Abstract
Objectives
Lymphoma is a common and clinically important malignancy in cats. Development of a second malignancy has been reported previously in 7–14% of cats with small-cell gastrointestinal (GI) lymphoma. The aim of our study was to describe the incidence, clinical signs, clinicopathologic data, response to therapy and outcomes in cats diagnosed with large-cell lymphoma following treatment for small-cell GI lymphoma.
Methods
Medical records from a single referral specialty hospital were reviewed for all cats with lymphoma diagnosed between 2008 and 2017. The cases with a diagnosis of small-cell GI lymphoma followed by a diagnosis of any large-cell lymphoma and complete outcome data were selected for further review.
Results
Seven hundred and forty cats with a diagnosis of lymphoma were identified. Twelve cats (12/121) treated for small-cell GI lymphoma followed by a diagnosis of any anatomic form of large cell lymphoma were identified. Nine cats met the study inclusion criteria and were used in analyses. Mean event-free survival time from small-cell GI lymphoma diagnosis until diagnosis of large-cell lymphoma was 543 days, with a median survival time of 615 days. Mean event-free survival time from large-cell lymphoma to death was 55 days, with a median survival time of 24.5 days. Hematocrit, albumin and total protein were significantly decreased when cats developed large-cell lymphoma compared with their values at the time of small-cell lymphoma diagnosis.
Conclusions and relevance
Large-cell lymphoma occurred in 9.9% (12/121) of cats treated for small-cell GI lymphoma. Feline practitioners should include large-cell lymphoma on their list of differential diagnoses in cats diagnosed with small-cell GI lymphoma developing weight loss, anemia, hypoalbuminemia and hypoproteinemia.
Introduction
Lymphoma is a common and clinically important malignancy in cats. Since the decline in feline leukemia virus, the gastrointestinal (GI) tract has emerged as the most commonly affected anatomic location.1–3 While multiple pathologic entities exist, the disease has been previously classified into two broad forms of lymphoma: indolent, or small-cell lymphoma, and the more aggressive, intermediate-to-large-cell lymphoma.3–5
Indolent GI lymphoma is characterized by lamina propria (mucosal) or transmural infiltration of the GI tract by a monomorphic population of well-differentiated, small lymphocytes.3–6 Lymphocytes can be arranged in nests and plaques, and demonstrate occasional epitheliotropism, which can be diagnosed with endoscopic or full-thickness biopsies.3–6 This type of lymphoma typically carries a favorable prognosis.7–11 Originally, indolent small-cell GI lymphoma was thought to account for about 10% of cases.1,3,11 However, recent data suggest that previous studies underestimated disease occurrence, and current reports indicate that small-cell GI lymphoma represents 36–75% of all feline GI lymphoma cases.4–6 The disease affects older cats, with a median age of 12.3–14 years.7,9–13 The current standard of care is a combination glucocorticoids and chlorambucil (Leukeran; GlaxoSmithKline) therapy. Several dosing regimens exist, with response rates ranging from 69–96%.7,9,10 Therapy is usually well tolerated with minimal toxicities.7,8,11 Reported median survival times (MSTs) range from 786–1317 days.7,10,11
The presence of neoplastic large, immature lymphocytes (greater than the size of a neutrophil on cytologic evaluation, or nuclei greater than twice the size of a red blood cell with histopathology) on pathologic evaluation is consistent with intermediate-to-high-grade lymphoma.3,4 This diagnosis carries a poor prognosis.9,12,14–17 Recommended chemotherapy protocols vary, but commonly multi-agent chemotherapy regimens – often variations of CHOP (doxorubicin [Adriamycin; Pfizer], vincristine [Oncovin; Hospira], cyclophosphamide [Cytoxan; Baxter] and glucocorticoids]) – are recommended.12,14–17 Remission rates range from 30–80%, with expected MSTs ranging from 97–213 days,12,14–17 or longer for cats in a complete remission.14–17
Secondary malignancies in patients with small-cell GI lymphoma have been previously reported with rare instances being large-cell lymphoma.4,7,11 Despite the known occurrences of large-cell lymphoma in cats previously treated for small-cell GI lymphoma, to our knowledge, there are no existing data regarding such cases. Thus, the goals of this study include describing a population of cats undergoing therapy for small-cell GI lymphoma that develop large-cell lymphoma and undergo treatment for the second malignancy. Reported here are the clinical signs and clinicopathologic data at the time of both small- and large-cell lymphoma diagnoses, chemotherapy protocols, comorbidities, response rates, toxicities and survival of cats affected with small-cell GI lymphoma followed by large-cell lymphoma.
