Abstract
Case series summary
Cutaneous mastocytosis is a disorder rarely reported in veterinary dermatology and usually described as ‘urticaria pigmentosa’. This study aimed to evaluate the diagnosis, treatment and outcome of 13 affected cats, selected from the files of a private referral dermatology practice within a period of 14 years. Breeds of the affected individuals included Sphynx (n = 9), Devon Rex (n = 2) and Sphynx/Devon Rex crossbreeds (n = 2). Females (n = 9) were over-represented and the median duration of clinical signs prior to diagnosis was 8 months. The clinical presentation of these 13 cats was compared with cases reported in the veterinary literature and classified according to the current human consensus on cutaneous mastocytosis. Three clinical forms could be distinguished in cats: (1) large papular lesions and wheals, typically localised to the head, shoulders, ventral neck and axillae, and which may spontaneously resolve (termed polymorphic maculopapular cutaneous mastocytosis); (2) erythematous dermatitis, characterised by small maculopapular lesions often associated with crusts and with a poorer prognosis (termed monomorphic maculopapular cutaneous mastocytosis); and (3) more chronic dermatitis characterised by lichenification and hyperpigmentation, similar to the human condition ‘urticaria pigmentosa’ (termed pigmented maculopapular cutaneous mastocytosis). Histopathology was performed in eight cases and revealed a superficial-to-deep dermatitis characterised by infiltrates of mast cells and eosinophils. The response to various treatments, including antihistamines, steroids and ciclosporin, was variable.
Relevance and novel information
This article reports 13 new cases of feline cutaneous mastocytosis, confirming the clinical presentation and apparent breed predisposition. The feline maculopapular cutaneous mastocytosis seems to be clinically very close to the human form. This study proposes a new classification system for the feline disease based on the current human consensus, clinical presentation and prognosis, with three different subforms: polymorphic maculopapular cutaneous mastocytosis with eventual spontaneous regression; monomorphic maculopapular cutaneous mastocytosis with chronic evolution; and pigmented maculopapular cutaneous mastocytosis.
Introduction
Cutaneous mastocytosis (CM) has been reported in cats as a maculopapular eruption, often associated with significant pruritus. Cats are presented with dermatological lesions only, consisting of papules, nodules and/or inconsistent hyperpigmentation.1–4 The most frequent term used to describe this feline disease is ‘urticaria pigmentosa’.1–5
Mastocytosis is also reported in humans. It is characterised by the expansion of clonal mast cells (MCs) in different organs. Two main subtypes are described: CM and systemic mastocytosis (SM). In the latter condition, skin lesions are present in association with MC accumulation in other organs, mostly the bone marrow and gastrointestinal tract. CM usually has a benign course but may sometimes progress to SM.6,7 The clinical course ranges from ‘asymptomatic’ to highly aggressive in some systemic forms. Recently, an international task force has proposed a consensus on the classification and criteria regarding diagnosis of the human disease. 8
The goal of this paper is to report 13 new cases of feline CM (FeCM). Taking into account recent advances in human medicine we make the correlation to mastocytosis in cats and suggest a new classification for the feline disease.
Case series description
Cases of FeCM were retrospectively selected from the files of our dermatology referral practice between 2003 and 2017. These cases were selected based on the typical clinical presentation and breed, exclusion of other dermatoses (feline food and non-food hypersensitivities, flea bite hypersensitivity, ectoparasitoses, dermatophytosis).9,10 Specifically, tests/trials included exclusion of food allergy through an 8 week elimination diet using hydrolysed protein diet, no apparent seasonal pruritus, negative Wood’s lamp and acetate tape impression, clear supportive fine-needle aspiration cytology (predominant population of MC and excluding bacterial infection) and/or histological findings (haematoxylin–eosin, periodic acid–Schiff and Giemsa-stained sections). Histology was conducted by the same pathologist (MH). Clinical criteria for inclusion, based on previous studies, included macules, papules (with or without crusts), nodules, hyperpigmentation, lichenification, excoriations, pruritus and early onset.1,3 The evolution of the disease, the treatments administered and the responses to therapy were reviewed.
Thirteen cases of FeCM were selected (Table 1), consisting of nine Sphynx, two Devon Rex and two Sphynx Devon Rex crossbreeds. The median age at presentation to us was 15 months (range 3–60 months), while the median estimated time before final diagnosis was 8 months. Four of the cats were males; nine were females. On general physical examination, the cats were healthy with abnormalities restricted to the skin.
