Abstract
Practical relevance:
Infectious disease in feline patients often presents a diagnostic challenge. This article reviews the most relevant viral, bacterial and protozoal diseases and their cutaneous manifestations. Many of the diseases discussed have overlapping presentations or may mimic more common noninfectious disease processes. The purpose of the article is to reinforce knowledge of common and uncommon infectious diseases, help practitioners identify possible infectious dermatoses, create a comprehensive and prioritized differential list, and provide guidance for the diagnosis of these diseases. A working knowledge of these clinical syndromes is important if what is thought to be a case of a common disease does not respond to conventional management.
Audience:
This review is aimed at veterinarians who treat cats and especially those with an interest in feline dermatology. Tables are included to allow the reader to formulate a concise list of differential diagnoses for clinically similar presentations. The diagnostic approach to a case of ulcerative facial dermatitis is reviewed in a Case Notes quiz.
Evidence base:
This article includes up-to-date information regarding dermatologic manifestations of less commonly encountered feline cutaneous infectious diseases. Information has been drawn from the published, peer-reviewed literature and the most recent textbook chapters with a particular aim of describing and differentiating clinical lesions and the diagnostic approach to cutaneous disease, especially in unusual cases.

Viruses – and associated cutaneous disease

Feline immunodeficiency virus
Feline immunodeficiency virus (FIV) is a retrovirus that results in an increased risk of neoplasia, and susceptibility to infection.1–3 There are no specific dermatologic conditions associated with FIV status; however, affected cats may present with skin infection or cutaneous neoplasia. Many opportunistic infections (including cryptococcal and mycobacterial infections) have been reported in FIV-positive cats, but there does not appear to be an increased prevalence of most infections compared with healthy cats. Although infection rates may be similar, FIV-positive cats may have more severe and prolonged clinical illness.4,123,129,131 FIV-positive cats have a five- to six-fold greater risk of developing multicentric or extranodal lymphoma, although cutaneous epitheliotrophic lymphoma remains uncommon. Other cutaneous tumors, especially Bowenoid in-situ carcinoma (BISC)/squamous cell carcinoma (SCC), have been reported with high rates (see ‘Bowenoid in-situ carcinoma’ box on page 1150).5–8

Feline leukemia virus
Feline leukemia virus (FeLV) is an immunosuppressive, oncogenic gamma retrovirus that is passed between cats after prolonged contact.9,10 Immunosuppression is more severe than with FIV infection and FeLV-positive cats are at a greater risk of opportunistic infections and neoplasia.5,11–13 Similar to FIV-positive cats, the clinical course of opportunistic infections may be more severe. 14 Otitis externa and skin infections due to other underlying causes may be persistent or recurrent.9,15
FeLV infection is associated with a number of different neoplasms in cats and increases the risk of developing lymphoma by 60-fold. However, an association between FeLV status and cutaneous lymphoma is questionable.5,16,17 FeLV status has also been associated with the development of BISC/SCC (see box above). 18
Cutaneous horns are a clinically distinct entity in FeLV-positive cats (see box below). Another clinical syndrome, termed giant cell dermatosis, occurs rarely in FeLV-positive cats. Affected cats are pruritic with scaling, crusting, alopecia and variably ulcerated lesions most frequently affecting the head and face. Less common sites have included the trunk, legs and footpads. In all reported cases, the affected cats either presented with systemic signs or deteriorated quickly after diagnosis and died or were euthanized.23,24 Histopathologic features include syncytial cell formation and giant multinucleated keratinocytes. Positive staining for FeLV viral antigen, gp70, can be found throughout the epidermis and follicular outer root sheath. 22 The differential list should include all other causes of head/neck ulcers and crusts (see Table 1).
Differentiation of ulcerative and crusting dermatoses of the head and neck