Materials and methods
The medical record database of the Animal Medical Center was searched from January 2008 to October 2017 for cats with a lymphoma diagnosis. Figure 1 illustrates the case selection. GI lymphoma was defined as documented lymphoma via histopathology, cytology and/or molecular clonality testing (PCR for antigen receptor rearrangement [PARR]) within the stomach, small intestine and/or large intestine. Small-cell GI lymphoma diagnosis required histopathologic confirmation.4,6,18,19 Cats diagnosed with large granular lymphocyte lymphoma were included in the large-cell lymphoma group. The medical records of cats that underwent treatment for small-cell GI lymphoma were reviewed for a diagnosis of large-cell lymphoma. To be included in this study, cats were required to be treated for both small- and large-cell lymphoma and have complete follow-up.

Feline lymphoma diagnoses from Animal Medical Center between January 2008 and October 2017. GI = gastrointestinal
Medical records of the 12 cats treated for small-cell GI lymphoma subsequently diagnosed with large-cell lymphoma were reviewed. Clinicopathologic data and diagnostic imaging results for each cat were collected from two unique times; time of small-cell GI lymphoma diagnosis and time of large-cell lymphoma diagnosis. Data included age, body weight, clinical signs, clinicopathologic abnormalities, diagnostic imaging abnormalities, diagnostic method(s), chemotherapeutic protocol(s), response to therapy and survival time. Maximum body weight while on small-cell GI lymphoma therapy was recorded for each cat as a surrogate data point for response to therapy. 20 Maximum body weights were used to calculate a median maximum body weight. Median maximum body weight was then compared with median body weights at time of large-cell lymphoma diagnosis.
Screening for comorbidities was based on available clinical pathology data (pancreatic lipase, cobalamin levels, urinalysis and thyroid levels) when available, thoracic radiographs and ultrasound examinations. Chemotherapy toxicity was retrospectively assessed and graded based on the Veterinary Co-operative Oncology Group’s Common Terminology Criteria for Adverse Events (VCOG-CTCAE version 1.1). 21 No post-mortem examinations were available.
Response to initial therapy was based on resolution of clinical signs previously attributed to small-cell GI lymphoma. In following the previous established parameters, a complete response was defined as 100% resolution of clinical signs for ⩾30 days. A partial response was defined as ⩾50% but <100% improvement of clinical signs for ⩾30 days. Non-responders had <50% improvement of clinical signs, a response of <30 days or progressive disease.10,11
Continuous data were assessed for normality with the Shapioro–Wilk test. Baseline descriptive statistics were normally distributed and reported as mean ± SD. Between-group analyses of continuous variables were performed using the paired t-test (two-tailed). Analyses for proportions of categorical variables were evaluated with Fisher’s exact analysis (SigmaPlot13; Syst Software). Event-free survival comprised the time interval from the date of therapy for cases in the small-cell lymphoma cohort to the first date that each case developed large-cell lymphoma. Post-event survival comprised the time from the first date of treatment for each case in the large-cell lymphoma cohort to end date. End date was defined as death (natural death or death by euthanasia from intractable cancer or cancer-related complications). Patient(s) remaining alive at study completion were right censored. Owing to varied study enrollment and study end dates, the mean between-cohort survival times estimated by univariate Kaplan–Meier method were compared by ANOVA as the error residuals were normally distributed. All analyses were carried out with SAS 9.4 and deemed significant at P <0.05.
Results
Twelve of 121 cats (9.9%) treated for small-cell GI lymphoma were subsequently documented to have large-cell lymphoma. Three cats did not meet the inclusion criteria and the data from nine cats are reported here. All included cats had a biopsy confirmed diagnosis of small-cell GI lymphoma and a cytologic diagnosis of large-cell lymphoma. No PARR data were available for these cats at the time of small-cell or large-cell lymphoma diagnosis. Figure 2 represents the typical diagnostic histopathologic features of small-cell GI lymphoma. Figure 3 shows the typical diagnostic cytologic features of large-cell lymphoma. Select data for each cat from the time of small-cell GI lymphoma diagnosis are listed in Table 1. Interestingly, we also identified 11/290 cats (3.7%) that were affected by both small-cell GI lymphoma and large-cell lymphoma simultaneously. As these cats did not meet our inclusion criteria, they were not included in data analysis.
