Abstract
Objectives
The pharmacokinetics of praziquantel and pyrantel pamoate has never been reported in cats. The present study was designed to establish the plasma concentration–time profile and to derive pharmacokinetic data for a combined formulation of praziquantel and pyrantel in cats, after a single, oral administration.
Methods
Twenty-two clinically healthy adult cats were used, each receiving a single oral dose of praziquantel (8.5 mg/kg) and pyrantel (100 mg/kg). Blood samples were collected at regular time points up to 48 h post-dosing. Plasma concentrations of praziquantel and pyrantel were measured using a liquid chromatography–mass spectrometry–high-throughput screening method.
Results
Clinical examination of all cats did not reveal any side effects after oral administration of these medications. The terminal half-life for praziquantel and pyrantel was 1.07 and 1.36 h, respectively. Praziquantel peak concentration (Cmax) was 1140 μg/ml, reached at 1.22 h. The plasma concentrations of pyrantel after oral administration were low with a mean Cmax of 0.11 μg/ml, reached at a Tmax of 1.91 h. Pyrantel showed a very limited absorption as pamoate salt, suggesting permanence and efficacy inside the gastrointestinal tract, where the adult stages of most parasitic nematodes reside.
Conclusions and relevance
Pyrantel showed a very limited absorption as pamoate salt. Praziquantel was rapidly absorbed following oral administration and the concentrations achieved suggest that praziquantel could be an effective and safe medication in cats. Although some resistance problems are arising as a result of their long use, these anthelminthic products can still play a major role in parasitic control, especially in geographical areas where the high cost of newer treatments or necessity of parenteral administration could decrease the number of treated animals.
Introduction
Helminth infections are a major animal and human health concern, with hookworms infecting large numbers of people worldwide, especially those of low economic status. 1 Dogs and cats play a major zoonotic role, both in transmitting parasites that use companion animals as their definitive and paratenic host and in mechanically transmitting and spreading the dissemination range of an array of human-specific parasites.2–4 As an example, a recent feline study in metropolitan Rio de Janeiro revealed an 89.6% prevalence of gastrointestinal helminth parasites in cats. 5
Praziquantel shows a broad anthelmintic profile of action against parasites known as ‘cestodes’ or tapeworms. The common tapeworm of dogs and cats, Dipylidium caninum, is the usual target, although praziquantel is also effective against trematodes and less common types of tapeworms such as Taenia species and the more dangerous Mesocestoides species.6,7
Pyrantel is usually available as tartrate and pamoate salts, which have different pharmacokinetic properties. Pamoate salt is almost insoluble in water and poor bioavailability from the gastrointestinal tract has been reported for humans, pigs, horses, donkeys and water buffalos, and most is excreted unchanged in the faeces.8–12 Reduced systemic absorption of the pamoate form potentially increases the availability in the lumen of the intestine, which is considered an advantage for young animals. 9 Pyrantel is a broad-spectrum anthelmintic, effective against roundworms (Ascaris species), threadworms (Enterobius species), hookworms and trichostrongylus in humans, domestic animals and horses. These salt formulations of pyrantel are not effective against whipworms. The pharmacokinetics of pyrantel has been described in horses, 10 donkeys, 11 pigs and dogs.9–13 In the case of praziquantel, pharmacokinetics have been reported for water buffalo, 14 dogs, 15 rats, sheep and donkeys.16,17 Although the efficacy of these two drugs has been demonstrated,18,19 considering the extensive use of combinations of prazi quantel and pyrantel in dogs and cats, pharmacokinetic information might help to optimise treatments and minimise the possible adverse effects in cats.
The objectives of the present study were to establish the plasma concentration–time profile and to derive pharmacokinetic data for a combined formulation of praziquantel and pyrantel in cats after a single oral administration.
Materials and methods
The study was conducted in compliance with applicable national legislation and was reviewed and approved by the Bioethics Committee of the University of Agricultural Sciences and Veterinary Medicine of Cluj-Napoca.
Animals
Twenty-two clinically healthy adult cats weighing 2–3.2 kg and aged 1.8–3.6 years were included in this study. The cats were submitted to a prestudy medical examination 7 days prior to the beginning and at the end of the study. During the administration days, all cats received water in a standardised way, with regard to the composition and time of administration. The animals fasted for at least 10 h prior to the study drug administration and up to 4 h after.
Experimental design
Each animal received together a single oral dose of 8.5 mg/kg praziquantel and 100 mg/kg pyrantel (Aniprantel Cattabletta; VimSpectrum).
Blood samples (2 ml) were collected from the cephalic vein, into heparinised tubes, at 0 (pre-treatment), 0.5, 1, 1.5, 2, 4, 8, 12, 24, 36 and 48 h post-dosing. Samples were centrifuged at 480 G for 10 mins and the plasma was stored at −20ºC until assayed.
Analytical method
Plasma concentrations of praziquantel and pyrantel were measured using a liquid chromatography–mass spectrometry (MS)–high-throughput screening (HTS) method. The system was equipped with a triple quadrupole mass spectrometer (Quantum Access MAX) connected to a computer with a ThermoXcalibur software 2.2 (Thermo Fisher Scientific). Praziquantel and pyrantel pure standards (Sigma-Aldrich) were used for quality controls.
