Abstract
Objectives
The objective of this study was to evaluate the effect of different needle sizes used to obtain blood via jugular venipuncture in cats on routine measures of hemostasis.
Methods
This was a prospective, observational, randomized, clinical study carried out at a university teaching hospital. Twenty healthy, client-owned cats were used. Each cat had blood collected via direct venipuncture from both jugular veins. Sampling of the right and left jugular vein was randomized to be collected with either a 22 G or a 25 G needle, respectively, and routine coagulation variables and platelet count were performed on all samples. Values were analyzed for differences in needle size, and site of sample collection.
Results
There was no difference between the two needle gauges in activated partial thromboplastin time, platelet count, fibrinogen degradation products, or fibrinogen, or between sampling from the left and right jugular vein. Prothrombin time (PT) was significantly higher when drawn from a 25 G needle (11.7 s) compared with a 22 G needle (11.4 s) (P = 0.01), but not different in left vs right jugular vein samples. Bland–Altman analysis of PT comparing for 25 G minus 22 G needle vs the average, calculated a mean bias (95% limits of agreement) of 0.49 s (–1.4 s to 2.4 s).
Conclusions and relevance
This study of 20 healthy cats found that the use of either a 25 G or 22 G needle for jugular venipuncture did not introduce any clinically meaningful difference in routine coagulation variables or platelet count.
Introduction
Acquired and inherited bleeding disorders are often found in cats. 1 Diagnostic evaluation of bleeding disorders in cats would commonly include platelet count, prothrombin time (PT), activated partial thromboplastin time (aPTT), and measurements of fibrinogen and fibrinogen degradation products (FDPs). 2 These tests have more potential pre-analytical variables than routine hematology and biochemistry testing, owing to the need to inhibit and later reactivate clot formation.
A smaller diameter needle exerts higher shear forces as blood is collected, and high shear forces can activate both platelets and coagulation factors; as a consequence, the result of coagulation variables and platelet count may be altered.3,4 The risk of post-venipuncture hemorrhage increases as the needle size increases in coagulopathic patients, 5 and the ideal situation would be to use the smallest needle size that does not affect coagulation testing. In humans, platelet count but not PT or aPTT has been shown to differ with varying needle sizes. 6 In dogs, no differences were found in those variables when comparing 21, 23 and 25 G needles. 7 The recommended needle gauge used for venipuncture in human medicine is 21 G, when possible. 6 There are no evidence-based recommendations in feline medicine, but there have been suggestions in the literature to use 21 G needles.2,8,9
Feline platelets have greater aggregability in response to shear stress than human and canine platelets, and it is unknown how various needle sizes will affect tests of hemostasis in cats.10,11 The objective of this study was to evaluate the effect of needle sizes commonly used to obtain blood via venipuncture in cats on routine measures of hemostasis. We hypothesized that the samples drawn through 25 G needles would have a lower platelet count, shorter PT and aPTT times, and higher fibrinogen and FDPs than samples drawn through the larger 22 G needles.
Materials and methods
To determine the number of cats needed, a power analysis was performed using platelet count, PT and aPTT to detect a 10% difference for coagulation panel values and a 15% difference in platelet count with an alpha of 0.05 and a power of 80%. Power analysis suggested that 20 cats needed to be included in the study. Twenty cats, weighing >4 kg, and deemed healthy on the basis of their owner reporting no medical problems, normal physical examination and unremarkable routine complete blood count and biochemistry panel were recruited and enrolled in the study. The origin of the cats was the community, with volunteers solicited from the veterinary school population (students, residents, staff and faculty). The study was approved by the Institutional Animal Care and Use Committee at the University of California, Davis, and inclusion of an animal in the study was dependent upon receiving the owner’s written consent.
Each cat had two samples collected, one from the right and one from the left jugular vein. The order of needle gauge used for collection was randomized, and venipuncture from the right jugular vein was always performed first. The venipuncture sample had to be a clean stick, as defined as no more than one redirection of the needle once inserted through the skin in order to puncture the jugular vein and obtain blood. If a clean stick could not be achieved, all samples were discarded, and repeat sampling was not attempted in that cat for 7 days. If an adequate blood sample could not be obtained on the second attempt, no further attempts were tried for that cat, and a new volunteer was located.