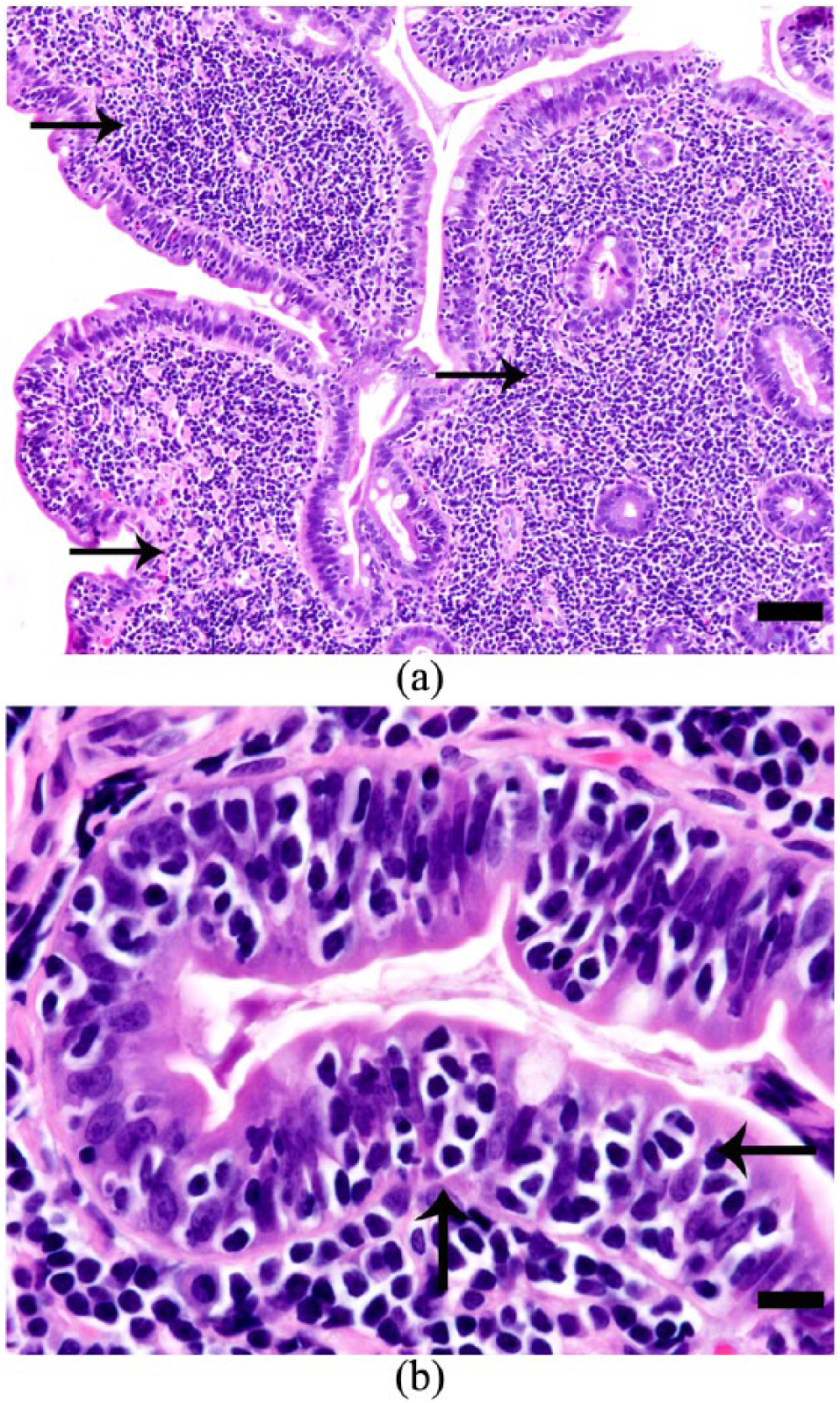
(a) Photomicrograph of a section of small intestine from a cat with epitheliotropic small-cell gastrointestinal lymphoma. The lamina propria is infiltrated and expanded by a proliferation of small lymphocytes, which widen the villi, separate the crypts and form sheet-like regions throughout the lamina propria (arrows). The cells are also present in nests and plaques in the overlying mucosal epithelium (hematoxylin and eosin × 40; bar 20 μm). (b) Photomicrograph of a section of small intestine from the same cat as in (a), at higher magnification showing epitheliotropism (arrows) with formation of nests (>5 clustered lymphocytes within a single enterocyte and plaques [>5 adjacent epithelial cells overrun by lymphocytes]) (hematoxylin and eosin × 100; bar 10 μm). Photomicrographs courtesy of Dr Taryn Donovan DVM, DACVP, Animal Medical Center, New York, NY, USA
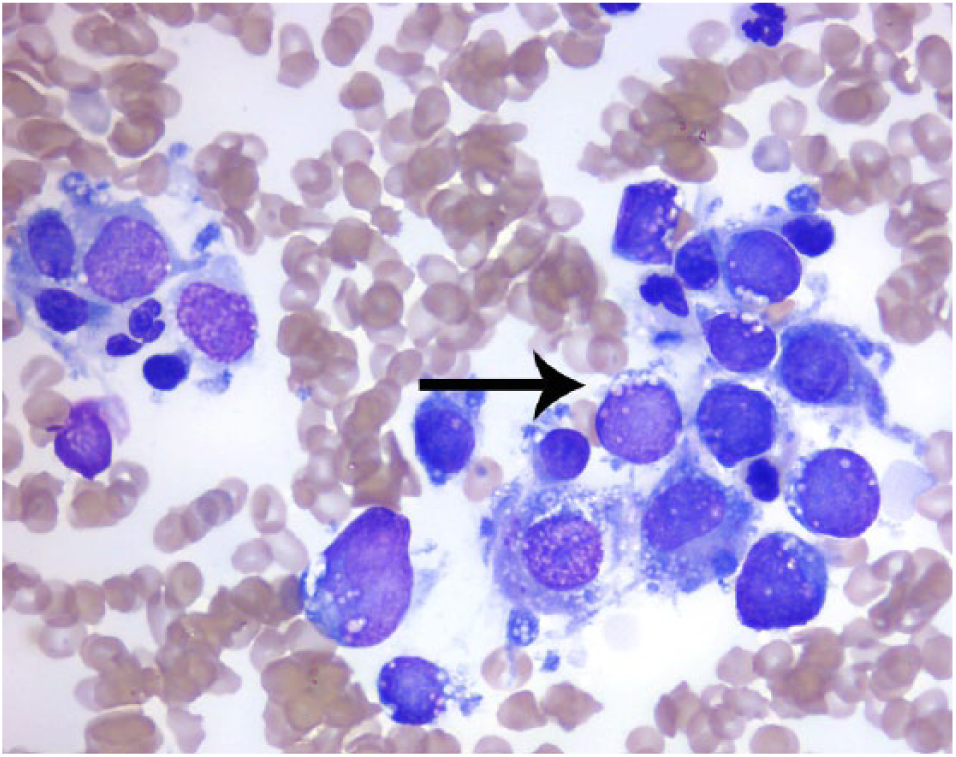
Photomicrograph of a cytologic sample of an intestinal mass fine-needle aspiration cytology from a cat with large-cell lymphoma. Large lymphoid cells (larger than a neutrophil) have a small amount of moderately basophilic cytoplasm, which exhibits punctate vacuoles (arrow). Nuclei are irregularly ovoid, eccentrically located, with a finely stippled chromatin pattern. (Hematoxylin and eosin, × 100). Photomicrograph courtesy of Dr Andrea Siegel DVM, DACVP, IDEXX Laboratories, New York, NY, USA
Signalment, selected clinical pathologic data, abdominal ultrasound abnormities, response to treatment, and survival at the time of small-cell gastrointestinal lymphoma diagnosis

All continuous values are expressed as median values
A = inappetence; B = vomiting; C = diarrhea; D = lethargy; E = weight loss; HCT = hematocrit; LSA = lymphoma; FS = female spayed; P = prednisolone; Ch = chorambucil; CR = complete response; PR = partial response; MC = male castrated; LN = lymph node; L = L-asparaginase; CHOP = combination chemotherapy: cyclophosphamide, doxorubicin, vincristine and prednisolone; NR = non-responder; RI = reference interval
The study included eight domestic shorthair cats and one domestic longhair cat. The most common anatomic locations of small-cell GI lymphoma diagnosis were the duodenum (8/9 cats) and ileum (6/9 cats). Anatomic areas sampled were not comprehensive for all cats. Vomiting was the most common clinical sign (8/9 cats). Other clinical signs included weight loss, diarrhea, inappetence and lethargy, and are detailed in Table 1. In all cases, clinical signs were classified as chronic by persisting for ⩾30 days.
