Abstract
Objectives
Feline injection site sarcoma (FISS) is a rapid growing locally aggressive tumor with a low metastatic rate. Its histologic features are clearly defined, but there are few studies regarding its immunohistochemical characteristics. The present study investigated the immunohistochemical characteristics of 21 cases of FISS.
Methods
FISSs from 12 male and nine female cats, 20 mixed-breed and one Siamese, were included in the study. After histopathological diagnosis, additional histologic sections were immunostained for vimentin, cytokeratin, desmin, S100 protein, viral feline leukemia virus (FeLV) particles, cyclooxygenase 2 (COX-2) and c-KIT. Positive and negative controls were adopted accordingly. Immunostainings were classified as positive or negative according to the number of positive cells from a total of 1000 cells per tumor section.
Results
Histopathologic diagnosis of the tumors revealed 18 (85.7%) fibrosarcomas and three (14.3%) other sarcomas; four fibrosarcomas (22.2%) were grade III, five (27.8%) were grade II and nine (50.0%) were grade I. Two sarcomas were grade III and one was grade II. Seventeen (81%) tumors were negative for desmin. All samples were positive for vimentin. Twenty tumors (95.2%) were positive for S-100 protein. Positivity for c-KIT was observed in four (19%) samples; COX-2 was positive in 13 (61.9%) and FeLV viral particles were positive in nine (42.9%) FISSs.
Conclusions and relevance
Immunohistochemical findings of FISSs revealed positive immunostainings for desmin, vimentin, S-100 protein, c-KIT, COX-2 and FeLV viral particles.
Introduction
Feline injection site sarcoma (FISS) is a rapidly growing neoplasm with a variable, often prolonged, latency period and aggressive local behavior, despite low metastatic potential. 1
From the histologic perspective, FISS is a mesenchymal neoplasm with different histologic types. Undifferentiated high-grade pleomorphic sarcoma, osteosarcoma, chondrosarcoma and rhabdomyosarcoma have been described; however, fibrosarcoma is the most commonly reported.2–4
Inflammatory and immunologic reactions at injection sites maybe a predisposing factor for the development of these soft tissue sarcomas. 2 The inflammatory infiltrate includes macrophages often with cytoplasmic material, giant cells, lymphocytes and mixed neutrophils and eosinophils. Multiple cytokines, growth factors and mutations in tumor suppressor genes have been identified in these tumors. 5
Despite well-established histologic characteristics, molecular expression of FISS requires further investigation. Immunohistochemistry is widely employed in research settings and is increasingly used for diagnostic purposes and therapeutic biomarker assessment. 6 Immunohistochemical characterization of neoplasms may bring significant contributions to differential diagnosis, prognosis, overall survival and support therapeutic decisions.
This study aimed to investigate immunohistochemical expression of vimentin, cytokeratin, desmin, S100 protein, feline leukemia virus (FeLV) viral particles, cyclooxygenase 2 (COX-2) and c-KIT in FISSs.
Materials and methods
Twenty-one sarcomas, characterized as injection site tumors based on location, history and histopathologic findings, obtained from male and female cats referred to the surgery department of the School of Veterinary Medicine and Animal Science, University of São Paulo (SVMAS-USP) were used in this study. The research protocol was approved by the bioethics committee (protocol 2126/2011) of the SVMAS-USP. Free and informed owner consent was obtained in all cases.
Data regarding disease progression, tumor gross appearance (affected site, consistency, macroscopic invasion of surrounding structures and surface characteristics – alopecic, ulcerated vs non-ulcerated) and tomographic features were extracted from patient history, as well as physical and tomographic examination. Only patients with a history of previous vaccination in the tumor location were included.
Neoplasms were surgically resected; surgical specimens were then fixed in 10% formalin and sent to the Animal Pathology Service of the Veterinary Hospital of the SVMAS-USP for diagnosis. Representative slices of the neoplasms were routinely processed for paraffin embedding. The 5 µm sections were then stained with hematoxylin and eosin and observed with a Nikon microscope. Histologic analysis and grading were based on the classifications of the World Health Organization. 7 Immunohistochemistry was carried out at the Laboratory of Experimental and Comparative Oncology, SVMAS-USP, Department of Pathology. Immunostained cells were manually counted under light microscopy (× 40 magnification) using Image Pro Plus software (Image Pro Plus 4.5; Media Cybernetics).