Signalment, lesions, pruritus, diagnosis method, classification subform and follow-up of 13 cases of feline cutaneous mastocytosis from 2003–2017

mMPCM = monomorphic maculopapular cutaneous mastocytosis; pMPCM = polymorphic maculopapular cutaneous mastocytosis; pigMPCM = chronic pigmented maculopapular cutaneous mastocytosis; F = female; E = erythema; P = papules; LP = large papules; W = wheals; LTF = lost to follow-up; M = male; FS = female sterilised; MS = male sterilised; H = hyperpigmentation
Three clinical presentations were observed: in six cases (five Sphynx, one cross-breed) the lesions were very ‘polymorphic’, consisted of large wheals and 0.5–1.0 cm papules, and were mainly located on the front of the body (head, shoulders, ventral neck, axillae; Figures 1 and 2) without any hyperpigmentation. Three cats (two Sphynx, one crossbreed) presented with a ‘monomorphic’ clinical picture characterised by erythema, small papules that could be coalescent – often associated with crusts (secondary to pruritus) – and sometimes 0.5–1.0 cm papules (Figures 3 and 4); lesions were mostly generalised. Four cases (two Sphynx, two Devon Rex) presented with more chronic and generalised lesions, mainly consisting of erythematous, crusted coalescing papules, wheals, excoriations, hyperpigmentation and lichenification (Figures 5 and 6); this last clinical form, the only one with clear hyperpigmentation, was typically previously described as ‘urticaria pigmentosa’.
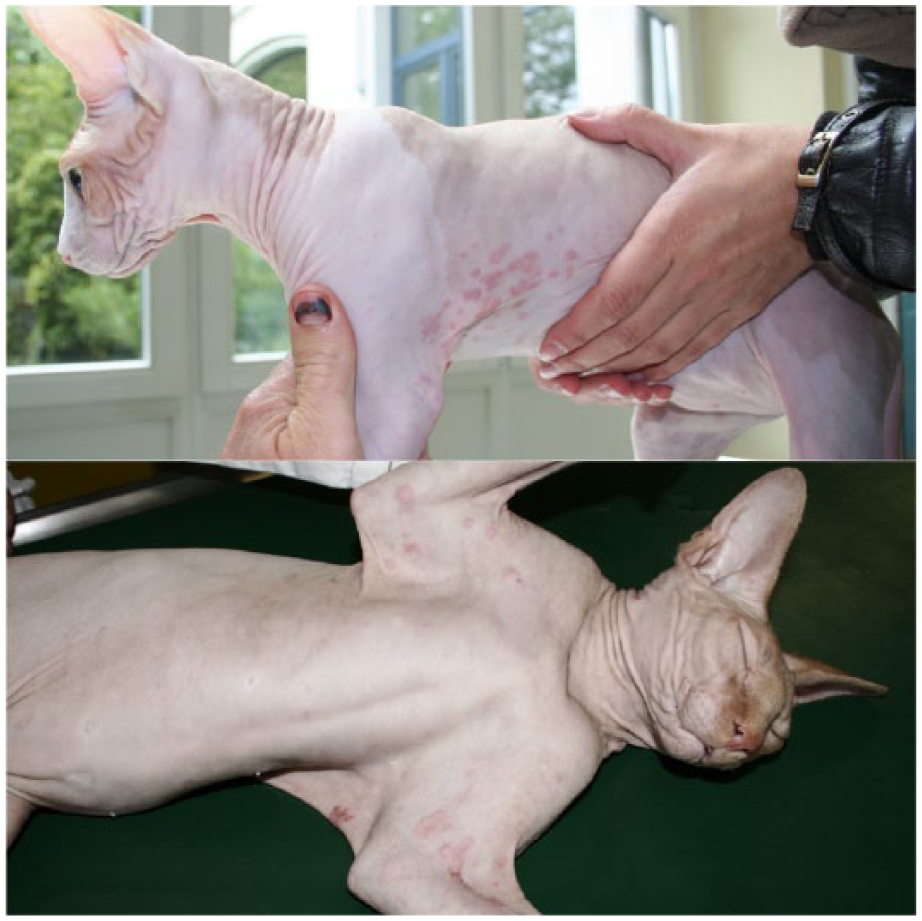
Cases 1 and 2: papules and wheals/large papules in two Sphynx cats localised on the axilla. Some crusts associated with pruritus are observed (polymorphic maculopapular cutaneous mastocytosis)

Case 3: large papules on the (a) axilla and (b) head in a Sphynx kitten (polymorphic maculopapular cutaneous mastocytosis [pMPCM]). (c) Global aspect of pMPCM in a Sphynx kitten

Case 7: global aspect of monomorphic maculopapular cutaneous mastocytosis in a Devon Rex

Case 9: papules and crusts on the (a) face, and (b) chest and limbs in a Sphynx. Note the self-traumatic lesions (monomorphic maculopapular cutaneous mastocytosis)