SCC/BISC = squamous cell carcinoma-Bowenoid in-situ carcinoma; CNS = central nervous system; FeLV = feline leukemia virus
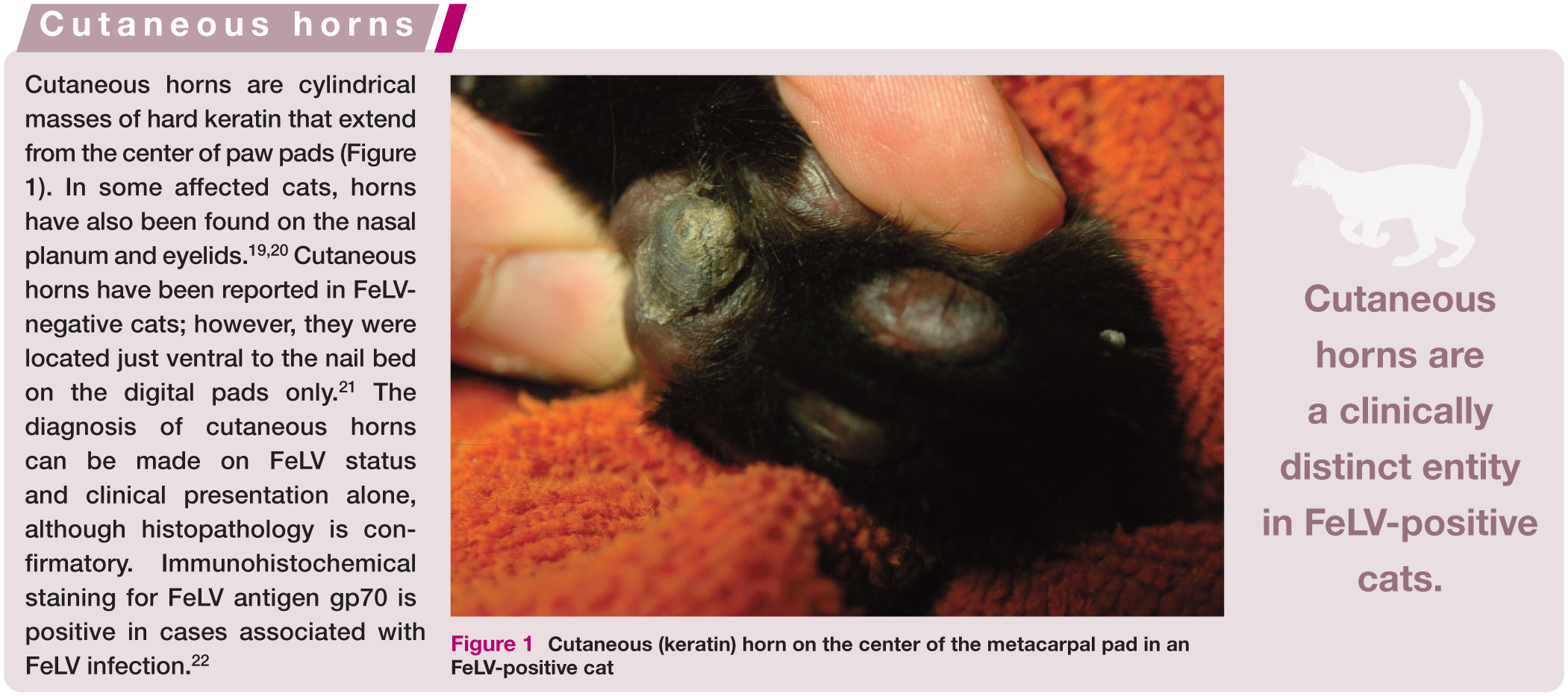
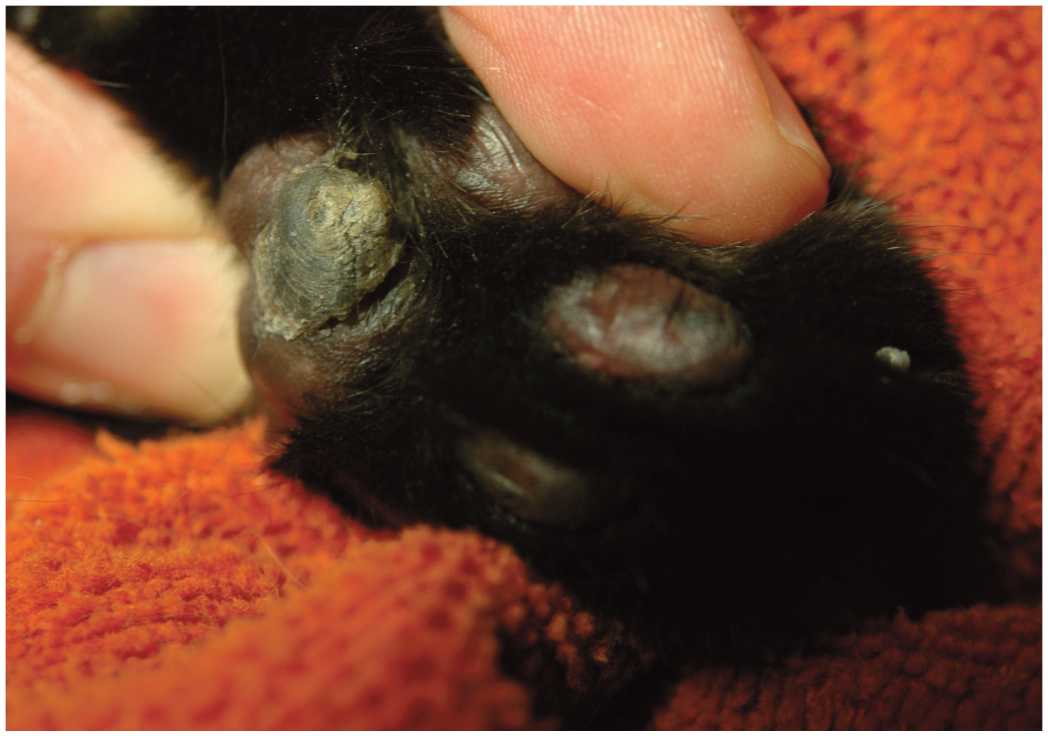
Cutaneous (keratin) horn on the center of the metacarpal pad in an FeLV-positive cat

