Abstract
Objectives
The objectives for this study were to compare the body composition of adult indoor neutered domestic cats with outdoor intact cats with an ideal body condition score using dual-energy x-ray absorptiometry, and to report the body composition findings of free-roaming cats, as this has not been previously reported. Most domestic house cats differ from free-roaming cats as they are confined indoors and neutered. Indoor neutered cats have reduced activity and hormonal alterations that may result in lower muscle mass and higher body fat percentages vs outdoor intact cats, despite similar body condition scores.
Methods
Twenty-one outdoor intact cats (10 male, 11 female) were selected from a trap–neuter–return program and 16 indoor neutered domestic cats (10 male, six female) were client-owned. Inclusion criteria included an estimated age between 1 and 6 years, complete blood count, biochemistry panel, urinalysis, total thyroxine, feline leukemia virus/feline immunodeficiency virus screening and a body condition score of 4–5/9.
Results
Indoor neutered cats had a higher body fat percentage (22.1% [range 17.3–28.2%]) than outdoor intact cats (17.3% [range 10.0–33.6%]; P = 0.002). Indoor neutered male cats had a higher body fat percentage (P <0.001) than outdoor intact cats. No difference in body fat percentage was observed in female cats (P = 0.159). Indoor neutered domestic cats had a higher bone mineral density than outdoor intact cats (P = 0.023).
Conclusions and relevance
The results of this study suggest indoor confinement and neutering increase body fat percentage and bone mineral density in cats with an ideal body condition score.
Introduction
Normal weight obesity is a condition where people maintain a normal weight, having a low body mass index (18.5–24.9 kg/m2), but have a high percentage of body fat, usually >30%. 1 These people are at an increased risk for developing cardiovascular disease, metabolic syndrome, type 2 diabetes mellitus, hypertension and dyslipidemia.2–5 One study evaluating the body composition of lean indoor cats with a body condition score (BCS) of 5 on a 9-point scale found a mean body fat percentage (%BF) of 31.8%, which is higher than the accepted ideal (15–24%).6–8 Indoor spayed or castrated cats with an ideal BCS and higher body fat percentage may share similar characteristics to people with normal weight obesity.
As of 2009, approximately 80% of household cats in the USA were spayed or castrated. 9 Owners in the USA and Australia also reported that approximately 32% of household pets were either overweight or obese. 10 The prevalence of overweight or obese cats has been reported in other nations, ranging from 11.5–63%.11,12 Neutering is a risk factor for obesity, 13 and research reveals that cats will have increased calorie intake, BCS and body fat percentages, as well as various metabolic changes, such as increased blood leptin and blood glucose, following an ovariohysterectomy.14–17 Furthermore, maintenance energy requirements are also reported to decrease by as much as 25% from the current National Research Council recommendations after an ovariohysterectomy. 18
The combined effect of neutering, a sedentary life indoors and elevated body fat percentages may place domestic cats at a higher risk for obesity-related disease, including insulin resistance and diabetes mellitus, even at an ideal body condition.
In contrast, free-roaming or feral cats may represent a population of cats at low risk for the development of obesity-related disease. In 2000, it was estimated that there were approximately 73 million feral or abandoned free-roaming cats in the USA. 19 Access to food sources from both humans and the natural environment offer a wide variety of situations where cats may forage, hunt or travel various distances for food. This increased activity level and exposure to a variety of food sources likely affects their body composition. We hypothesize that adult outdoor intact (OI) cats will have a lower body fat percentage than indoor neutered (IN) cats with similar BCSs (4/9 or 5/9) using dual-energy x-ray absorptiometry (DEXA). DEXA is widely used in veterinary research to evaluate body composition in cats. This methodology has been validated for use in cats when compared with chemical analysis of the carcass. 20 The objectives of this study were to compare the results of body composition using DEXA in adult IN domestic cats with OI free-roaming cats with an ideal BCS, and to report the body composition findings of free-roaming cats as this has not been previously reported.