Several comorbidities were identified at the time of small-cell GI lymphoma diagnosis. Six cats were categorized by the International Renal Interest Society (IRIS) as stage 1, two stage 2 and one stage 3 chronic kidney disease (CKD). Two were diagnosed with pancreatitis based on elevated feline pancreatic lipase results. In one cat, biopsy of the pancreas was consistent with nodular hyperplasia and mild chronic pancreatitis. The other cat did not have a pancreatic biopsy. Serum cobalamin concentration had been measured in eight cats and 4/8 of cats had a value ⩽400 ng/l.10,22 Cobalamin supplementation was provided to cats with hypocobalaminemia, 23 although regimes varied.
All nine cats diagnosed with small-cell GI lymphoma received glucocorticoid therapy (2 mg/kg/day [range 1–2.7 mg/kg/day]) for at least 1 month. Two cats received only glucocorticoid therapy. Six cats were treated with adjuvant chlorambucil.7,9,10 Mean chlorambucil dose was 1.1 mg/kg/week (range 0.8–1.4 mg/kg/week). One cat was treated with CHOP injectable chemotherapy, 17 and developed progressive disease within 3 weeks. This cat was subsequently switched to single-agent mechlorethamine (compounded by BestPetRX). Six of nine cats experienced a complete or partial response. Chemotherapy toxicities were rare. One cat developed a grade 3 neutropenia after 79 days of chlorambucil therapy, 21 at which point chlorambucil was discontinued. No other grade 3 or higher related toxicities were reported. One cat is still alive 900 days following the diagnosis of small-cell GI lymphoma and was censored from survival data. Of the remaining cats, the mean event-free survival time from diagnosis of small-cell GI lymphoma until development of large-cell lymphoma was 543 ± 378 days (Figure 4). Median survival time from small-cell lymphoma until death was 615 days (range 204–1267 days).

Kaplan–Meier survival curves estimating the event-free survival (EFS) proportion and post-event survival (PES) proportion (y-axis) against time (x-axis). EFS comprised a cohort of nine cats with small-cell lymphoma and was calculated as the time from initial onset of therapy to the time that large-cell lymphoma was diagnosed (red line). PES was calculated for these same nine at the time that each was diagnosed with large-cell lymphoma to the time of death (blue line). Eight of these nine cats died from cancer or related complications. EFS was significantly longer compared with PES (P = 0.0008)
In these cats, different clinical situations emerged that led to further diagnostic testing and ultimately a diagnosis of large-cell lymphoma. Relapse of GI clinical signs was the most common reason for additional testing and five cats were noted to have a new intestinal mass. Other causes included azotemia in two cats and voice change and dyspnea in one cat.
Select data for each cat from the time of large-cell lymphoma diagnosis are listed in Table 2. Five of nine cats were anemic at the time of large-cell lymphoma diagnosis. The decrease in hematocrit between small-cell GI lymphoma and large-cell lymphoma diagnoses was statically significant (P = 0.06). Five cats had IRIS stage 2 CKD at the time of large-cell lymphoma diagnosis, two cats were stage 4 and one cat was stage 1. One cat did not have renal parameters available for evaluation. The difference in IRIS stage between small- and large-cell diagnosis was not statically significant (P = 0.153). Biochemical profiles, including liver enzymes, were available for eight cats. Three cats had liver enzyme elevation. Only one cat had cytologic evaluation of the liver, consistent with large-cell lymphoma; however, this cat had normal liver enzymes. Mean total protein decreased significantly (P = 0.04) from the time of small-cell GI lymphoma diagnosis and large-cell lymphoma diagnosis. Mean albumin decrease was also statistically significant (P = 0.003) between diagnoses. Two cats had developed hyperthyroidism between the diagnosis of small-cell GI lymphoma and large-cell lymphoma.
