Abstract
Practical relevance:
Ciclosporin (CsA) is a systemic immuno-modulatory drug widely used to treat immune-mediated diseases in humans and veterinary species. CsA was registered for use in cats in the USA and Europe in 2011, and is indicated for the treatment of chronic allergic dermatitis at a recommended daily dose of 7 mg/kg PO.
Audience:
This review will be of interest to all veterinarians working with cats, given the wide range of potential applications of CsA and its safety profile. Although the drug is currently only licensed to treat chronic allergic dermatitis in cats, a small number of reports describe its use in non-dermatological conditions.
Evidence base:
This article reviews the mechanism of action, pharmacokinetics, drug interactions, adverse effects and clinical use of CsA, both for the licensed indication and for off-label use in the feline patient. Information presented has been summarised from the existing literature on CsA, with specific interest in studies carried out in cats. For its licensed indication, chronic allergic dermatitis, evidence provided includes randomised, placebo or prednisolone-controlled studies (EBM grade I) and prospective or retrospective open trials.
Discovery and development
Ciclosporin (CsA) is a systemic immuno-modulatory drug licensed for use in humans and veterinary species. It was registered for use in the cat in the USA and Europe in 2011, and is indicated for the treatment of chronic allergic dermatitis.

CsA is a cyclic undecapeptide metabolite of the fungus Tolypocladium inflatum Gams1,2 and its molecular weight is 1203 Daltons. 3 It was discovered in 1971, during a search for new antibiotics produced by fungi. CsA and other metabolites were initially screened for antifungal activity, but their efficacy was poor. Its immunosuppressive, non-cytotoxic activity was discovered and reported in 1976. The first human trial started at the end of the same year.4,5 In the USA, it was approved in 1983 for the prevention of graft rejection following transplantation in human patients, and in 1984 synthetic CsA was produced. 4
CsA is fat soluble, extremely hydrophobic, and is poorly absorbed after oral administration. It has been produced in different formulations over time, with the aim of improving its absorption and bioavailability. The first commercially available CsA formulation, only registered for humans, was a solution containing ethanol and corn oil as excipients (Sandimmune; Novartis Pharma). The CsA formulation currently used in humans, dogs and cats is a microemulsion, which enhances oral absorption of the drug.

In most studies published before 2011, micro-emulsified CsA in soft gelatin capsules, registered for the treatment of atopic dermatitis in dogs, was used in feline patients at a daily dose of one 25 mg capsule/cat. In older studies investigating the use of CsA in feline renal transplant recipients, variable dosages were used, according to the different formulations available at the time. Dosages will be specified in this review, unless microemulsified CsA as the oral solution registered for cats was used at the recommended dosage of 7 mg/kg PO q24h.
Mechanism of action
CsA is a systemic drug that exerts its effects mainly on the cell-mediated immune system, while humoral immunity is less affected.1,6 Its mechanism of action is depicted in Figure 1. CsA suppresses the production of interleukin (IL)-2 and the expression of the IL-2 receptor on T lymphocytes, resulting in inhibition of T cell proliferation and activation. It also inhibits the production of IL-4, IL-5, IL-6, IL-8, IL-13, granulocyte-macrophage colony-stimulating factor (GM-CSF), tumour necrosis factor alpha (TNF-α) and interferon gamma (IFN-γ). This leads to a number of different anti-inflammatory effects, including reductions in mast cell degradation, keratinocyte proliferation and cytokine production, as well as decreased tumouricidal and superoxide activity by macrophages. Other effects include reduced expression of intercellular adhesion molecule 1 and leukoctye trafficking in endothelial cells, and inhibition of antigen-presenting cell function (Figure 2).1,6,7