Feline papillomavirus
Papillomaviruses are small, ubiquitous and hardy, non-enveloped, double-stranded DNA viruses. To date, four unique papillomaviruses have been identified and sequenced in cats (Felis catus papillomavirus [FcaPV] 1–4).27–31 A fifth virus, previously designated feline sarcoma associated papillomavirus (FeSarPV), amplified from feline sarcoids, is now thought to be a bovine papillomavirus (BPV-14). 32 In cats, papillomaviruses are associated with the following syndromes: oral papilloma (FcaPV-1, FcaPV-2), exophytic papilloma (FcaPV-1), viral plaques (FcaPV-1, FcaPV-2), BISC (FcaPV 1-4) and feline sarcoids (BPV-14).25,33–35
FcaPV-2 has been strongly implicated in the pathogenesis of a high proportion (approximately 60%) of nasal planum SCCs (see BISC box above). A recent study demonstrated a significantly longer median survival time in cats with p16-positive SCCs (consistent with a papillomavirus etiology) when compared with p16-negative SCC, making identification of the etiology clinically relevant. 36
Oral and cutaneous papillomas have been rarely reported in domestic cats. Oral papillomas may present as multiple raised pale nodules or plaques on the ventral surface of the tongue and may mimic feline eosinophilic granulomas or feline oral SCC. 33 Two reports of cutaneous papillomas have been published and, in both, lesions appeared as exophytic nodules in sites of previous skin damage.37,38
Viral plaques are uncommon. No breed or sex predilection has been identified. Adults are typically affected, but reported ages have ranged from 7 months to 15 years. Immunosuppression seems to play a role and viral plaques may represent precursors to SCC (see BISC box on page 1150). Viral plaques are small (<8 mm), pigmented or non-pigmented lesions that often have a scaly, crusted or irregular surface and are found on haired skin.39,40 Differential diagnoses include actinic keratosis, BISC or SCC.
Feline sarcoids or fibropapillomas are rare papillomavirus-induced neoplastic proliferations of fibroblasts associated with BPV-14 that may be acquired after exposure to cattle. 32 Sarcoids are slowly growing, firm, exophytic alopecic nodules that may become ulcerated. The nodules are singular or multiple and are most common on the nose, upper lip and digits of young cats.41–43 Differential diagnoses include other cutaneous neoplasms as well as inflammatory and infectious abscesses and granulomas.
Biopsy typically allows diagnosis of papillomaviral etiology. Papillomavirus-infected cells (termed koilocytes) are characterized by cytoplasmic clearing, pyknotic nuclei and increased blue–grey cytoplasm.39–44 The presence of papillomavirus can be confirmed with immunohistochemical staining to detect the L1 antigen. Molecular techniques including PCR have been used to detect viral DNA in lesions where viral replication is not active.34,45
Feline calicivirus
Feline caliciviruses are non-enveloped, single-stranded RNA viruses that are common upper respiratory pathogens of cats. Most field strains produce relatively mild disease. However, highly virulent strains cause high fevers, severe respiratory disease and cutaneous signs. 46 Mortality for these strains ranges between 33% and 60%, which is in contrast to less virulent stains which produce high morbidity but low mortality. Dermatologic lesions associated with highly virulent calicivirus infection are striking and include edema of the head and limbs, skin ulcers and crusting (often of the nose, lips, periocular region, ears and footpads). Petechiae and ecchymoses have been noted in some coagulopathic cases.47–49 In any adult cat with acute systemic illness including respiratory signs, and head and limb edema, suspicion for feline calicivirus should be high. Diagnosis is based on virus isolation or PCR from oropharyngeal or conjunctival swabs. 50
Feline herpesvirus-1
Feline herpesvirus-1 (FHV-1) is widespread in the feline population. Infected cats become lifetime carriers, with inactive virus inhabiting the trigeminal ganglion. Recrudescence and increased viral shedding has been associated with periods of stress, illness and treatment with immunosuppressive medications (including glucocorticoids). 47 Although over 90% of healthy, owned cats (US population) have been found to be seropositive to the virus, the incidence of clinical disease is low, and even fewer cats will ever show signs of herpesvirus dermatitis.51,52
Most cats affected by herpesvirus dermatitis are adults (average 4.5 years). 53 There is no breed or sex predilection and patients may or may not have a history of respiratory or ocular disease. Affected cats develop small vesi-les; however, primary lesions are quickly disrupted, resulting in erosions, ulcers and crusts that can be painful or pruritic. Lesions are predominantly found on the face (especially the nasal planum [Figure 2a], bridge of the nose, perinasal or periocular areas), but have also been reported on the trunk and limbs.53–55