Materials and methods
Animals
OI cats were selected from a trap–neuter–return (TNR) program at the University of Tennessee College of Veterinary Medicine. Collection of animals took place at events held on 21 July and 8 September 2012. Cats were humanely trapped over a 24 h period from the surrounding urban, suburban and rural areas and transported to a University of Tennessee veterinary surgical facility. Investigators and volunteers visually inspected cats prior to anesthesia while still in their traps to avoid injury. If a cat visually appeared to be an adult (>1 year of age based on size and the presence of mature features), in ideal body condition (BCS 4–5/9), and free of injury or physical illness, it was tagged for further examination following sedation.
BCS was determined in anesthetized cats by one or two investigators (MC and/or AW) and cats were selected for inclusion following confirmation that they: had a BCS of 4–5/9; were aged between 1 and 6 years of age (based on physical examination findings of mature features including dentition); and were intact and free of injury or physical illness by physical examination. Female cats were excluded if they were lactating or found to be pregnant before or during ovariohysterectomy. A laboratory evaluation, including a complete blood count (CBC), serum biochemistry, total thyroxine (TT4), urinalysis and testing for feline immunodeficiency virus (FIV) antibodies and feline leukemia virus (FeLV) antigens, was performed. Following castration or ovariohysterectomy, cats selected for study inclusion were transported by motor vehicle approximately 1 mile under veterinary or veterinary technician supervision to the University of Tennessee Veterinary Medical Center, where a DEXA scan was performed. Animals were included for final analysis if they were negative for FeLV antigens, negative for FIV antibodies, their TT4 was within the reference interval (RI), and they had unremarkable CBC, serum biochemistry and urinalysis results.
IN cats were recruited from employees and students associated with the University of Tennessee College of Veterinary Medicine. BCS was determined by one or two investigators (MC and/or AW). Cats were selected for inclusion if they were aged 1–6 years, spayed or castrated, had an unremarkable physical exam, TT4 within the RI, negative FeLV and FIV status, unremarkable CBC, serum biochemistry and urinalysis, a BCS of 4–5/9 and were housed exclusively indoors.
Protocols for both IN and OI cats, including protocols for the TNR program, were approved by the University of Tennessee Knoxville Institutional Animal Care and Use Committee.
Analytical procedures
Blood was collected by jugular venipuncuture and urine by gentle bladder expression in OI cats under anesthesia. Jugular venipuncture and cystocentesis was used to collect blood and urine in awake IN cats. Serum, whole blood and urine collected from OI cats was stored for approximately 48 h at 1.6°C prior to analysis. Analyses of laboratory samples from IN cats were performed the same day as collection. CBC was analyzed by Siemens ADVIA 120 Hematology System, serum biochemistry by Roche Cobas 6000 and urinalysis by Bayer Clinitek 500 at the Clinical Pathology Laboratory, College of Veterinary Medicine, University of Tennessee. TT4 analysis was analyzed by CobraII Auto-Gamma Counter at the Endocrinology Laboratory, College of Veterinary Medicine, University of Tennessee. FeLV and FIV status was determined by SNAP FIV/FeLV Combo Test (IDEXX Laboratories).