For the first blood collection, a 6 ml plastic syringe and needle was used, and 6 ml blood was obtained using the needle gauge randomized as #1 (either 25 G [Monoject Covidien 25 G × 5/8”] or 22 G [Monoject, Covidien 22 G × 1”]). Immediately post-collection, the needle was removed from the syringe, the rubber stopper of the blood tubes removed and 1.8 ml blood placed into a 3.2% sodium citrate tube (Vacuette 3.2%; Greiner Bio-One) for analysis of PT, aPTT, fibrinogen and FDPs (Stago STA Compact Coagulation Analyzer; Diagnostica Stago). The tubes were stored on ice during transit and analyzed immediately. The remaining blood was placed in an EDTA-containing tube (Covidien EDTA [K3] 7.5%) for a complete blood count, including a platelet count (Siemens Bayer Advia 120 Hematology System), and a serum tube (Covidien) for biochemistry analysis (Roche Cobas c501 Analyzer; Roche Diagnostics). The tubes were inverted 10 times for proper mixing of the blood and anticoagulant. Samples were submitted to the University of California, Davis William R Pritchard Veterinary Medical Teaching Hospital clinical laboratory for analysis. Blood smears were evaluated by light microscopy for platelet clumps. The venipuncture site had manual pressure placed for 30 s or until hemostasis was achieved.
For the second blood collection, 4 ml of blood was obtained with a 6 ml plastic syringe and placed into the sodium citrate and EDTA-containing tubes as described above, using the alternate-sized needle. A chemistry panel was only run for the first blood draw, otherwise the exact same tests were performed. Both samples from each patient were obtained within 5 mins of each other.
Data were assessed for normality by the Shapiro–Wilk normality test. Normally distributed data are reported as mean ± SD and non-normally distributed data reported as median and interquartile range (IQR). Normally distributed data were compared using a paired t-test, and non-normally distributed data were compared via Wilcoxon matched-pairs signed rank test. The influence of needle gauge size (22 G vs 25 G) and first vs second jugular venipuncture were analyzed as stated above. The proportion of samples with platelet clumps was compared with Fisher’s exact test. A P value <0.05 was considered significant. Agreement between measurement techniques was evaluated by Bland–Altman analysis with the difference vs average used to calculate the bias of 25 G and 22 G needles or right vs left jugular venipuncture if statistical significance on univariate analysis was found.
Results
Thirty cats were enrolled in the study. Ten cats were excluded owing to an inability to draw both samples in sufficient volumes. The median age was 5.4 years (range 1–15 years) and median body weight was 5.1 kg (range 4.1–8.1 kg). There were 12 castrated males and eight spayed females.
On manual microscopic evaluation of blood smears, three (15%) samples drawn with a 22 G needle showed platelet clumping, whereas six (30%) samples drawn with a 25 G needle showed clumping (P = 0.45). The distribution of platelet counts, when all samples that were not clumped were evaluated, is shown in Figure 1. When automated platelet counts were available for paired samples, the mean ± SD platelets were 278 ± 100 × 1000/μl and 288 ± 87 × 1000/μl for 25 G and 22 G needles (n = 13), respectively (P = 0.15), and 290 ± 91 and 277 ± 97 platelets × 1000/μl for right and left jugular venipuncture (n = 13), respectively (P = 0.06).

Scatter plot of automated platelet count when clumping was not present for the differing needle sizes used for collection and venipuncture site. The bar represents the mean value
The median for PT was 11.7 s (IQR 11.3–12.2 s) for 25 G needles and 11.4 s (IQR 11.0–12.1 s) for 22 G needles (P = 0.01). The median for PT was 11.6 s (IQR 11.1–12.1 s) for right jugular venipuncture and 11.6 s (IQR 11.1–12.2 s) for left jugular venipuncture (P = 0.33). The median aPTT was 12.7 s (IQR 12.0–13.7 s) for 25 G needles and 13.5 s (IQR 12.5–14.0 s) for 22 G needles (P = 0.06). The median for aPTT was 12.6 s (IQR 11.9–13.8 s) for right jugular venipuncture and 13.6 s (IQR 12.6–14.0 s) for left jugular venipuncture (P = 0.44).
For fibrinogen, the mean ± SD value was 130 ± 34 mg/dl and 134 ± 23 mg/dl for 25 G and 22 G needles, respectively (P = 0.35). The median for fibrinogen was 138 mg/dl (IQR 115–156 mg/dl) and 137 mg/dl (IQR 113–150 mg/dl) for right and left jugular venipuncture, respectively (P = 0.40) All FDP values reported were <5 μg/ml.
Bland–Altman analysis comparing the difference of 25 G minus 22 G needle vs the average calculated a mean bias for PT of 0.49 s (95% confidence interval [CI] –1.4 to 2.4s), as shown in Figure 2(a), for aPTT of –0.42 s (95% CI –2.7 to 1.8s) (Figure 2b), for platelet count of 9.9 × 1000/μl platelets (95% CI –55 to 33 100/μl) (Figure 2c) and for fibrinogen of –4.1 mg/dl (95% CI –42 to 34 mg/dl) (Figure 2d). Bland–Altman analysis was not performed for FDP as all values were <5 μg/ml.