Additional 5 µm histologic sections were obtained in silanized slides from all tumors, which were submitted to immunostaining to detect vimentin, cytokeratin, desmin, S100 protein, viral FeLV particles, COX-2 and c-KIT expression in FISSs. Tissue sections were dewaxed in xylene, dehydrated in graded alcohols and washed in 0.01 M phosphate-buffered saline (PBS; pH 7.2 FISS). Tissue sections were dewaxed in xylene, dehydrated in graded alcohols and washed in 0.01 M for 30 mins. Before incubation with antibodies, antigen recovery was performed in a microwave heating at maximum power for three cycles of 5 mins in citrate buffer (pH 6) at 80°C. Tumor sections were incubated overnight with the respective antibodies, diluted as described in Table 1.
Primary antibodies, sources and dilutions

COX-2 = cyclooxygenase 2; FeLV = feline leukemia virus
Immunohistochemistry was performed using the streptavidin–biotin–peroxidase method (LSAB + System-HRP; Dako), and the antibodies are listed in Table 1. A cocktail of biotinylated anti-mouse, anti-rabbit and anti-goat immunoglobulins, 1 diluted in PBS, was used as a secondary antibody. This mixture was applied and then tissue sections were incubated for 30 mins. After washing in PBS, the tissue sections were incubated for 30 mins in streptavidin-conjugated horseradish peroxidase in Tris–HCl buffer containing 0.015% sodium azide. 2 To identify sites of primary antibody binding, 3,3 o-diaminobenzidine tetrahydrochloride dissolved in PBS, with 0.5% of H2O2, was used as a chromogen, and hematoxylin was used for counterstaining. Expressions of vimentin, cytokeratin (DAKO Z0622, wide spectrum), desmin, S100 protein, viral FeLV particles, COX-2 and c-KIT in FISS were assessed by determining the percentage of immunoreactive cells from a total of 1000 cells, as previously described. 6 Immunoreactivity was graded as follows: positive staining was defined as >10% immunoreactive cells; negative staining was defined as <10% immunoreactive cells or an absence of detectable staining. 8
Positive controls for the immunostainings were found in the surrounding tissues for vimentin (mesenchymal cells), cytokeratin (epithelial cells, like epidermis, sebaceous or apocrine glands), desmin (smooth muscle cells from arteries) and S100 protein (melanocytes or pericytes). For COX-2, a positive canine mammary tumor was used as the control, and for c-KIT a positive mast cell tumor (MCT) was used as the control. Negative controls were obtained by omitting the primary antibody in additional sections of each of the 21 FISSs. The criteria for selecting the areas was the presence of areas with the best representative sample of the tumor. Two observers were involved in the assessment. The discrepancies were analyzed by further assessing reports and reviewing slides to look for sources of error such as interpretation of morphology or immunohistochemistry. The morphologic interpretation and/or panel of immunohistochemical stains were interpreted and compared by the referring pathologists and the final pathologist.
Results
There were 12 male and nine female cats (one Siamese, 20 mixed breed). Mean weight of the cats was 5.28 kg and mean age was 9.4 years. CT FISS volume ranged from 1.15–355.6 cm3 (mean volume 115.34 cm3); CT assessment revealed neoplastic infiltration of adjacent tissues in 55% of cases. Clinical and physical characteristics of 21 cases of FISSs are described in Table 2.
Clinical and physical presentation of 21 cases of feline injection site sarcomas

M = male; TA = thoracoabdominal; X = alive at the end of study; HL = hindlimb; NI = not informed; F = female; C/IS = cervical/interscapular; (−) = absence; (+) = presence
Histological grades I, II or III were attributed to nine (50.0%), five (27.8%) and four (22.2%) fibrosarcomas, respectively. Two sarcomas were grade III and one was grade II. Neoplasms were classified either as fibrosarcomas (18/21; 85.7%) or sarcomas (3/21; 14.3%).
Immunohistochemistry revealed 17 (80.9%) desmin-negative tumors. Vimentin was positive in all samples. Twenty tumors (95.2%) were positive for S-100 protein. Positivity for c-KIT was observed in four (19.0%) samples; COX-2 was positive in 13 (61.9%) and FeLV viral particles were positive in nine (42.9%) FISSs (Figures 1 and 2).