Mechanism of action of CsA. CsA enters the cytoplasm of T cells and binds to the receptor cyclophilin. The drug–receptor complex secondarily binds to calcineurin, inhibiting its action. In the absence of CsA, calcineurin binds to nuclear factor of activated T cells (NF-AT), which in turn enters the nucleus and binds to activator protein 1 (AP-1), a transcription factor. This complex induces the transcription of cytokine genes by the T cell. IL = interleukin; IFN-γ = interferon gamma; TNF-α = tumour necrosis factor alpha; GM-CSF = granulocyte–macrophage colony-stimulating factor

Cellular targets of CsA. IL = interleukin; GM-CSF = granulocyte–macrophage colony-stimulating factor; TNF-α = tumour necrosis factor alpha; IFN-γ = interferon gamma
Very few studies have been published specifically addressing the effects of CsA in feline species. In the cat, CsA has been shown to suppress in vitro lymphoblast transformation after stimulation with the mitogens concanavalin A and pokeweed within 7 days of administration, with return to a normal response within 7 days after withdrawal. Large individual variations in CsA serum trough levels were observed in both studies.3,8 In feline peripheral blood mononuclear cells (PBMCs), CsA suppresses the expression of mRNAs for IL-2, IL-4, IL-10, GM-CSF, IFN-γ and TNF-α, and the number of IL-2 secreting lymphocytes, in a dose-dependent manner.9,10 In feline intestinal mucosal biopsies, CsA is able to reduce neutrophil infiltration. 11

Therapeutic monitoring
Due to the extreme variability in absorption and metabolism, monitoring the concentrations of CsA in the blood has been recommended. Knowing the blood concentration may help clinicians adjust the dose, in order to maintain ‘effective’ concentrations. Therapeutic monitoring may also be useful to identify cats at a higher risk of adverse effects or opportunistic infections.15,19 Monitoring is also suggested in patients treated concurrently with imidazoles (see section on drug interactions). CsA concentration should be evaluated in whole blood rather than plasma, because the drug concentrates within blood cells. 20 Ideally, testing should be carried out after 2 weeks of treatment and, where available, high performance liquid chromatography is a better method than immunoassay for evaluating CsA whole blood concentrations. 21
In older studies using CsA twice daily in feline transplant patients, the recommendation was to adjust the dose in order to maintain the 12 h trough blood levels at 300–500 ng/ml. 22 Trough level is defined as the lowest concentration reached by a drug before the next dose is administered, so if CsA is administered once daily, the blood sample should be taken 24 h after the dose. Since trough plasma levels vary widely between tested cats, it has been suggested that blood levels of CsA 2 h after administration (peak concentration) may be more useful for monitoring both therapeutic drug levels and toxicity; however, therapeutic peak concentrations have not been established.15,20
Unfortunately, the correlation between clinical efficacy of CsA and trough blood levels is usually poor. 23 Because CsA concentrates in the skin, reaching higher levels compared with blood,12,14 the current opinion is that when treating skin diseases with CsA, monitoring trough CsA levels is not particularly useful for evaluating the drug’s effectiveness and such monitoring is not considered mandatory, unless adverse effects are observed.
An alternative, although not commercially available, method for assessing the effectiveness of CsA involves pharmacodynamic assays, which evaluate the effect of a drug on its target cells. In human medicine, a large number of immunological markers of the immunosuppressive activity of CsA have been studied and validated for pharmacodynamic monitoring.20,24 In veterinary medicine, a few studies have investigated the expression of specific cytokines by either PBMCs or lesional skin of dogs treated with CsA, revealing decreased expression of IL-2, IL-4 and IFN-γ.25–27 The reduced expression of these biomarkers was very marked with high doses of CsA and still present at the recommended dosage for dogs (5 mg/kg PO q24h), showing that CsA may have immunosuppressive effects even at dosages considered to be low. 28 No studies on pharmacodynamic assays of CsA in the cat have been published yet. However, it is likely that the immunosuppressive effects may be similar to those observed in humans and dogs.