A DEXA scan was performed on a Hologic Discovery A Dual Energy Bone Densitometer X-Ray Unit (Model QDR4500) with software package (Apex, version 2.3; Hologic). OI cats were anesthetized with a combination of tiletamine HCl (250 mg free base) and zolazepam HCl (250 mg free base) reconstituted with 4 ml ketamine HCl (100 mg/ml), 1 ml xylazine HCl (100 mg/ml) and 4.25 ml buprenorphine (0.3 mg/ml), given intramuscularly at doses of 0.2 ml for cats weighing <2.3 kg, 0.35 ml for cats 2.3–4.5 kg and 0.5 ml for cats 4.5–6.8 kg. Yohimbine HCl 0.5 mg was given to OI cats following procedures for reversal. IN cats were anesthetized with an intramuscular injection of dexmedetomidine 5–8 μg/kg, butorphanol tartarate 0.4 mg/kg and ketamine 6 mg/kg. Atipamezole 0.05–0.08 mg/kg was given intramuscularly for reversal of IN cats following procedures. Cats were placed in ventral recumbency under anesthesia with forelimbs extended cranially along with hindlimbs and tail extended caudally. Data recorded included bone mineral content (g), bone mineral density (BMD) (g/cm2), fat mass (FM) (g), lean soft tissue mass (LSTM) (g) and total mass (TM) (kg). Percent body fat (%BF) was calculated by
Statistical analysis
Measures of body composition were compared using SigmaStat and SPSS (IBM SPSS Statistics Version 25) statistical software. All data were tested for normality using the Kolmogorov–Smirnoff goodness-of-fit test. Student’s t-test was used to compare normally distributed data and reported as mean ± SD. Mann–Whitney rank sum test was used to compare non-normally distributed data and reported as median and range. Multiple group comparisons were also performed for all dependent measures of interest. If the dependent measure was found to be normally distributed, a one-way ANOVA was performed with Fisher’s least significant difference post-hoc tests. If the dependent measures were found to be non-normal, a Kruskal–Wallis test was performed with Mann–Whitney rank sum test post-hoc paired comparisons.
Results
Thirty OI cats were selected for the study. Nine OI cats were excluded owing to positive FIV status (n = 5), positive FeLV status (n = 1), positive FIV and FeLV status (n = 1), hyperglycemia with glucosuria and ketonuria (n = 1) and lack of laboratory data, including CBC, serum biochemistry, TT4 and FIV/FeLV status (n = 1). Of the 21 remaining OI cats, 11 were female and 10 were male. Two OI cats had a BCS of 4/9, and the remaining 19 cats had a BCS of 5/9. TT4 was missing from one OI cat included for analysis.
Sixteen IN cats met the inclusion criteria for analysis: six spayed females and 10 castrated males. Median age for all IN cats was 4 years (range 1–6 years). All IN cats had an average BCS of 5/9. TT4 was within the RI for all cats, and none of the cats tested positive for FIV or FeLV.
The two groups did not differ by BCS (P = 0.631). DEXA data are provided in Tables 1–4. IN cats had a greater total mass (TM; kg) than OI (median IN 4.26 [range 3.17–5.51]; OI 3.32 [range 2.50–6.86]; P = 0.004). IN cats had a higher %BF than OI (median IN 22.1% [range 17.3–28.2]; OI 17.3% [range 10.0–33.6]; P = 0.002). OI cats had a lower BMD (g/cm2) than IN cats (median OI 0.365 [range 0.32–0.47]; IN 0.381 [range 0.36–0.45]; P = 0.023).