Briefly, plasma samples (200 µl) were vortexed with internal standard (indapamide), centrifuged at 4300 G for 10 mins and 180 µl supernatant was transferred to chromatographic vials and injected (5 µl) into the high-performance liquid chromatography (HPLC)–MS–HTS. Aqueous 0.2% formic acid was used to dilute samples. The online extraction method (HTS-Turbo Flow) used a Turbo Flow Column Cyclone-P 0.5 × 50 mm (Thermo Fisher Scientific). The HPLC method used a Hypersil Gold column (50 × 2.1 mm, 1.9 μm; Thermo Fisher Scientific). For the MS, an ionisation source (heated electrospray ionisation) with positive polarity was used. The plasma calibration curves were linear over the concentration range of 20–400 ng/ml (correlation coefficients >0.9978) for praziquantel and pyrantel. The limit of quantification was 20 ng/ml for pyrantel and praziquantel. The limit of detection was 6 ng/ml for pyrantel and 20 ng/ml for praziquantel. Both the inter- and intra-day precisions were <5.8% for praziquantel and <7.6% for pyrantel at three quality-control levels (0.05, 2 and 30 μg/ml).
Pharmacokinetic analysis
A non-compartmental model using the Kinetica software program (version 5.0; Inna Phase) was used to determine pharmacokinetics parameters.
Statistical analysis
Descriptive statistical parameters (mean ± SD) were calculated. Harmonic means were calculated for the half-lives of elimination and absorption. 20 The statistical software used was SPSS (version 20.0; IBM). Data were evaluated for normality using the Shapiro–Wilk test and equality of variance using Levene’s test.
Results
The mean ± SD plasma concentrations of praziquantel and pyrantel following oral administration are plotted in Figure 1 and pharmacokinetic parameters (mean ± SD) from both praziquantel and pyrantel after oral administration are summarised in Table 1. Plasma concentrations of both drugs were not detected 12 h post-administration. Clinical examination of all cats after oral administration of this combination therapy did not reveal any side effects. The terminal half-life (t1/2λz) for praziquantel and pyrantel was 1.07 and 1.36 h, respectively. Praziquantel peak concentration (Cmax) was 1.14 ± 0.61 μg/ml, reached at a Tmax of 1.22 ± 0.49 h. Pyrantel Cmax was 0.11 ± 0.66 μg/ml at a Tmax of 1.91± 1.19 h.
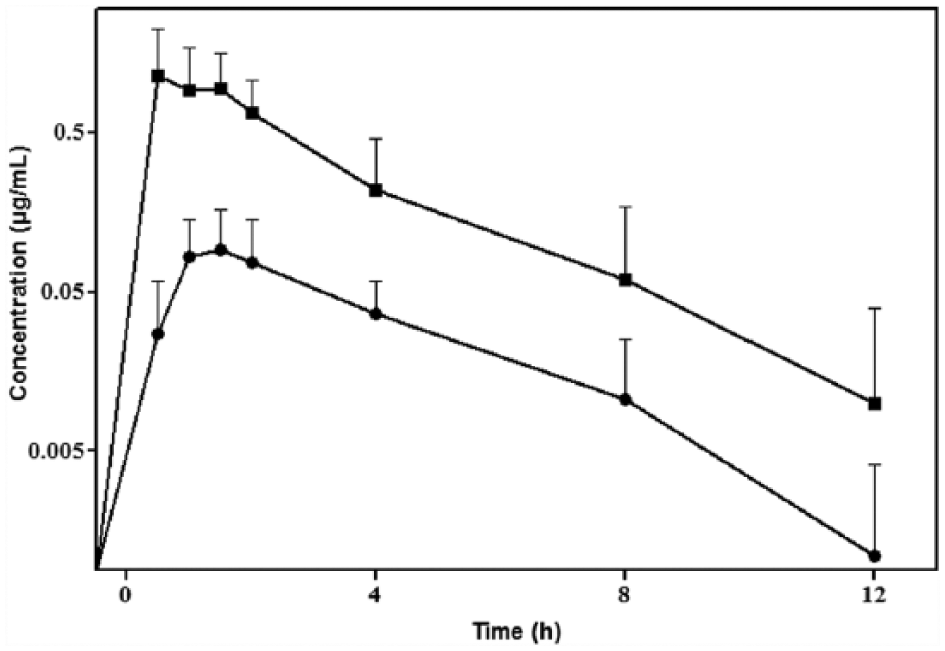
Semi-logarithmic plot of plasma concentrations (mean ± SD) of praziquantel (8.5 mg/kg;■) and pyrantel (100 mg/kg; ●) after a single oral administration in cats (n = 22)
Pharmacokinetic parameters of praziquantel and pyrantel in cats after oral administration (n = 22)

Data are mean ± SD
Harmonic mean
t½λz = the elimination half-life associated with the terminal slope (λz) of a semi-logarithmic concentration–time curve; Ka = absorption constant; t½ka = absorption half-life; AUC0–∞ = the area under the plasma concentration–time curve from zero to infinity; AUC0–last = the area under the plasma concentration–time curve from zero to the last point of concentration; MRT = mean residence time; Cmax = the peak or maximum plasma concentration following extravascular administration; Tmax = the time to reach peak or maximum plasma concentration following extravascular administration
Discussion
Praziquantel and pyrantel pamoate have been used as anthelmintic drugs for the past few decades in different domestic animals. However, despite the wide use of this combination in companion animals, there are very limited pharmacokinetic data for these two drugs, and no pharmacokinetic studies have been reported for this combination in cats.