Case 10: generalised papules and erythema in a Devon Rex (monomorphic maculopapular cutaneous mastocytosis)
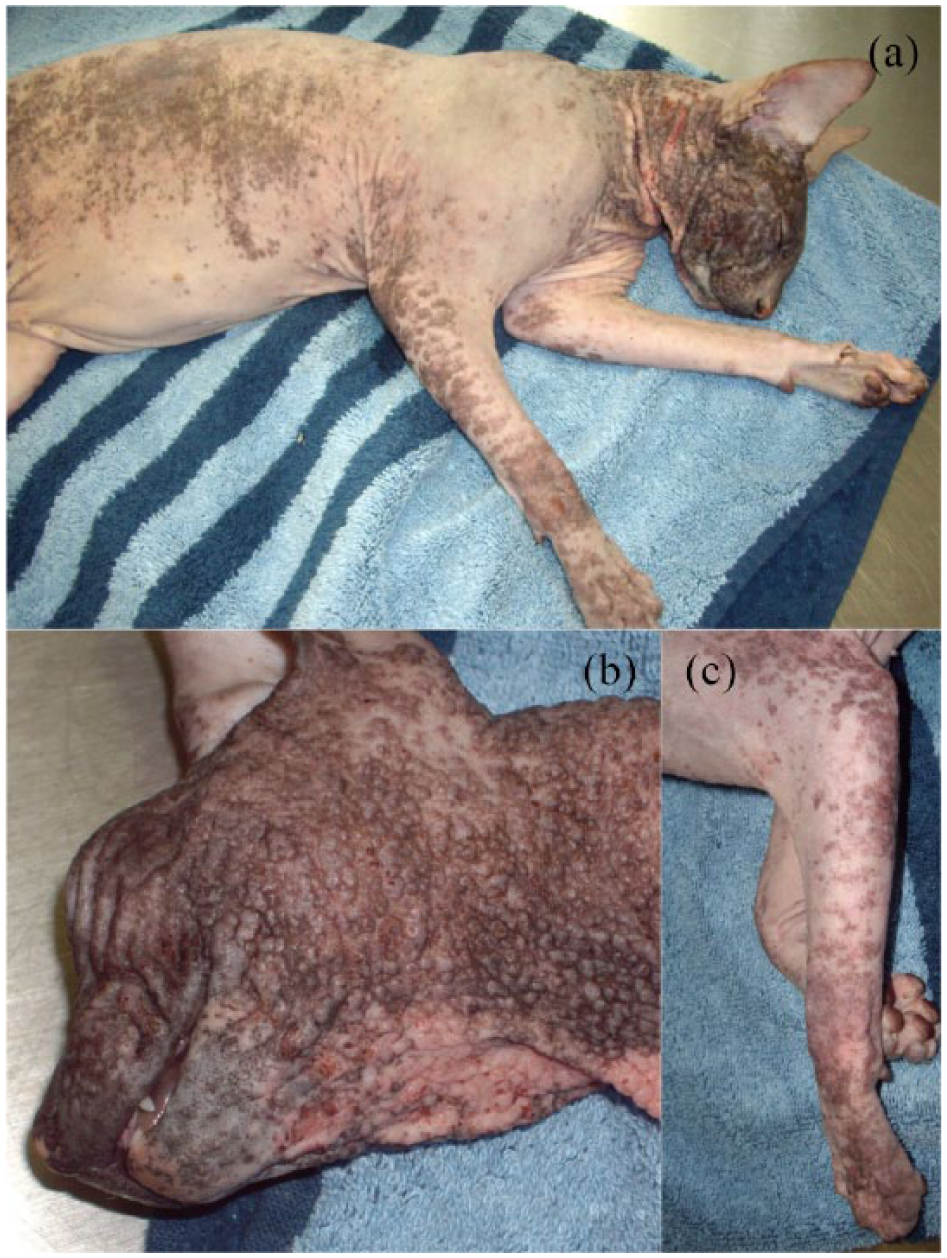
Case 12: classical (a) global aspect, (b) face and (c) forelimb of a Sphynx with chronic pigmented maculopapular cutaneous mastocytosis, previously called ‘urticaria pigmentosa’. Hyperpigmentation is associated with chronicity. Note the excoriation secondary to pruritus at the same location as the primary lesions
Pruritus was observed in 12/13 cats and subjectively estimated by the owners at a median of 4 on a scale of 0–5. The three clinical presentations had a median pruritus index of 3.25, 2.5 and 4.5, respectively (Table 1).
Interestingly, all of the six cats with polymorphic lesions resolved spontaneously, with a resolution time varying from a few weeks to 4 years. In contrast, none of the other cats had complete resolution and all of them needed long-term therapy to stabilise the lesions and/or pruritus (Table 1).
Case 8 was the only one with a clear bacterial overgrowth on acetate tape cytology. Antibiotic therapy (doxycycline 10 mg/kg q24h) resolved the bacterial overgrowth but not the other lesions, which needed additional treatments. For some cats, antihistamines (cetirizine 2.5 mg/cat q12h) were able to control the clinical signs within 1 month, with definitive cure or flares after cessation. Other cats required methylprednisolone (1 mg/kg q24h then tapered) and eventually ciclosporin (7 mg/kg q24h) to be stabilised (Figures 7 and 8). One cat (case 10), which showed good improvement with ciclosporin, also showed excellent improvements in terms of both lesions and pruritus with doxycycline (10 mg/kg q24h) without any sign of bacterial overgrowth prior to therapy but was then lost to follow-up. Three cases (cases 7, 11 and 13) did not respond to any treatment (alone or in combination), including masitinib at a dose of 12.5 mg/kg/day, and were then lost to follow-up after several months of trials (Table 2). Neither topical nor systemic essential fatty acid supplementation were tried.

