Abstract
Objectives
The first objective was to assess correlation between free thyroxine (fT4) measurements by equilibrium dialysis (fT4ED; Antech Diagnostics) and a chemiluminescent enzyme immunoassay (fT4CEIA; IMMULITE 2000 Veterinary Free T4 [Siemens Healthcare Diagnostics Products]) in hyperthyroid, otherwise healthy, cats before (T0), and 1 month (T1) and 11–23 months (T2) after radioactive iodine (131I) therapy. The second objective was to determine correlation between thyroid status based on fT4 (by both techniques) and the gold standard, thyroid scintigraphy.
Methods
Thyroid status, including thyroid-stimulating hormone (TSH), total thyroxine (TT4) and fT4 serum concentrations, were assessed in 45 client-owned hyperthyroid cats before (T0), and 1 month (T1) and 11–23 months (T2) after 131I therapy. fT4 was determined by a chemiluminescent enzyme immunoassay (CEIA) and equilibrium dialysis (ED). Quantitative thyroid scintigraphy (with sodium 99m-Tc-pertechnetate) was performed at T2.
Results
Spearman correlation between fT4CEIA and fT4ED was 0.81, 0.88 and 0.79 at T0, T1 and T2, respectively. fT4CEIA was consistently lower than fT4ED, with a median difference of −5.4 pmol/l (P <0.001) and −4.9 pmol/l (P <0.0001) at T1 and T2, respectively. At T2, all cats were identified as euthyroid based on thyroid scintigraphy. None of the cats were identified as being hypothyroid, based on serum TT4 and TSH measurements. Nine of 22 (40.9%) cats had an fT4CEIA below the reference interval (RI) at T2, whereas only 2/22 (9.1%) cats had an fT4ED concentration below the RI at T2.
Conclusions and relevance
Good correlation exists between both assays at T1 and T2, but a significant systematic difference is noted at both time points. This could be an indication for reconsideration of the current RI, although further studies are warranted for assessing test accuracy (in otherwise healthy cats and cats with non-thyroidal illness). At this time, routine use of fT4CEIA after 131I therapy is not advised in feline patients.
Introduction
Equilibrium dialysis (ED) is considered to be the gold standard for determination of serum free thyroxine (fT4) concentration as interference from alterations in metabolism, hormone binding to plasma carrier proteins (eg, albumin), transport into cells, intracellular binding, effects of binding proteins inhibitors (certain drugs or present in non-thyroidal disease) and, less important in cats, circulating thyroid hormone antibodies are mostly eliminated.1 However, this method is expensive (at our institution the cost of fT4 after ED measurement compared with measurement by chemiluminescent enzyme immunoassay [CEIA] is 100% higher) and time consuming, and hence not widely available. Measurement of serum fT4 by a CEIA is a direct or single assay. Thus, a preincubation step or preliminary isolation of the free fraction (after dialysis or column chromatography) is not required, resulting in a more time-efficient and cost-effective technique for use in practice. Studies assessing the use of fT4CEIA measurement in cats are scarce. 1
Up to 10% of all hyperthyroid cats and 30% of cats with early or mild hyperthyroidism have total T4 (TT4) concentrations within the reference interval (RI). Serum fT4 after ED concentrations are more consistently (over 98% of cases) elevated in otherwise healthy hyperthyroid cats than serum TT4 concentrations.2–4 Furthermore, fT4 is influenced to a lesser extent by non-thyroidal illness than TT4. 5 When hyperthyroidism is suspected based on the cat’s history and physical examination but not supported by TT4 measurement the combination of measuring TT4 and free T4 by ED can be useful. 2 Moreover, measurement of fT4 can play an essential role in monitoring hyperthyroid cats after treatment. Determination of the true thyroid status after therapy with radioactive iodine (131I) for hyperthyroidism is essential, particularly in cats with concurrent chronic kidney disease. 6
The objective of this study was two-fold. The first objective was to correlate fT4 after ED and CEIA results before and after treatment with 131I. The second objective was to compare thyroid status based on fT4 (ED and CEAI) with the gold standard, quantitative thyroid scintigraphy 11–23 months post-131I treatment.