(a) Herpesvirus dermatitis on the nasal planum of a cat, characterized by a full-thickness ulcer affecting haired and non-haired skin. Note the increased corneal opacity on the left as a result of herpesvirus stromal keratitis. (b) Histopathologic image of herpesvirus dermatitis. Eosinophilic inflammation is seen surrounding hair follicles, and there are large intranuclear inclusion bodies within keratinocytes (arrows)
Histopathology is required for diagnosis. Histologically, the lesions are characterized by ulceration and necrosis of the epidermis, with eosinophilic inflammation. Intranuclear inclusion bodies can be identified within keratinocytes (Figure 2b). 56 Given the strong eosinophilic component in these cases, and difficulty identifying inclusion bodies, these lesions may be mistaken for allergic dermatitis or eosinophilic granuloma complex. Confirmatory testing is often warranted and may include PCR or immunohistochemistry (the current diagnostic standard). 57–59 Differential diagnoses for herpesvirus dermatitis include other ulcerative and crusting diseases of the head and neck (see Table 1).

Feline cowpox virus
Cowpox virus is a double-stranded DNA virus that affects cats, cattle, humans, dogs and other mammals. Disease is most commonly reported in the UK and Western Europe;65–67 there have been no reports from North America. The virus is spread through bites of rodent reservoir hosts or inoculation into wounds. Rural cats that hunt tend to be affected, with most infections occurring in the summer and fall. 67 Human infections are frequently reported in association with cats.68–71
After inoculation, local replication results in a primary lesion. Secondary lesions occur distantly following transient viremia. Initial lesions are erythematous, ulcerative, crusted macules or plaques, whereas secondary lesions comprise papules or nodules that may ulcerate. 72 In some instances, severe necrotizing facial dermatitis has been reported. 73 Rarely, affected cats may develop other systemic signs including necrotizing pneumonia that can result in death.74,75
Only cats that live in or have recently traveled to the UK or Western Europe should be considered suspects. Diagnosis can be achieved through identification of the virus in tissue samples. 76 Lesions are characterized by extensive ulceration and necrosis, with ballooning degeneration of keratinocytes. Brightly eosinophilic cytoplasmic inclusion bodies may be identified within keratinocytes. 56
Differential diagnoses include atypical bacterial and fungal infections, dermatophytosis, eosinophilic granuloma complex and neoplasia (SCC, lymphoma). If the disease presents mainly on the head or neck, all other causes of ulcerative facial dermatitis must be considered (Table 1).
Bacteria – and associated cutaneous disease

Mycobacteria
Mycobacteria are aerobic, non-motile, pleiomorphic, acid-fast bacteria that cause a number of clinical syndromes. They can be divided into two main types – slow-growing fastidious species and rapid-growing species – based on the relative ability and speed with which they grow in culture.
The two types of infections have distinctly different clinical presentations: slow-growing species are typically associated with development of dermal or subcutaneous nodules, whereas rapid-growing species most commonly affect the panniculus and subcutaneous adipose tissue. Some species within each category are associated with unique clinical syndromes, including historical data, body site distribution and prognosis. 77 These features are discussed briefly below. The reader is encouraged to refer to previously published articles in this journal for more detailed information on specific species.
Slow-growing species
Slow-growing mycobacterial species within the Mycobacterium tuberculosis complex (MTBC), including Mycobacterium tuberculosis, Mycobacterium bovis and Mycobacterium microti, cause feline tuberculosis. These infections most commonly present as dermal nodules with or without ulceration and/or submandibular lymphadenopathy. 78 However, infection may also affect other body systems including the respiratory and alimentary tracts.77,79,80 The presence of respiratory, gastrointestinal or cutaneous signs may reflect the mode of transmission. However, most cats are thought to become infected through bites from prey reservoir hosts, resulting in cutaneous lesions on the face and limbs. Dissemination from cutaneous lesions or other sites may occur. 79 Cats are most susceptible to M microti and M bovis; M tuberculosis infections are rare. Cases are most frequently reported in Great Britain where M bovis is endemic in badgers and cattle and M microti in rodents.81,82 However, MTBC infections occur worldwide.78,83–86
The Mycobacterium avium complex (MAC), including M avium and Mycobacterium intracellular, are slow-growing saprophytic species that mainly cause infections in immunocom-promised patients.79,87,88 MAC infections may cause cutaneous nodules or disseminated disease.79,89 Disseminated MAC infections have been reported more commonly in Siamese and Abyssinian cats.90,91

