Abstract
Objectives
Thoracic CT is commonly performed in cats and has been shown to be more sensitive than radiography for the detection of lymphadenopathy. However, no studies to date have reported the normal CT appearance of feline intrathoracic lymph nodes. The aim of this study was to describe the distribution, number, size and attenuation of presumed normal intrathoracic lymph nodes in cats.
Methods
Retrospective analysis of the imaging archive in a teaching hospital identified cats that had undergone thoracic CT and were deemed to have no evidence of either intra- or extrathoracic disease that would influence the size of the intrathoracic lymph nodes. Lymph nodes were classified into lymph centres based on anatomical references. Descriptions of the position of each node, number of nodes in each centre, size of each node and attenuation pre- and post-contrast were recorded. Statistical analysis aimed to assess for correlations between these factors and the age, weight and sex of the subjects.
Results
Twenty cats were included in the study. One or more lymph nodes were identified in the sternal (n = 19/20), cranial mediastinal (n = 14/20) and tracheobronchial (n = 15/20) centres, while none were identified in other locations. Size varied between lymph centres and within lymph centres where more than one lymph node was identified. Tracheobronchial and sternal nodes were consistent in location, while mediastinal nodes were variable. All nodes were round to ellipsoid in shape and showed moderate enhancement post-contrast. No significant correlations were found with age, weight or sex.
Conclusions and relevance
This study describes the CT characteristics of presumed normal intrathoracic lymph nodes in cats, and only identified nodes in the sternal, cranial mediastinal and tracheobronchial locations. This study serves as a reference point for CT analysis of feline thoracic lymph nodes.
Introduction
Thoracic CT is known to provide more comprehensive clinical information than radiographic studies of the thorax,1,2 and is increasingly used in the investigation of thoracic disease in companion animals. Radiography is known to be insensitive for the assessment of tracheobronchial and cranial mediastinal lymph nodes, 3 whereas the greater contrast resolution of CT and the removal of superimposition allow for individual assessment of these structures.2–6
Four intrathoracic lymph centres are described in cats: the dorsal thoracic (comprising intercostal and aortic nodes), ventral thoracic (comprising sternal, xiphoid and phrenic nodes), cranial mediastinal and bronchial (comprising pulmonary and left, middle and right tracheobronchial nodes) 7 centres. The presence and number of lymph nodes within these lymph centres is known to be variable. 7 The CT appearance of presumptively normal intrathoracic lymph nodes has been described in dogs;4,8 however, despite there being several studies describing varying degrees of intrathoracic lymphadenopathy in cats,2,5,6 no studies to date describe the normal CT characteristics of these lymph nodes.
The aim of this study, therefore, was to describe the CT characteristics of intrathoracic lymph nodes in cats that were deemed to have no evidence of disease likely to affect their appearance.
Materials and methods
Study design
This was a retrospective descriptive study, for which ethical approval was granted by the University of Bristol Animal Welfare and Ethical Review Body.
Subject selection
The medical database of the Langford Vets Small Animal Referral Hospital was searched for cats that underwent thoracic CT and bronchoscopy between November 2011 and June 2018. For cats to be included in the current study, they were required to have a normal thoracic CT, an unremarkable bronchoscopy, a negative bronchoalveolar lavage and a final clinical diagnosis consistent with no thoracic disease. Cats were excluded if abnormalities of the cervical region, body wall or cranial abdomen were identified, or if systemic inflammatory, infectious or immune-mediated disease was detected owing to the potential for these abnormalities to influence the morphology of the thoracic lymph centres.
Image acquisition
All studies were acquired using a Siemens Somatom Emotion 16 (Siemens) with the following helical acquisition parameters: kV 130, reference mAs 50 (CARE Dose4D), slice thickness 1.5 mm, pitch 0.8 and rotation time 0.6 s. Intravenous contrast medium (iopamidol) was used if it was clinically indicated, and this was administered at a standard dose of 600 mg I/kg at a rate of 2 ml/s. Pre- and post-contrast studies were reconstructed in soft-tissue algorithms with the following parameters: window level 50, window width 400 and slice thickness 1.5 mm.
