Abstract
Objectives
The objective of this study was to evaluate itraconazole 10 mg/ml oral solution for the treatment of Microsporum canis infection using an alternating-week pulse therapy regimen in a controlled laboratory setting.
Methods
Eighty cats with experimentally induced infections were randomly assigned to treatment (itraconazole vs control [sterile water]), administered 5 mg/kg PO q24h for 1 week on alternate weeks for 5 weeks, followed by a 4 week follow-up period. Topical therapeutic treatment was not administered. Cats were individually housed in stainless steel cages that were cleaned and disinfected daily. Study measures included weekly fungal cultures, clinical lesion scores, Wood’s lamp examination and periodic laboratory monitoring. Mycological cure was defined as two consecutive negative cultures.
Results
Itraconazole-treated cats had significantly greater (P = 0.0003) mycological cure compared with untreated controls (24/40 [60%] vs 1/40 [2.5%], respectively) and all of these reached clinical cure and had negative final Wood’s lamp examinations. Furthermore, 36/40 (90%) treated cats had at least one negative fungal culture by the end of the study vs only 3/40 (7.5%) control cats. For both treatment groups, prevalence of clinical cure peaked at the end of the study (week 9), with 39/40 (97.5%) itraconazole-treated cats and 6/40 (15%) control cats achieving clinical cure. Wood’s lamp negative examination rates were significantly greater (P <0.0001) for itraconazole-treated cats compared with controls (39/40 cats [97.5%] vs 6/40 [15%], respectively) and followed the same pattern of improvement as primary clinical lesions.
Conclusions and relevance
In this controlled study, orally administered itraconazole using a 5 mg/kg pulse-dose treatment regimen reduced the time to mycological cure and increased both mycological and clinical cure rates compared with untreated controls.
Introduction
Feline dermatophytosis is a superficial, contagious skin infection that is spread most commonly via direct contact with another infected animal, but can be spread by traumatic fomite inoculation. 1 In >90% of feline dermatophytosis cases, Microsporum canis is the identifiable causative agent.1,2 Although typically self-limiting by 4 months after the onset of clinical lesions, treatment of affected cats is recommended to shorten the clinical course of disease and reduce the risk of spreading infection.1,3 Current treatment recommendations include the use of systemic antifungal drugs to eradicate the infection within the intrafollicular portion of the hair and concurrent topical therapy to disinfect the haircoat and environmental cleaning to remove infective material.1,4
Itraconazole has been shown to be effective when administered as pulse-dose therapy (week on–week off) for systemic treatment of human dermatophytosis because it binds tightly to keratin and persists in the stratum corneum for up to 4 weeks after discontinuation of oral administration. 5 Concentrations above minimum inhibitory concentration levels (0.1 µg/ml) were detected in cat hair after cats received 5 mg/kg q24h for three alternating weeks. 6 This finding, and a favorable safety profile, made it an attractive alternative to griseofulvin and ketoconazole. Between 1995 and 2016, there were 12 reports on itraconazole use to treat feline dermatophytosis;7–18 however, only two were controlled studies and neither used a pulse-dose therapy regimen.11,13
The efficacy of the widely recommended pulse-dose therapy regimen for cats has not been documented in a controlled study using an experimentally induced challenge model and large numbers of animals. Furthermore, there is a lack of controlled research evaluating the sole effect of systemic treatment. The objective of this study was to evaluate itraconazole 10 mg/ml oral solution for the treatment of M canis infection using an alternating-week pulse therapy regimen at a daily dose of 5 mg/kg.
Materials and methods
The study protocol was reviewed and approved by the site’s Institutional Animal Care and Use Committee. Client-owned animals were not used in this study; test facility ownership consented to use of animals.
Experimental design
This study was carried out over 9 weeks using a randomized complete block design with two treatments: (1) itraconazole oral solution (Elanco Animal Health) and (2) control (sterile water). The experimental unit for all measures was an individual cat. The study period involved a regimen of oral treatment administration once daily for 1 week on alternate weeks for 5 weeks (three total weeks of dosing). A 4 week follow-up period followed the end of the treatment period to allow time for clinical and mycological cure and additional data collection.