Materials and methods
Animals
Fifty client-owned, previously reported hyperthyroid cats presented for treatment with 131I at the Small Animal Department of Ghent University were prospectively enrolled in this study. 7 Local ethical committee approval (Ghent University EC 2017/72) and the signed informed consent of the owners was obtained. The cats were diagnosed with hyperthyroidism based on clinical signs, increased serum TT4 concentration (at the referring veterinarian) and increased technetium uptake in one or both thyroid glands on scintigraphic scan (thyroid:salivary ratio >1). Patients were excluded when any clinically relevant systemic disease other than hyperthyroidism was present or if the use of any medication within the 2 weeks prior to enrollment (except for antithyroid medication) was reported. Antithyroid medication was stopped at least 10 days prior to treatment with 131I. Cats were treated with an individually determined dose of 131I (mean dose 142.6 MBq [3.9 mCi]; range 67.7–455.1 MBq [1.8–12.3 mCi]) based on the severity of clinical signs, serum TT4 and thyroid:salivary gland ratio. 8
Sampling
The cats were evaluated before (T0), 1 month (T1) and 11–23 (T2) months after treatment with 131I. Full health screening was performed, including medical history, physical examination, systolic blood pressure, complete blood count (CBC), biochemistry profile, urinalysis, urine culture and echocardiography at all three time points. 9 At T0, abdominal ultrasonography and standard two-view thoracic radiographs (lateral and ventrodorsal projections) were performed. All blood samples were centrifuged within 30 mins of collection, and serum separated and stored at 4°C until sent to commercial laboratories within 24 h of collection. Samples for thyroid-stimulating hormone (TSH) were stored at −80°C and sent for analyses all together at the end of the study. Serum TT4 concentration was measured on an automated biochemistry analyser using a homogenous enzyme immunoassay (DRI T4 assay; Microgenics) with an RI of 10–45 nmol/l. Serum TSH was determined using a canine TSH CEIA (Immulite Canine TSH; Siemens Healthcare Diagnostics Products) with an RI of 0.02–0.3 ng/ml. fT4 was determined by a CEIA (fT4CEIA; IMMULITE 2000 Veterinary Free T4; Siemens Healthcare Diagnostics Products) and ED (fT4ED; Antech Diagnostics) with an RI of 9–33.5 pmol/l and 9–30 pmol/l, respectively. 1 All analyses were performed by IDEXX Laboratories.
Thyroid status
Thyroid status determined at T1 was based on the combination of TT4 and TSH measurements. Hypothyroidism was defined as a low TT4 value (<10 nmol/l) in combination with an elevated TSH value (>0.3 ng/ml). Euthyroidism was defined as a TT4 and TSH within the RI (10–45 nmol/l and 0.03–0.3 ng/ml, respectively). Subclinical hypothyroidism was defined as an elevated TSH value (>0.3 ng/ml) in combination with a TT4 within the RI (10–45 nmol/l). Hyperthyroidism was defined as an elevated TT4 value (>45 nmol/l).
Quantitative thyroid scintigraphy with sodium 99m-Tc-pertechnetate was used as gold standard to determine thyroid status at T2. Scintigraphic diagnosis of hypothyroidism was based on a lowered value of at least three of four of the following variables (see Table 1): thyroid:salivary gland ratio, percentage of thyroidal uptake of the administered 99m Tc-pertechnetate, thyroid:tracheal background ratio and calculated thyroid volume. 10
Results (mean value followed by range of results) of quantitative thyroid scintigraphy at T2 (11–23 months after radioactive iodine therapy)

Data are mean (range)
T:S ratio = thyroid: salivary gland ratio; T:TB ratio = thyroid:tracheal background ratio; TcTU = Tc-pertechnetate
Statistical analysis
The two techniques for the determination of fT4 were compared by the signed rank test with cat as a stratification factor. Spearman rank correlations coefficients were derived between the measurements of the two techniques. All analyses were performed at the three different time points separately.