Image evaluation
Images were evaluated using dedicated DICOM viewing software (Horos v2.0.1, The Horos Project, www.horosproject.org). Lymph nodes were categorised into ventral thoracic, dorsal thoracic, cranial mediastinal and bronchial lymph centres, as well as subgroups (eg, tracheobronchial vs pulmonary), based on anatomic classifications, 7 and for each group the number of visible nodes was recorded. For each individual node, the following information was recorded by a single board-certified radiologist: description of location, height, width, length and attenuation pre- and post-contrast. Multiplanar reconstruction was used for size measurement, with individual height, width and length measurements made on whichever plane image allowed for most confident identification of the node margins. Attenuation values were obtained by positioning an ellipsoid region of interest (ROI) over each lymph node and the mean, minimum and maximum Hounsfield units (HU) were recorded.
Statistical analysis
Statistics were performed in Microsoft Excel. The Shapiro–Wilk’s test (α = 0.05) was used to assess for normal distribution of continuous data. Age and weight were compared against nodal size, number at each location and attenuation pre- and post-contrast using Pearson’s correlation. A two-tailed Student’s t-test was used to look for significance of these correlations and the P value was set to 0.05. Categorical data (sex) were compared against nodal size, number at each location and attenuation pre- and post-contrast using an independent Student’s t-test, with the P value set to 0.05. Body weight was also compared between male and female cats using an independent Student’s t-test with a P value of 0.05.
Results
Seventy-two cats were identified to have undergone both thoracic CT and bronchoscopy in the selected time period, 24 of which were reported to have no thoracic disease. Four cats identified to have normal thoracic CT scans on the basis of their clinical records were excluded following initial image review; two were noted to have mass lesions in the caudal cervical region, one had a mass at the thoracic inlet and one had diffuse lesions consistent with inflammatory nodules affecting the cutaneous and subcutaneous tissues. Therefore, 20 cats met the study criteria, including 15 domestic shorthairs, two domestic longhairs, and one each of Persian, Russian Blue and Egyptian Mau breeds. Age and weight values were normally distributed. Ages ranged from 1.75 to 21 years (mean age 7.5 years). Weights ranged from 2.81 to 5.70 kg (mean 4.31 kg). The sex distribution was as follows: 12 female neutered, seven male neutered and one male entire. Pre- and post-contrast studies of the thorax were available in 15/20 cats; three cats only had pre-contrast studies performed, with the remaining two only having post-contrast studies. The final clinical diagnoses were as follows (in descending order of frequency): inflammatory rhinitis (n = 5), skeletal injuries secondary to trauma (n= 3), endocrine disease (hyperaldosteronism, hyperthyroidism) (n = 2), nasal polyp (n = 1), maxillary neoplasia (n = 1), chronic ureteral obstruction (n = 1), chronic kidney disease (n = 1), inflammatory enteropathy (n = 1), intracranial meningioma (n = 1), lack of recurrence/metastasis from previous inguinal haemangiosarcoma (n = 1), idiopathic hypercalcaemia (n = 1), pure red cell aplasia (n = 1) and Heinz body anaemia of unknown origin (n = 1). No patients were identified to have significant abdominal pathology that would be expected to affect the sternal and cranial mediastinal lymph nodes, and patients with pathology in the head were not identified to have significant cervical lymphadenopathy.
A total of 78 intrathoracic lymph nodes were identified, consisting of 32 sternal, 19 cranial mediastinal and 27 tracheobronchial nodes, with one or more nodes identified at each of these locations in 19/20, 14/20 and 15/20 cats, respectively. No lymph nodes were identified in the dorsal thoracic (aortic and intercostal) lymph centre, and no xiphoid, phrenic or pulmonary nodes were visualised. In the three cases where no post-contrast images were available, two cats had no visible cranial mediastinal lymph nodes and two had no visible tracheobronchial lymph nodes. Information regarding the nodal number and size at each location are displayed in Tables 1 and 2.