Case 11: evolution of maculopapular lesions before and after 1 month of ciclosporin treatment in a Devon Rex (neck)

Case 11: evolution of maculopapular lesions before and after 1 month of ciclosporin treatment in a Devon Rex (ears)
Response to treatment and follow-up of 13 cases of feline cutaneous mastocytosis from 2003–2017
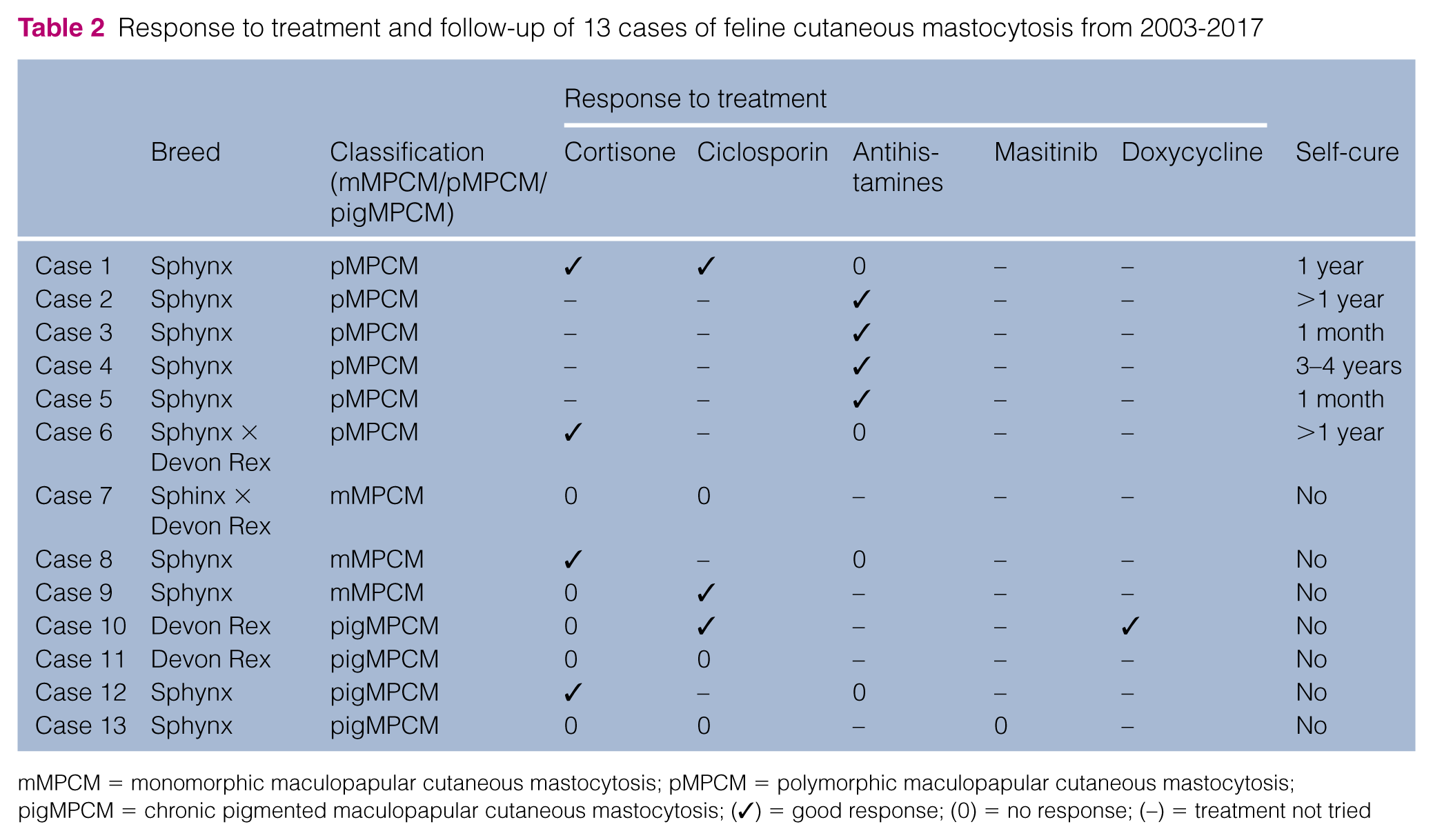
mMPCM = monomorphic maculopapular cutaneous mastocytosis; pMPCM = polymorphic maculopapular cutaneous mastocytosis; pigMPCM = chronic pigmented maculopapular cutaneous mastocytosis; (✓) = good response; (0) = no response; (–) = treatment not tried
Histopathological analysis was performed on eight cases (Table 1) when cytology was not informative. The inflammatory distribution of these lesions varied from interstitial to perifollicular or even nodular, multifocal to diffuse and from superficial to deep with inflammatory cell infiltrate composed predominantly of a mixture of eosinophils and MCs. The numbers of leukocytes varied from low to high, and varied both between cases and also between different biopsy sites within the same individual (Figure 9). In some, but not all, cases, the MCs formed aggregates (Figure 10). With the chronic progression of the dermatitis and secondary changes, neutrophils, macrophages, plasma cells and lymphocytes could also be observed, but no flame figure formation was seen. Ectatic lymphatic vessels containing protein-rich plasma and numerous neutrophils and eosinophils within hyperplastic arteries and veins were frequently seen (Figure 10). No difference was observed between the mPMCM and pPMCM clinical forms. The pigmented form showed an acanthosis and more severe leukocytic infiltrates with larger aggregates of MCs.