Duration between diagnosis of small-cell gastrointestinal (GI) lymphoma and large-cell lymphoma, anatomic location of large-cell lymphoma, selected clinical pathology data, abdominal ultrasound abnormalities, response to treatment and survival at the time of large-cell GI lymphoma diagnosis

All continuous values are expressed as median values
Patient was alive at time of manuscript submission at 131 days post-large-cell diagnosis
A = inappetence; B = vomiting; C = diarrhea; D = lethargy; E = weight loss; F = abdominal mass; G = bilateral renomegaly; H = dyspnea and voice change; BW = body weight; LSA = lymphoma; HCT = hematocrit; PR = partial response; L = L-asparaginase; COP = combination chemotherapy; CHOP = combination chemotherapy of cyclophosphamide, doxorubicin, vincristine and prednisolone; NR = non-responder; L-CCNU = 1-(2-chloroethyl)-3-cyclohexyl-1-nitrosourea or lomustine; LN = lymph node; P = prednisolone
All nine cats underwent treatment for large-cell lymphoma and received glucocorticoid therapy (2 mg/kg/day). Two cats received only glucocorticoids and survived 14 and 20 days, respectively, and one also received L-asparaginase (Elspar; Lundbeck) 24 but survived 2 days. Four cats were treated with L-asparaginase and CHOP. 17 One cat was treated with L-asparaginase and a COP protocol. 16 One cat was treated with L-asparaginase and lomustine. 25 The two cats with renal lymphoma had cytosine arabinoside (Cytarabine; Pfizer) incorporated into their chemotherapy protocol. 26 Three of nine cats treated for large-cell lymphoma after treatment for small-cell lymphoma had a documented response to treatment. One cat is still alive (131 days following large-cell lymphoma diagnosis) and is receiving treatment for renal large-cell lymphoma at the time of manuscript preparation. The mean post-event survival for the eight other cats from the time of large-cell GI lymphoma diagnosis until death was 55 ± 66 days (Figure 4). Median survival time was 24.5 days (range 2–183 days).
Discussion
The data presented here represent the first report focused on cats diagnosed with small-cell GI lymphoma that subsequently developed the secondary malignancy of large-cell lymphoma. This occurred at an incidence of 9.9% in a large population of cats diagnosed with small-cell GI lymphoma. Secondary malignancies are a well-documented phenomenon in human patients.27–30 The risk of a second malignancy in human patients treated for lymphoproliferative disorders approaches 15% at 20 years’ survival.27–30
While the true frequency of secondary malignancies in veterinary medicine is unknown, it is likely underestimated. Previously reported secondary malignancies do exist in veterinary patients undergoing cancer treatment, including transitional cell carcinomas associated with cyclophosphamide administration and radiation therapy-induced sarcomas.31–33 Second malignancy development has been reported in 7–14% of small-cell GI lymphoma cats and our data are consistent with that finding.4,7,11 Reported secondary malignancies in such patients have included mammary carcinoma, gastric mast cell tumor, carcinomatosis, oral squamous cell carcinoma, colonic mucinous adenocarcinoma and large-cell lymphoma.7,11 It appears that there is nearly equal frequency of carcinomas and large-cell lymphoma in previously treated cats with small-cell GI lymphoma.
Though not the focus of this paper, an interesting fact worth mentioning is the rate of simultaneous small-cell GI and large-cell lymphoma found in our original patient search. We identified 11/290 cats (3.7%) that were affected by both small-cell GI lymphoma and large-cell lymphoma simultaneously (Figure 1). Moore et al identified a similar rate of affected cats at 3.3%. 4
The results of this study indicate that a dramatic change in the clinical status of cats undergoing treatment for small-cell GI lymphoma preceded the diagnosis of large-cell lymphoma. Cats lost weight, became anemic, exhibited lower albumin and total protein values at the time of large-cell lymphoma diagnosis. Monitoring trends in laboratory data (hematocrit, albumin and total protein) and changes in body weight are simple and inexpensive screening techniques available to every veterinarian. Early detection of changes could lead to an earlier diagnosis and treatment of large-cell lymphoma, or detection of other comorbidities such as pancreatitis, CKD or hyperthyroidism capable of causing similar abnormalities. In the cats ultimately diagnosed with large-cell lymphoma, early detection could hopefully improve survival compared with that reported here.