Number of cats categorised by the number of identified lymph nodes within each lymph centre

Percentages represent the number of cats in that category within that lymph centre (ie, left to right totals 100%)
LN = lymph node
Mean lymph node dimensions (mm) at each lymph centre

The sternal lymph nodes were relatively consistent in location, ranging from immediately dorsal to the mid-second sternebra to the mid-fourth sternebra, with the majority (n = 22/32) being located dorsal to the third sternebra. The most common position of the nodes was slightly to the left of midline (n = 14/32) even if there was only one node present. Midline (n = 11/32) and slightly right-sided positions (n = 7/32) were also identified. All nodes were closely associated with the internal thoracic blood vessels (Figure 1). All nodes were ellipsoid with the greatest length being in the craniocaudal orientation. Where more than one sternal node was visualised, they were mildly to moderately variable in size.

Post-contrast axial soft tissue reconstruction at the level of the sternal lymph nodes. Right is to the left of the image. The arrows indicate two sternal nodes to the left and right of the midline, situated immediately ventral to the internal thoracic vessels
The cranial mediastinal lymph nodes were more variable in location but were largely found in close association with either the venous structures (brachiocephalic, jugular and cranial vena cava) or the trachea (Figure 2). Three were located more dorsally and laterally (Figure 3) but were still contained within the cranial mediastinum and were therefore not considered to be consistent in location with the dorsal thoracic lymph centre (aortic or intercostal nodes). Compared with the sagittal midline, more nodes were located to the left (n = 8/19) than the right (n = 4/19) with the remainder being located centrally enough that they were considered to be midline (n = 7/19). Overall, the shape was more variable, showing a mixture of rounded/spherical and ellipsoid nodes. Where more than one cranial mediastinal node was identified the sizes were moderately variable. Several small, ill-defined, soft-tissue-attenuating foci were identified within the cranial mediastinum in some patients, but these were hard to define and measure accurately, and were therefore not included.

Post-contrast axial soft tissue reconstruction at the level of the cranial mediastinum. Right is to the left of the image. The arrow indicates a single lymph node that is situated dorsolateral to the cranial vena cava, and immediately ventral to the common carotid arteries
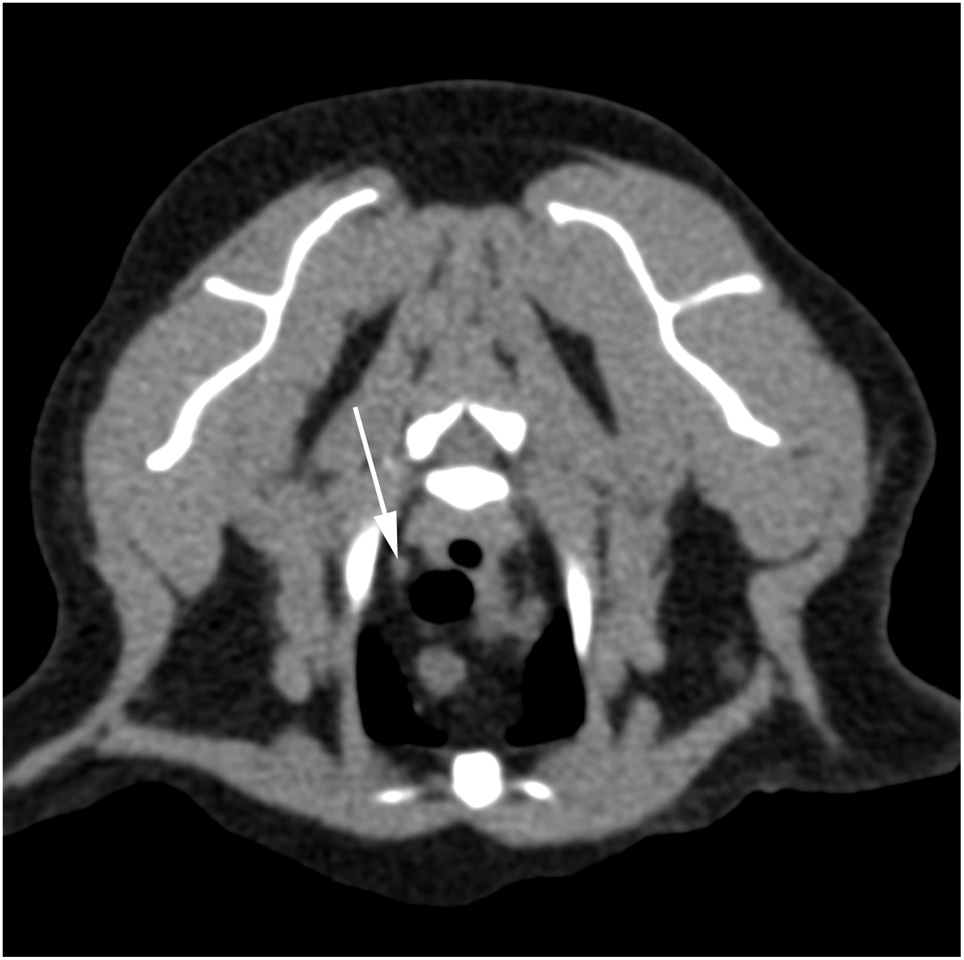
Pre-contrast axial soft tissue reconstruction at the level of the cranial mediastinum. Right is to the left of the image. The arrow indicates a single lymph node within the mediastinum that is more dorsally placed than most, situated immediately dorsolateral to the trachea