(a) Illustration of polymorphic maculopapular cutaneous mastocytosis (case 6) with a mild periannexial, superficial-to-deep inflammatory distribution and mild oedema. (b) More superficial, almost lichenoid, infiltrate (although other areas within the same case were more deeply infiltrated) in chronic pigmented maculopapular cutaneous mastocytosis (case 13). The epidermis shows more acanthosis than in the other forms (haematoxylin and eosin × 40)
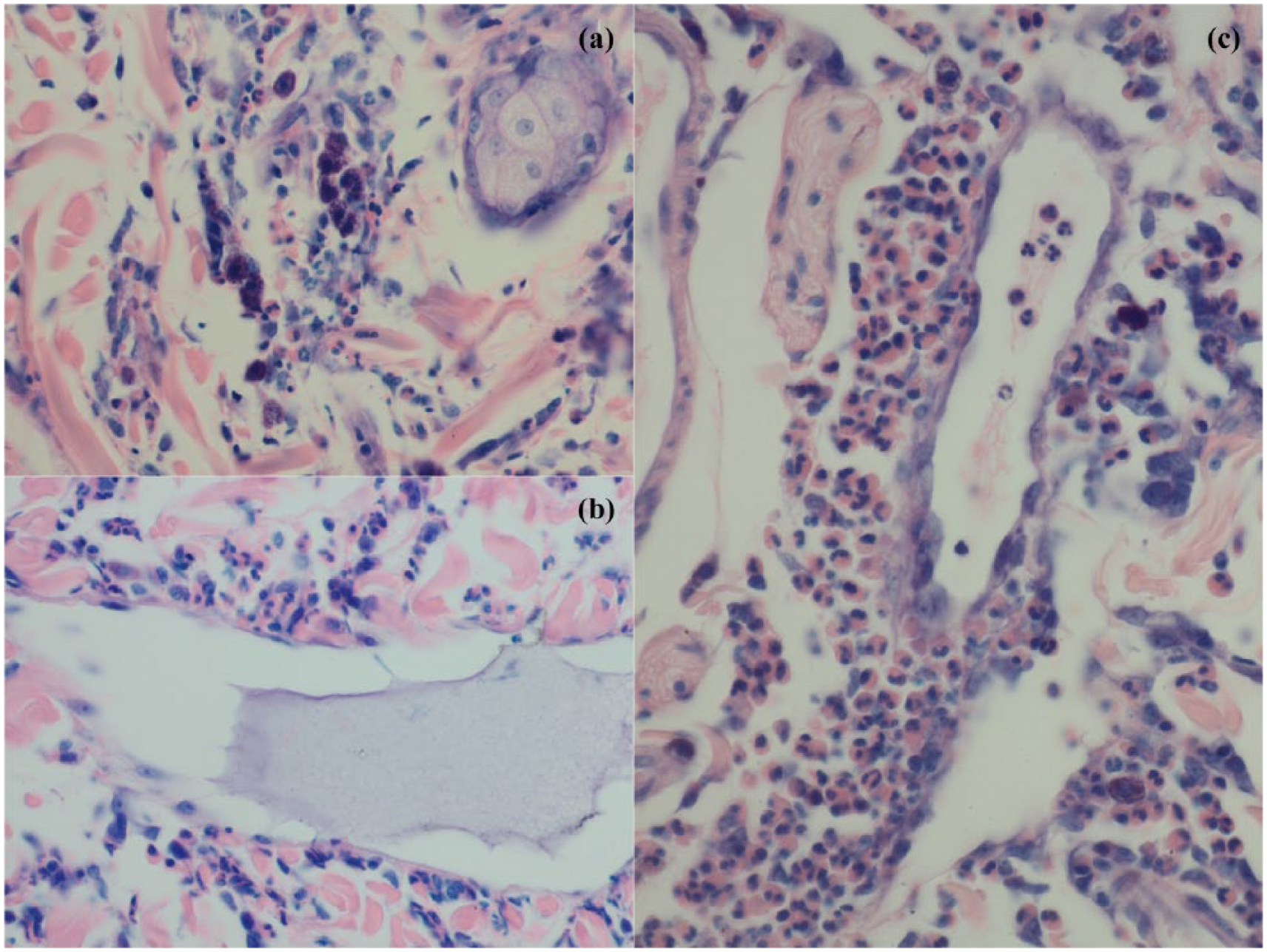
Case 3: (a) predominant eosinophilic infiltrate with mast cell aggregates (Giemsa × 400); (b) dilated lymphatic vessel with rich plasmatic fluid (haematoxylin and eosin × 400); (c) severe perivascular eosinophilic and mast cell infiltrate (Giemsa × 400)
Discussion
FeCM is a rare skin disorder characterised by a pathological infiltrate of increased numbers of MCs in the skin. Owing to its resemblance to the human condition, it is interesting to compare it with the classification and description of the human disease.8,11 –13
The classification of the disease in humans is traditionally divided into cutaneous and systemic forms (CM and SM). The systemic form is associated with a MC infiltrate in the bone marrow and/or other organs, with frequent cutaneous involvement. SM is mostly observed in adults, has a potentially poor prognosis and has to be differentiated from the MC sarcoma, which is characterised by solid tumour, mostly in the bone marrow. 14 In the veterinary literature, only one case of feline SM has been reported. The 15-year-old domestic mediumhair cat showed an onset of multiple skin lesions and, owing to a poor response to treatment, was euthanased. SM was then diagnosed at necropsy; involvement of the spleen, liver and bone marrow was observed. 15 It is thus important to always consider the possibility of underlying SM in cats presenting with CM.
In human CM (HuCM), the clinical signs vary widely according to the age of onset (childhood or adulthood) and from one patient to another. In some cases, the lesions potentially cover the entire body. 8 HuCM has recently been divided into three subforms: maculopapular CM (MPCM), diffuse CM (DCM) and cutaneous mastocytoma (for patients showing ⩽3 nodules). 8 MPCM, representing 90% of the human cases, is subdivided into the monomorphic (mMPCM) and polymorphic variants (pMPCM).8,11,16,17 The nodular form (mastocytoma) occurs usually at birth or during the first weeks of life, with a frequent spontaneous regression during the first years of life. Most cases of MPCM in children also regress around puberty. However, there is a risk of a systemic involvement.
DCM is a very unusual condition in humans, more frequently observed in neonates and infants, and consisting of generalised erythema, pachydermia and, frequently, blisters.11,13,15 –19
In adults, often presenting with mMPCM, the clinical signs are very typical: small, round, brown or red monomorphic lesions, previously named ‘urticaria pigmentosa’ (Figure 11). The lesions usually start on the thighs, axillae or lower trunk, and spread after several years throughout the rest of the body, except for the facial skin, which is typically spared. 8