Animals and housing
A total of 87 healthy purpose-bred domestic shorthair cats, 8–9 weeks of age, were available for selection of 80 cats for inclusion in the study. Study cats were housed individually in stainless steel cages within two rooms with 40 cats per room. Each room contained eight banks of six close-sided, stainless steel cages, each measuring 71.1 cm × 61.0 cm × 55.9 cm (length × width × height), with five cats per bank.
Each cage and equipment were cleaned and disinfected (or disposed of) once daily. Caretakers and investigators changed gowns and gloves between handling cats to minimize the risk of cross-contamination. Cats were fed ad libitum commercial diets suitable for their age and weight.
Infection protocol
At the end of the 1 week acclimation period, using procedures adapted from a previous study, cats were experimentally infected over 2 days with a highly fluorescing field-isolated strain of M canis that previously showed evidence of inducing clinical disease. 19
Prior to inoculum application, the right lateral flank was clipped free of hair, a circle approximately 3 cm in diameter was drawn on the skin at the intended site of application, the skin was gently scarified with the edge of a plastic knife and 200 µl inoculum containing approximately 105 spores was applied to the skin and rubbed into the area with a glass rod. An occlusive bandage was then applied to the inoculation site and the edges secured with surgical tape.
After the occlusive bandage was removed 72 h post-application, the inoculation site of each cat was covered with a cotton stockinette, which remained in place until the first day of treatment. The infection was allowed to propagate for 4 weeks prior to initiation of treatment.
Inclusion criteria, randomization and dosing of treatments
Cats were evaluated for study inclusion within a 2 day period prior to first treatment. For inclusion, cats were required to be fungal culture positive for M canis, score >0 on Wood’s lamp examination at any location on the hair shaft, have a visible or palpable clinical lesion, show microscopic evidence of fungal arthrospore invasion of hairs at the challenge site as determined via direct examination using potassium hydroxide, and be amenable with study procedures. Eighty cats met all criteria and were enrolled in the study and each was randomly assigned to a cage in one of two animal rooms, and to one of 16 banks of cages using PROC PLAN of SAS (SAS Institute). Within the rooms, banks of cages were randomly assigned to a treatment group, resulting in four cage banks per a treatment per room. Each treatment group consisted of 40 cats.
The treatment period lasted 5 weeks, during which cats received either itraconazole administered at 5 mg/kg body weight (ie, 0.5 ml/kg body weight) or sterile water administered at 0.5 ml/kg body weight. To follow the pulse-dose therapy regimen, cats were treated orally once daily for 1 week on alternate weeks over 5 weeks for a total of three weekly treatment cycles. Cats were not fasted prior to treatment administration. Dosage volumes were adjusted for individual cats based on body weight measurements collected prior to each weekly treatment cycle. If cats vomited or regurgitated at any time within 2 h of treatment, a second full treatment dose was administered.
Monitoring schedule
General health was observed twice daily throughout the study. Blood samples were collected and analyzed for changes in hematology at the start of study, after last treatment administration, and at the end of study.
Fungal cultures and scores
Samples for fungal culture were obtained once during acclimation, 2 weeks postinfection, at the start of treatment and then once weekly until week 9 (end of study). Samples were obtained using the toothbrush fungal culture technique, brushing the infection site 20 times using an individually packaged toothbrush.1,13,17–19 The bristles of the toothbrush were then gently inoculated onto the surface of a 90 mm diameter Dermatophyte Test Medium plate (DTM) supplemented with chloramphenicol, cycloheximide and gentamicin (DTMccg) (Sporometrics) for incubation at 26 ± 2°C for 3 weeks. Results were recorded as no growth, contaminant growth, or positive for M canis based upon microscopic confirmation. For cultures positive for M canis, the number of colony-forming units (cfu) per plate was recorded for semi-quantitative analysis of fungal culture results.3,11,13,17,18,20 Each culture was given a pathogen score (P-score) based on cfu/plate using the following scale: P0 = no colonies, P1 = 1–5 cfu/plate, P2 = 6–10 cfu/plate and P3 = >10 cfu/plate.3,11,13,17,18,20 Cats with active infections are characterized by fungal cultures with high cfu/plate (ie, P3).