The tracheobronchial lymph nodes showed the most consistency in location when identified (Figure 4). The left node was located lateral to ventrolateral of the angle between the trachea and the left principal bronchus and ventromedial to the aortic arch, with the right similarly located on the contralateral side typically caudal to the termination of the azygos vein. The middle node was positioned immediately caudal to the carina between the principal bronchi. This was dorsoventrally flattened but never clearly V-shaped. The left node was most commonly visualised (n = 12/27), followed by the middle (n = 9/27) and right nodes (n = 6/27). Again, both rounded and ellipsoid nodes were identified. Right and left nodes were typically very similar in size, with the middle nodes being slightly larger, in cases where more than one of the nodes was visible.

Post-contrast axial soft tissue reconstructions immediately (a) cranial and (b) caudal to the carina. Right is to the left of the image. In (a), the right tracheobronchial node (white arrowhead) is situated between the right cranial lobar artery laterally and the trachea medially. The left node (white arrow) is situated ventrolateral to the trachea and is bound by the main pulmonary trunk ventrally and the aorta dorsally. In (b), the middle tracheobronchial node (black arrow) can be seen ventral to the oesophagus and is bound laterally by the two principal bronchi
All nodes were subjectively assessed to be largely of homogeneous soft tissue attenuation, with a few nodes showing mild heterogeneity owing to the inclusion of fat, and objective attenuation values pre- and post-contrast are displayed in Table 3. A large degree of overlap was seen between the ranges of the pre- and post-contrast values, but all mean values showed a moderate to marked increase post-contrast. The highest pre-contrast attenuation (76 HU) was identified as an outlier, with the remaining nodes all having an attenuation of less than 55 HU. In cases where both pre- and post-contrast sequences were acquired, some nodes were identified only in the post-contrast sequence. In these cases, the numbers of nodes identified only in the post-contrast sequence for each lymph centre were as follows: sternal (n = 1/23), cranial mediastinal (n = 6/16) and tracheobronchial (n = 13/23). All nodes that were identified pre-contrast were also identified post-contrast.
Mean lymph node attenuation values (Hounsfield units) at each lymph centre

n = number of measured lymph nodes
No statistically significant correlations were found when comparing age or weight with number, size or attenuation. Male neutered cats were found to have greater sternal nodal length than female neutered cats, as well as tracheobronchial nodal width, length and pre-contrast attenuation, as displayed in Table 4. No significant difference was found between male and female weights (P = 0.26). Only one male entire cat was included in the population and was therefore not included in these comparisons.