The similarities in this group of cats to cats reported in prior publications makes the data reported here useful to feline practitioners as we expect other veterinarians will diagnose large-cell lymphoma in cats undergoing treatment for small-cell GI lymphoma. The distribution of GI lymphoma in our population of cats reflects the previously reported frequency in the literature. Our data revealed that 41.7% of all GI lymphoma cases were diagnosed as small-cell GI lymphoma. This frequency falls into the previously reported range of 36–75%.4,6 Cats in this study also reflect prior reports of anatomic distribution of small-cell GI lymphoma. Shown here, 7/9 cats had histologically documented small-cell lymphoma in more than one anatomic location, with the small intestine being most common.4,8 Our nine patients demonstrated a high overall response rate, consistent with previous reports of small-cell GI lymphoma cats.7,10 Chlorambucil chemotherapy-related toxicities were rare, as expected.7,8,11 CKD was a common comorbidity in these cats, 34 and was evident in all nine cats included in this study. Over time, IRIS stage progressed in these cats; however, no statistically significant progression could be proven. A chemotherapy response was noted in 3/9 cats diagnosed with large-cell lymphoma, similar to that reported by Collette et al. 17 Survival data reported here also support previous publications of an abbreviated survival in cats diagnosed with large-cell lymphoma that do not achieve complete remission.14–17
There were several differences noted between our data and previously reported data. A noteworthy difference in our study cats is the lower median age at diagnosis of small-cell GI lymphoma. Cats diagnosed with small-cell GI lymphoma subsequently developing large-cell lymphoma were typically afflicted with small-cell GI lymphoma earlier in life than previously reported. The nine cats in this study had a median age of 11.4 years at the time of their small-cell GI lymphoma diagnosis compared with reported median ages of 12.3–14 years.7,10,11,13
Furthermore, our cats had an abbreviated median survival time vs previous reported survival times for cats with small-cell GI lymphoma.7,10,11 The mean event-free survival time from small-cell GI lymphoma to large-cell lymphoma diagnosis was 543 ± 378 days. Median survival time was 615 days (range 204–1267), and when the abbreviated post-event survival time is considered, the overall survival of these cats appears less than expected. Based on our study results, younger cats on therapy for small-cell GI lymphoma should be monitored closely for clinical signs and clinicopathologic changes consistent with possible large-cell lymphoma.
In addition, our reported frequency of secondary large-cell lymphoma is nearly double that of the existing literature. 11 Pope et al found only 5.4% of small-cell GI lymphoma cats were subsequently diagnosed with large-cell lymphoma. 11 It is unknown why our cats had a higher rate of large-cell lymphoma. This could be due to differences in patient populations, geographic locations, chemotherapy protocol utilization, or to other reasons not yet identified.
Another potential difference was the most commonly affected anatomic location of small-cell GI lymphoma; however, not all cats had a biopsy obtained from all regions of the intestine. In our study duodenum (8/9 cats) and ileum (6/9 cats) were commonly affected. This contrasts with Moore et al, 4 who reported jejunum was the most commonly affected region of the GI tract. 4
It is interesting to speculate on the causes of the second tumors in this group of cats. One plausible reason is that cats treated successfully for their small-cell GI lymphoma diagnosis lived long enough to reach an age whereby they were at risk for developing large-cell lymphoma as a de novo entity.