Rapid-growing species
Infections caused by rapid-growing species are typically opportunistic, arising as a result of inoculation of mycobacteria into tissue through wounds or contamination of surgical sites in healthy animals. Mycobacterial panniculitis is a distinct clinical presentation of infection by rapid-growing species. This syndrome commonly affects the inguinal fat pad, but has also been described on the flanks and around the base of the tail. Subcutaneous tissue becomes thickened and adherent to the dermis, resulting in the development of nodules and draining tracts (Figure 3a). Systemic spread is rare, but affected cats may be febrile, lethargic, anorexic and painful.79,106–108 Differentials include nocardiosis and lymphan-giosarcoma.

(a) Mycobacterial panniculitis caused by a fast-growing Mycobacterium species that originated from a spay incision infection. Note the serpiginous tracts radiating away from the center of the lesion and serosanguineous fluid discharging from fistulas. (b) Mycobacterium smegmatis from a culture sample. x 100, modified Ziehl-Neelsen stain
Diagnosis is based on the cytologic and histologic appearance of organisms, culture and molecular techniques. Negative staining bacilli are detected on cytologic or biopsy samples, and will show positive staining when an acid-fast stain is applied (Figure 3b). Mycobacterial species may be cultured using specialized media. MTBC and slow-growing non-tuberculous mycobacteria (including MAC) may take many weeks to grow and some fastidious species will not grow at all. 81 For species identification, molecular diagnostics (PCR, MALDI-TOF) are available through specialized laboratories.79,96,98
Identification of mycobacterial species can be challenging. However, identification can help direct antimicrobial choices and provide prognostic information. Additionally, since MTBC infection represents a public health risk, it is important to rule these out as causative organisms.
Nocardia and Actinomyces species
Nocardia species are aerobic, branching, filamentous, gram-positive, partially acid-fast bacteria. A number of different species have been isolated from cats.109–113 These organisms are found ubiquitously in the environment, associated with decaying organic matter, dust and fecal matter. 114 Actinomycetes are anaerobic or microaerophilic, filamentous, gram-positive, non-acid-fast bacteria. They are considered normal inhabitants of the oropharynx and gastrointestinal tract. 115
Disease occurs when Nocardia or Actinomyces species are inoculated into the skin from bite wounds or penetrating foreign bodies. Infections occur in otherwise healthy animals, although immunosuppression may play a role in the development of chronic infections. 116 Actinomycetes are most commonly isolated from cutaneous non-healing wounds and abscesses; intra-abdominal and thoracic infections have also been reported.117–120 Nocardiosis is occasionally associated with severe disseminated disease and pneumonia; however, the cutaneous/subcutaneous form is the most common (>75% of reported infections) (Figure 4a). 112

(a) Nocardia species infection affecting the base of the tail and caudal lumbar region, likely due to fight wounds. This close-up view shows coalescing ulcers and fistulae with serous discharge. (b) Filamentous acid-fast Nocardia species on a cytology preparation. x 100, modified Ziehl-Neelsen stain. Courtesy of Dr Stephen Cole
Nodules and abscesses, with ulcers, draining tracts and cellulitis, will appear anywhere the bacteria are inoculated, but occur most commonly on the extremities, head and neck, and inguinal area. So-called ‘sulfur granules’ may be present in association with Actinomyces species (see box above). 115 Infection in the inguinal region may be extensive and mimic rapid-growing mycobacterial panniculitis.112,121


Diagnosis is often made through a combination of cytologic, histologic and microbiologic techniques. Filamentous bacteria may be identified on cytologic samples (Figure 4b). Histopathology reveals nodular to diffuse pyogranulomatous dermatitis and panniculitits, with or without sulfur granules. Special stains (gram, Brown–Brenn) can be utilized to better visualize the bacteria and a Fite–Faraco modified acid-fast stain may be useful to differentiate Nocardia from Actinomyces species.114,115 Splendore–Hoeppli reaction may be identified surrounding the organisms in tissue samples (see box above). 122 Culture may be necessary to differentiate Actinomyces species from Nocardia species (see box below for comparison of features). Differential diagnoses include other causes of cutaneous nodules and draining tracts (see box on left).