Statistically significant differences between male and female neutered cats

Data are mean ± SD
HU = Hounsfield units
Discussion
Despite the widespread use of CT for imaging feline thoracic disease, this is the first study that aimed to define the appearance and distribution of intrathoracic lymph nodes in cats with no evidence of thoracic or other contributing pathology. Although the patients included in this study were undergoing CT as part of a clinical investigation, the final diagnoses were not considered to be conditions that would adversely affect the thoracic lymph nodes, and these cases were therefore considered to be acceptable for the purposes of the study.
It is well known that there is individual variation in the presence and specific location of certain lymph nodes and the frequency of cats in this study without visible cranial mediastinal (30%) or tracheobronchial (25%) lymph nodes is consistent with this. Nonetheless, it is also possible that greater numbers of lymph nodes were present but could not be clearly defined on the CT images owing to limitations of spatial resolution such as partial volume effect and/or effacement by adjacent anatomy. Varying factors influenced the degree to which small lymph nodes were visible, including the presence and extent of mediastinal fat, which served to separate and highlight soft tissue structures. It was also noted by the authors that identification of lymph nodes, particularly those of mediastinal and bronchial centres, was easier in post-contrast compared with pre-contrast images. Pre-contrast, identifying the margins of nodes that lay adjacent to vascular structures was difficult due to effacement. In the cases where both pre- and post-contrast images were available, measurements were performed on the post-contrast series due to greater observer confidence in nodal margination. Some measurements were able to be performed in the three cats where post-contrast images were not available; however, both cranial mediastinal and tracheobronchial nodes were only recorded in one case each (two separate cats). While these cases may not have possessed CT-detectable lymph nodes in these centres it is possible that contrast administration would have allowed the identification of nodal structures, and this should be taken into consideration when planning CT studies in clinical cases. Despite the difficulties encountered, we feel that inclusion of the three cats without post-contrast series is warranted, as cases will arise in clinical practice where contrast may not be administered, either because of clinical contraindication or clinician choice.
The sternal nodes were most commonly identified with 95% of the study population having at least one visible. They were also relatively consistently located, with only mild variation in their craniocaudal distribution along the dorsal surface of the sternum. The sternal lymph centre has previously been reported to consist of only one node in cats,6,9 but our population had a similar number of individuals with one (40%) and two (45%) distinct nodes, and 10% were identified as having three separate nodal structures. The tracheobronchial nodes were highly consistent in location when visualised, whereas the cranial mediastinal nodes showed the most variation in location.
No lymph nodes meeting the anatomical criteria for the dorsal thoracic lymph centre were identified in our study. The intercostal lymph nodes are described as being inconstant and are not identified in a large proportion of cats, 7 and the thoracic aortic lymph nodes are anatomically very small, and therefore may be missed on CT studies owing to the limitations of spatial and contrast resolution. We therefore propose that if nodes are seen in these described locations they are likely to represent lymphadenopathy.
There was not found to be a large variation in node shape, with most being slightly ellipsoid, as seen in many other locations. 10 The previously described characteristic V-shape of the middle tracheobronchial lymph node was not appreciated in any of the cats included, again likely owing to limitations in spatial resolution.
Interestingly, while nodes were subjectively assessed to be of soft tissue attenuation objective measurements showed marked variation, with pinpoint values as low as –24 HU and up to 76 HU in pre-contrast images. The lower end of this scale is likely due to the inclusion of perinodal fat within the ROI when performing objective measurements, or fat deposits within the nodes themselves. The value of 76 HU was a statistical outlier that was present in a sternal lymph node, a possible explanation for which is the inclusion of blooming artefact from the adjacent sternebra. The increased mean attenuation post-contrast was an expected finding and the lower values within the post-contrast ranges again likely represent the inclusion of fat within the ROI.