Total mass (TM), bone mineral content (BMC), bone mineral density (BMD), lean soft tissue mass (LSTM), fat mass (FM) and body fat percentage (%BF) values for adult male outdoor intact cats

Total mass (TM), bone mineral content (BMC), bone mineral density (BMD), lean soft tissue mass (LSTM), fat mass (FM) and body fat percentage (%BF) values for adult male indoor neutered cats
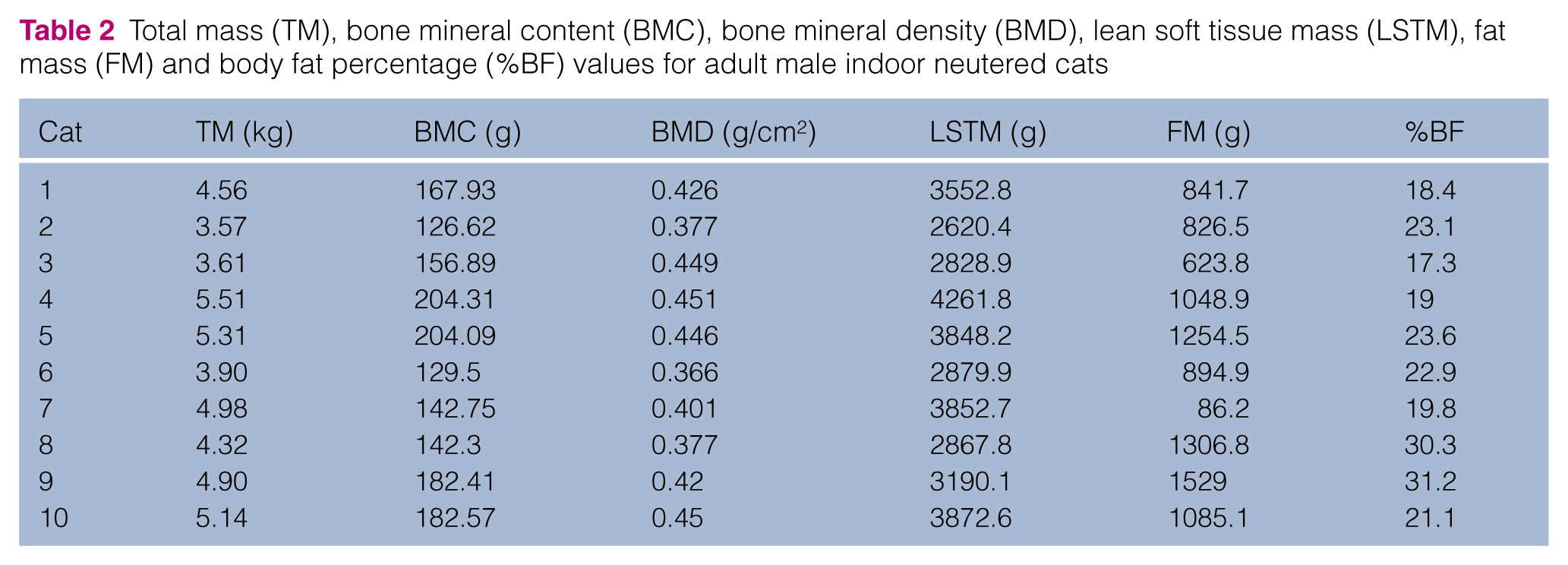
Total mass (TM), bone mineral content (BMC), bone mineral density (BMD), lean soft tissue mass (LSTM), fat mass (FM) and body fat percentage (%BF) values for adult female outdoor intact cats
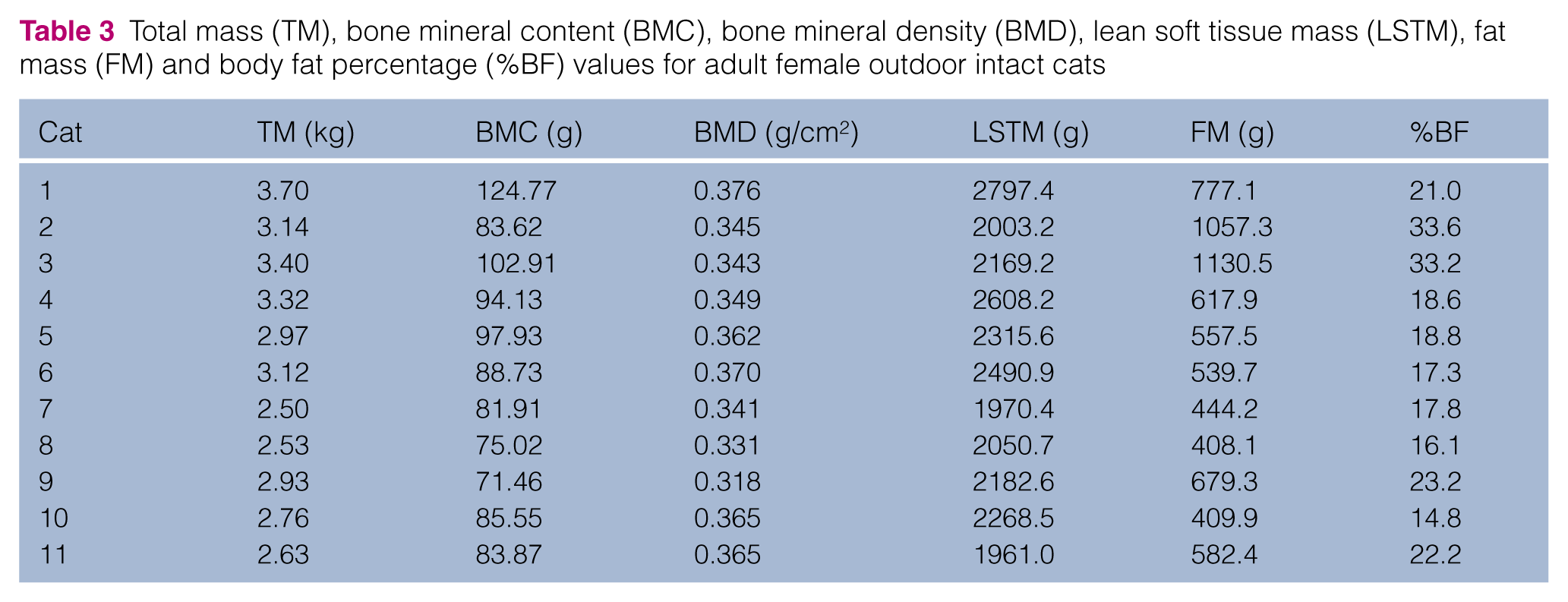
Total mass (TM), bone mineral content (BMC), bone mineral density (BMD), lean soft tissue mass (LSTM), fat mass (FM) and body fat percentage (%BF) values for adult female indoor neutered cats

IN females had a greater TM than OI females (mean IN 3.82 ± 0.44 kg, OI 3.00 ± 0.38 kg; P = 0.001). TM for IN and OI males was not significantly different (mean IN 4.58 ± 0.70 kg, OI 4.31 ± 1.45 kg; P = 0.604). Body fat percentage did not differ significantly in females (median IN 23.2% [range 19.7–28.2], OI 18.7% [range 14.8–33.6]; P = 0.159) but was significantly higher in IN than OI males (mean IN 22.67 ± 4.75%, OI 14.89 ± 2.78%; P <0.001). BMD was lower in OI females than in OI males (P = 0.009) and IN males (P < 0.001).
Discussion
Ideal body weight OI cats had a lower body fat percentage than ideal body weight IN cats. While this was significant for males in our study population, this was not significant for females. Mean %BF of male and female client-owned domestic cats with a BCS of 5/9 has been previously reported as 30.1 ± 4.1% and 31.6 ± 4.6%, respectively. 7 Male cats in our IN population had a mean %BF of 22.7%, whereas female IN cats had a median %BF of 23.2%, and both were lower than previously reported. The median age in the previously reported study was 6 years (range 3–13 years). The more mature age of these cats vs the relatively young adult cats included in this study (median 4 years, range 1–6 years) may account for the lower %BF. Body condition scoring is a somewhat subjective process and investigators in the separate studies may differ in the 5/9 scoring techniques. While the average %BF of indoor cats in our study was still within what is considered an ideal range for cats (20–24%),6–8,21 the combined effects of neutering, indoor confinement and age likely contributed to the slight increase in %BF when compared with intact outdoor cats. However, the results of our study were unable to demonstrate that IN cats with ideal body conditions suffer from normal weight obesity as a higher %BF would be expected.