Papules with hyperpigmentation in a classic case of human monomorphic maculopapular cutaneous mastocytosis. Courtesy of Dr MA Morren, Department of Dermatology UZ Leuven, Belgium. Verbal informed consent was obtained from the patient for their anonymised image to be published in this article
In children, pMPCM is more frequently observed, exhibiting different sizes of skin lesions (mostly large), with a brown or red colour and variable shapes (sharp, indistinct, elevated or flat; Figure 12). The type of the lesions can evolve, ranging from nodule to macule, usually flattening in time. The distribution of lesions is usually asymmetrical and involves the head, neck and extremities. A characteristic feature is the presence of brown lesions on the lateral parts of the forehead. 8 This form usually has a good prognosis, with a progressive spontaneous improvement often before puberty.
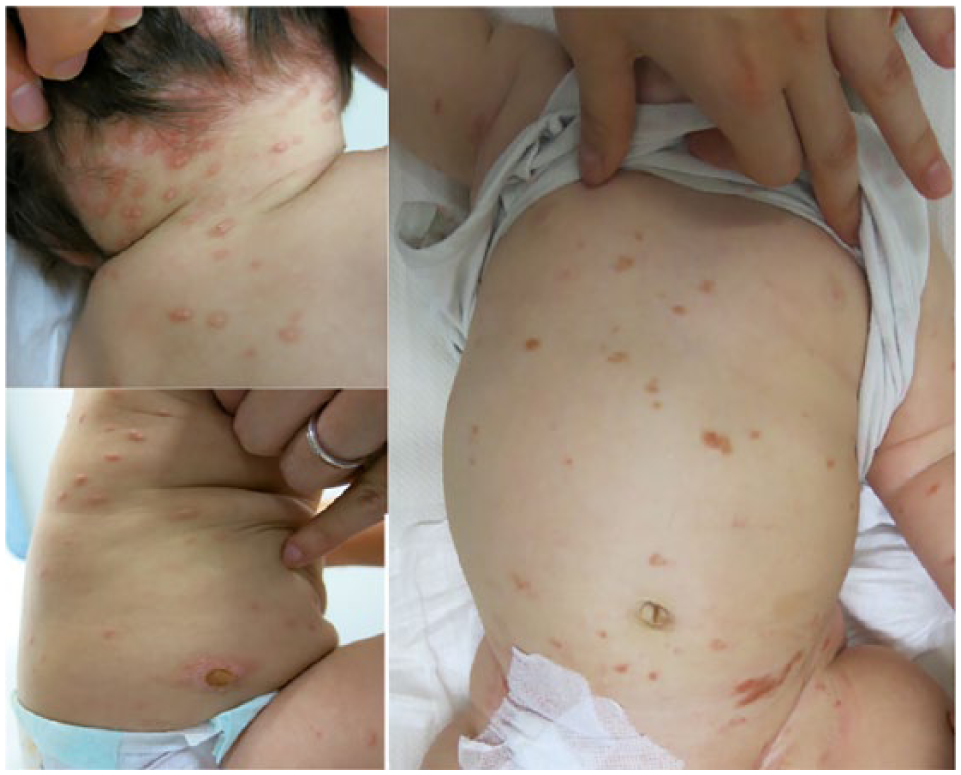
Papules, macules and nodules found in a paediatric form of polymorphic maculopapular cutaneous mastocytosis. Courtesy of Dr MA Morren, Department of Dermatology UZ Leuven, Belgium. Verbal informed consent was obtained from the patients’ parents/guardians for their anonymised images to be published in this article
All cats included in this study showed a maculopapular form of CM. Unlike the human cases, we never observed vesicles in any of case of feline MPCM. Three clinical presentations were observed (Table 3): (1) a dermatitis with large papular lesions, as well as wheals, mainly localised to the head, shoulders, ventral neck and axillae associated with moderate pruritus (this clinical form resembles the human pMPCM); (2) a small maculopapular erythematous dermatitis often associated with crusts and moderate pruritus, very similar to the human mMPCM, except for the lack of hyperpigmentation; (3) a highly pruritic, more chronic dermatitis similar to the human adult monomorphic subform, which was historically called ‘urticaria pigmentosa’ (although this term is no longer used nor recommended according to the latest definition of urticaria [a transient apparition of wheals and/or angioedema], 20 which does not correlate with the chronic history of the disease). A bilaterally symmetrical linear distribution of lesions along the flanks could be observed in some cats. We suggest the term of ‘pigmented MPCM’ or pigMPCM to describe this chronic feline subform.
Summary of the lesions, pruritus and prognosis observed in the different clinical forms of feline maculopapular cutaneous mastocytosis (MPCM)

In children, pruritus often occurs after the appearance of lesions, which are secondary to release of MC mediators. 6 The chronology of the clinical signs is hard to estimate in cats, although it seems that most of the cats had lesions before pruritus was observed by the owners, which is consistent with the human disease.