Although a previous study investigating the appearance of feline abdominal lymph nodes showed a correlation between age and both size and number of certain lymph nodes, 10 no such correlations were found in the current study. This may be explained by the age range, with the youngest subject being 1.75 years, while the youngest in the previous study was 1 year old. Abdominal lymph nodes in puppies have been shown to be larger than in adult dogs,11,12 but to our knowledge this has not been described for thoracic lymph nodes in cats.
Male neutered cats were found to have greater sternal nodal length measurements and greater tracheobronchial width, length and pre-contrast attenuation measurements than female neutered cats, despite there being no difference in body weight between these two groups. It is interesting to note that differences in size were only seen in certain dimensions, rather than in all nodes and all dimensions, and therefore the clinical significance of these findings is questioned. The increased pre-contrast attenuation of the tracheobronchial nodes in males vs females is considered to be due to the larger size, and therefore greater accuracy of placement of the ROI over these nodes.
There are several limitations to this study. Only a small number of cases were available for inclusion and the population was taken from a single centre. Although a large number of feline thoracic CT scans were performed in the chosen time period, the number of included cases was largely restricted by the requirement to have an unremarkable bronchoscopy and a negative bronchoalveolar lavage; however, this was considered to be a necessary restriction to prevent inadvertent inclusion of cats with pathologically enlarged lymph nodes. A previous publication aiming to establish normal reference intervals for feline abdominal lymph nodes included similarly small case numbers, 10 which likely represents the low number of clinically driven CT studies that are performed and considered to be within normal limits.To our knowledge, the only previous study that reports CT-measured intrathoracic nodes was an experimental study that assessed sternal and tracheobronchial lymph node sizes in a population of six cats prior to and after infection with Aelurostrongylus abstrusus. 6 Interestingly, the size measurements recorded in the previous study are slightly larger than those reported here. Also, the attenuation values were higher in the previous study both pre- and post-contrast, which may well reflect the larger size of the lymph nodes and a subsequent ability to use a larger region of interest. The reason for these discrepancies is unknown; however, the population included in the current study is considered to be a fair representation of the breeds presented clinically to our institution, and this may indicate potential variation between breeds that would need larger case numbers for further investigation.
Owing to the retrospective nature of the study, raw CT data were not available for alternative reconstructions using different slice thicknesses or sharpness settings. The slice thickness of the images available for review was set at 1.5 mm. This is the standard reconstruction for clinical assessment of feline thoracic studies at our institution and was therefore considered to be most clinically applicable. It is possible that thinner slice reconstructions may give better accuracy for lymph node measurement, but this would need to be assessed in future studies.
Finally, cytology and/or histopathology was not performed on any of the measured lymph nodes, and therefore it cannot be definitively concluded that these were all lymphoid tissue, either normal or abnormal. At the time of investigation these structures were subjectively deemed to be within normal limits and therefore there was no clinical indication for sampling to be performed, particularly given the technical challenges and risks associated with sampling of such small structures. However, we feel that all reasonable efforts were made to distinguish the structures as lymph nodes based on their anatomical location and the clinical experience of the observer.
Conclusions
This study provides data regarding the CT appearance of presumed normal intrathoracic lymph nodes in cats that have not been previously described. The variability seen between the numbers and location of these lymph nodes is not only consistent with previous anatomical reports, but also reflective of the difficulties in accurately assessing such small anatomical structures. Nonetheless, the data presented in this study should be considered when assessing clinical cases for intrathoracic lymphadenopathy and may help guide the decision as to when sampling for cytology or histopathology is considered. We recommend that 1.5 mm slice reconstruction is likely to be satisfactory for thoracic lymph node assessment, and that imaging should include both pre- and post-contrast images unless significant contraindications exist. Future research is proposed to assess changes in diseased populations, with the hope of establishing optimal cut-off values for normal lymph node characteristics.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work involved the use of non-experimental animals only (owned or unowned) and followed established internationally recognised high standards (‘best practice’) of individual veterinary clinical patient care. Ethical approval from a committee was therefore not necessarily required.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work for the procedure(s) undertaken. No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
