Abstract
Objectives
The aim of this study was to describe the cellular and stromal components of the hepatic progenitor cell niche in feline hepatic lipidosis (FHL).
Methods
Immunohistochemical staining for the progenitor/bile duct marker (K19), activated Kupffer cells (MAC387), myofibroblasts (alpha-smooth muscle actin [α-SMA]) and the extracellular matrix component laminin were used on seven liver biopsies of cats with FHL and three healthy cats. Double immunofluorescence stainings were performed to investigate co-localisation of different cell types in the hepatic progenitor cell (HPC) niche.
Results
HPCs, Kupffer cells, myofibroblasts and laminin deposition were observed in the liver samples of FHL, although with variability in the expression and positivity of the different immunostainings between different samples. When compared with the unaffected cats where K19 positivity and minimal α-SMA and laminin positivity were seen mainly in the portal area, in the majority of FHL samples K19 and α-SMA-positive cells and laminin positivity were seen also in the periportal and parenchymatous area. MAC387-positive cells were present throughout the parenchyma.
Conclusions and relevance
This is a preliminary morphological study to describe the activation and co-localisation of components of the HPC niche in FHL. Although the HPC niche in FHL resembles that described in hepatopathies in dogs and in feline lymphocytic cholangitis, the expression of K19, α-SMA, MAC387 and lamin is more variable in FHL, and a common pattern of activation could not be established. Nevertheless, when HPCs were activated, a spatial association between HPCs and their niche could be demonstrated.
Introduction
Feline hepatic lipidosis (FHL) is considered the most common hepatobiliary disease in cats.1–3 Clinical signs include lethargy, anorexia, weight loss, icterus and often vomiting and constipation. Histologically, it is characterised by the excessive accumulation of triglycerides in more than 60% of hepatocytes, resulting in secondary impairment of liver function and intrahepatic cholestasis.2,4 The histological feature of FHL is that of simple steatosis and the current literature suggests that the disease lacks necroinflammatory lesions and fibrosis.4,5 FHL often presents as an acute critical syndrome that can result in the cat’s death if therapeutic measures are not rapidly implemented. Its progression into steatohepatitis and chronic liver failure has not been documented in cats.1,2
Mature liver cells, ie, hepatocytes and cholangiocytes, have a remarkable capacity to proliferate and restore liver function in homeostasis and in response to injury.6–8 Three decades ago ‘the streaming liver hypothesis’ was proposed based on pulse-chase experiments: periportally derived hepatocytes migrate along the central vein to the hepatic vein axis. 9 Recent cell-tracing experiments demonstrated a population of proliferating and self-renewing cells adjacent to the central vein in the liver lobule. These cells appear to be able to replace all hepatocytes in the liver lobule during homeostatic renewal. 10 These experiments helped the understanding of liver regeneration and the importance of specific hepatic stem- or progenitor cells (HPCs), as recently reviewed.11,12
Irrespective of the origin of HPCs, once the ability of hepatocytes to restore liver mass and function is exhausted or impaired, HPCs become activated, as has been described for several species.8,13–18 These quiescent HPCs reside in the smallest branches of the biliary tract, also known as the canal of Hering, and are recognised by their morphological appearance, portal location, and expression of specific markers, such as cytokeratin 7 (K7) and keratin 19 (K19).6,14,19–21 Once activated, HPCs proliferate and their expansion results in the formation of histological structures known as ductular reactions, also known as bile duct proliferation. 14
There are few descriptions of HPCs in cats.15,22,23 The first study that reported on progenitor cells in healthy cats was the study by Ijzer et al in 2009. 15 Those progenitor cells were described as small, periportally located cells positive for K7, with a morphology and location identical to resident HPCs located in the canal of Hering, as described in humans and dogs.14,15,24 The same study identified the presence of ductular structures (ductular reaction) in different types of feline liver disease (acute hepatitis, neutrophilic and lymphocityc cholangitis and lipidosis), and characterised their histological and phenotypical features similarly to what has been described in humans and dogs.8,13,15–18,23–28 However, the simple description of the ductular reaction does not further implicate the activation and co-localisation of the HPCs and their niche. 29
The balance between HPC self-renewal, proliferation and differentiation is determined by the interaction between the cells and stroma present in the microenvironment; ie, the ‘HPC niche’.30–32 In rodent models, humans and dogs, important cellular components in this niche include HPCs, hepatic stellate cells (HSCs), their differentiated counterparts, alpha-smooth muscle actin (α-SMA)-positive myofibroblasts and macrophages/Kupffer cells with laminin as the main stromal component.6,7,19,24,33 Laminin is important for HPC proliferation and maintenance of the progenitor phenotype. Once the HPCs have left the laminin-enveloped niche, they can differentiate into a hepatocyte phenotype. 34
In humans and dogs the ductular reaction and the degree of HPC and its niche activation is directly related to the severity of disease,17,35,36 indicated by the amount of hepatocyte loss, the amount of inflammation and the extent of fibrosis, and it is seen in diseases like acute hepatitis, chronic hepatitis, biliary disease and hepatic tumours.13,17,24,27,28,36–42
In a recent publication, the remodelling and activation of the HPCs and their niche was described in feline lymphocytic cholangitis. 43 The characteristics of the HPCs and their niche in feline lymphocytic cholangitis share similarities to the HPCs and their niche in sclerosing cholangitis in human medicine.