In humans, it appears that it is the chronic inflammation that causes the pigmentation, mostly in the monomorphic variant, although a link between the presence of MC and hyperpigmentation has been suggested, possibly owing to the secretion of stem cell factor by the MCs, stimulating the production of melanin by melanocytes.21,22 Feline pigMPCM could be a chronic form of mMPCM.1,3
To our knowledge, only one case of feline DCM has been reported. The cat was a 1-year-old domestic shorthair with pruritic papulocrustous lesions progressing from the head to the entire body. Involvement of the lymph nodes and absence of response to treatment led to euthanasia in that case. 23
In humans, diagnosis of MPCM is based on a combination of two major criteria: the presence of typical clinical signs and Darier’s sign. Skin biopsies are frequently required, but not mandatory, for the final diagnosis in human patients, as well as the absence of criteria indicating systemic involvement. 6 The biopsies provide histological evidence of focal MC infiltrates in the dermis (using tryptase or KIT immunohistochemical [IHC] staining techniques), or the detection of an activating KIT mutation (encoding c-Kit, a receptor tyrosine kinase for MC growth factor) by PCR in lesional skin tissue.6,24 In HuCM, the average number of MCs found is commonly increased in the lesional dermis, although there is an important variation of this number, potentially overlapping those in healthy skin. 8
Similarly, diagnosis of FeCM is based on typical clinical signs and exclusion of other dermatoses. Dermatophytosis, for example, has been reported in three cases that clinically mimicked MPCM but were diagnosed with histopathology. 25 In the same way, MC tumour (MCT) may initially present as papules but is typically not pruritic and found in older cats.26 –28 The useful Darier’s sign, observed in children, is most frequently negative in cats.3,4 In the cases described, biopsies were often refused by the owners, particularly for aesthetic reasons, as many of the affected cats were hairless breeds and biopsy would result in a visible scar. Histological findings in FeCM are variable in severity and aspect, but they all show an increased number of MC and eosinophils, sometimes associated with other types of leukocytes. Tryptase IHC has been validated to assess the presence of MC in feline tissues, as well as KIT IHC in feline MCT, although Giemsa staining remains the routine test used.29,30 Similarly, KIT mutations have been detected by PCR in feline MCT.31,32 Therefore, evaluation of aberrant KIT expression, as well as mutation, should be studied in FeCM. It is important to note that, as with human medicine, a diagnosis of FeCM cannot be made upon histology alone; indeed, MC and eosinophil counts can also be elevated in other pathologies such as allergic diseases. 33 The diagnosis should be made via a combination of typical clinical presentation in a breed-predisposed kitten or young cat, the exclusion of other dermatoses, cytological/histological assessments and absence of criteria indicating systemic involvement (eg, fatigue, gastrointestinal issues, abnormal blood chemistry results, etc). Hence, pathologists should be supplied with gross lesion descriptions and the clinical differentials.