Results
Animals
Fifty client-owned previously reported hyperthyroid cats presented for 131I treatment were prospectively enrolled in this study (Figure 1). 7 Five cats were excluded at T0 because of owner non-compliance (n = 1), congestive heart failure (n = 1), thyroid carcinoma with lung metastasis (n = 1) and insufficient sampling due to aggressive behaviour (n = 2). Of the 45 included cats at T0 and T1, 21 were castrated males, 23 were spayed females and one was an intact female. Represented breeds included 42 domestic short- or longhair cats, one British Shorthair, one Norwegian Forest Cat and one Chartreux. Median age was 13 years (range 7–16 years). Twenty-one of 45 (47%) patients were previously treated for hyperthyroidism with methimazole (Felimazole; Dechra) (n = 17), with carbimazole (n = 1), with a combination of unilateral thyroidectomy and methimazole (n = 1) or with Hill’s Y/D diet (n = 2). A total of 25 cats were presented for long-term follow-up (ie, T2, 11– 23 post-131I treatment). Of these 25 cats, three cats were excluded because of thyroid hormone supplementation (n = 3).

Study design.
Thyroid status
Serum TT4, fT4ED, fT4CEIA and TSH concentrations at T0, T1 and T2 are summarised in Figure 2.
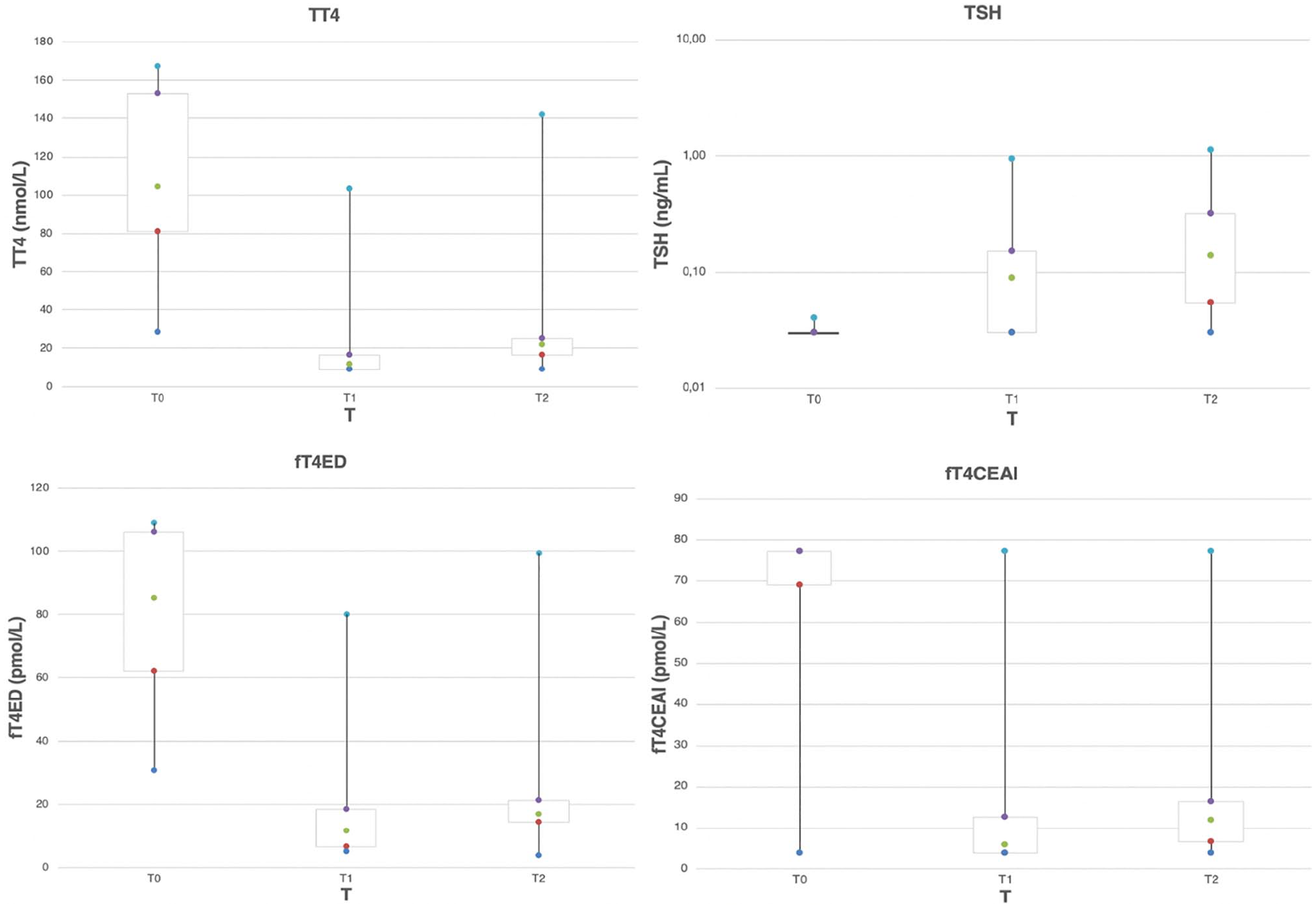
Box plot results of total thyroxine (TT4), thyroid-stimulating hormone (TSH), free thyroxine by equilibrium dialysis (fT4ED) and free thyroxine by a chemiluminescent assay (fT4CEAI) at the three study points (T0, T1 and T2 [before, 1 month after and 11–23 months after radioactive iodine therapy, respectively]). Boxes represent the interquartile range from the 25th (red dot) to the 75th percentile (purple dot). The green dot represents the median value in each box. The dark and light blue dots represent the main body of the data (highest and lowest value)