Drug interactions
CsA is metabolised by the cytochrome P450 enzymatic system and any drug able to induce or inhibit these enzymes may respectively increase or decrease CsA metabolism, thus decreasing or increasing CsA blood concentration. CsA is also both a substrate for and an inhibitor of P-glycoprotein, which is an efflux pump able to protect the blood–brain barrier from adverse effects of potentially neurotoxic drugs. CsA shows interactions with many drugs: in humans, 853 drugs are reported. 29 The most commonly reported interactions of CsA with other drugs in cats and humans are summarised in Table 1.
Drugs most commonly reported as interacting with CsA in cats and humans

Very few studies have been performed to specifically address this issue in feline species. CsA has been shown to interact with both ketoconazole and itraconazole; administration of ketoconazole or itraconazole at 10 mg/kg PO q24h approximately doubles the CsA blood concentration, allowing for a significant reduction of the CsA dose and of the cost of treatment.21,31 The interaction with clarithromycin was studied recently. Administration of this antibiotic significantly increased the oral bioavailability of CsA, reducing the CsA dose required to maintain therapeutic levels by 65%. 30 Robson 32 anecdotally reported that concurrent administration with metoclo-pramide reduced CsA concentrations in 3/8 cats. Co-administration of CsA with macro-cyclic lactones does not seem to be associated with neurological adverse effects in cats. 33

Adverse effects
When using CsA at the recommended daily dose (7 mg/kg PO or, before 2011, 25 mg/cat), the most commonly reported adverse effects have been gastrointestinal (GI).33,35–40 In a study specifically addressing adverse effects in cats treated with CsA, vomiting occurred in 12% of cats and soft stools or diarrhoea in 16% of cats. 38 GI adverse effects are often temporary and many disappear within the first few weeks of treatment. Anecdotally, it has been suggested that metoclopramide 0.3–0.4 mg/kg given orally 20–30 mins before CsA administration, or maropitant 1 mg/kg PO q24h, or freezing the capsule may be used to prevent GI upset.7,32 However, metoclopramide in cats may reduce CsA concentration. 32
Anorexia has been reported in 2% of cats in one study 38 and in 10% of cats in another study. 33 Anorexia may be a serious problem in cats, as it can eventually lead to hepatic lipidosis.6,32,33,38–40 Weight loss has been described in 5–16% of cats.33,38 Some studies reported weight loss at the beginning of CsA therapy, with return to normal weight over the following months after CsA dose reduction.33,39
Gingival hyperplasia, possibly due to the stimulating effect of CsA on fibroblast proliferation consequent to increased transforming growth factor beta production, 41 has been reported rarely in cats.3,6 Recently, systemic CsA has been reported to be a significant risk factor for the development of acute bullous keratopathy in cats. 42
Other reported adverse effects are polyphagia, polydipsia, hyperactivity, 37 hypertrichosis, 43 weight gain, dental tartar and gingivitis, otitis, inflammatory bowel disease, urinary tract infection, cataract, hyperthyroidism and transient inappropriate urination. 38 However, most of these adverse effects are rare and not likely to be directly caused by CsA. 38
In experimental studies using CsA at higher doses (up to five times the normal dose), peripheral lymphadenopathy was reported, 44 and death occurred in one cat with a very high trough CsA concentration and bone marrow hypocellularity. 40 In three cats undergoing renal transplantation and given CsA at 5 mg/kg PO q12h, together with prednisone at 0.25 mg/kg PO q12h, haemolytic uraemic syndrome has been described. 45 CsA was deemed responsible for inducing this syndrome in two of these cases.
Haematological concentrations
In most studies, cats receiving systemic CsA have undergone haematology and biochemistry testing before, during and after the study, in order to identify significant alterations. Relative leukopenia, lymphopenia, neutropenia and eosinopenia, although with values still within normal reference intervals, have all been observed. 39 Biochemistry showed mild increases of total bilirubin, glucose and blood urea nitrogen (BUN) and mild reductions of alanine aminotransferase activity, alkaline phosphatase activity and albumin, but values were also within normal reference intervals.33,39
In experimental studies using CsA at higher doses (up to five times the normal dose), aplastic anaemia, lymphopenia, prolonged activated partial thromboplastin time, reduced total proteins and increased cholesterol, creatinine and BUN were also occasionally reported.40,44
Opportunistic infections
CsA is an immunosuppressive drug, and opportunistic infections may develop in treated patients. The most commonly reported infection in cats is toxoplasmosis (see box).