Photomicrographs of immunohistochemical expression of vimentin, desmin and c-KIT in feline injection site sarcomas. (a,b) Positive immunostaining for vimentin (× 400 magnification); (c,d) negative (× 40 magnification) and positive immunostaining for desmin; (e–g) positive immunostaining for c-KIT (× 400); (h) positive endothelial cells immunostaining for c-KIT (× 400 magnification)
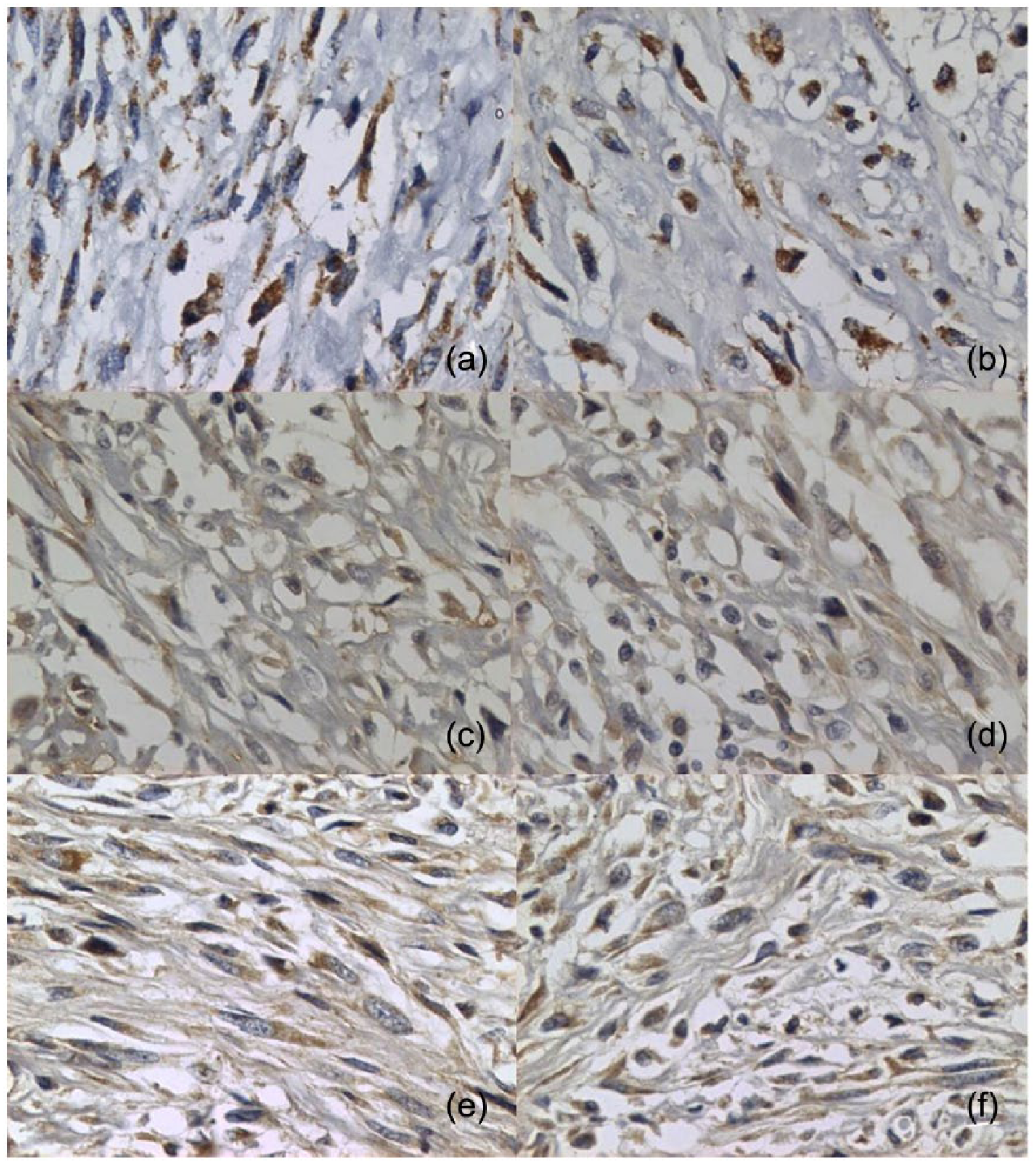
Photomicrographs of immunohistochemical expression of cyclooxygenase 2 (COX-2), feline leukemia virus (FeLV) and S-100 protein in feline injection site sarcomas. (a,b) Positive immunostaining for COX-2; (c,d) positive immunostaining for FeLV; (e,f) positive immunostaining for S-100. × 400 magnification
Discussion
FISSs include several histologic types, such as fibrosarcoma, rhabdomyosarcoma, extraskeletal osteosarcoma, chondrosarcoma and histiocytic sarcoma, with aggressive behavior and invasion of adjacent tissues in all cases. 5 Neoplasms included in this study were either fibrosarcomas or sarcomas; lack of some histological types may have been the consequence of insufficient material owing to the small number of tumors.