Mycological cure was defined as two consecutive weekly fungal culture scores of P0 at any time during the treatment or follow-up periods with all subsequent fungal culture scores not exceeding P1. Time to mycological cure was calculated as the number of weeks between the start of treatment and the second of the two consecutive negative fungal scores of P0. The assumption was made that subsequent scores of P1, particularly in the absence of any new clinical lesions or changes in Wood’s lamp examination findings, resulted from environmental contamination or lack of disinfection of the haircoat rather than true relapse or reinfection.
Primary lesion scores
Clinical lesions were evaluated on the same schedule as fungal cultures under room lighting and with the aid of a Wood’s lamp. Assessments included scoring of erythema, induration (dermal thickening) and scale/crust (Table 1). 20 Full resolution (ie, scores of 0) of erythema, induration and scale/crust was considered a ‘clinical cure’.
Description of erythema, induration, scale/crust and Wood’s lamp scoring system

Wood’s lamp scores
Wood’s lamp examinations were performed on the same schedule as fungal cultures, using a hand-held plug-in electric Wood’s lamp with built-in magnification. Hair fluorescence was scored at three locations: base, mid-shaft and tip using a 4-point scale (Table 1).13,20 It has been shown that as the hair continues to grow after the organism has been killed, fluorescence will first disappear at the base of the hair, followed by the mid-shaft and eventually from the tip.21,22 Therefore, a negative Wood’s lamp examination was defined as: yes (score of 0 on the base and mid-shaft of the hair) or no (score of 1, 2 or 3 on the base and/or mid-shaft of the hair).
Statistical analysis
For fungal culture scores and Wood’s lamp scores, differences between the two treatments were evaluated using a generalized linear mixed model using GLIMMIX of SAS, assuming a binomial distribution and using a logit link.
For ‘time to mycological cure’, values were evaluated as ordinal outcomes. Cats that did not reach mycological cure by the study conclusion were considered censored and assigned to the category ‘>9 weeks’. Differences between the two treatments were evaluated using a generalized linear mixed model using GLIMMIX of SAS, assuming a multinomial distribution and using a cumulative logit link.
The models included treatment group as a fixed effect and room and cohort nested in room as random effects. A P value <0.05 was determined significant.
For lesion scores (erythema, induration, scale/crust), the effectiveness of itraconazole in improving the total lesion score for each of the assessed primary lesions at each examination day was calculated according to the formula:

Results
The experimental challenge procedure used was effective at inducing clinical disease in all 80 cats, as all had positive fungal culture scores of P3, had positive Wood’s lamp examinations and had clinical lesions prior to treatment initiation.
Over the course of the study, there was a slightly greater incidence of vomiting (5 vs 4, respectively) and diarrhea (8 vs 4, respectively) in treated vs control cats. Two itraconazole-treated cats had multiple episodes of mild, transient hypersalivation during the study.
Additionally, there were statistical differences (P <0.05) for alanine aminotransferase, calcium, potassium, sodium and total bilirubin. However, treatment differences in clinical pathology values before and after treatment with itraconazole appeared to be transient and of little, if any, clinical significance, and, despite differences, most values remained within normal laboratory reference intervals for the age of the cats.
Mycological cure
The median and mean fungal culture score at the start of the study for both groups was 3. Median and mean fungal culture score decreased for the itraconazole-treated cats from week 4, and reached ‘0’ and ‘0.25’, respectively, at week 8. The mean and median for the control group remained at 3 throughout the study (Figure 1).

Effects of itraconazole treatment on median fungal culture score
Itraconazole-treated cats had significantly greater (P = 0.0003) and faster (P = 0.0003) mycological cure rates compared with controls (Table 2). At the end of the follow-up period (week 9), 24/40 (60%) itraconazole-treated cats had reached mycological cure vs only 1/40 (2.5%) control cats. Furthermore, 36/40 (90%) treated cats had at least one negative fungal culture by the end of study vs only 3/40 (7.5%) control cats.