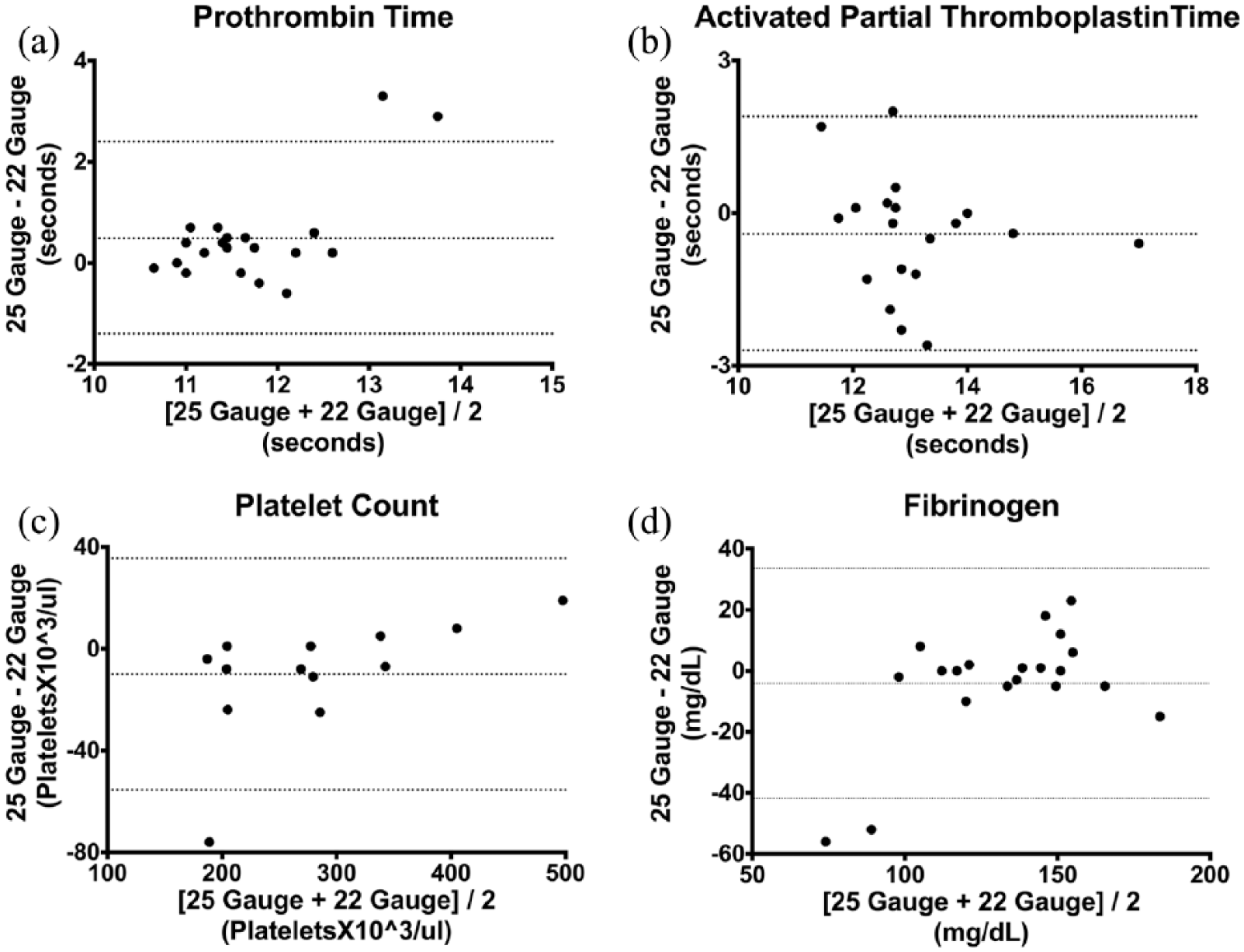
Bland–Altman plot of (a) prothrombin time, (b) activated partial thromboplastin time, (c) platelet count and (d) fibrinogen, comparing 25 G and 22 G needles. The horizontal dashed lines represent the mean bias and 95% confidence interval
Discussion
We found no difference between the two needle gauges used for sampling, or sampling from the left vs right jugular vein, for platelet count, proportion of samples with platelet clumping, aPTT, FDPs or fibrinogen. PT was statistically significantly longer when drawn from a 25 G needle compared with a 22 G needle, yet this value did not appear to be clinically significant on Bland–Altman analysis. Additionally, the mean biases for aPTT, platelet count and fibrinogen were not considered clinically significant either when evaluated by Bland–Altman analysis.