A serum tryptase dosage is usually carried out for the classification of human SM; in HuCM, it ranges from normal to mildly increased. 34 To our knowledge, there is no study of feline serum tryptase levels assessing its value in the diagnosis of feline mastocytosis.
Paediatric mastocytosis is secondary to a mutation of KIT (exon 8, 9 or 17 – the most common being D816V or p.Val816Asp.). 35 Most often, this mutation is somatic; only in exceptional cases (2–4%) does it involves the germ line and a familial occurrence is present. In cats, the KIT mutations in MCT are distributed in exons 6, 8, 9 and 11, which correlates with the findings in childhood HuCM. 31 However, to our knowledge, these mutations have not been confirmed in FeCM. A genetic cause is also probable in cats as FeCM is mainly observed in the same breeds (Sphynx, Devon Rex).
In human medicine, there is no consensus regarding the treatment of CM. Indeed, as HuCM is most often a self-resolving disease treatment is mainly symptomatic, primarily aimed at controlling pruritus or blistering; H1 and H2 antihistamines are given orally in addition to systemic glucocorticoids, which are not frequently used, and topical cromolyn sodium. 13 In asymptomatic patients, antihistamines may be prescribed as prophylaxis. The patients are advised to avoid as much as possible any situation or agent able to provoke a reaction: sudden changes in temperature (bath, sports), ‘histamine-releasing’ food (nuts, strawberries, seafood, tomatoes, etc), ‘histamine-enriched’ food (canned food, wine, cheese, etc), drugs (morphine derivatives, antibiotics, anaesthetics, non-steroidal anti-inflammatory drugs, etc), and any bee sting or snake bite.
In cats, different therapies have been recommended with various results, such as systemic glucocorticoids, antibiotics (amoxicillin/clavulanic acid) and/or blackcurrant seed oil, and also antihistamines (hydroxyzine/ceterizine) in addition to shampoo or ciclosporin.1,3,36 In those reports, response to therapy ranged from good (remission), mild (relapses controlled with glucocorticoids) or absent (Table 4). In our study, there was no recommended drug associated with a better response. Antihistamines were efficient (4/13) but only in pMPCM cases. Steroids demonstrated a similar efficacy (4/13), as did ciclosporin (3/13). Doxycycline also gave good results in one cat (case 10), probably more because of its inhibitory effect on MC activation rather than its antibiotic properties, as no bacterial infection was found on the cat treated. 37 Owing to an effect on c-Kit, inhibitors of tyrosine kinases are, theoretically, a possible treatment. However, the efficacy of the different tyrosine kinase inhibitors is related to the specificity of the mutations. Indeed, masitinib has shown promising results in humans, whereas imatinib requires a KIT mutation analysis as D816V mutant MCs are resistant to the drug.38,39 In the one cat in which masitinib was trialled, it had no effect. KIT mutations were not assessed in any of our cases.
Review of 10 cases of feline maculopapular cutaneous mastocytosis previously described in literature

F = female; MN = male neutered; EFA = essential fatty acids
The clinical course, as well as the prognosis, of FeCM is as described in humans. In our 13 cases, it varied in severity from nearly ‘asymptomatic’ to highly aggressive. According to Hartmann et al, 8 the course of the disease in children is usually temporary, starting within the first 6 months of age, sometimes even at birth, and regressing during puberty in most cases. We show the same range of onset age (7 months), which is younger than previous reports (Table 4). Similar cases of spontaneous regression were observed in cats with a disease occurring in kitten to young adults and associated with good prognosis.1,3,4 Here, the six cats with pMPCM had spontaneous regression. This is similar to the human pMPCM, which seems to have a better prognosis. 40 Nevertheless, in the remainder of the cats, the disease was not cured and progressed to a chronic dermatitis (Table 3). The authors did not observe any progression to systemic involvement in any of the 13 cases.
Conclusions
In many aspects, FeCM is similar to the human condition. Using the current human classification of the disease, we recommend using the term MPCM in order to distinguish it from DCM. According to the lesions observed in cats, MPCM should be divided into three subforms: pMPCM, mMPCM and pigMPCM (previously known as urticaria pigmentosa). Further larger studies should be carried out to evaluate this classification’s appropriateness, mainly to confirm the good prognosis of pMPCM, as well as study the underlying genetics and diagnostic methods of the disease.
Footnotes
Acknowledgements
The authors would like to thank Dr O Smith for assistance with English proofreading.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