Effects of itraconazole treatment on time to mycological cure (number of cats cured/week)

Data were censored at 9 weeks. The category ‘>9’ represents animals not cured by the end of study
Lesion scores
The first cat with a clinical cure was recorded at week 2 and week 6 for the itraconazole-treated group and control group, respectively. For both groups, the prevalence of clinical cure peaked at the end of study (week 9) with 39/40 (97.5%) itraconazole-treated cats and 6/40 (15%) control cats achieving clinical cure.
After completing the 5 week treatment period, improvement in primary lesion scores for the itraconazole-treated group, relative to controls, was >90%. By the end of the study, 40/40 (100%), 39/40 (97.5%) and 40/40 (100%) itraconazole-treated cats had scores of ‘0’ for erythema, induration and scale/crust, respectively, vs 28/40 (70%), 10/40 (25%) and 21/40 (52.5%) control cats, respectively (Figure 2).
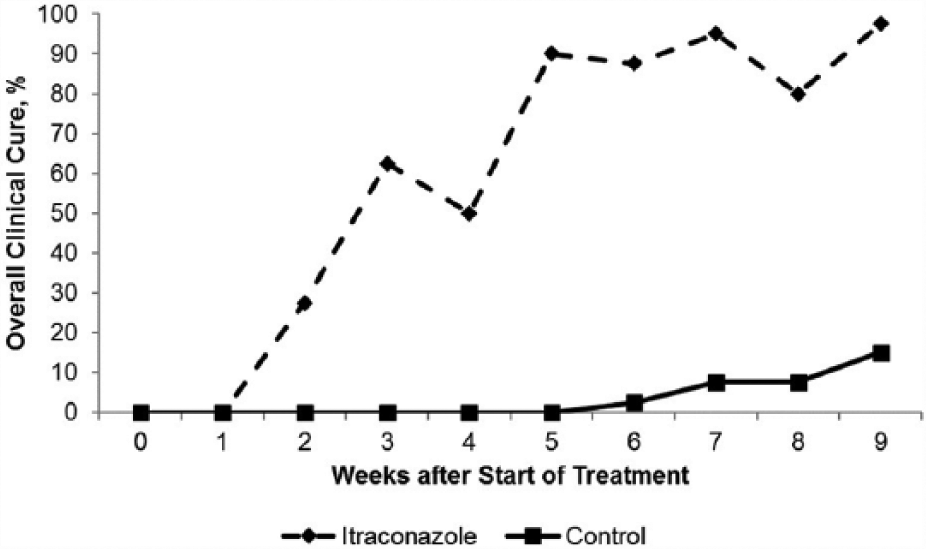
Effects of itraconazole treatment on overall clinical cure
Wood’s lamp examinations
By week 9, the incidence of negative Wood’s lamp examinations was significantly greater (P <0.0001) for itraconazole-treated cats (39/40 cats [97.5%]) compared with controls (6/40 [15%]) and followed the same pattern of improvement as primary clinical lesions (Figure 3). The first negative Wood’s lamp examinations were recorded at week 1 for the itraconazole-treated group (9/40 cats; 22.5%) and week 7 for the control group (1/40 cats; 2.5%) (Figure 3). Statistical separation (P <0.0001) between groups was achieved for Wood’s lamp scores at the base of the hair 2 weeks after the start of treatment. By week 4, all 40 itraconazole-treated cats had negative Wood’s lamp scores at the base of the hair shaft, and remained negative throughout the rest of the study. Fluorescence at the hair tips was noted in cats that had reached mycological cure; hair tip fluorescence was greater (P <0.05) for the itraconazole-treated group.