Fungi – and associated cutaneous disease

Cryptococcus and Aspergillus species
Cryptococcus species are dimorphic basidiomycetous fungi and the cause of the most common systemic mycosis of cats. There are at least 19 species described; of these, Cryptococcus neoformans and Cryptococcus gattii cause the majority of disease. Infection typically occurs through inhalation of spores from environmental sources. 124 C neoformans is found in soil contaminated with pigeon excrement or decaying plant material. 125 C gattii is found ubiquitously in the environment. 126
Affected cats are typically young adults with access to the outdoors. However, approximately 25% of affected cats in the US are reported to be indoor-only. 127 After inhalation, spores within the nasal cavity spread through the bloodstream to other tissues or ascend to the central nervous system through the cribriform plate. 128 Cats can present with nasopharyngeal, central nervous system, ocular, cutaneous and/or systemic disease. While nasal cavity disease is most common (occurring in 40–90% of cases), cutaneous signs are reported in approximately half of cases, with the majority having signs localized to the nasal planum or bridge of the nose.127,129–131 Classically, single or multiple fluctuant to firm nodules present on the bridge of the nose, which may ulcerate or develop draining tracts (Figure 5a). Similar cutaneous lesions may present elsewhere as well.127,129 Sino-nasal/sino-orbital aspergillosis has been reported uncommonly, but with similar clinical signs, and may be included as a differential especially when facial deformation is noted. Affected patients commonly present with nasal discharge and sneezing. Exophthalmos and paranasal swelling is seen frequently in patients with sino-orbital disease, and draining tracts have been reported in patients with sinonasal disease. 132