fT4CEIA was consistently lower than fT4ED at T0, T1 and T2. At T1, a median difference of −5.4 was noted (P <0.001). The median difference at T2 was −4.9 (P <0.0001). Spearman correlation between fT4CEIA and fT4ED was 0.81 (95% confidence interval [CI] 0.68–0.89) at T0; 0.88 (95% CI 0.78–0.93) at T1; and 0.79 (95% CI 0.55–0.91) at T2 (Figure 3). None of the cats with fT4CEIA within the RI had decreased fT4ED at T1 or T2. The correlations between TT4 and fT4CEIA were equal to 0.767 (95% CI 0.609–0.867), 0.692 (95% CI 0.5000–0.819) and 0.567 (95% CI 0.204–0.803) at T0, T1 and T2, respectively. Correlations between TT4 and fT4ED were equal to 0.702 (95% CI 0.512–0.827), 0.684 (95% CI 0.488–0.814) and 0.773 (95% CI 0.522–0.901) at T0, T1 and T2, respectively (Figure 4).

Correlation between free thyroxine (fT4) measured by equilibrium dialysis (fT4ED) and fT4 by chemiluminescent enzyme immunoassay (fT4CEIA) combined over the three time points

Correlation between free thyroxine (fT4) measured by equilibrium dialysis (fT4ED), fT4 by chemiluminescent enzyme immunoassay (fT4CEIA) and total thyroxine (TT4) combined over three time points
At the time of presentation (T0), TT4 and fT4CEIA were elevated in 43/45 (95.6%) cats. fT4ED was elevated in 44/45 (97.8%) cats. T4 was within the RI for three cats at T0, within the RI for two, and below the RI in one cat for fT4CEIA and below the RI in one cat for fT4ED.