In human and rodent models of non-alcoholic fatty liver disease (NAFLD) steatosis is sufficient to inhibit replication of mature hepatocytes and to trigger the activation of HPCs.17,18 There is growing evidence that steatosis per se is associated with the development of steatohepatitis and fibrosis, and should not be considered just an innocent bystander in liver disease.44–46
To our knowledge, there is no literature available for cats on the changes of the HPCs and their niche in FHL. Therefore, the aim of this study was to describe the cellular and stromal pattern of the HPC niche in FHL, with emphasis on MAC387-positive Kupffer cells, α-SMA-positive myofibroblasts and the important extracellular matrix component laminin. To substantiate the immunohistochemical data we further used double immunofluorescence to investigate possible co-localisations of the various cell types within the HPC niche.
Materials and methods
Samples of diseased livers were obtained from cats submitted for routine post-mortem examination. Normal livers from healthy cats were obtained from fresh cadavers of non-liver-related studies as required under the University 3R policy. Projects were approved by the Animal Welfare Committee on Experimental Animal Use, as required by Dutch legislation.
Seven liver specimens (formalin-fixed and paraffin-embedded) with a histological diagnosis of FHL were selected from the archives of the Department of Pathobiology of the Faculty of Veterinary Medicine, Utrecht University, The Netherlands. The seven FHL samples demonstrated a severe degree of lipidosis (>70% of hepatocytes involved), based on histological evaluation performed by a board-certified veterinary pathologist (GCMG). In one sample extensive lipidosis coincided with a histological diagnosis of peracute hepatitis. No other liver comorbidities were present in the six other FHL samples. Diagnoses were based on criteria as reported previously by the World Small Animal Veterinary Association. 47
Tissue samples from three healthy cats were taken post-mortem and processed similar to the FHL samples. Sections (3 μm) of all samples were cut for haematoxylin and eosin and immunohistochemical and immunofluorescence staining. The samples were confirmed histologically normal by the same board-certified veterinary pathologist (GCMG).
Immunohistochemistry
All FHL and control samples were examined immunohistochemically for the expression of K19 (marker of bile duct and progenitor cells), 27 MAC387 (myelomonocytic antigen; marker of infiltrated and resident macrophages),22,48 α-SMA (marker of activated hepatic stellate cells/myofibroblasts),27,34 laminin (extracellular matrix component and part of the HPC niche) 34 and Ki67 (proliferation marker), 23 to characterise the HPC niche in cats (Table 1).
Antibodies and processing characteristics as applied in the immunohistochemistry and double immunofluorescence of liver samples of cats with hepatic lipidosis

Mouse-derived antibodies were monoclonal, whereas the rabbit-derived antibodies were polyclonal
K19 = keratin 19; Prot K = proteinase-K; O/N = over night; MAC387 = anti-macrophage antibody 387; α-SMA = alpha-smooth muscle actin; RT = room temperature; LAM = laminin; Ki67 = proliferation marker; panCK = pancytokeratin, widespectrum cytokeratin marker
The immunohistochemical procedure was performed as described previously.23,49 Briefly, sections were deparaffinised and rehydrated in xylene and a graded alcohol series, respectively. After antigen retrieval, endogenous peroxidase was inhibited with a ready-to-use peroxidase block (Dako). Sections were blocked with 10% normal goat serum and the primary antibody was diluted in antibody diluent (Dako) and incubated for 1 h at room temperature or overnight at 4°C, depending on the marker (Table 1). EnVision horseradish peroxidase-labelled secondary antibodies (anti-rabbit or anti-mouse; Dako) were used and 3,3’-diaminobenzidine was used as the substrate for visualisation. Sections were counterstained with haematoxylin. As a negative control, the primary antibody was omitted and the IgG1 isotypes served as internal non-specific controls for each other. For washing steps PBS/Tween 0.1% was used.