Effects of itraconazole treatment on negative Wood’s lamp examination
Discussion
Current industry treatment protocols for feline dermatophytosis all recognize the need for concurrent systemic and topical antifungal therapy. 4 Systemic therapy eradicates the infection from within the hair follicle where topical applications are unlikely to penetrate. Topical therapy kills viable fungal spores adhered to the haircoat that could potentially result in positive fungal culture scores and act as a potential source of infection for in-contact animals and people. In the present study, 11/16 itraconazole-treated cats that did not reach mycological cure had fungal culture scores of either P0 or P1 over the final 2 weeks of study. It is likely that concurrent topical therapy would have eradicated some of these positive culture scores by eradicating the haircoat of spores, and would have resulted in a greater number of cats achieving mycological cure before the end of study.
The reason for the lack of complete mycological cure in 16 itraconazole-treated cats could be attributed to multiple factors, including concluding the study before an additional final culture could be obtained, environmental contamination and/or lack of disinfection of the haircoat, or non-response to treatment. The latter point appears relevant to one cat that had lesions and was Wood’s lamp and culture positive (P2) at the end of study. For the other 15 cats that did not achieve mycological cure, persistence of spores on the haircoat in the absence of topical therapy or reinfection from a contaminated environment may have also contributed to the persistent culture-positive status in some of the cats with fungal culture scores of P1 or cats that showed ‘flip flopping’ culture results (between P1 and P0). Fluctuating culture results in cats at the end of treatment is common.3,13
In this study, clinical cure was achieved in 39/40 (97.5%) itraconazole-treated cats vs only 6/40 (15%) control cats. The rapid clinical response observed in itraconazole-treated cats was apparent as early as 1 week into treatment and speaks to the rapid antifungal effects of itraconazole. Itraconazole-treated cats began to achieve clinical cure as early as week 2 of the study, whereas the first control cat did not achieve clinical cure until week 6 of the study. Similar to previous studies, clinical cure in the current study was achieved earlier and cure rates were greater than mycological cure rates, suggesting that clinical cure will often precede mycological cure.8,11,13
Wood’s lamp examination findings in this study were similar to those reported in both experimental and spontaneous disease studies and were helpful in monitoring the experimental infections.3,11,13 Much of the previous research, however, reported results only as positive or negative based on fluorescence on the hair shaft, without accounting for the hair-shaft location of the fluorescence.3,7,8,11,17,18 As the hair continues to grow after the organism has been killed, fluorescence will first disappear at the base of the hair, followed by the mid-shaft and eventually from the tip.21,22 Thus, in the absence of fluorescence at the base of the hair, fluorescence mid-shaft or at the tip of the hair is not a sign of active infection. Wood’s lamp examinations revealed rapid resolution of fluorescence at the base and mid-shaft of the hairs as early as week 1 of treatment. In addition, previously infected, but culture-negative, hairs often had residual fluorescence on the hair tips, as noted in other studies.21,22 The high rate of negative Wood’s lamp examinations at the base and mid-shaft of the hair and the rapid rate at which scores improved in the itraconazole-treated cats lends to the high level of effectiveness of itraconazole in the present study.
Conclusions
This is the first controlled study to closely monitor the effects of oral itraconazole administered at 5 mg/kg using a pulse-dose (week on–week off) regimen, in the absence of topical antifungal therapy, for the treatment of dermatophytosis. The results demonstrate that itraconazole produced clinical cure and facilitated and reduced the time to mycological cure compared with untreated controls. The results demonstrate the potential for systemic itraconazole use as a cure for M canis dermatophytosis, and underscore the need for adjunctive topical therapy to eliminate infection and reduce the risk of exposure of in-contact animals and people.
Footnotes
Acknowledgements
The authors thank Dr Manuelle De Bock for her help in designing and conducting the pilot study that preceded this research. Additionally, the authors thank Dr Steve Radecki for performing statistical analysis and Dr William Ryan for reviewing and editing this paper prior to submission. The authors also thank the staff at Kingfisher International for caring for the animals and conducting the study.
Author note
Data are on file at Elanco Animal Health.
Conflict of interest
Chris Puls, Aaron Johnson, Kelly Rosenkrans and Lisa Young are employees of Elanco. Karrie Young and Jonathan Hare had no conflict of interest outside of being contracted researchers to conduct the study. Karen Moriello was a paid consultant for Elanco.
Funding
This study was funded by Elanco Animal Health.