One month after treatment (Table 2) with 131I, 1/45 cats (2.2%) was classified as hyperthyroid, 40/45 cats (88.9%) as euthyroid, 3/45 cats (6.7%) as subclinical hypothyroid and 1/45 cats (2.2%) as hypothyroid based on serum TT4 and TSH measurements. Persistently elevated values of fT4ED and fT4CEIA were noted in 2/45 (4.4%) cats; one of these cats had an elevated TT4 and was judged to be persistently hyperthyroid. Subclinical hypothyroidism was defined as a normal serum TT4 measurement in combination with elevated serum TSH (>0.3 ng/ml). The one hypothyroid patient based on serum TT4 (9 nmol/l) and TSH (0.95 ng/ml) concentrations had an fT4CEIA value below the RI (<3.9 pmol/l), but fT4ED in this patient was within the RI (10.7 pmol/l). Of the three cats with subclinical hypothyroidism, one cat was identified as being hypothyroid by fT4ED and fT4CEIA, one cat was identified as being euthyroid by fT4ED and fT4CEIA, and the last cat was identified as being euthyroid by fT4ED and hypothyroid by fT4CEIA. Based on fT4CEIA, 11/45 cats (24.4%) were classified as having suboptimal thyroid function while having fT4ED within the RI at T1. Of these cats 10/11 (90%) had normal thyroid status based on the combination of serum TT4 and TSH measurements. One of 11 cats was determined to be hypothyroid and one of 12 cats was determined to be subclinical hypothyroid based on serum TT4 and TSH measurements.
Thyroid status 1 month after radioactive iodine treatment (T1) based on the combination of serum total thyroxine (TT4) and thyroid-stimulating hormone (TSH), TT4, TSH, free thyroxine (fT4) measured by equilibrium dialysis (fT4ED) and fT4 by chemiluminescent enzyme immunoassay (fT4CEIA) (n = 45)

Data are n (%)
At T2, thyroid scintigraphy classified all 22 cats as being euthyroid after 131I treatment at T2 (Table 3). Thyroid:salivary gland ratio was normal in all cats (mean 0.94; range 0.49–1.51 [RI 0.48–1.9]). Mean thyroid:tracheal background ratio was 2.5 (range 1.47–3.40 [RI 1.6–6.4]) and abnormal in only 2/22 cats (9%). Mean calculated thyroid volume was 492.40 (range 159.14–2798.99 [RI 300–1000]). In contrast, percentage Tc-pertechnetate (mean 0.20%; range 0.02–0.40% [RI 0.21–3.9%]) was within the RI only in 6/22 (27%) cats (Table 1). Based on the combination of serum TT4 and TSH measurements, no cat was classified as hypothyroid (TT4 below the RI, TSH above the RI). Based on serum fT4CEIA, at T2, 9/22 cats (40.9%) were identified as having subnormal thyroid function. Only two of those 22 (9%) cats had an fT4ED below the RI (Table 3). No cat had elevated fT4ED or fT4CEIA values at T2.
Thyroid status 11–23 months after radioactive iodine treatment (T2) based on the combination of serum total thyroxine (TT4) and thyroid-stimulating hormone (TSH), free thyroxine (fT4) measured by equilibrium dialysis (fT4ED), fT4 by chemiluminescent enzyme immunoassay (fT4CEIA) and quantitative thyroid scintigraphy (n = 22)