Feline herpesvirus infection (FHV-1) reactivation has been reported in cats experimentally infected and treated with CsA, methylpred-nisolone acetate or placebo. In most of the cats showing clinical signs of FHV-1 infection, these were mild and self-limiting; however, one cat with a very high CsA blood concentration developed severe clinical manifestations. 51 Conversely, CsA may have antiviral properties: it has been shown to suppress in vitro FIV production and apoptosis in infected cells, as well as feline infectious peritonitis (FIP) coronavirus replication.52,53
Urinary tract infections (UTIs) were investigated in 33 cats treated with either CsA or glucocorticoids for more than 3 months and no association between any therapy and UTI could be demonstrated. 54
Many other infectious diseases in cats receiving CsA have been reported. However, these occurred as single cases and the role of CsA could not be proven with certainty. Moreover, these diseases occurred in cats treated with different dosages of CsA. Reported diseases include Mycobacterium avium complex infection,43,55 severe pneumonia caused by Salmonella species, 56 nasal nodular cryptococ-cosis, 57 dermatophytosis, 58 actinobacillosis, pyelonephritis, retroperitoneal abscess and septicaemia, upper respiratory tract infection, oral abscess, chronic stomatitis, FeLV infection, FIP, fungal pinna dermatitis, septic peritonitis and cholangiohepatitis.23,40,44,59
Malignant neoplasia
The development of neoplasia has been reported in cats treated with CsA. The first report described a cat, immunosuppressed with CsA at 7.5 mg/kg q12h and prednisolone at an initial dosage of 2 mg/kg q24h, which developed FeLV-associated lymphosarcoma 6 months after transplantation surgery. 60 Two retrospective studies on feline transplant patients, treated with combinations of CsA and prednisolone at various dosages, were subsequently published: the first study reported malignant neoplasia in 9/95 (9.5%) cats, while the second reported malignant neoplasia in 11/45 (24%) patients.61,62 Types of malignant tumours included GI or multicentric lymphoma, bronchogenic adenocarcinoma, hepatic adenocarcinoma, gastric or intestinal adenocarcinoma, tonsillar or pharyngeal squamous cell carcinoma, pharyngeal plasmacytoma, transitional cell carcinoma and unspecified malignant round-cell neoplasia.61,62 The authors observed that transplanted cats had more than six times higher odds of developing malignant neoplasia compared with control cats, but the specific role of CsA remains undetermined. 62 Several human studies have also confirmed an increased incidence of post-transplant malignant neoplasia; however, no increased risk could be demonstrated when patients were immunosuppressed with CsA compared with azathioprine.63,64
In cats treated with CsA at the recommended dose for allergic or immune-mediated diseases, malignant neoplasia appears to be a rare occurrence.38,43
Vaccinations
A single study 65 evaluated the effectiveness of vaccinations in cats treated with CsA. Cats were given CsA at a very high dosage (24 mg/kg PO q24h) for 56 days and compared with placebo-treated cats. All cats had been previously vaccinated against calicivirus (FCV), FHV-1, feline parvovirus (FPV), FeLV and rabies, and were given a booster of the same vaccines 4 weeks after initiation of CsA administration. The booster vaccinations were successful in all treatment and placebo group cats. However, a novel vaccination for FIV given at the same time as the boosters did not induce adequate protection in CsA-treated cats. This suggests that booster vaccinations are effective in cats on CsA therapy, but primary immune responses may be impaired. Vaccination with live vaccines is not recommended during CsA therapy. 34
Clinical uses
CsA is licensed only for the treatment of chronic allergic dermatitis in cats, and all other uses mentioned in this review are off-label. The recommended dosing for CsA is shown on the left. In some of the studies using CsA off-label, the drug was administered at a different dose and this will be noted where relevant. CsA can be given to cats either mixed with a small amount of food or directly into the mouth.