Another possibility is that the combination of development of a malignancy at a younger age and exposure to chemotherapy led to an increased risk of a second hematopoietic malignancy later in life. In our study, six cats underwent chlorambucil treatment for their small-cell GI lymphoma diagnosis and one received cyclophosphamide. Chlorambucil and cyclophosphamide are nitrogen mustard derivatives, members of the alkylating agent group of cytotoxic drugs. Their mechanism of action involves covalent bonds between alkyl groups within DNA. This binding leads to inter- and intra-strand cross-links and, ultimately, cell damage. If these alkylating agents induced DNA damage leading to cellular mutations instead of cellular death, a second malignant population of immature lymphocytes could arise. In people, younger patients treated with alkylating agents are at greater risk of developing of a second malignancy and our cats were younger than the typical cats reported with GI small-cell lymphoma.28,29,35 Human secondary leukemia occurs, on average, 5–7 years post-alkylating agent therapy,30,35 and is commonly associated with specific cytogenetic abnormalities in chromosomes 5 or 7.36,37 It is currently unknown if other species experience similar genetic changes as a sequela to chemotherapy.
A final hypothesis to explain the development of a second tumor in these cats could be that proliferation of small-cell lymphoma cells may ultimately lead to mutation(s) allowing for a more aggressive lymphoma phenotype that would then be classified as a large-cell lymphoma. Given that lymphoma is a hematopoietic malignancy, these mutated aggressive cells feasibly could localize in any area of the body as the disease progresses explaining why we diagnosed large-cell lymphoma in multiple anatomic locations. Advanced molecular testing (PARR or immunohistochemistry) on serial biopsy samples of cats with suspected secondary malignant lymphoma could help further investigate these theories. If molecular data had been available for these cats and had confirmed T-cell genotype for the small-cell GI lymphoma, the most common finding in this tumor, 3 and a B-cell genotype was found in the large-cell lymphoma, then one could hypothesize a new malignancy had arisen. If both tumors were found to be the same phenotype (ie, both T-cell), then one hypothesis to explain the finding would be the small-cell GI lymphoma had become a more malignant version of the original tumor. Confirmation of these hypothesis would require further investigation into the sequential chromosomal and genetic changes of these paired tumors.
This study has several limitations, all related to its retrospective design and small case numbers. This study likely underestimates the true occurrence of small-cell GI lymphoma with subsequent large-cell lymphoma in cats. Cases included in this study were required to have a definitive histopathologic or molecular diagnosis of small-cell lymphoma. Several excluded cats were noted to have a presumptive diagnosis of inflammatory bowel disease prior to the diagnosis of large-cell lymphoma.6,18,19 Some of these cats received glucocorticoid therapy and several cases developed large-cell lymphoma. We suspect that diagnostic evaluation in such cats would have led to the inclusion of additional cases. Thus, our large-cell lymphoma secondary malignancy incidence of 9.9% is likely a conservative one. Anatomic areas sampled in our study cats were not comprehensive and true disease burden could be under-represented in cats with small-cell GI lymphoma. Comprehensive histopathologic evaluation of intestinal biopsy samples is recommended for cats with chronic weight loss and GI signs. In cases with biopsies supportive of possible lymphoma, such as epithelitropism or lymphocytes arranged in nests and plaques,4,6 consultation with a pathologist regarding additional testing should be considered. Another limitation of our small case numbers is that our cats were not classified as mucosal or transmural small-cell GI lymphoma. A previous report indicates transmural small-cell GI lymphoma may carry a worse prognosis. 4 There were no standard staging protocols performed on these patients and full disease burden is likely under-reported. Because adverse events associated with chemotherapy were abstracted from the medical record, we believe they, too, may be underestimated. Therapy was not standardized in these patients. The multiple protocols administered limit our ability to recommend an optimal treatment protocol for these cats.
Conclusions
Large-cell lymphoma occurred in 9.9% of cats treated for small-cell GI lymphoma. Feline practitioners should have large-cell lymphoma on their list of differential diagnoses in cats diagnosed with small-cell GI lymphoma at a younger age, with weight loss or marked changes in hematocrit, albumin, total protein or in cats with a new intestinal mass. The survival time in cats undergoing treatment for large-cell lymphoma after treatment for small-cell lymphoma appears less than that expected for cats diagnosed with only large-cell lymphoma that receive treatment.
Footnotes
Acknowledgements
The authors would like to thank Dr Philip Fox of the Animal Medical Center in New York, NY, USA, and Kenneth E Lamb, PhD, of Lamb Statistical Consulting and Medical Editing in West Saint Paul, MN, USA, for providing statistical analysis of the data in this paper. Additionally, the authors could like to thank Dr Taryn Dovovan of the Animal Medical Center in New York, NY, USA, for her assistance in the preparation of this manuscript.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