The stainings for K19, Ki67, α-SMA and laminin were scored by one operator at high (× 40) magnification. Five random fields were observed for positively stained hepatocytes and myofibroblasts, and the number of positive cells/fields were reported. Only the cells that displayed nuclei on the section were considered. A distinction was made within initial scoring for portal, periportal and parenchymal cell positivity for K19, α- SMA and laminin. To perform a cell count for MAC387, ImageJ was used, a Java-based image processing program from the National Institutes of Health. Images of a representative slide were made using an Olympus ColorviewIIIu digital camera and an Olympus BX41 microscope. Cells that stained positive for MAC387 were reported as total cells per total area analysed (mm2) for each sample.
Double immunofluorescence staining
To evaluate the spatial relationship between macrophages, activated HSCs, laminin, and HPCs double immunofluorescence staining was performed on three samples of cats with FHL showing varying K19 positivity. The decision not to include double immunofluorescence on healthy samples was related to the fact that the positivity to α-SMA and laminin in healthy samples was almost negligible and mainly seen around the portal area (bile ducts/blood vessel), as also described in previously performed studies.15,29
Staining was performed using the aforementioned markers for macrophages (MAC387), activated stellate cells (α-SMA) and laminin. To detect HPCs, panCK, a broad-spectrum cytokeratin and well-validated HPC marker, was used (Table 1). 50 Immunofluorescence staining was performed as described for the immunohistochemical procedure. Sections were incubated with the primary antibodies in a parallel approach overnight at 4°C. The secondary antibodies (goat–anti-mouse AlexaFluor-488 and goat–anti-rabbit AlexaFluor 568; Invitrogen) were applied 1:100, diluted in antibody diluent (Dako). Sections were counterstained using 4′,6-diamidino-2-phenylindole 1:2000 for 10 mins at room temperature, and mounted using Fluorsave. In the negative controls the first antibody was omitted and replaced with an aspecific isotype control.
Slides were analysed with a Leica DMRE fluorescent microscope with Photometrics Coolsnap CCD digital photo camera and CellB software (AnalySIS; Olympus).
Results
Immunohistochemistry
The results of the immunohistochemical staining for normal liver and FHL samples are summarised in Tables 2 and 3, and Figure 1 depicts examples of each staining. Negative controls remained unstained, indicating a specific signal for each antibody (data not shown). Samples were considered for the study when at least five portal areas were evaluated.
Keratin 19 (K19) and alpha-smooth muscle actin (α-SMA)-positive cells and their distribution per portal area in feline hepatic lipidosis and in unaffected liver samples

Cells were evaluated at high magnification (× 40) and their numbers are reported as cells/fields
Ki67- and MAC387-positive cells and laminin-positive staining in feline hepatic lipidosis and in unaffected liver samples

For Ki67, cells were evaluated at high magnification (× 40) and their numbers are reported as cells/fields; MAC387 was evaluated with ImageJ and reported as number of total cells per total area (mm2)

The activated feline hepatic progenitor cell (HPC) niche immunohistochemically stained for keratin 19 (K19), MAC387 (macrophages), alpha-smooth muscle actin (α-SMA; stellate cells), laminin (LAM) and Ki67 (hepatocyte regeneration). The figure shows (a,e,i,o,s) one unaffected cat C2 and selected feline hepatic lipidosis cases with different marker expression: (b,f,l,p,t) F1; (c,g,m,q,u) F5; and (d,h,n,r,v) F6
K19
In unaffected livers, K19 – indicating bile duct and/or progenitor cells – was present in bile ducts and in a variable number of small epithelial cells located in the periportal area (Table 2; Figure 1) confirming the validity of the anti-K19 antibody. No ductular reactions were observed. Of the seven cats with lipidosis, four showed an increase in the number of K19-positive cells in the (peri)portal area and clusters of cells in the parenchymal area (Table 2; Figure 1). In 3/7 cases an increased number of cells positive for K19 staining were seen in the (peri)portal areas and a positive staining for parenchymal ductular reactions was observed in one case.
α-SMA
α-SMA-positive staining, indicating the presence of HSCs and differentiated smooth muscle cells. In healthy livers very few cells stained positive and mainly in the portal area, around bile ducts and in the smooth muscle of blood vessels (Table 2; Figure 1). In 4/7 FHL samples, α-SMA expression was strongly increased in both the periportal area and throughout the parenchyma. In 3/7 FHL samples, α-SMA-positive cells were increased both in the portal and periportal area when compared with the unaffected samples.