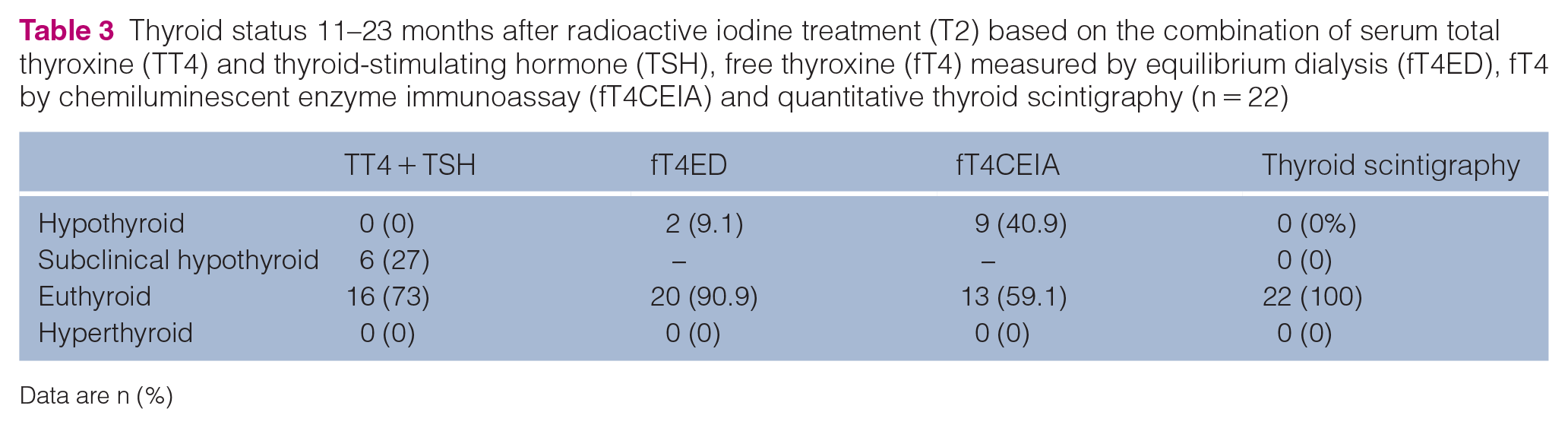
Data are n (%)
Discussion
Several methods are used for determination of serum fT4 concentrations (eg, ED, chemiluminescent assays, radioimmunoassays).4,11,12 The performance of these tests is variable, complicating the interpretation of fT4 measurements.3,4,13,14 fT4 measurement assays have to overcome large variations in the concentrations and affinities of serum T4-binding proteins in a population. No method for fT4 measurement directly measures the actual unbound hormone in the serum, making minimal displacement of serum protein-bound T4 into the free phase in the sample critical.15,16
fT4 concentrations are only truly determined after ED or ultrafiltration.11,12,17 Non-dialysis techniques for fT4 measurement are often less accurate, underestimate the fT4 concentration and offer little, if any, advantage over TT4 measurement.18,19 However, determination of fT4ED is complicated, expensive and time-consuming in comparison with measurement by CEIA. For this reason, measurement of fT4CEIA would offer an advantage for use in practice. As no dialysis step is required, measurement of fT4 by non-dialysis techniques possibly does not reflect the true fT4 fraction, but rather a proportion of the TT4 concentration. In the absence of binding kinetics alterations (non-thyroidal disease, use of certain drugs), fT4 measurement by dialysis and non-dialysis techniques most likely are correlated. 12 This correlation could be lost when T4 binding properties are altered.
In this study, no cats with non-thyroidal illness that required medications other than antithyroid medication were included. Thus, conclusions regarding correlation can only be made in otherwise healthy cats.
In this study, fT4CEIA correlated well to fT4ED (based on Spearman correlation). However, there was a consistent difference in fT4 values between both techniques at both timepoints. Given this difference, the current RI should be revisited. The current RI (9–33.5 pmol/l) used for fT4CEIA (Immulite 2000 Veterinary Free T4) was established by Peterson et al in 2011. 1 Fifty-three healthy cats were included. Euthyroidism was confirmed in these cats based upon history, physical examination, CBC, serum biochemistry and serum TT4 measurement. Technetium scan was performed in 10/53 of these cats, confirming euthyroidism. Establishment of this RI was performed in a different laboratory. So, a large number of cats (including healthy elderly cats, hyperthyroid cats and hyperthyroid cats after 131I treatment) would be necessary to establish a current RI for this assay, taking cats with various degrees of non-thyroidal illness into account. At this moment, fT4CEIA offered no advantage at both T1 and T2 over TT4 determination.