No differences were found between the two needle gauges for the number of samples with platelet clumps or for platelet numbers. This is in contrast with a study in humans, where platelet counts were found to be higher when blood was drawn with a 21 G needle than when a 23 G or 25 G needle was used. 6 The difference in platelet count identified in the human study was not considered clinically significant, as Bland–Altman analysis found the mean bias for platelet count to be <6000/μl. As only platelet count and number of samples with platelet clumps were assessed, we cannot rule out that there would be a difference in platelet activation associated with different needle sizes that was not identified. More sensitive methods for testing platelet function and activation were beyond the scope of this study.
To our knowledge, no studies in other species have found any difference in PT on blood drawn with different needle sizes,6,7 and the finding that PT is longer with smaller gauge needles is unexpected. In theory, using a smaller-diameter needle requires a higher vacuum, which will exert higher shear forces on the blood, and more activation of platelets and coagulation factors, potentially leading to a shorter PT.3,4 There are multiple considerations for this unexpected finding.
One possible explanation could be that the 22 G needle was longer (1 inch vs the 5/8 inch 25 G needle), resulting in the blood being drawn through a longer needle being exposed to a larger surface area, causing more activation of coagulation. However, the larger size needle would also be expected to allow for more rapid collection of blood, so the surface exposure time should be lower in the 22 G needle group. Additionally, as this result was found in the PT, and not the aPTT values, this explanation is unlikely.
A second possible explanation is that this is a false-positive finding (type I statistical error), meaning that the null hypothesis (that there is no difference) was inappropriately rejected. There is some evidence for this possibility in that there were two values in the Bland–Altman analysis that had 25 G–22 G needle differences of approximately 3 s. If these outliers (greater than 1.5× IQR) were removed from analysis there would no longer be a statistically significant difference. As the data set was small, we could not confidently identify outliers, and the whole data set was presented, but in our opinion, this is the most likely explanation for this finding.
A Bland–Altman analysis of PT comparing for 25 G minus 22 G needle vs the average calculated a mean bias of 0.49s (95% –1.4 to 2.4). This suggests that there would be no clinical relevance to the different results seen with different needle size used, because the interpretation of a PT value within that range would not change. It is therefore important to emphasize that the statistical difference in PT found between the two needle gauges in this study is most likely not clinically significant.
No difference was found between the two different needle gauges for aPTT, and this finding is consistent with other studies in dogs and humans.6,7 The FDP concentration was under 5 μg/ml for all samples in the study. We could not find any other studies of FDPs with various needle gauges, but our findings are consistent with a study in dogs, which evaluated D-dimer values, where no difference was found when blood was drawn from different bore catheters. The lack of a difference in fibrinogen concentrations is consistent with the findings in the same study. 12
In our opinion, it is reasonable to extrapolate these results from normal cats to cats with hemostatic disorders, although future studies of cats with hemostatic disorders would be needed to verify this assumption. It is possible that patients with hemostatic disorders might have testing altered by different needle size used for collection, and the choice of needle size used in those patients will be based on multiple factors.
There are multiple limitations to this study. First is the small number of cats included, with the possibility of false-negative findings (type II statistical error). It should be noted that there were two cats with a clinically relevant difference in PT (approximately 3 s), which may reflect collection technique or individual variability. For the analysis of platelet count, paired samples were only available for 13 cats, owing to platelet clumps, thereby reducing the ability to detect a difference if one was truly present. The pre-hoc power analysis was based on preliminary data, and designed to calculate the number of cats needed to detect a clinically significant difference in findings. The differences between needle sizes in this study population were smaller than in our power calculations and, on post-hoc analysis, the number needed to find a statistical difference between needle sizes was calculated to be 156, 292, and 3212 cats for PT, aPTT and platelet count, respectively. Although a larger population of tested cats may have allowed detection of a statistical significance, the difference between needle sizes would not be likely to alter clinical interpretation.
We did not investigate the potential differences between blood drawn with other needle gauges, such as 21 G or 20 G, which are also commonly used in practice, as jugular venipuncture in three separate areas in a cat would have been difficult to achieve. A separate group of 22 G vs 20 G needles was not possible owing to limited funding, but remains an area for future research. It is unknown if different needle gauges will affect the results of more novel hemostatic tests, such as viscoelastic tests, measurements of thrombin generation or platelet function tests.
Conclusions
This study of 20 healthy cats found that the use of either a 25 G or 22 G needle for venipuncture did not introduce any clinically meaningful difference in routine coagulation variables or platelet count. This is important, because in addition to decreased patient comfort, there is a risk of hemorrhage when larger needle gauges are used. Factors that will influence the needle gauge used for venipuncture in a clinical setting include the minimum amount of blood required, phlebotomist preference and suspected risk of hemorrhage with venipuncture. Based on the results of this study, both 25 G and 22 G needles can be used for venipuncture for routine coagulation tests and platelet count in cats.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This project was supported by the Center for Companion Animal Health (CCAH), School of Veterinary Medicine, University of California, Davis.
