 mm block length and critical lengths between 20 and 25 mm using lidocaine HCl at varying concentrations. The current study describes a 20 mm target stain length, as do most cadaveric studies.8,15,16 The dose rate of 0.2 ml/kg body weight was extrapolated from Campoy et al 15 and aimed to minimise the risks of collateral nerve staining when using larger volumes as documented by Anson et al. 3
As with most cadaveric studies, major limitations to this study include undetected neurovascular damage and translating how nerve staining (block) relates to nerve desensitisation clinically. Using this modified lateral approach, theoretical damage may be possible to the axillary artery, its continuation as the brachial artery and the cranial and caudal circumflex arteries. Using frozen-thawed cadaveric specimens is known to alter nerve echogenic characteristics. 2 Despite this, all four nerves were adequately visualised prior to aliquot injection. Characteristics of solution behaviour in terms of distribution through tissue previously frozen, when compared with that of live tissue, remains largely unclear. Reuptake of solution by lymphoid (major) and venous plexuses (minor) is postulated to be a cause for reduced clinical efficacy of previously evaluated cadaveric blocks. 17 Successful and repeatable regional desensitisation in live animals, however, does tend to suggest translatable nerve staining characteristics. For example, an ultrasound-guided axillary brachial plexus block successfully stained 6/6 RUMM nerves in cat cadavers 3 and produced visually complete motor and sensory block to the antebrachium in 6/8 cases clinically. 6 Therefore, only live models can compare clinical efficacy between this novel approach and traditional RUMM approaches.
Conclusions
The lateral approach to an ultrasound-guided axillary RUMM block is feasible in cats and may be a useful alternative to current approaches for distal brachial and antebrachial desensitisation. Both cranial and caudal approaches produced similar results; however, further research in live animals will determine its clinical applicability.
Footnotes
Acknowledgements
The authors would like to acknowledge The University of Sydney, University Veterinary Teaching Hospital Camperdown post-mortem staff for their commitment to research and assistance with specimen management.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work involved the use of non-experimental animals only (including owned or unowned animals and data from prospective or retrospective studies). Established internationally recognised high standards (‘best practice’) of individual veterinary clinical patient care were followed. Ethical approval from a committee was therefore not necessarily required.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedure(s) undertaken (either prospective or retrospective studies). No animals or humans are identifiable within this publication and therefore additional informed consent for publication was not required.