MAC387
In healthy livers variable amounts of MAC387-positive cells were found spread throughout the parenchyma as single cells with no clear spatial relationship with the portal areas (Table 3; Figure 1). All FHL samples also demonstrated variable amounts of MAC387-positive cells. In these samples, the MAC387-positive cells formed clusters varying in number from 2–3 to 4–6 cells per cluster, mostly located in close proximity to the portal area.
Laminin
Weak laminin staining was observed in and around the portal areas in healthy livers (Table 3; Figure 1). Four of five FHL samples showed an increase in laminin positivity in the periportal area, ceasing towards the central vein. The central areas in all samples were negative for laminin. One FHL sample remained negative for the laminin staining.
Ki67
Ki67 positivity was minimal in healthy liver. Three FHL samples showed a distinctive increase in Ki67-positve cells, whereas four remained negative (Table 3; Figure 1).
Double immunofluorescence staining
Double immunofluorescence showed that α-SMA and laminin expression were increased and in close proximity to the panCK/K19-positive cells. Laminin strictly co-localised with HPCs, whereas α-SMA was also located at other histological sites. MAC387-positive clusters of cells were found adjacent to K19-positive cells (Figure 2).
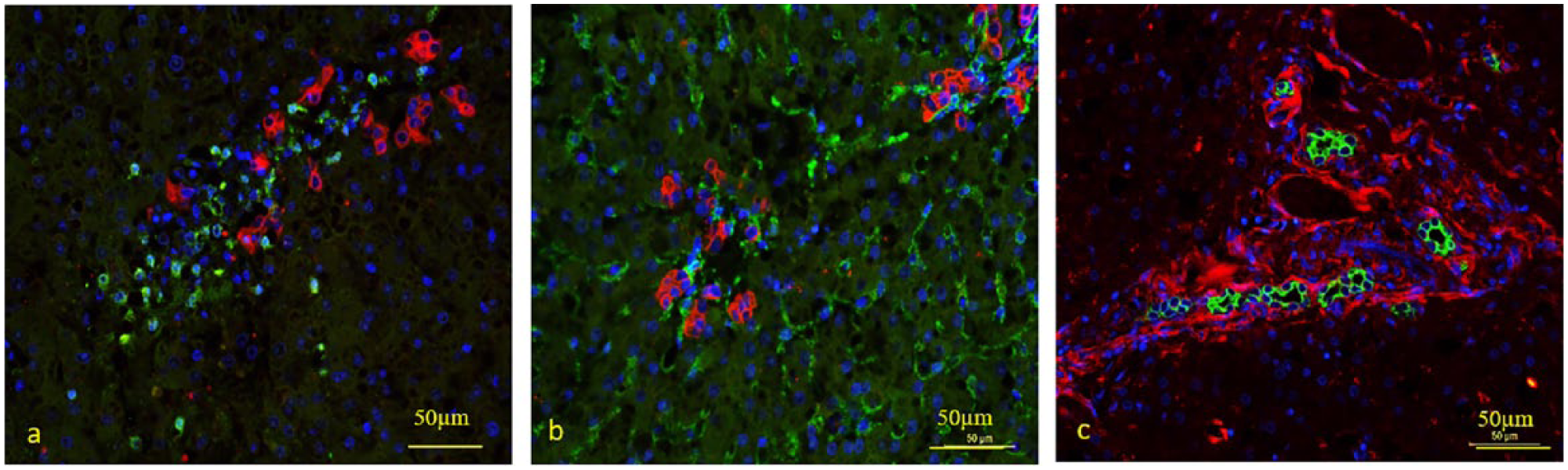
Hepatic progenitor cells (HPCs) co-localised with macrophages, stellate cells and laminin (LAM) in examples of feline hepatic lipidosis. Double immunofluorescence staining with pancytokeratin (panCK; HPCs) and MAC387 (macrophages), α-smooth muscle actin (α-SMA; stellate cells) and LAM, respectively. (a) panCK/MAC387: increase in macrophages (green) and clustering of these cells in close proximity to the HPCs (red); (b) panCK/SMA: increase in α-SMA (green) positivity in parenchyma and around the HPCs (red); (c) panCK/LAM: the LAM (red) can be seen as sheets around the HPCs (green)
Discussion
This study characterised the feline HPC niche by immunohistochemical and immunofluorescence staining in a common feline hepatic disease, FHL. The results of the study demonstrate that ductular reactions observed in the feline HPC niche in FHL contain Kupffer cells, activated HSCs (myofibroblasts) and the extracellular matrix component laminin. This is comparable with other hepatopathies in humans and dogs, such as acute and chronic hepatitis, hepatobilary diseases and hepatic carcinoma.13,15,16,24–27,32,49 It is also in line with the HPC activation in feline lymphocytic cholangitis, a chronic feline hepatobiliary disease. 43
In human, rat and murine models of impaired hepatocyte replication in NAFLD, simple steatosis is enough to inhibit replication of mature hepatocytes and to trigger the activation of HPCs.17,18 Owing to elevated levels of serum-free fatty acids and insulin resistance, lipotoxicity is promoted, reactive oxygen species are formed, and hepatic inflammation and hepatocyte apoptosis are induced.18,44,45 This results in an inhibition of the replication capability of the hepatocytes and stimulation of the activation of the HPCs. Liver steatosis seems to affect the gravity and progression of more chronic liver disease.44–46 Although a liver with steatosis is less protected against inflammation and fibrosis, only a small population of people affected by liver steatosis will further develop steatohepatitis and fibrosis.44–46
However, we were unable to demonstrate a definite pattern between the histopathological presence of lipidosis and the activation of the HPCs or the upregulation of the niche components in all cats with hepatic lipidosis. Different degrees of HPC activation in FHL were suggested by the variability in the expression and positivity of the different immunostainings in the different samples. Despite the presence of severe steatosis (>70% of hepatocytes affected), this might be explained by the fact that hepatic lipidosis in cats often has an acute onset and progression due to a negative energy balance resulting from a heterogeneous group of underlying diseases, which may have different secondary effects on the liver. In the cases with only a minimal number of cells staining positive, the damage to the hepatocytes could have been minimal or not severe enough to induce hepatocyte apoptosis/death and secondary activation of HPCs.