(a) Cryptococcosis affecting the bridge of the nose and resulting in facial deformity. (b) Appearance of Cryptococcus species on a cytologic sample from a cat. Numerous organisms are present within a foamy macrophage (arrow) and free in the sample. Organisms may vary in size (4–15 urn) and have a thick, non-staining capsule. x 100, Wright-Giemsa stain. Courtesy of Dr Koranda Walsh
Diagnosis can be made by identifying the organism in cytologic or tissue samples. Cryptococci are non-staining pleomorphic yeast-like organisms, 4—15 μm in diameter. They show narrow-based budding and are surrounded by a refractory capsule (Figure 5b). In biopsy samples, these organisms can be identified using special stains (PAS, GMS or Fontana-Masson). Confirmatory culture should be performed using biopsy samples. 124 Latex agglutination tests for capsular antigen in serum can be used to identify affected cats and monitor response to therapy. Infected cats typically have high titers, though titers can vary greatly between patients. 133 Cryptococcosis should, therefore, not be ruled out based on low or negative titers, which may occur in some patients with primary ocular or CNS disease.127,134
Aspergillosis is typically diagnosed by identification of Aspergillus organisms on cytologic or histologic examination. Samples may be obtained using fine-needle aspiration or biopsy of the lesion. These findings must be confirmed by growth of the fungi in culture.132,135
Blastomyces and Histoplasma species
Blastomyces dermatitidis is a dimorphic fungus found as a soil contaminant in endemic areas (Southeastern USA, around the Great Lakes, and Ohio and Mississippi river valleys). 136 Although disease is more common in dogs, infections are occasionally reported in cats from endemic areas. Respiratory, ocular and cutaneous signs are found most commonly, although neurologic signs, including mentation changes, ataxia and circling, are present in up to 41% of affected cats.137–140 Dermatologic lesions are nodules with or without draining tracts, and are most commonly reported on the head, limbs or trunk.138,139
Histoplasma capsulatum is a dimorphic fungus found in nitrogen-rich soil (especially that contaminated by bat or bird excrement). It has been reported on almost all continents, but is endemic in the Midwestern and Southern United States. Histoplasmosis is more commonly reported in cats than blastomycosis. 141 Cats most frequently present with respiratory (40%) and ocular signs (25%). The skin is infrequently affected but cutaneous nodules can occur. 142
Suspicion of blastomycosis and histoplasmosis should increase in patients presenting with cutaneous lesions and other systemic signs. The organisms may be detected on cytology or histopathology. Histoplasma species are often abundant in macrophages and are small (2–4 μm) round or oval bodies with a clear halo (Figure 6). Blastomycetes are thick walled, broad-based budding yeasts approximately 8–12 μm in diameter. In biopsy samples, organisms are best found using periodic-acid-Schiff (PAS) or Gomori’s methenamine silver (GMS) stain. Culture should only be performed in reference laboratories with appropriate facilities as the mycelial form in culture poses a risk for human infection.136,141 Antibody and antigen serum and urine testing has been utilized in cases of histoplasmosis and may be useful for monitoring clinical progression.143,144 Similarly, a urine antigen test is available and useful for diagnosis and monitoring of blastomycosis. 145 However, since there may be cross-reactivity between systemic mycoses, these results should be interpreted carefully. 146
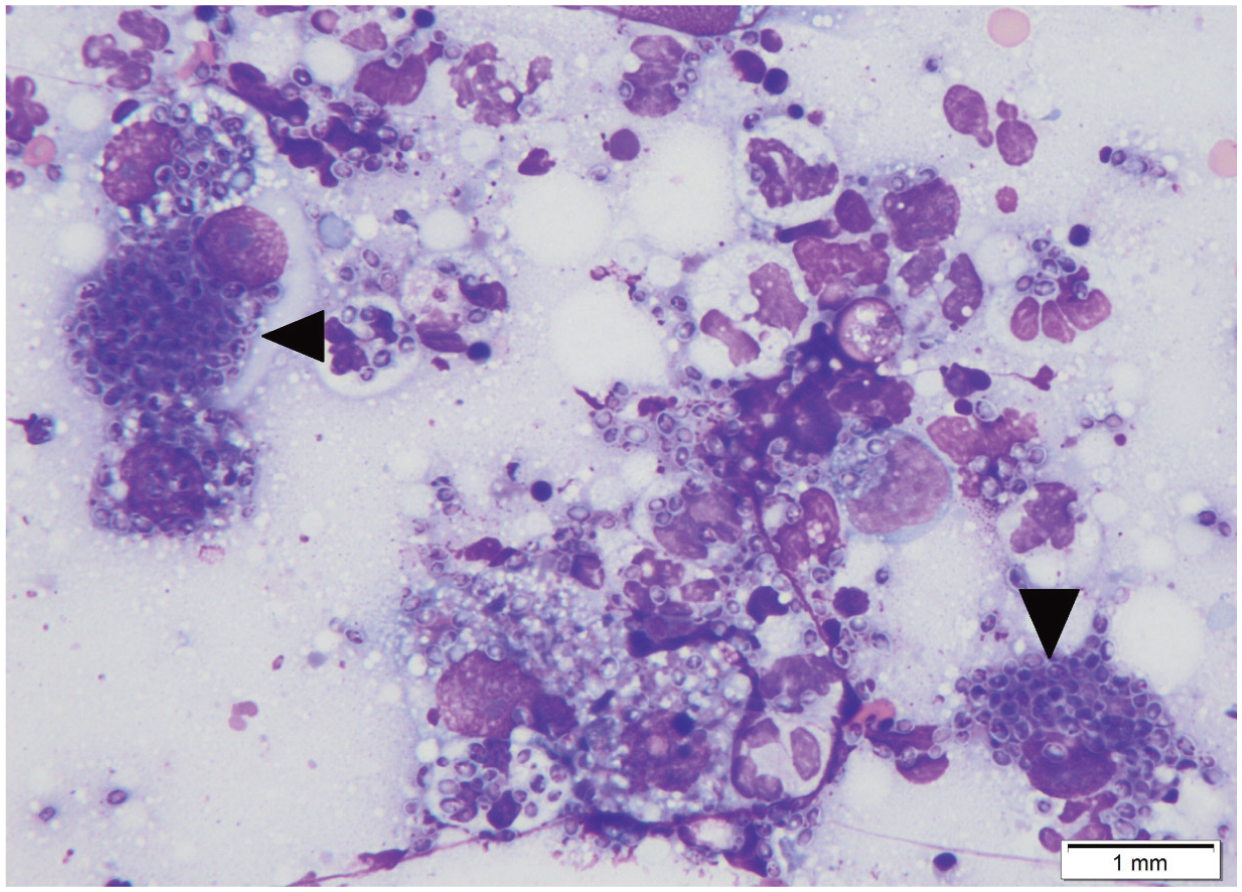
Appearance of Histoplasma capsulatum on cytology. Organisms are numerous within macrophages and multinucleated cells (arrows). These are small (2-4 um) round or ovoid yeasts with a thin cell wall. x 100, Wright-Giemsa stain. Courtesy of Dr Koranda Walsh
Sporothrix species
Sporotrichosis is caused by dimorphic saprophytic fungi within the Sporothrix schenckii complex (which include at least six species). Sporothrix species favor warm, humid climates and soil rich in decaying organic matter. Many mammals, including humans, are susceptible to infection. Cats are considered highly susceptible and may facilitate spread of the fungi in endemic areas. 147 Large outbreaks in humans have been closely associated with disease in cats in Brazil.148–150 Sporothrix species infections occur worldwide, although the disease is considered endemic in Mexico, Brazil, Uruguay, Peru, China, Japan, India and South Africa. The disease is uncommon in the United States and Europe, although sporadic outbreaks have occurred.151,152
Young adult intact male outdoor cats with a history of fighting are at the highest risk of developing sporotrichosis. Infection occurs when fungi are inoculated into skin wounds or through inhalation of spores. Affected cats may display a wide spectrum of signs, ranging from subclinical to severe. The overwhelming majority of patients show cutaneous lesions, which appear as small papules or nodules that may ulcerate and drain. Large areas of ulceration and necrosis that expose underlying muscle and bone may also be seen (Figure 7a). Lesions are most common on the head (especially the nose) and limbs, although some cases show mucosal involvement.153,154 Evidence of dissemination has been reported in 50–70% of cats; lungs, liver, spleen, kidneys, lymph nodes and/or testicles may be involved.155,156 Respiratory signs are the most common extracutaneous signs.153,157