Measurement of fT4 can aid in the diagnosis of hyperthyroidism in cases where TT4 (of all hyperthyroid cats and 30% of cats with early or mild hyperthyroidism have TT4 concentrations within the RI) is normal but where a high clinical suspicion of hyperthyroidism exists.2,3,4 Furthermore, fT4ED is less influenced by non-thyroidal illness than TT4 (serum TT4 concentration can be suppressed into the RI in hyperthyroid cats with non-thyroidal illness). 5 In 95% of cats with occult hyperthyroidism serum fT4 concentration is high.3,4
In this study, fT4ED measurement identified 97.8% of cats with hyperthyroidism vs 93.3% of cats for both fT4CEIA and TT4 measurement at T0. However, it should be noted that despite a higher sensitivity of fT4 (98%) measurement in comparison with TT4 (91%) measurement for the diagnosis of hyperthyroidism, specificity was lower for fT4 (93%) than for TT4 (100%) in a previous study.3,13,20 In this previous study, 14/221 cats with non-thyroidal illness had a fT4 value above the RI. 3 A more recent study reported a false-positive rate of 33% when fT4ED is used for the diagnosis of hyperthyroidism. 1 Thus, in most cases measurement of TT4 will be sufficient for diagnosis of hyperthyroidism. In cats highly suspected of hyperthyroidism but with serum TT4 concentrations within the RI fT4ED can be of added value. Measurement of fT4ED should always be interpreted in combination with TT4 measurement. This is especially important in cats with concurrent mild non-thyroidal illness. 3
Naturally occurring hypothyroidism in cats is rare. 21 In contrast, iatrogenic hypothyroidism is a known complication of treatment with 131I in hyperthyroid cats. This complication is reported in 3–79% of cats treated with 131I. However, most institutions report a prevalence of <9%.4,6 Development of iatrogenic hypothyroidism in azotaemic cats after 131I treatment is associated with reduced survival times, hence the importance of correctly identifying these patients. 6 A recent article indicates that TSH measurement best identifies feline iatrogenic hypothyroidism and is able to differentiate iatrogenic hypothyroidism from non-thyroidal illness in azotaemic cats after 131I treatment. 9 The value of fT4 by ED or by CEIA measurement for the determination of thyroid status after treatment with radioactive iodine remains unclear. Measuring serum fT4 in combination with serum TT4 is recommended in cats with non-thyroidal illness for the diagnosis of hypothyroidism.10,22 However, fT4 can be lowered, as well as elevated, in cats with non-thyroidal illness,3,19 making TSH measurement possibly more reliable. 10
In this study, thyroid status at T1 was determined based on TT4 and TSH serum concentrations. However, for true determination of thyroid status and consequent comparison of thyroid status based upon fT4ED or fT4CEIA, TSH stimulation testing or thyroid scintigraphy should be considered. At T2, all cats in this study were found to be euthyroid, based on thyroid scintigraphy. When comparing determination of thyroid status by fT4 measurement with scintigraphy, sole measurement of fT4CEIA does not seem to be reliable for establishing the diagnosis of iatrogenic hypothyroidism in these patients (fT4CEIA correctly identified thyroid status in only 59.1% of cats). In contrast, fT4ED measurement correctly identified the thyroid status in 90% of cats.
The significance of the systematic difference between both techniques can be interpreted two-fold. Firstly, a significant part of this cat population after 131I therapy was misclassified as being hypothyroid, based on fT4CEIA measurement. This could possibly be an indication for revision of the current reference interval. Secondly, the misclassification of patients based on fT4CEIA measurement could also indicate inherent limitations in the testing methodology. Further studies are warranted to evaluate the routine use of the CEIA for determination of fT4. At this moment, fT4CEIA offers no advantage over TT4 determination post-131I therapy.
This study has several limitations. Only a small number of cats was included. Determination of thyroid status by fT4ED and fT4CEIA could not be compared with the gold standard, scintigraphy, at T1. Nevertheless, scintigraphy was available for long-term follow-up in these cats, the most important time point for the determination of thyroid status in these cats after 131I treatment.
Conclusions
Measurement of serum fT4ED and CEIA correlated well at the three time points in this study. At both T1 and T2 a systematic difference between fT4ED and ft4CEIA measurement was noted. Revision of the current RI could avoid misclassification of cats after 131I therapy. Further studies are warranted to determine if fT4CEIA measurement is an accurate test for iatrogenic hypothyroidism in our feline patients after 131I therapy.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by IDEXX Laboratories.
Ethical approval
This work involved the use of non-experimental animals (owned or unowned) and procedures that differed from established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient. The study therefore had ethical approval from an established committee as stated in the manuscript.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedure(s) undertaken (either prospective or retrospective studies). No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