FISS histology has been widely investigated; a peripheral inflammatory infiltrate composed of lymphocytes and macrophages, granulation tissue and multinucleated giant cells have been typically described. 7 Malignant cytologic features such as high mitotic index and cell pleomorphism are more commonly seen in these neoplasms vs sarcomas unrelated to injection. 1
Well-described histologic findings of FISSs, supported by a central area of necrosis and the presence of inflammatory cells, including multi-nucleated giant cells, which have phagocytized a grayish-blue material consistent with the aluminum-based vaccine adjuvant in some cases,2,9 provide a basis for differential diagnosis with other types of sarcomas. In contrast, the molecular characteristics of FISSs have not been widely investigated. Immunohistochemical features can provide additional information regarding tumor biology to support prognosis and decision-making on target therapy protocols.
A study describing the immunohistochemical characteristics of FISSs reported positive immunostaining for vimentin in most tumor samples (4/5). 2 A second study of FISSs reported that all fibrosarcomas were immunoreactive for vimentin, and 28/44 (63.6%) were reactive for alpha-smooth muscle actin (SMA). 10 Anti-vimentin antibodies are known to be specific for mesenchymal cell filament protein. 11 According to previous studies,2,10 all FISSs in this study were vimentin positive. Immunoreactivity for vimentin in FISSs is helpful to define their origin and detail of morphologic characterization.
Desmin and SMA staining of neoplastic FISS cells, particularly of peripheral cells, has been described. 12 Expression of desmin in FISSs was less common than smooth muscle actin. 13 Positive staining for vimentin associated with immunoreactivity to one or more muscle markers (eg, desmin, SMA) is consistent with myofibroblast reactivity. 2 Positive staining for desmin was observed in four (19.0%) fibrosarcomas in our study, similar to the proportion of expression described by Aberdein et al. 13 Given that all desmin-positive stainings were grade I or II and desmin was absent in myoepithelial cells, 14 we can exclude staining of undifferentiated cells/myoepithelial cells, respectively. Thus, desmin expression associated with vimentin in these tumors suggest reactivity of myofibroblasts, reflecting a continuous inflammatory response that characterizes these neoplasms. Although implicated in various processes, including wound healing, proliferative disorders and the desmoplastic response to certain neoplasms, the origin and exact role of these cells remain unclear.2,9,10
S-100 protein is commonly expressed by a variety of cell types; however, the S-100B isotype is more abundant in glial cells of the central and peripheral nervous system, melanocytes, chondrocytes and adipocytes. 15 Most specimens in our study (20/21; 95.2%) were S-100 protein-positive. Given the low specificity of this protein, positivity was thought to reflect the presence of adipocytes rather than neural sheath origin, as immunostaining suggests.
The KIT receptor (CD117) is a transmembrane protein with tyrosine kinase activity encoded by the oncogene c-KIT. c-KIT gene expression has been described in several cell types, including Cajal, mast cells, interstitial 16 and endothelial cells. 17 This receptor is thought to be associated with immature cell growth and differentiation. KIT protein expression has been reported in neoplastic cells of canine gastrointestinal stromal tumors and feline MCTs. 18
Mutations in the proto-oncogene c-KIT occur frequently in naturally developing MCTs in dogs and aggressive mastocytosis in humans, causing functional KIT dysregulation. 18 Smith et al reported KIT expression in 26% of soft tissue sarcomas in cats. 19 c-KIT labeling was documented in 19% of neoplasms in our study. Endothelial cell c-KIT expression has not been systematically evaluated in feline cancer. In the present study, endothelial cell c-KIT expression was observed in some FISSs. c-KIT-positive endothelial cells have not been considered for immunohistochemistry classification of the tumors. Endothelial and tumor c-KIT expression in FISSs scan suggest associations between c-KIT and proliferation and invasiveness. Together, these findings suggest that FISSs may be considered c-KIT-positive tissues, with potential contributions to FISS diagnosis, prognosis and therapeutic decision-making.