Sporotrichosis affecting the bridge of the nose in a cat. Note the deep ulcer with exposure of underlying bone.
Diagnosis is based on identification of the organism in affected tissues or fungal culture. Cytologic analysis of exudate may reveal the presence of large numbers of organisms. Sporothrix species are cigar-shaped yeasts (4–6 x 5–9 μm) with narrow-based budding, often found within neutrophils and macrophages (Figure 7b). 147 Organisms may be identified in close to 80% of cytologic samples and are typically abundant in biopsy samples as well (Figure 7c).158,159 Fungal culture (biopsy samples are preferred) can be performed to isolate the fungi. An ELISA for antibody against cell wall proteins, with a high sensitivity and specificity, has been developed but is not widely used. 160

Protozoa – and associated cutaneous disease

Toxoplasma gondii
Toxoplasmosis is a systemic coccidial disease for which cats are the definitive host. Cutaneous lesions have been reported in only a few cases and are characterized by single or multiple nodules that may ulcerate. In these cases, organisms have been visible on cytologic samples and biopsy specimens.161–163 Importantly, severe disseminated infections have been reported in association with immunosuppressive states induced by concurrent diseases and medications such as corticosteroids and ciclosporin modified.164,165 It has been clearly demonstrated that cats which become infected by Toxoplasma gondii after commencing ciclosporin therapy are at the highest risk of developing severe or disseminated disease. 166
Leishmania species
Leishmaniosis is a protozoal infection that typically affects humans and dogs in endemic areas. The disease is transmitted by blood-sucking sand flies and occurs where these flies are found in high numbers (the Middle East, South and Central America, the Mediterranean basin and Portgual). 167 Although disease is rare in the USA, Leishmania mexicana is considered endemic in Texas. 168
Cats are considered resistant to disease and reports of infections are infrequent. In Southeast Brazil, surveillance has revealed low numbers of positives on samples from healthy cats (4—6%).169,170 However, when cats with dermatologic lesions were examined from a similar area, close to half (27/55) were found to be positive. These results indicate that patients presenting with dermatologic lesions in endemic areas should be closely evaluated for Leishmania species. 171
Cats with cutaneous leishmaniosis typically present with focal alopecia, nodules, crusting and scaling, most often on the head (especially the pinna), limbs and trunk. Systemic signs, particularly lymphadenopathy, dehydration, weight loss and diarrhea, often accompany skin lesions.171–175
This disease should be considered as a differential for head/neck ulcers in patients that live in or have traveled to endemic areas. The most straightforward method of diagnosis is via identification of the organisms within aspirates or tissue samples (spleen, lymph nodes, bone marrow or liver). 171 Samples from affected skin can be submitted for histopathologic analysis and immunohistochemistry. Histologic diagnosis has relatively low sensitivity; however, immunohistochemistry may help to identify additional cases.175,176 Serology (ELISA and immunofluorescence antibody test) shows poor correlation with parasitological diagnosis and may underestimate infections in cats due to differences between individual immune responses to the disease. 177
Key Points
Many of the Cutaneous manifestations of inflections disease are uncommon. It is important always to look for the common first, and then consider other differentials when conventional therapy is not working.
When approaching atypical cutaneous disease in cats, start by identifying the main reaction pattern to narrow down the differential list.
Diagnosis can be difficult - diagnostic tests likely to give the most information should be pursued first. Cytologic examination should always be performed prior to biopsy collection.
Consider biopsy early in cases that do not follow the typical pattern.
Consider FIV/FeLV status in cats with skin lesions consistent with squamous cell carcinoma or Bowenoid in-situ carcinoma.
Keep in mind regional variations in disease.

Footnotes
Conflict of interest
The authors declare that there is no conflict of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this article.