The pharmacokinetics of praziquantel have been studied in dogs, monkeys, sheep and rats, 17 water buffalo, 14 and in combination with pyrantel in dogs. 15 Pyrantel pharmacokinetics have been studied in humans, pigs, horses and donkeys.9–12,21
After oral administration of the pyrantel and prazi quantel combination, the praziquantel half-life (t½λz = 1.07 h) in the present study was shorter than that reported in water buffalo (t½λz = 6.35 h) 14 and humans (2.5 h). 22 It has been reported that there is an extensive hepatic first-pass effect of praziquantel after oral administration in other species like cattle and water buffalo with a high liver oxidative metabolism.14,22 Data from the present study (short half-life, mean residence time and Cmax) suggest an important first-pass effect of praziquantel in cats that might contribute to the low bioavailability of praziquantel. However, low bioavailability is mainly owing to its poor hydrosolubility. Praziquantel is a class II compound (high permeability, low solubility) and thus presents poor solubility in water and, consequently, low absorption through the gastrointestinal tract. However, further studies would be necessary to confirm these aspects. Praziquantel rapidly distributes throughout the body and across the blood–brain barrier in other animal species, and lethal effects have been described in cats at injected doses of 200 mg/kg. 23 Although the dose administered in this study (8.5 mg/kg) is far lower than this lethal dose of 200 mg/kg, no neurological signs were observed in any cat included in this study, suggesting a wide margin of safety for praziquantel.
The pyrantel pamoate half-life after oral administration of 100 mg/kg in the present study was 1.36 h, shorter than that reported with the same pamoate salt in horses (t½λz = 13.43 h) 10 and donkeys (12.39 h). 11 Pyrantel pamoate salt has been widely used in small animals. This salt is largely insoluble in water and undergoes very little absorption.9,24,25 It is excreted largely unchanged in the faeces, keeping a high concentration throughout the gastrointestinal tract. In fact, in the present study data the low plasma levels of pyrantel suggest that the majority of pyrantel remains in the gastrointestinal tract following oral administration as a pamoate salt. The plasma concentrations of pyrantel after oral administration were low with a mean Cmax of 0.11 μg/ml, similar to a peak concentration of 0.09 μg/ml reported in horses. 10 However, Tmax was 1.91 h, shorter than that for the same salt in horses (7.5 h). The difference in Tmax values between these two species could be owing to the difference in the anatomy of the gastrointestinal or in the study design, especially with respect to the number of blood samples taken around the Tmax. According to the higher doses reported in other studies, in our study (100 mg/kg) the bioavailability of pyrantel seems to be very low compared with other species, thereby increasing the safety not only for adult cats, but for young cats as well, providing that the effect produced by pyrantel is inside the gastrointestinal tract. Pyrantel pamoate plasma concentrations have shown greater persistence after oral administration in other species. In the present study, a mean residence time of 2.99 h and the lower concentrations of pyrantel reflect a marked low bioavailability and shorter persistence in cats.
Most pharmacokinetic studies of praziquantel or pyrantel have been performed with individual drug administration and there is a paucity of data regarding the combination of these two drugs. One of the most important requirements to be considered regarding the administration of a drugs combination is the similarity of their pharmacokinetic profiles. In this case, although the site of action of praziquantel (systemic) and pyrantel (local) are different, similar elimination parameters in cats are additional factors to be considered in the present combination.
Conclusions
This study reports the pharmacokinetic profile of praziquantel and pyrantel after oral administration for the first time in cats. Pyrantel showed a very limited absorption as pamoate salt, suggesting permanence and efficacy inside the gastrointestinal tract, where the adult stages of most parasitic nematodes reside. Praziquantel showed rapid absorption and the concentrations reached suggest that it could be effective and safe in this species. Although some resistance problems are arising as a result of their long-term use, these anthelminthic products can still play a major role in parasitic control, especially in geographical areas where the high cost of newer treatments or the necessity of parenteral administration could decrease the number of treated animals.
Footnotes
Acknowledgements
The authors would like to thank the Vim Spectrum Group for the financial support and data provided.
Conflict of interest
This study was sponsored by Vim Spectrum, Hungary. LG is an employee of Vim Spectrum. The author meets the criteria for authorship as recommended by the International Committee of Medical Journal Editors and participated in the analysis and interpretation of the data.
Funding
This work was supported by a privately funded research grant: ANIMAL-MED Kft, GERMANY, study code VIM–ANIPR–10.11; number 3940/24.04.2012.