At present, recommended pretreatment examinations include complete blood count, biochemistry panel, urinalysis and serology for FeLV and FIV. 7 Serology testing for toxoplasmosis is not essential, but might be helpful as seronegative cats may require more careful management to prevent infection during treatment. Any complicating infection should be treated before commencing CsA administration.
During CsA treatment, physical examination, body weight check, haematology and biochemistry should be performed every 6 months.6,32,44
Chronic allergic dermatitis
Many studies have reported on the effectiveness of CsA in chronic allergic dermatitis (Figure 3), and these are summarised in Table 2. CsA treatment was also used to validate systems for scoring the severity of lesions in allergic cats.71,72

Feline atopic syndrome. (a) Generalised self-induced alopecia and (b) 1 month after initiation of CsA therapy
Studies on the use of CsA in feline chronic allergic dermatitis
Some cats on Sandimmune solution, some on Atoplus capsules
AD = atopic dermatitis; EG = eosinophilic granuloma; EGC = eosinophilic granuloma complex; EP = eosinophilic plaque; HD = hypersensitivity dermatitis; IU = indolent ulcer; MD = miliary dermatitis; NR = not reported; OT = open trial; P = pruritus; Pred = prednisolone; PS = plasmacytic stomatitis; PT = pilot study; RCT = randomised controlled trial; REV = review; RS = retrospective study; S-A = self-induced alopecia; FEGEPESI = feline eosinophilic granuloma, eosinophilic plaque, extension and severity index
Pemphigus complex
The first report of the use of CsA in cats for the treatment of pemphigus foliaceus (PF) (one cat) and pemphigus erythematosus (PE) (one cat) at 15 mg/kg PO q24h was published in 1989. The PF cat showed a partial and temporary response, while the PE cat showed complete resolution maintained with continuation of CsA. 73 A retrospective study in 2012 evaluated CsA in 15 cats with PF, both as an adjuvant and sole drug, in comparison with chlorambucil. All the cats treated with CsA at 5 mg/kg PO q24h were eventually maintained on CsA only and glucocorticoids were either not used from the beginning or discontinued.
In the chlorambucil group, only 1/6 cats could be weaned off glucocorticoids. 43 CsA in feline PF (Figure 4) appears to be a promising drug; however, a controlled, prospective study should be conducted to confirm its effectiveness.
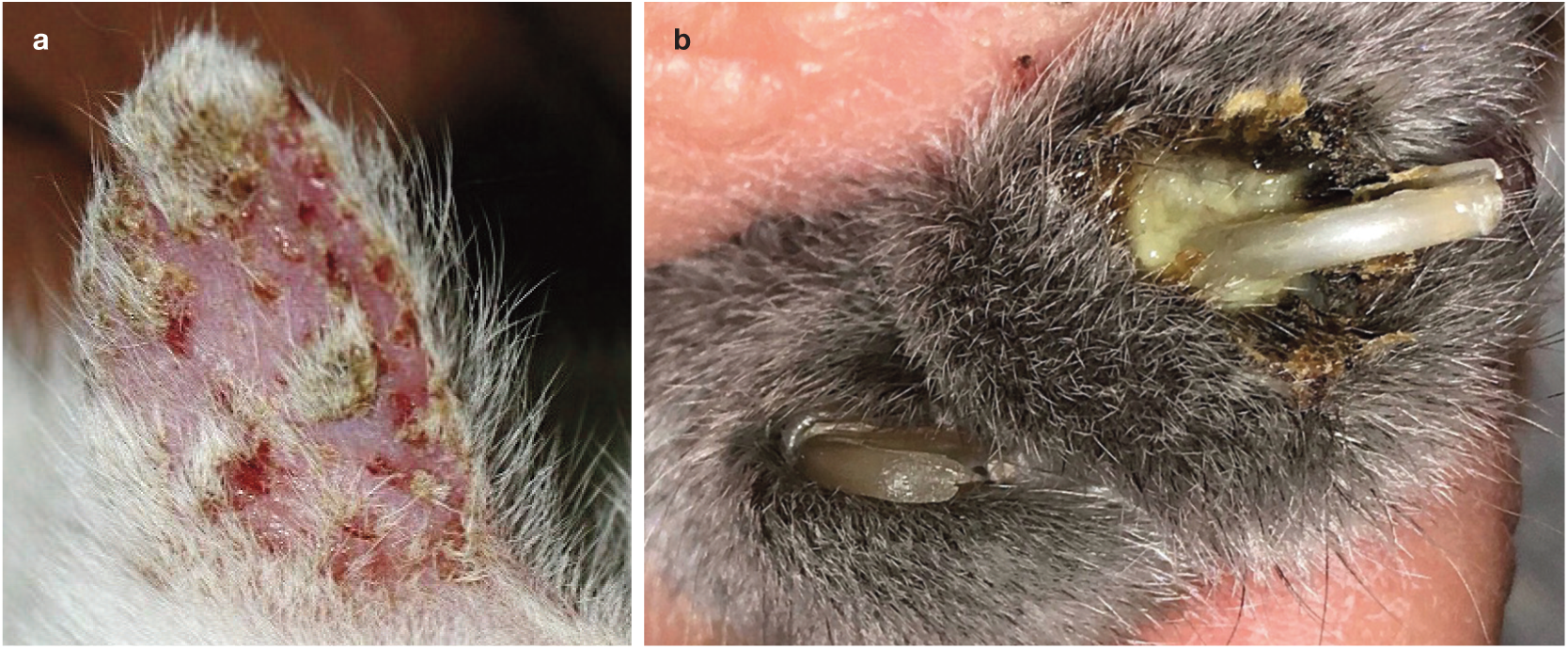
Feline pemphigus foliaceus. (a) Multifocal crusting covering small erosions on the pinna. (b) Caseous material in the claw fold
Miscellaneous skin diseases