The %BF of IN males in this study is similar to the %BF of males with a BCS of 5/9 in a previous study to validate the BCS method (21.8 ± 1.7%). 6 Female cats with a BCS of 5/9 were not reported in the previous study, and only one female cat was reported as having a BCS of 6/9. However, regression analysis predicted a %BF of 37.8%. Cats included in the Laflamme study were of an unreported neuter status; 6 therefore, the presence of intact animals may have influenced the results. Current guidelines for weight management in domestic animals recommends an ideal %BF of 20–24% for animals with a BCS of 5/9. 8 Results of this study would support this recommendation.
The BMD of OI females was significantly lower than both OI and IN males. Sex differences in BMD have been previously described in the cat. Like our study, the BMD, as assessed by DEXA, of 1–2-year-old intact male cats was statistically higher than 1–2-year-old intact female cats from a closed breeding colony. 22 A more recent study using quantitative CT to assess trabecular BMD did not find any significant sex differences. 23
The BMD of IN cats was significantly lower than OI cats in this study population. This finding was unexpected as higher levels of circulating sex hormones (ie, testosterone and estrogen) are positively associated with BMD in people. 24 BMD was increased by 16% when comparing 6–10-month-old cats with 1–2-year-old cats. 24 A limitation of this study was the inability to obtain an accurate age of the OI cats. We estimated the age of OI cats based on a physical examination, although it is possible that some of the cats were <1 year of age. Free-roaming cats are typically not expected to have a long life span due to their environment; therefore, only young adult cats in both the OI and IN groups were selected.
Diet can potentially impact the musculature, body fat and BMD of cats. The diet history of OI cats was impossible to obtain and therefore could not be compared with IN cats. The OI cats came from known colonies in the surrounding area, with access to a wide variety of local wildlife in addition to food provided by humans. The nutrient profile of free-roaming feral cats is estimated to be approximately 52% metabolizable energy (ME) protein, 46%ME fat and 2% nitrogen-free extract with adequate amounts of mineral and trace element consumption. 25 Analysis of feral cat stomach contents from the urban San Francisco Bay area found that 41/68 (60%) cats contained prey, 63% of which were small rodents; however, prey only represented approximately 28% of the metabolizable energy of the feral cat’s diet. 26 OI cats in our study came from urban, suburban and rural areas, and their diet intake likely varied in relation to their proximity to humans. IN cats in our study were more likely to be on a complete and balanced diet with a consistent intake of calcium and phosphorus at recommended ratios, which may have impacted their BMD when compared with OI cats.
We acknowledge several limitations with this study. A limitation of DEXA is that it assumes the hydration of lean soft tissue mass as 0.73 ml/g in all animals under all conditions.27,28 OI cats may have been subclinically dehydrated confounding our results. Dehydration could occur because of the trapping process during summer months. Further, the sterilization procedure, particularly the ovariohysterectomy of OI cats, may have contributed to further body fluid loss. Among the females, the small sample size of IN cats (n = 6) may have limited the ability of achieve significance. The BCS was obtained in OI cats under anesthesia for the safety of investigators. IN cats were awake and in a standing position for BCS assessment. Differences in obtaining BCS in these two groups may have affected the results, although a systematic approach to BCS was employed by the investigators to minimize any of these differences.
Conclusions
Observed differences in body composition between OI and IN cats are most likely related to indoor confinement and neutering. Normal weight obesity was not observed in the study. Moreover, an ideal %BF of 20–24% for young adult, healthy IN cats is still recommended.
Footnotes
Acknowledgements
We would like to thank the Companion Animal Initiative of Tennessee and volunteers who assisted with data collection: Ashley Cox, Gina Galyon, Susan Sanchez, Seth Patterson, Laura Burgette, Jessica Pruitt and Amy Macintire. Additionally, the authors would like to thank Josh Price and Dr Jonathan Lustgarten for their assistance with statistical analysis.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by a grant from Hill’s Pet Nutrition.