Nevertheless, when HPCs were activated, a spatial association between HPCs and their niche could be demonstrated. The result of the double immunofluorescence in FHL cases with an enhanced number of progenitor cells suggested that a relationship was present between the HPCs, Kupffer cells/macrophages and HSCs/myofibroblasts
Stellate cells/myofibroblasts are the main producers of extracellular matrix components in the liver and could be responsible for the enhanced laminin deposition. 34 Recently, it has been suggested that the activated HPCs produce their own laminin sheet to perpetuate their proliferation and maintain their undifferentiated phenotype.34,50–52 The close anatomical relationship between HPCs and laminin suggests that this may also be true in cats. In situ hybridisation for laminin mRNA can resolve this issue.
This is the first study to characterise the feline HPC niche, HPC activation and the relation of HPCs with FHL. The low number of FHL cases and the lack of longitudinal samples limits the power of the study. Staining differences may have been affected by variations in fixation and/or storage time. However, the quality control with omission of the first antibody resulted in negative staining for all antibodies that were used. Furthermore, the staining patterns were not identical, despite the use of similar IgG isotypes and the same secondary antibody in the stainings for K19, MAC387 and Ki67. This strongly argues against aspecific staining patterns.
We did not use liver samples collected during life, because histological biopsies are not routinely taken in cats suspected to have hepatic lipidosis.1–3 High-quality biopsies of sufficient size are therefore difficult to obtain. 53 Furthermore, liver diseases in cats are often associated with coagulation disturbances and haemorrhage is a potential complication.1,3,52,53 In particular, serial histological liver biopsies for the evaluation of disease progression are difficult to obtain owing to the reservations of veterinarians and the understandable reluctance of pet owners. 52 In addition to this, histological liver biopsies are also not necessary for a diagnosis of FHL. 3 Because of the absence of sequential sampling and the descriptive nature of the immunohistochemistry, this study does not provide dynamic information and limits conclusions on cause–effect relationships. Despite these restrictions, we were able to highlight some interesting aspects of the HPCs and their niche in FHL.
Conclusions
Based on this study the composition of the feline HPC niche in FHL and its observed cellular and stromal interactions resemble that of normal and diseased livers in other companion animals like dogs and in cats with lymphocytic cholangitis. However, the feline HPC niche in FHL shows variability between cats regarding the expression of K19, α-SMA, MAC387 and laminin, and a common pattern of activation could not be established. Nevertheless, when HPCs were activated, a co-localisation between HPCs and their niche could be demonstrated. It remains speculative whether this variation is caused by the duration of lipidosis, variations in the underlying disease or the effect of the therapeutic measures initiated by the referring clinician.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
LCP receives grants form the Netherlands Organisation for Health Research and Development (NWO ZON/MW numbers 92003538 and 16004121) for liver progenitor cell research.