Many different feline dermatological immune-mediated diseases have been treated with CsA off-label and with various doses and protocols. However, this has been described only in single cases or small groups of patients and the results must be viewed with caution. Diseases successfully treated or controlled with CsA include pseudopelade, 58 feline urticaria pigmentosa (Figure 5), 74 idiopathic facial dermatitis of Persian cats, 75 granulomatous folliculitis and furunculosis, sebaceous adenitis, 76 feline lymphocytic mural folliculitis, 77 exfoliative dermatitis not associated with thymoma, 78 an atypical form of feline eosinophilic dermatosis (Figure 6), possibly of genetic origin, 79 and feline plasma cell pododermatitis. 80

Feline urticaria pigmentosa. Note the linear distribution of the lesions

Feline pedal eosinophilic dermatosis
Transplants
CsA has been used to prevent organ rejection in feline transplant patients for more than 25 years.81,82 In an experimental study using the gracilis musculocutaneous flap as an allograft, six cats were treated with CsA (trough whole blood levels 750 ng/ml for 70 days, then 500 ng/ml for 30 days) in combination with prednisolone at 0.5 mg/kg PO q24h, while six other cats were left untreated. In the treated cats, the flaps survived for more than 100 days while the grafts of the untreated cats were rejected within 2 weeks. 60
The first large case series of renal transplants was published in 1997. 23 Immunosuppressive treatment protocols in this series were based on administration of either CsA at 7.5 mg/kg PO q12h or microemulsified CsA at 3 mg/kg PO q12h, always in association with prednisolone at 0.125–0.25 mg/kg PO q12h. Seventy-one percent of the cats survived until discharge from the veterinary hospital. Eight cats died within a year of immunosuppression-related causes such as infectious or neoplastic diseases. In a second case series, cats were treated with CsA at doses able to maintain target whole blood trough levels at 300–500 ng/ml and prednisolone at 0.25 mg/kg PO q12h; 77.5% of cats survived until discharge, with 65% of cats alive 6 months postoperatively. 59
Haematological diseases
Treatment with CsA and prednisolone of severe, non-regenerative anaemia due to pure red blood cell aplasia of autoimmune origin was reported in a young cat in 1998. 83 After this case report, three retrospective studies on pure red blood cell aplasia supported the use of CsA (5–20 mg/kg PO q24h) combined with various immunosuppressive drugs (prednisolone 3–4 mg/kg PO q24h, methylpred-nisolone 2 mg/kg PO q24h, dexamethasone 0.6–0.8 mg/kg q24h or recombinant human erythropoietin 150 U/kg SC three times weekly) to treat this rare condition. Most cats required long-term immunosuppressive treatment to maintain the disease in remission.84–86
Two single case reports described the use of CsA in combination with immunosuppressive drugs at similar dosages to treat idiopathic, possibly immune-mediated, thrombocytopenia and immune-mediated granulocytopenia associated with thymoma.87,88
Feline stomatitis
CsA has been reported to be effective in feline gingivostomatitis. 89 A randomised, double blinded, placebo-controlled clinical trial of CsA in cats with chronic refractory stomatitis confirmed CsA efficacy at 2.5 mg/kg PO q12h, with a mean improvement of 52.7% after 6 weeks in the treated group compared with 12.2% in the placebo group. 90