COX-2 expression is related to tumor growth and angiogenesis, and has been reported in several human neoplasms. 20 With regard to animal medicine, COX-2 expression has been demonstrated in intestinal carcinoma, 21 transitional cell carcinoma of the urinary bladder, 22 squamous cell carcinoma, 23 mammary and renal carcinoma,24,25 and FISS. 26 COX-2 expression is induced by inflammatory stimuli and reflects cancer development and progression. 27 COX-2 staining in 61.9% of samples in this study and in 97% of samples in the study of Magi et al suggest a potential role of this enzyme in FISS progression. 26 The expression of COX-2 in FISSs may contribute to the understanding of tumor behavior and may become a potential target therapy to treat FISSs with a non-steroidal anti-inflammatory drug.
Fibrosarcomas that are associated with FeLV are caused by FeSV, a recombinant virus that develops de novo in FeLV-A-infected cats by recombination of the FeLV-A genome with cellular oncogenes. Fibrosarcomas caused by FeSV tend to grow rapidly, often with multiple cutaneous or subcutaneous nodules that are locally invasive and metastasize to the lung and other sites. Solitary fibrosarcomas in older cats are not caused by FeSV. They are usually classified as FISSs caused by the granulomatous inflammatory reaction at the injection site, commonly occurring after inoculation of adjuvant-containing vaccines. 28
Kidney et al failed to demonstrate endogenous FeLV (enFeLV) virus RNA content in FISSs; 29 therefore, their study provided no basis for involvement of enFeLV replication or expression to FISS development. The study of Deim et al involving aluminum-induced FISS in two cats reported that one of the animals was positive for FeLV. 30 Positive FeLV viral particles staining was documented in 42.9% of samples in our study. Despite this, the cats with nine virus-positive tumors had not been FeLV vaccinated and/or were FeLV positive.
enFeLV sequences are found in the genomes of domestic cats. 31 Although transcription and translation of enFeLV proviruses were detected in various tissues and cell lines, no infectious viruses are produced, owing to mutations within essential parts of the viral genome. 32 Cats with high enFeLV loads have transiently higher proviral, plasma viral and FeLV p27 loads than cats with low enFeLV loads. 33 The transcription of enFeLV has been observed in feline lymphomas. 34 Kidney et al compared enFeLV content in feline vaccine and non-vaccine-site-associated sarcomas by use of reverse transcriptase PCR, but the amount of enFeLV RNA within the long terminal repeat and env gene was not significantly different between the two groups. 29 Our study presented positive staining immunohistochemistry for FeLV in FISS. Thus, we speculate the potential impact of enFeLV on the formation of FISSs. Therefore, different methods to detect the provirus (FeLV DNA) and the possible mechanisms that could have induced malignant transformation into FISSs are warranted. Owing to the laborious and/or cost-intensive character of most of these methods, they were not suitable for this study.
The limitations of this study are largely related to the small number of cases included. A larger number of cases would enable statistical tests to be performed to further investigate associations of immunopositivity for different markers with different tumors and histological grade. Previous studies have described the immunohistochemical characteristics of FISSs; therefore, to the best of our knowledge, this is the first report to show FeLV viral particle immunohistochemistry expression in FISS, suggesting the possible involvement of enFeLV and FISS development. Owing to the small number of animals, further studies are warranted to clarify the immunohistochemical characteristics of FISSs and provide information for the diagnosis and treatment. Additionally, different methods to detect the proviral (FeLV DNA) and the possible mechanisms that could have induced malignant transformation into FISS are warranted.
Conclusions
Some FISSs showed immunohistochemical staining for vimentin, desmin, S100 protein, viral FeLV particles, COX-2 and c-KIT. Overall, the immunostaining results can be useful for establishing prognosis and therapeutic approaches for this tumor type.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by grants from Fundação de Amparo a Pesquisa do Estado de São Paulo (FAPESP) for which the authors are truly grateful