Feline asthma
Information on the use of CsA to treat feline asthma is scarce and contradictory. In one experimental study, CsA (trough levels between 500 and 850 ng/ml) given to Ascaris suum-experimentally sensitised cats prior to antigen challenge was able to inhibit airway hyper-responsiveness, IL-2 production in PBMCs, increase of eosinophils in broncho-alveolar lavage fluid and airway remodelling. 91 However, a second study showed that CsA administration did not inhibit early phase bronchoconstriction and did not inhibit mast cell degranulation within airway smooth muscle occurring after antigen stimulation in A suum-experimentally sensitised cats. 92 A single case report described the clinical use of CsA at 4 mg/kg PO q24h in a 15-year-old cat suffering from feline asthma and concurrently affected by diabetes mellitus and congestive heart failure. 93
Miscellaneous immune-mediated diseases
One case report described treatment with CsA at 25 mg/cat q24h, associated with dexa-methasone for the first month only, in an FIV-and FeLV-positive cat affected by chronic progressive polyarthritis. 94 Only anecdotal reports are currently available on the use of CsA in the treatment of feline inflammatory bowel disease. 95
Prevention of adverse reactions to drugs
The use of CsA to control acute carrier-induced hypersensitivity reactions due to the chemotherapeutic agent docetaxel has been reported in cats with various neoplastic diseases. 96
Key Points
CsA is a calcineurin inhibitor and very effective in the treatment of allergic dermatitis in cats. It has also been used to manage a variety of immune-mediated diseases in feline species, often with very promising results, although prospective controlled studies are lacking for most diseases.
CsA is safe: the most commonly reported adverse effect is GI upset, which is often temporary and self-resolving. Being an immunosuppressive drug, CsA may increase susceptibility to opportunistic infections and neoplasia; however, this appears to be rare when the drug is used at recommended doses.
Cats should be tested for FIV and FeLV before initiating treatment. Owners should be instructed to keep their cat indoors and not to feed raw meat to prevent toxoplasmosis in seronegative cases.
Routine booster vaccinations can be given to cats on CsA, and have been shown to induce a protective antibody response.
CsA absorption is variable and this is reflected in CsA blood concentrations, which do not correlate with the clinical response, at least in dermatological patients.
CsA blood level determination is not considered mandatory, but it may be useful to identify cats at higher risk of opportunistic infections.
Caution should be used when the concurrent use of CsA with other drugs is required, due to its numerous pharmacological interactions.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
